Abstract & Commentary
New Recommendations for HIV Testing
Detecting HIV early creates opportunity for treatment, reduces transmission risk
By Stan Deresinski, MD, FACP, FIDSA
Dr. Deresinski is Clinical Professor of Medicine, Stanford University, Editor of Infectious Disease Alert.
Centers for Disease Control and Prevention and Association of Public Health Laboratories. Laboratory testing for the diagnosis of HIV infection: updated recommendations. Available at http://www.cdc.gov/hiv/pdf/HIVtestingAlgorithmRecommendation-Final.pdf
Recommendations for testing for HIV infection have continually evolved as the result of improving technology and clinical knowledge. The latest generation assays allow earlier detection of infection and avoid the frequent false-negative and indeterminate results seen with use of Western blot or immunofluorescence assays (IFA) for confirmation in patients with early infection. This is of importance because of the recognition that the risk of transmission is highest during the acute early phase of infection and because treatment is beneficial in all phases of infection, including the early phase. Furthermore, while HIV-2 infection remains rare in the U.S., use of the HIV-1 Western blot misclassifies the majority as HIV-1 infection. Western blot and IFA tests are no longer included in the testing algorithm.
As a consequence of this evolution, CDC and the Association of Public Health Laboratories have provided new recommendations for HIV testing of serum or plasma that supersede previous ones (See algorithm page 122). The changes take into full account the sequence of appearance of laboratory markers in the course of HIV-1 infection. Thus, HIV-RNA becomes detectable by nucleic acid amplification tests (NAT) approximately 10 days after infection and increases to very high concentrations. Four to 10 days after viral RNA is detectable, 4th generation immunoassays are able to detect P24 antigen, but this is transient as the result of the appearance of antibody — unless special methods to disrupt antigen-antibody complexes are used. IgM antibody may be detected by 3rd and 4th generation immunoassays 3-5 days after P24 antigen is first detectable and 10-13 days after the appearance of viral RNA. The sequence can be more broadly considered to consist of an initial eclipse period when no markers are detectable, a seroconversion window between initial infection and first detection of antibodies, acute HIV infection describing the interval between HIV RNA and antibody detectabilities, and established HIV infection beginning with a fully developed IgG antibody response.
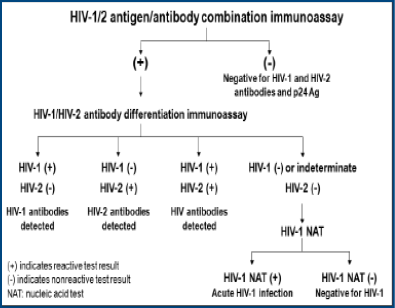
Initial testing should be with an FDA-approved antigen/antibody combination (4th generation) immunoassay that detects antibodies to both HIV-1 and HIV-2 as well as HIV-1 p24 antigen. If this test is negative, no further testing is indicated. If the test is reactive, additional testing with an FDA-approved antibody immunoassay that differentiates antibody to HIV-1 and HIV-2 should be performed. If the 4th generation immunoassay is reactive but the antibody differentiation test is negative or indeterminate, an FDA-approved HIV-1 NAT should be performed.
COMMENTARY
These recommendations emerge from the continuing advance in the technological and clinical aspects of testing for HIV-1 infection. The elimination of Western blot and IFA testing for confirmation of the presence of specific antibody is important given the subjectivity and both indeterminate falsely negative results seen with these assays. The ability to detect HIV-1 infection in its earliest phase provides the opportunity to initiate treatment which is clinically beneficial to the patient and reduces the risk of transmission during a time when viral loads reach their highest levels in plasma in the absence of therapeutic intervention.

