Prostate Health to a T? Tea and Tomatoes for Cancer Chemoprotection
Prostate Health to a T? Tea and Tomatoes for Cancer Chemoprotection
By Susan T. Marcolina, MD, FACP, and George Heffner, DO. Dr. Marcolina and Dr. Heffner are physicians at the HealthPoint Community Health Clinic in Kent, WA; they report no financial relationship to this field of study.
Prostate cancer is the most common non-cutaneous cancer and is the second leading cause of cancer death for men in the United States.1 There are significant differences in outcomes for localized vs. advanced disease, with 5-year survival rates of 100% vs. 31%, respectively.2 As a result, screening programs with prostate-specific antigen (PSA) were instituted and promoted on the assumption that early detection and treatment would be the optimal means to decrease the morbidity and mortality of advanced disease. For various reasons discussed below, this screening program has fallen short of expectations. It may now be time, especially in view of the fact that dietary factors may have important implications for mitigating the course of prostate disease, to re-emphasize alternative strategies for improving prostate cancer outcomes.
PSA Screening in the United States
Approximately 75% of men in the United States have had at least one PSA blood test.3 In association with this high rate of screening there has been a dramatically increased incidence of prostate cancer from 1 in 11 in 1980 to 1 in 6 today,4 yet there has not been a significant decrease in mortality compared with the United Kingdom, where PSA has not been universally adopted.5 Two prostate cancer screening trials reported in this past year showed disappointing results. The European Randomized Study of Screening for Prostate Cancer (ERSPC) showed that, although PSA-based screening decreased the rate of prostate cancer death by 20%, it was associated with a risk of overdiagnosis (defined as diagnosis of prostate cancer in men who would not have clinical symptoms in their lifetime), which was estimated to be 50% in the screened group.6 In the United States, the PLCO Cancer Screening Trial Project showed that the rate of prostate cancer death was not significantly different between the screened (with PSA levels and digital rectal exam) and control groups.7
The ERSPC Trial revealed a significant cost to decreasing mortality in that more than 1,000 men had to be screened and 48 treated to prevent one prostate cancer-related death. It is important to note that surgical, radiation, and hormonal treatment modalities for prostate cancer can have problematic side effects including urinary and fecal incontinence, sexual dysfunction, and infections that can severely affect quality of life. An assessment of the risk/benefit of treatment should be considered, particularly in men with limited life expectancies who may never experience clinical symptoms of prostate cancer.
The Screening Utility of PSA
The Prostate Cancer Prevention Trial (PCPT) was a phase III, randomized, double-blind, placebo-controlled trial of more than 18,000 men older than age 55, which evaluated the role of finasteride (a 5-alpha reductase inhibitor) vs. placebo for the prevention of prostate cancer. Prostate biopsies from patients with a range of PSA levels, including many below the cutoff of 4.0 ng/mL, were examined.8 Interestingly, they found that there was no level of PSA below which cancer was not found. As a matter of fact, in a cohort study of more than 5,500 men from the placebo arm of the PCPT trial (88% had a PSA level £ 4.0 ng/mL), 22% had biopsies positive for prostate cancer, 11% of which were in patients with a PSA level of 0-1 ng/mL.9 Because the PSA test cannot discriminate between non-cancer disease (benign prostatic hypertrophy and prostatitis) and cancer, its use as a screening tool is limited.10,11
Recommendations for Screening for Prostate Cancer
In recognition of the evidence that treatment for prostate cancer detected by screening can cause moderate to substantial harm, particularly in men with comorbidities that may more imminently affect their life expectancies, the U.S. Preventive Services Task Force (USPSTF) has concluded that the benefits of screening for prostate cancer in men younger than age 75 are uncertain or indeterminant, an I-level statement. This means that, according to current clinical evidence, the balance of benefits and harms cannot be ascertained. Therefore, if screening for prostate cancer is offered to patients, they should understand the uncertainty regarding the balance of benefits and harms. There is moderate certainty, however, that for men older than age 75, the harms of screening for prostate cancer outweigh the benefits; therefore, the USPSTF discourages prostate cancer screening in this group of patients. This is a D-grade statement for practitioners.12
Dietary Factors as Chemoprevention
Since prostate cancer has a long latency period, this provides an important opportunity to treat premalignant lesions. It has become increasingly evident over the past 15 years that dietary constituents have a unique ability to target multiple deregulated signaling pathways without interruption of normal processes. This has led epidemiologists to suggest that lifestyle and dietary choices may have important effects upon the development of prostate cancer. Migration studies highlight this role of dietary and lifestyle influences on prostate cancer development. Asian men who relocate to the United States and adopt a Western lifestyle and diet have a significantly higher risk of prostate cancer in comparison to their native Asian counterparts.13
Chemoprevention is the use of natural or synthetic agents that reverse, inhibit, or prevent cancer development in cancer-free individuals.14 Such agents will be utilized as prevention strategies if they have a favorable side effect profile, are broadly accessible, and if patients most at risk can be targeted for treatment through a combination of risk factors identified in their history and clinical characteristics15 (outlined in Table 1) and the use of specific serum biomarkers, which can be followed serially to determine effects.
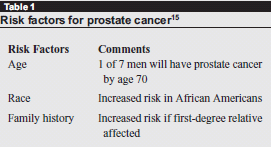 |
Epidemiologic studies have identified green tea polyphenols and lycopene as possible chemopreventive agents to mitigate the risk of prostate carcinoma.
Green Tea Polyphenols for Chemoprevention
Tea (Camellia sinensis) is the most consumed beverage worldwide after water and has been enjoyed by Asian populations for more than 5,000 years. Green tea, as opposed to other types of tea, is prepared by steaming freshly harvested leaves to prevent oxidation. Black tea, in contrast, is prepared by allowing the leaves to wilt, which causes oxidation of the polyphenols in the leaves.
Native Asian men with a high dietary intake of green tea have a low incidence of prostate carcinoma; therefore, green tea catechins (GTCs) have been proposed as chemoprotective. GTCs are released as the leaves are steeped in boiling water; they are water-soluble polyphenolic substances, the most abundant of which is epigallocatechin-3-gallate (EGCG), present in a concentration of approximately 100-150 mg per cup of brewed green tea.13,16 EGCG is the most studied catechin and manifests its significant growth-inhibitory properties in prostate cancer cells by interfering with signaling pathways responsible for cancer cell proliferation.
EGCG inhibits the 5-alpha reductase enzyme, which catalyzes the formation of testosterone to the more potent androgen dihydroxytestosterone. In vitro studies show selective and potent pro-apoptotic activity on prostate cancer cell lines, without toxicity on normal prostate cell lines.17
Polyphenon E (chemically defined, standardized extract of green tea polyphenols, primarily EGCG) has been shown to significantly decrease serum levels of several biomarkers, including hepatocyte growth factor (HGF), vascular endothelial growth factor, insulin-like growth factor-1, and insulin-like growth factor binding protein-3, in patients with localized prostate cancer taking this extract for a median of 34.5 days while awaiting prostate resection. HGF is a serum marker for prostate cancer; high levels are associated with metastatic disease and decreased survival.18,19 It is secreted by the stromal fibroblasts and activates tumor cell proliferation.
Bettuzzi et al, in a small, randomized, double-blind, placebo-controlled proof-of-principle trial of 60 Italian men with a diagnosis of high-grade prostate intraepithelial neoplasia, a known precursor to prostate cancer, demonstrated that treatment with 600 mg/d of GTC capsules for 1 year resulted in a statistically significant 10-fold decrease in progression to prostate cancer compared with the placebo arm (1/30 [3%] incidence vs. 9/30 [30%] incidence, respectively; P < 0.01).20 Although there was no significant change in PSA levels between the groups, the GTC-treated group tended to have lower values. Additionally, the GTC-treated men had statistically significant improvements in lower urinary tract symptoms as evidenced by improved International Prostate Symptom Scores without adverse effects reported.21
Lycopene/Processed Tomato Food Products for Chemoprevention
Lycopene is a non-provitamin A carotenoid antioxidant found in foods such as tomatoes, guavas, and watermelon and is the most efficient free-radical scavenger of this group of compounds.22 Tomatoes and tomato products constitute about 80% of this micronutrient in the United States.23 Absorption of this nutrient is increased by processing, heat, and the presence of fat.24 Tomato products consumed in oil, such as pizza and spaghetti/tomato sauce, are particularly bioavailable lycopene sources.
As a known antioxidant, lycopene may protect prostate tissue from oxidative damage by limiting cellular free radical exposure. Whole foods, such as tomatoes and tomato products, also contain other carotenoids and phytochemicals that may confer additional protection. Such foods may potentially modify risk in certain men with genetic polymorphisms of the manganese superoxide dismutase gene, which may put them at risk for prostate cancer.25 In vitro studies show that lycopene also inhibits the growth of human prostatic epithelial cells.26
In epidemiological studies, regular intake of lycopene in the form of tomato-based food sources and high blood levels of this carotenoid have been associated with a reduced risk of developing prostate cancer, particularly the more aggressive histologic variants.27,28 Not all studies, however, have shown a correlation between intake of lycopene-containing foods with decreased prostate cancer risk.
A prospective dietary intake assessment of more than 25 tomato-containing foods in almost 30,000 men over an average of 4 years in the PLCO cohort showed no overall association of intake of tomatoes and lycopene with prostate cancer, although inverse associations were suggested for some processed tomato products commonly cooked with fat. Subgroup analysis showed a trend toward lower risk, though not statistically significant, of histologically aggressive prostate cancer in men with increased consumption of pizza (3 1 serving weekly vs. < 0.5 servings/month) and spaghetti/tomato sauce (3 2 servings per week vs. < 1 serving/month). Men who had family histories of prostate cancer also trended toward a benefit (that was nonsignificant statistically) with increased pizza and spaghetti/tomato sauce consumption.29
Schwarz et al conducted a small, randomized, placebo-controlled chemopreventive study in 40 males (average age, 67 years) with benign prostatic hypertrophy,30 a common disease affecting men older than age 55 years and a common risk factor for prostate carcinoma.31 The patients were treated with 15 mg/d of an oral synthetic lycopene supplement as a powdered formulation (LycoVit®) with lunch or placebo for 6 months. The 6-month lycopene supplementation resulted in significantly increased plasma levels for the treated patients without adverse effects. Compared to the placebo group, PSA levels significantly decreased in the subjects treated with lycopene (P < 0.05) who also had a commensurate significant improvement in lower urinary tract symptoms (improved International Prostate Symptom Score questionnaire) compared to the placebo group.30
Chemoprevention Dosage
A synthetic lycopene supplement such as LycoVit has excellent bioavailability and is identical to a tomato-based supplement.32 At the dose of 15 mg/d used in clinical studies for 6 months, it was safe and well-tolerated. Processed tomato food products consumed in oil such as pizza and spaghetti/tomato sauce are particularly bioavailable (and popular) sources of lycopene. Two servings of a tomato sauce-based dish weekly may decrease the risk for prostate cancer in men with a family history or men with underlying genetic polymorphisms of superoxide dismutase, which increases their susceptibility to oxidative damage.25 Food sources of lycopene are listed in Table 2.33
 |
Clinical studies of green tea catechins for prostate carcinoma have used a standardized green tea catechin product called Polyphenon E in a dosage of 600 mg/d for 1 year20 or 800 mg/d over a median period of 34.5 days in patients with localized prostate cancer awaiting prostatectomy.18 This product is formulated to have low caffeine levels and is currently being used in Phase I/II cancer chemoprevention trials throughout the country.
Adverse Effects
The most common side effect of GTCs noted in clinical trials has been jitteriness, thought to be a result of caffeine intake.34 They should not be taken with monoamine oxidase inhibitors. Standardized pharmaceutical-grade preparations used in clinical studies such as Polyphenon E have very limited caffeine content, but it is important to check the content of each individual over-the-counter green tea supplement. The independent laboratory ConsumerLab analyzes over-the-counter products for contamination and accuracy of labeling and can be consulted before recommending specific supplements (www.consumerlab.org).
There have been case reports of green tea extract dietary supplements causing liver toxicity beginning from 5 days to 4 months after initiating use.35 Generally, liver function tests revert to normal after discontinuation of the green tea extracts, although one person required liver transplantation. Such toxicity has not been reported with consumption of green tea beverages. It is possible that certain extraction processes, such as ethanolic extractions, may have contributed some toxic compounds that caused the hepatic injury. Green tea contains a small amount of vitamin K that could counteract the blood-thinning effects of warfarin; however, large quantities of tea beverages would be required to provide a clinically significant amount of vitamin K.
Conclusion
Modest regular consumption of antioxidant-rich food such as tomato sauce products or green tea beverages may be a low-risk way for high-risk individuals to mitigate the chance of developing prostate cancer.
Recommendation
Men at high risk for prostate cancer, including African Americans and men with positive family histories, may consider a therapeutic indulgence in 2 weekly servings of a tomato sauce dish, or 1-2 cups of green tea daily.
References
1. Jemal A, et al. Cancer statistics, 2009. CA Cancer J Clin 2009;59:225-249.
2. Ries L, et al. SEER cancer statistics review. Available at: http://seer.cancer.gov/csr/1975_2005/. Accessed Dec. 12, 2009.
3. Sirovich BE, et al. Screening men for prostate and colorectal cancer in the United States: Does practice reflect the evidence? JAMA 2003;289: 1414-1420.
4. Seidman H, et al. Probabilities of eventually developing or dying of cancerUnited States, 1985. CA Cancer J Clin 1985;35:36-56.
5. Oliver SE, et al. Comparison of trends in prostate-cancer mortality in England and Wales and the USA. Lancet 2000;355:1788-1789.
6. Draisma G, et al; ERSPC. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med 2009;360:1320-1328.
7. Andriole GL, et al; PLCO Project Team. Mortality results from a randomized prostate-cancer screening trial. N Engl J Med 2009;360:1310-1319.
8. Thompson IM, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med 2003; 349:215-224.
9. Thompson IM, et al. Assessing prostate cancer risk: Results from the Prostate Cancer Prevention Trial. J Natl Cancer Inst 2006;98:529-534.
10. Thompson IM, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level < or = 4.0 ng per milliliter. N Engl J Med 2004; 350:2239-2246.
11. Nadler RB, et al. Effect of inflammation and benign prostatic hyperplasia on elevated serum prostate specific antigen levels. J Urol 1995;154(2 Pt 1): 407-413.
12. U.S. Preventive Services Task Force. Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med 2008; 149:185-191.
13. Lee J, et al. Cancer incidence among Korean-American immigrants in the United States and native Koreans in South Korea. Cancer Control 2007;14:78-85.
14. Brawley OW. The potential for prostate cancer chemoprevention. Rev Urol 2002;4(Suppl 5):S11-S17.
15. The American Cancer Society. Cancer Facts and Figures 2008. Available at www.cancer.org/downloads/STT2008CAFFfinalsecured.pdf. Accessed Dec. 10, 2009.
16. Klein EA. Can prostate cancer be prevented? Nat Clin Pract Urol 2005;2:24-31.
17. Albrecht DS, et al. Epigallocatechin-3-gallate (EGCG) inhibits PC-3 prostate cancer proliferation via MEK-independent ERK1/2 activation. Chem Biol Interact 2008;171:89-95.
18. McLarty J, et al. Tea polyphenols decrease serum levels of prostate-specific antigen, hepatocyte growth factor, and vascular endothelial growth factor in prostate cancer patients and inhibit production of hepatocyte growth factor and vascular endothelial growth factor in vitro. Cancer Prev Res 2009;2: 673-678.
19. Humphrey PA, et al. Prognostic significance of plasma scatter factor/hepatocyte growth factor levels in patients with metastatic hormone-refractory prostate cancer: Results from cancer and leukemia group B 150005/9480. Clin Genitourin Cancer 2006;4:269-274.
20. Bettuzzi S, et al. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: A preliminary report from a one-year proof-of-principle study. Cancer Res 2006;66:1234-1240.
21. O'Leary MP. Quality of life and sexuality: Methodological aspects. Eur Urol 2001;40(Suppl 3):13-18.
22. Di Mascio P, et al. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch Biochem Biophys 1989;274:532-538.
23. Chug-Ahuja JK, et al. The development and application of a carotenoid database for fruits, vegetables, and selected multicomponent foods. J Am Diet Assoc 1993; 93:318-323.
24. Rao AV, Agarwal S. Bioavailability and in vivo antioxidant properties of lycopene from tomato products and their possible role in the prevention of cancer. Nutr Cancer 1998;31:199-203.
25. Li H, et al. Manganese superoxide dismutase polymorphism, prediagnostic antioxidant status, and risk of clinically significant prostate cancer. Cancer Res 2005;65:2498-2504.
26. Obermuller-Jevic UC, et al. Lycopene inhibits the growth of normal human prostatic epithelial cells in vitro. J Nutr 2003;133:3356-3360.
27. Mills PK, et al. Cohort study of diet, lifestyle, and prostate cancer in Adventist men. Cancer 1989;64: 598-604.
28. Giovannucci E, et al. Intake of carotenoids and retinol in relation to risk of prostate cancer. J Natl Cancer Inst 1995;87:1767-1776.
29. Kirsh VA, et al. A prospective study of lycopene and tomato product intake and risk of prostate cancer. Cancer Epidemiol Biomarkers Prev 2006;15:92-98.
30. Schwarz S, et al. Lycopene inhibits disease progression in patients with benign prostate hyperplasia. J Nutr 2008;138:49-53.
31. McVary KT. BPH: Epidemiology and comorbidities. Am J Manag Care 2006;12(5 Suppl):S122-S128.
32. Hoppe PP, et al. Synthetic and tomato-based lycopene have identical bioavailability in humans. Eur J Nutr 2003;42:272-278.
33. Mateljan G. The World's Healthiest Foods. 1st ed. Kihea, HI: The George Mateljan Foundation; 2007:740.
34. Pisters KM, et al. Phase I trial of oral green tea extract in adult patients with solid tumors. J Clin Oncol 2001;19:1830-1838.
35. Molinari M, et al. Acute liver failure induced by green tea extracts: Case report and a review of the literature. Liver Transplant 2006;12:1892-1895.
Prostate cancer is the most common non-cutaneous cancer and is the second leading cause of cancer death for men in the United States.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
