A(H1N1) 'Swine Flu' 2009 / 2010: Where We've Been, What We Now Know, Where We May Be Heading
A(H1N1) 'Swine Flu' 2009 / 2010: Where We've Been, What We Now Know, Where We May Be Heading
Author: James A. Wilde, MD, FAAP, Associate Professor of Pediatrics and Emergency Medicine, Medical College of Georgia, Augusta.
Peer Reviewer: Ghazala Q. Sharieff, MD, FACEP, FAAEM, FAAP, Division Director/Clinical Professor, Rady Children's Hospital Emergency Care Center and the University of California, San Diego; Director of Pediatric Emergency Medicine, California Emergency Physicians.
In the past nine months, the world has witnessed the outbreak of not one but two waves of pandemic influenza due to a new virus of swine origin. World public health authorities moved quickly to contain what appeared initially to be the severe pandemic that had been anticipated for so long.
Wave two has now subsided, giving public health authorities and health care providers alike the opportunity to assess the situation. The pandemic was not a hoax, as some have suggested, but it has been much lower in intensity than many expected. How did this happen? What have we learned about novel A(H1N1)? What is the likelihood that it will be back? This article considers these and other questions.
The Editor
Prelude to a Pandemic
On April 21, 2009, the Centers for Disease Control and Prevention (CDC) released a report about two patients in Southern California who had become ill as a result of infection from a previously unknown influenza virus.1 The new virus was a Type A, subtype H1N1, but distinct from the A(H1N1) human subtype that has circulated since the 1970s. It appeared that the origin of the virus was swine, so it became known initially as swine flu. Since then it has been referred to by a variety of names, including novel influenza H1N1 and influenza A(H1N1) 2009. The date of the first infection was March 28, and both patients recovered fully.
The first report was little noticed even in the medical world. A number of unusual sub-types of influenza originating in animals can occasionally be found in humans, usually as a result of close exposure to infected animals. There are human sub-types of flu and porcine sub-types of flu and avian sub-types of flu, and generally they stay within the species of origin, doing significant harm only to that species. The species barrier is only rarely breached, and usually not in a sustained fashion.
A recent major exception to that rule is high pathogenicity A(H5N1) avian influenza which burst on the world scene in Hong Kong in 1997 and subsequently spread throughout the entire Eastern Hemisphere.2 It has never been found in the Western Hemisphere. H5N1 "bird flu" received much attention from both the scientific community and the general public over the past 12 years because it spread so rapidly over a huge geographic area and because it continues to exhibit a mortality rate of over 60%. By comparison, the mortality rate from the Spanish influenza of 1918 was only 2%, but because Spanish flu had a very high attack rate, it led to the most deadly pandemic of any kind in world history.
The 20th century saw three major pandemics: A(H1N1) "Spanish flu" in 1918, A(H2N2) "Asian flu" in 1957, and A(H3N2) "Hong Kong flu" in 1968. The historical record shows that the world experiences on average three to five influenza pandemics per century. As a result, there was a renewed focus in preparation for an influenza pandemic at the state, national, and international level. The Strategic National Stockpile, a mechanism to store essential medical materials that would be needed during a pandemic, was built up with many supplies, most notably the antiviral medications oseltamivir (Tamiflu) and zanamivir (Relenza).
Although H5N1 avian flu remains a highly lethal infection for humans who are unlucky enough to become infected by it, by 2008 it was clear that it would not be the cause of our next pandemic.3 Three essential ingredients are needed for a pandemic to occur. First, a new virus must appear, one that has never been encountered by humans before. Second, there must be no pre-existing immunity to the new virus. Third, there must be sustained rapid spread from person to person. H5N1 met the first two criteria, but sustained rapid human-to-human spread has not been observed. Occasional person-to-person spread has been observed, particularly with intense exposure such as within a family unit, but casual contact does not appear to pose a risk for infection. As of 2010, fewer than 300 people worldwide are known to have died from H5N1 influenza, making it more of a pandemic for birds than for humans.3 The concern continues to be that a mutated form of H5N1 might emerge that does have the ability to spread quickly from person to person through casual contact alone. If that happens, a major pandemic is likely, with potentially catastrophic results.
Given this background information, the announcement by the CDC on April 23, 2009 that swine flu had been detected in six additional patients in Southern California and Texas caused alarm among flu experts.4 There was evidence in these cases for human-to-human spread. Initial genetic analysis by the CDC indicated that the virus had components from human, swine, and avian sources. On April 24, the Mexican government confirmed that at least seven cases had been detected in their country, with some deaths acknowledged. Swift genetic analysis by the CDC confirmed that the strains found in Mexico were identical to the strains found in the United States.4 Soon after, the Mexican government announced a cluster of 854 cases of pneumonia and 59 deaths in Mexico City thought to be due to H1N1. If these data were accurate, the mortality rate for this virus could be close to 8%, four times deadlier than the 1918 Spanish flu and almost as high as the 10% mortality seen with the aborted 2002 SARS epidemic. It appeared that the world might be witnessing the earliest stages of a new, deadly flu pandemic.
Events unfolded rapidly over the next few days. On April 26, 20 cases were confirmed in five U.S. states, including a cluster of students in New York who had recently traveled to Mexico for spring break. In response, the Department of Health and Human Services (HHS) declared a public health emergency in the United States, and 25% of the antiviral supplies in the Strategic National Stockpile were released to the states for distribution.5 By April 27, swine flu had been detected in at least five other countries, including New Zealand, Canada, UK, Spain, and Israel.6 This caused the World Health Organization (WHO) to raise the pandemic alert level from III to IV on the six-level scale, indicating that a new virus with pandemic potential had been detected with "increased" person-to-person spread.7
On April 29, just eight days after the world learned about swine flu, the WHO raised its pandemic alert level to V, indicating "significant" person-to-person spread of the virus.8 Analysis of novel A(H1N1) strains showed that the virus was completely resistant to the adamantane class of influenza antiviral medications (Amantadine, Rimantadine), but no resistance to the neuraminidase inhibitors oseltamivir or zanamivir was detected.9 A few days later, the CDC issued recommendations to prescribe oseltamivir or zanamivir to anyone with confirmed, probable, or suspected swine flu. The U.S. Food and Drug Administration (FDA) also issued an Emergency Use Authorization (EUA) for the use of oseltamivir in children younger than 1 year of age, an age group for which formal FDA approval had not yet been granted.10 The objective behind these policies was to try to limit the spread of the virus through the use of antiviral medications.
By the end of May 2009, there were 8,585 probable and confirmed cases of swine flu in the United States with 12 deaths, for a mortality rate of 0.14%.11 The Mexicans government now reported 4,910 confirmed cases and 85 deaths, for a mortality rate of 1.7%, still very high but much lower than the 8% rate reported earlier.12 There were also 2,673 cases of swine flu reported in 46 other countries around the world with only three deaths. It had become clear that except for the unusual number of deaths reported from Mexico, this virus was causing mortality in rates similar to or lower than mortality rates expected from seasonal flu. The data also indicated that the population most affected by the virus was the young; initial reports in the United States showed that the median age of those infected was 16 years, with very low attack rates for people older than age 50 years. This is highly unusual for influenza viruses, which infect 5%–10% of Americans each year and cause widespread morbidity and mortality in the elderly, who comprise over 90% of the 36,000 annual deaths due to flu.13
On June 11, the WHO raised the pandemic alert level to Phase VI, indicating sustained transmission of a novel influenza virus from person to person.14 The world was now officially in an influenza pandemic.
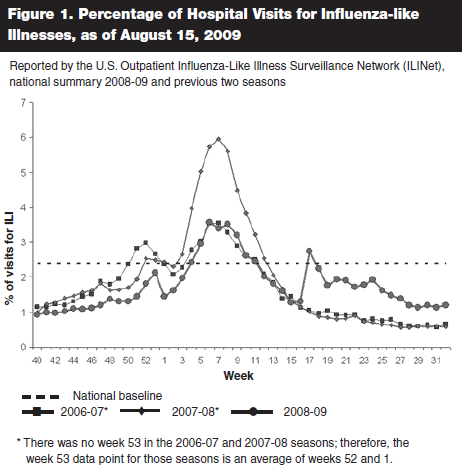
While H1N1 continued to spread throughout the United States and the rest of the world during the month of May, the sharp upturn in influenza-like illness (ILI) that was detected during the week ending May 2 (week 17 for CDC tracking purposes) was not sustained. By week 18 the national level of ILI had begun to decline, and by week 19 it sharply declined to below the epidemic threshold of 2.4%. (See Figure 1.) The explosive spread of infection feared by many in the influenza world did not occur, and it appeared that, in fact, the outbreak might be receding even as the pandemic was being officially recognized. In part due to these data, the CDC in June issued new recommendations to focus the use of antiviral medications on those who were hospitalized with likely swine flu and outpatients at high risk for complications from influenza infection. Treatment was no longer recommended for otherwise healthy outpatients even if swine flu was likely.15
The Summer Lull
Summer in the Northern Hemisphere, corresponding roughly to CDC weeks 22–34, brought continued slow spread of the virus. The overall rate of patients reported with ILI remained elevated compared to the rate normally seen for the time of year, but it was still hovering below the threshold that helps to define the epidemic period during a "regular" flu season. Throughout the summer, only a handful of U.S. states reported widespread infections, meaning at least half of the counties in the state had confirmed the presence of novel H1N1 during the preceding week.
The summer lull was viewed by many as a chance to better prepare for the anticipated return of swine flu in a second major "wave." It was unknown when this second wave would occur. Would it arrive in the winter, during the time when influenza normally circulates at epidemic levels? Or would it arrive at an earlier time, perhaps after the end of the flu season in the Southern Hemisphere, which generally runs from mid-May to mid-July? The other unknown was what strain of the new virus would predominate in our second wave. Influenza viruses are characterized by a high rate of mutation. One of the biggest concerns was that novel H1N1 would cause high levels of infection in the Southern Hemisphere during the May–July flu season and provide a breeding ground for mutated versions of the virus. The fear was that while the virus we knew so far showed low degrees of lethality, the virus to come might be significantly worse.
There was a concerted effort during our summer months to produce a vaccine against swine flu. If we could produce the vaccine quickly, there might still be a chance to avert a potential disaster when the virus returned in the fall or winter. This effort was being made not just in the United States but all over the world. Candidate strains were identified by the CDC as early as June, after which the long, slow process of producing vaccine began. In general, at least six months are required to produce sufficient amounts of vaccine after a new strain of influenza virus appears. The irony was that although we had extra time to prepare a vaccine, we had no way to know if the vaccine would still be effective if a mutated form of the virus appeared.
Vaccine mismatches happen with some degree of regularity due to the unpredictable nature of influenza virus mutations. The Fujian flu of 2003/2004 is a case in point. That year, candidate strains were chosen in early 2003 as always, based on the viruses that were circulating at the end of the Northern Hemisphere flu season and based on new strains detected in the Southern Hemisphere early in their flu season. Unfortunately, Fujian flu wasn't detected until the fall, too late to be incorporated into that year's flu vaccine. Fujian was an A(H3N2) virus that, like many H3N2 subtypes, caused severe illness. As predicted, the 2003/2004 was a particularly severe season. When the CDC retrospectively analyzed the mismatched flu vaccine against the Fujian strain in the spring of 2004, it was found to have less than 50% effectiveness.16
Flu Season in the Southern Hemisphere
While the swine flu continued to circulate in significant but relatively low numbers in the Northern Hemisphere during the months of June and July, flu season arrived in the Southern Hemisphere. Recognizing that the experience in the Southern Hemisphere might provide some insight about what might be coming in the fall and winter north of the equator, HHS began to gather data on the Southern experience. The countries surveyed included Argentina, Australia, Chile, New Zealand, and Uruguay due to their similarities to the United States with regard to demographics and economic development. A full report was issued on August 26 and is available online.17
There were a number of notable findings in this report. First, the experience in all countries was that influenza arrived as usual in May/June, peaked in mid-July, and decreased afterward. This pattern is consistent with typical flu seasons in the Southern Hemisphere, and showed that H1N1 2009 did not circulate for an unusual period of time. Figure 2 shows the data on visits to local physicians in New Zealand for ILI during the months of April to August, showing a typical eight- to 12-week period of heightened activity. The winter peak in activity during this time was three times that seen in the previous two seasons, indicating a more severe season than usual.17
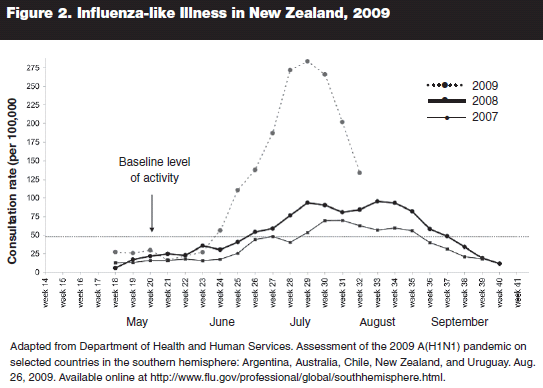
The predominant strain was 2009 A(H1N1) influenza, which accounted for 89% of all influenza viruses in the Southern Hemisphere by August. Notably, sequencing data indicate that the virus was genetically and antigenically stable during this time; there was no significant degree of mutation.17 Thus, the virus in late summer was still similar to the strains being used to produce vaccine throughout the world.
Clinical disease patterns resembled those from the United States and other countries to the north. 2009 A(H1N1) infections generally caused mild disease, with infections predominating in school-age children and adults younger than 65 years of age. Overall rates of hospitalization, severe illness, and death due to swine flu were similar to those observed thus far in the United States. Seventy-one percent of reported deaths occurred in adults 25–64 years old, with very few deaths reported in the over-65 category. The patients most at risk for complications included pregnant females and individuals with underlying medical conditions.
Antiviral medications were used in all five countries to treat various categories of patients, particularly those with medical conditions putting them at high risk for complications due to flu and those ill enough to be hospitalized. The virus remained sensitive to neuraminidase inhibitors throughout the season and no significant rate of resistance was found.
There were several implications of these findings for the expected second wave of infection in the Northern Hemisphere. It appeared that 2009 A(H1N1) had not mutated to a more deadly form during the time it circulated in the Southern Hemisphere. This meant that work could continue on vaccine development using strains selected early in the pandemic, with the growing likelihood that there would be a good match between the circulating strain and the vaccine strain. Furthermore, while certain subsets of the population were being affected more than would be expected during a normal seasonal flu outbreak, the overall lethality of the virus appeared to remain at or below that seen with seasonal flu.
The Second Wave Arrives
By August 2009 A(H1N1) had been circulating in the United States for well over three months, and an estimated 1 million people had been infected.18 CDC had recommended on July 24 that states discontinue reporting on individual cases of confirmed or probable swine flu and instead provide information on influenza-associated hospitalizations and deaths. During the period from April to the end of August, there were 9,079 laboratory confirmed hospitalizations and 593 deaths due to 2009 A(H1N1), a mortality rate of 6.5% in hospitalized patients but under 0.1% among all those estimated to be infected.19 Rates of ILI continued to hover somewhat over the levels usually seen at that time of the year, but were well below epidemic thresholds.
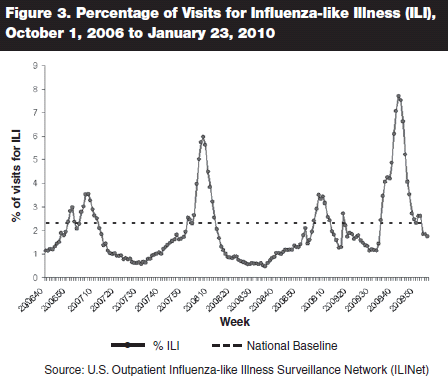
A sudden increase in ILI was noted first in Georgia, then in neighboring states in the Southeast United States, beginning around August 15.18 The Georgia State lab and CDC quickly confirmed that the increase was due to 2009 A(H1N1). Soon, rates of ILI increased to a level usually seen only during the peak periods of seasonal influenza in the winter, first in Georgia, then in other states. Over the next six weeks, more and more states reported increased levels of activity, peaking at 49 of 50 states in the continental United States reporting widespread infection as of the week ending October 16. Over the six weeks following October 16, swine flu decreased in activity at a rate similar to that observed in the Southern Hemisphere. (See Figure 3.)
Swine Flu: What We Now Know
During the first few months of the pandemic, most of the information on swine flu came from CDC and WHO briefings, MMWR dispatches, and online news reports regarding new information presented at national and international medical conferences. Beginning in the late summer, more and more information began to appear in medical journals throughout the world. The discussion below is a brief description of what we have learned about 2009 A(H1N1) up to the beginning of 2010.
The Virus. Genetic analysis of isolates from the original two infected patients in Southern California showed that the majority of the genes were similar to those of swine influenza viruses that have circulated in U.S. pig populations since 1999. However, two of the eight genes were from a Eurasian swine flu lineage. This combination of genes had never been found before in swine or human influenza viruses anywhere in the world.1 More detailed studies on isolates from Mexico and the United States were published in July. One revealed that 2009 A(H1N1) influenza virus has components that can be traced to humans, birds, and swine, with swine the most recent host for all eight genes.20 Very little genetic variability could be found in any of the isolates tested, suggesting that the introduction into humans may have been a single event.
Two publications in July pointed out that this virus and all viruses that have circulated in humans over the past 91 years can be traced to the 1918 A(H1N1) "Spanish flu" pandemic.21,22 The 1918 virus, thought to be derived from avian sources, was transmitted to pigs sometime before 1930 and remained relatively stable in the swine population until the late 1990s. Meanwhile, A(H1N1) viruses circulated in humans from 1918 until the outbreak of a new pandemic caused by A(H2N2) in 1957, disappeared for a time, then re-emerged in 1977. From 1977 to 2009, human A(H1N1) continued to evolve, leading to substantial divergence between human A(H1N1) and swine A(H1N1) viruses by the end of the century. Around 1998, swine A(H1N1) viruses reassorted with contemporary human A(H3N2) viruses and an avian influenza virus, creating a "triple-reassortant" A(H3N2) virus in swine populations throughout North America. Shortly after, similar triple-reassortant A(H1N1) was also detected, which evolved rapidly over the next 10 years. These events all served to create in swine a reservoir of viruses so antigenically distinct from their related human counterparts that a human pandemic could result.
Transmissibility. The basic reproductive number (R0) represents the number of infections caused by spread from a currently infected person who is introduced into a completely susceptible population.23 An R0 > 1 can support an epidemic while an R0 < 1 cannot. At an R0 of 2, spread of infection becomes logarithmic. The value for R0 is as high as 10 for varicella or measles, while values of 1.4 to 1.6 are typical for influenza. Soon after it was recognized that the 2009 A(H1N1) influenza virus was able to spread from person to person, work began on learning how transmissible it was. Early studies in Mexico and Peru showed that the R0 for this virus is probably between 1.2 and 1.7, making its transmissibility similar to seasonal flu.24,25 The R0 for the 1918 Spanish flu was probably 2.0 or higher.
As of June 1, 2009, 938 case reports of persons with confirmed or probable 2009 H1N1 influenza had been collected by the CDC. Analysis of those cases revealed that acute respiratory illness developed in only 78 of 600 household contacts (13%), and that no secondary respiratory illness developed in 72% of the 216 households.26 It was also found that household contacts younger than 18 years of age were twice as susceptible as those 19–50 years of age, and that those older than age 50 were the least susceptible. There was a mean of 2.6 days between onset of symptoms in an index patient and onset of symptoms in an infected household contact. A related study of an outbreak that occurred in a New York school from April 24 to May 8 showed a similar secondary attack rate in households of 17%.27 That study also showed a significantly higher attack rate for students than for school employees. These data confirmed that this flu virus was attacking the young in far greater numbers than those older than age 50, a pattern greatly different from that expected from seasonal flu.
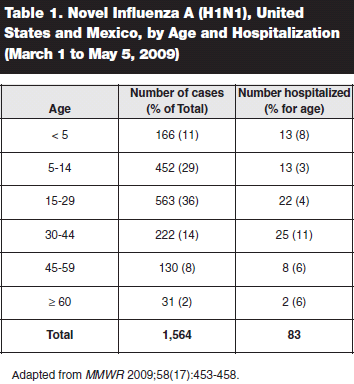
Epidemiology. Data on 1,591 confirmed 2009 A(H1N1) infections from Mexico and the United States during the period March 1 to May 5 revealed that two thirds of the infections were found in school-age children and adults younger than age 30, and another 10% of those infected were younger than 5 years of age.28 Only 10% of infected patients were older than age 45, and 2% were older than age 60. In contrast to seasonal flu, the virus was preferentially infecting the young, and those older than 50 years were largely spared. (See Table 1.)
A similar distribution of infections was shown in a report from the Chicago area.29 Investigators analyzed data from 1,557 patients with laboratory-confirmed 2009 A(H1N1) from April to July. The median age of confirmed cases was 12 years. The overall attack rate was highest among children age 5–14 years, at 147 per 100,000 population; next highest at 113 per 100,000 for children age 0–4; and lowest for those older than age 60, at 10/100,000. There was a consistent decrease in the attack rate for each successive age group older than the age of 14 years.
A study published in the New England Journal of Medicine in September shed some light on a possible reason for this unusual age distribution of infection.30 In this study from the CDC, investigators measured cross-reactive antibodies to 2009 A(H1N1) in stored serum samples from people who had been vaccinated with seasonal flu vaccine from 2005 to 2009, and from stored samples from patients who were vaccinated with the swine influenza vaccine of 1976. The analysis showed that people younger than 30 years of age had little to no evidence of pre-existing cross-reactive antibodies to 2009 A(H1N1), but at least 34% of those born before 1950 and 100% of those born between 1910 and 1929 had titres of 80 or more. Furthermore, 54% of patients vaccinated against the 1976 A(NJ/76) (H1N1) swine flu had evidence of cross-reactive antibodies against 2009 A(H1N1). Vaccination with contemporary seasonal flu vaccines did not lead to significant increases in cross-reactive antibodies against 2009 A(H1N1) in any age group. These data suggest that prior exposure to a 1918-like H1N1 virus had occurred in many older adults, and that this prior exposure resulted in cross-reactive antibodies against 2009 A(H1N1). This theory is also consistent with the information on the evolution of influenza viruses discussed in the previous section.
Symptoms. Two early reports on the swine flu showed a pattern of symptoms that was similar to seasonal flu, but with key differences. The first, released on April 30, was based on data collected primarily from a New York City high school with 2,686 students and 228 staff members.31 Between April 20 and April 24, at least 222 students and staff developed acute respiratory illnesses. There were 42 specimens collected on symptomatic patients in local physician offices and emergency departments, and 37 (88%) were later confirmed positive for swine flu by the CDC. Subjective fever was found in 96% of those with confirmed swine flu, and each of the following symptoms were found in at least 80%: cough (98%), fatigue (89%), headache (82%), sore throat (82%), runny nose (82%), chills (80%), or muscle aches (80%).31 These symptoms are typical for influenza in this age group. However, 55% reported nausea, and 48% reported diarrhea, which are relatively rare in seasonal influenza infections.32 Subsequent reports confirmed a relatively high rate of vomiting and diarrhea in patients with confirmed swine flu.28,33,34
Hospitalized Patients. In the May 8 MMWR report on the swine flu outbreak in a New York school, there was only one short hospitalization and no deaths among the 44 confirmed cases of 2009 A(H1N1), indicating that, at least in this low-risk population, the virus did not appear to be any more severe than seasonal flu. Subsequent data confirmed that hospitalization rates for school-age children were no higher overall than for seasonal flu.
Characteristics of patients who did require hospitalization have been detailed in several reports and have a consistent pattern: unusually high proportions of the young required inpatient management, two thirds had underlying medical conditions, about one quarter required management in an intensive care unit, and 7%–11% died.33,34 These data are in sharp contrast to data from several decades of surveillance for seasonal flu, which show that the overwhelming majority of hospitalizations due to influenza are in patients older than 65 years of age, with a secondary peak in hospitalizations for children younger than 5 years of age.35
In the first report, data were extracted from the charts of 272 patients hospitalized with confirmed swine flu in 24 states from May 1 to June 9.33 This represented about 25% of the 1,082 hospitalizations reported to the CDC during this period. Seventy-three percent had at least one underlying medical condition, including 60% of the children and 83% of the adults. The median time from onset of symptoms to hospital admission was three days. The median age of the patients was 21 years. Forty-five percent of the patients were children younger than 18 years; over one third were between 18 and 49 years of age; and only 5% were age 65 or older. Forty percent of those who had a chest radiography performed on admission had evidence for pneumonia. Sixty-five of the 272 patients (25%) were admitted to an intensive care unit, and 42 required mechanical ventilation (14%). Overall, 7% of admitted patients were pregnant females, a group that comprises only 1% of the general population.
A subsequent report on 1,088 patients hospitalized with confirmed swine flu in California from April to August showed remarkably similar results.34 In addition, while this study demonstrated a very high rate of admission for the young, particularly children younger than 1 year of age, hospitalized persons older than age 50 had the highest mortality. This study also found a strong association between morbid obesity and hospitalization.
A study from Argentina focused on hospitalizations for children infected by swine flu between May and July.36 Rates of hospitalization were double the rate expected from seasonal flu. Care in an intensive care unit was required for 19%, and 17% required mechanical ventilation. Overall, 5% died. The hospitalization rate was twice the rate observed during the 2008 seasonal flu. Hospitalization rates were particularly high for children younger than 1 year of age, after which rates dropped off sharply. The most striking finding in this study was that the mortality rate was 10 times higher in children with 2009 A(H1N1) than it was for the seasonal flu circulating in 2007.
Two studies examined patients admitted to intensive care units for confirmed 2009 A(H1N1) infection.37,38 Both reported mortality rates of about 17%. Both found an unusual concentration of patients in the pediatric or young adult age groups, with few patients older than age 65. Both reported a proportion of obese patients and pregnant patients that was significantly higher than in the general population.
Mortality. The early reports suggesting up to 8% mortality in Mexico proved to be overestimates. By May 8, the mortality rate in Mexico for confirmed cases had been revised down to 4.4%,39 and further data gathered to June 1 showed an overall mortality rate of about 2%.40 As of May 8, there were no deaths reported for the 309 persons with laboratory-confirmed infection outside of the United States and Mexico, and only two deaths out of the first 1,487 confirmed cases in the United States.
Annual mean influenza-associated mortality rates for underlying pneumonia and influenza deaths in the United States were estimated for various age groups in a study from 2003. This study showed a rate of 0.3 deaths due to flu per 100,000 in children younger than 1 year of age, 0.2 in children 1–4 years, 0.2 in patients age 5–49 years, 1.3 in those 50–64 years, and 22.1 for patients older than age 65.41 This study also showed that 90% of the influenza-associated deaths occur in persons older than age 65.
Data on mortality due to influenza in children have been collected nationally by the CDC only since the severe 2003–2004 Fujian flu epidemic, when a total of 154 deaths were recorded nationally for a mortality rate of 0.2 deaths per 100,000 children.42 In all subsequent years until 2009, the total number of deaths due to influenza in children have not exceeded that number. By comparison, the mortality rate for children with confirmed 2009 A(H1N1) influenza reported in a recent report from Buenos Aires from May to July was 1.1 per 100,000 children, five times the rate from Fujian flu and 10 times the rate for the less severe 2007 seasonal flu.36
In August, an analysis of the first 36 deaths in U.S. children from laboratory-confirmed 2009 A(H1N1) showed that two thirds of the children had high-risk medical conditions. Furthermore, 92% of the children with high-risk medical conditions had neurodevelopmental conditions.43
Testing. When 2009 A(H1N1) first appeared, and for a short time after, there was an urgent need to track progression of the pandemic through testing of subjects with flu-like illness. Some of this was done with point-of-care rapid influenza tests that were already available, and some was done with RT-PCR confirmatory assays that were available only through the CDC. Within only a few weeks of the appearance of the new virus, it was clear that spread throughout the United States had already occurred, and that the rise in ILI cases was due to swine flu. At that point, the CDC changed several of its recommendations about testing. Since it was no longer necessary or helpful to confirm every case, the CDC adopted a strategy of confirming only those cases of flu-like illness in hospitalized patients, which would permit public health authorities to monitor the severity of the pandemic as measured by hospitalization and death. Many state labs, which had obtained reagents for rRT-PCR testing from the CDC by July, adopted a similar strategy.
Early studies indicated that the available rapid tests lacked sensitivity in the detection of 2009 A(H1N1). Sensitivities of 60%–90% are reported for the detection of influenza by rapid tests during normal seasonal flu outbreaks.44 In contrast, rapid tests approved for the detection of seasonal influenza showed sensitivities of only 10%–70% in the detection of swine flu depending on the viral titer.45 Given this poor performance, the CDC issued recommendations to limit most influenza testing to nucleic acid amplification assays (rRT-PCR) in inpatients, and to manage outpatients based on their risk category and symptoms.46 The decision to use antiviral medications depended on symptoms and risk category, not on the result of a rapid test, so there was little benefit in the use of rapid tests in the management of outpatients.
Treatment. A number of studies have been published on the use of neuraminidase inhibitors in the treatment of both children and adults with acute influenza infection. In general, the literature shows a modest benefit in both groups if the medication is started within 48 hours of symptom onset.47,48 There is no published evidence that the use of neuraminidase inhibitors decreases significant morbidity or mortality in children, including children at high risk for complications due to flu. There are some studies showing reductions of morbidity and mortality in adults at high risk for complications, but those studies were in hospitalized patients or high-risk older adults.49 There have been no published studies showing a reduction in mortality in young, healthy adults who were treated with neuraminidase inhibitors. In both children and adults, the primary benefit from the use of neuraminidase inhibitors for the treatment of acute influenza is a 12–36 hour reduction in symptom duration.
Initial recommendations from CDC regarding the use of neuraminidase inhibitors for the control of 2009 A(H1N1) focused on treating anyone with confirmed, probable, or likely swine flu. Once spread throughout the nation was apparent, CDC issued new recommendations to treat two primary groups of patients: those who were ill enough to be hospitalized with flu-like illness, regardless of test results, and all outpatients with underlying medical conditions that put them at high risk for complications due to influenza infection.50 Treatment of ILI with antiviral medications was not recommended for the management of otherwise healthy outpatients who were not in any of the high-risk groups. These recommendations were revised several times, including a revision on September 22 that removed children between 2 years and 5 years of age from the list of groups at high risk.
Data reported early in the outbreak indicated that treatment with antiviral medications was beneficial to patients hospitalized with confirmed 2009 A(H1N1) infections, even if they had been symptomatic for more than 48 hours.33 In October, the FDA also issued an EUA for Peramivir, a new neuraminidase inhibitor available only for intravenous use but not yet FDA-approved.51
Vaccine. Trials of 2009 A(H1N1) influenza vaccine began in mid-summer and continued into the fall. Early reports showed the vaccine to be immunogenic in a single dose for adults, with low side-effect and adverse-event profiles.52,53 The vaccine was also found to be highly immunogenic and safe in a vaccine trial in infants and children.54,55 It is interesting to note that although that vaccine trial in children showed excellent vaccine response after only one dose of the vaccine, the U.S. Advisory Committee on Immunization Practices continues to recommend two doses of vaccine for children younger than 9 years of age.55 On Sept. 15, the FDA approved the manufacture of four monovalent vaccines against 2009 A(H1N1), and vaccines became widely available in the United States by mid October.56
There was considerable controversy around the vaccine from the start, in part because it arrived at a time when the second wave appeared to be already peaking, in part because the vast majority of those infected suffered only a self-limited illness, and in part because of concerns about vaccine safety. A national survey of parents conducted by the University of Michigan in August found that only 40% of parents intended to have their children vaccinated against 2009 A(H1N1).57 A survey conducted by the Associated Press in October found that 38% of parents were unlikely to give permission for their child to receive the vaccine.58
An article published in Lancet Infectious Disease in December 2009 added another wrinkle to the topic.59 The authors of this article point out that there is evidence that infection with influenza A viruses can induce at least partial protective immunity to unrelated influenza viruses. This "heterosubtypic immunity" may explain why people older than age 50 have such a low attack rate for A(H1N1) 2009. The authors of this article express concern that regular yearly influenza vaccination of immunologically naïve populations such as children may actually leave them more susceptible to future pandemic strains of influenza because they never have a chance to develop this heterosubtypic immunity through natural infection.
The Advisory Committee on Immunization Practices (ACIP) issued a statement in mid-summer identifying priority groups for vaccine.60 The intent was to provide vaccine first to those most at risk for infection and severe complications. When the vaccine did arrive in mid-October, it was available only in limited supplies, so the high-risk groups, including medical workers, were the first to receive the vaccine. Of the 24 million doses of vaccine administered in the United States by mid-November, 21 million doses (85%) went to members of the initial target groups.61
Vaccine manufacturers shipped 85 million doses of vaccine by December and 105 million doses by Jan. 4, 2010.62 Despite the plentiful supply of vaccine, however, vaccine coverage has been poor. As of Jan. 2, 2010, only 20.3% of the U.S. population (61 million persons) had received the vaccine. Vaccine coverage has been estimated to be 29.4% for children younger than age 18, 38% for pregnant females, 22.3% for healthcare personnel, and 11.6% for adults between 25 and 64 years of age with underlying high-risk medical conditions.61 So far, the 2009 H1N1 vaccine has shown a similar safety profile as seasonal flu vaccines, with no evidence of unexpected adverse events. The most common side effects have been mild, primarily limited to soreness, redness, tenderness, or swelling at the injection site.
Season Summary: Where We Are Today
The peak of the second wave of A(H1N1) 2009 occurred during the week ending Oct. 24, 2009, when widespread activity was reported in 49 states. The majority of U.S. states still reported widespread activity by November 21, but by Christmas week there were only seven states with that level of activity.
The weekly percentage of outpatient visits for ILI also peaked during the week of October 24. The peak rate of 7.7% ILI was higher than the peak rate during the 2006–2007 influenza season (3.5%) or the 2007–2008 influenza season (6%).63
A 2010 CDC report summarized flu activity from August 30 to January 9.64 Over 99% of specimens that were positive for influenza and subtyped during that period were confirmed 2009(H1N1). Influenza B accounted for 0.3% of identified isolates, and seasonal influenza A accounted for less than 0.1%. As of late January 2010 there was negligible seasonal influenza in circulation in the United States. Hospitalization rates since the second wave arrived in late August have been highest for children age 0–4 years, and generally declined for each successive age group. From the recent historical perspective, those rates are much higher than normal for children in the 0–4 year age group, and much lower than normal for adults older than age 65. Oseltamivir resistance has remained negligible. Only 52 oseltamivir-resistant viruses have been detected since April, and at least 77% of those patients had documented exposure to oseltamivir. In only three cases was there no known exposure to oseltamivir.
The impact of A(H1N1) 2009 was initially tracked by CDC and other public health authorities using only confirmed cases. By the middle of the summer, it became too cumbersome to continue this procedure, particularly since by then many health professionals were no longer performing confirmatory tests for outpatients with suspected infection. In July, experts from the CDC and the Harvard School of Public Health developed a model designed to estimate illness, hospitalization, and death due to the virus.65 Their model indicated that every reported case of H1N1 may represent 79 actual cases, and every reported hospitalization may represent 2.7 hospitalized patients. The model yielded estimates for total infected, total hospitalized, and total deaths.66 The mid-level estimated totals from April to December included 55 million infected Americans (18% infection rate), 246,000 hospitalizations, and 11,160 deaths. (See Table 2.)

While the overall rate of hospitalizations due to 2009 (H1N1) is in the range estimated for yearly seasonal flu, the breakdown by age is highly unusual. In a routine flu season, at least 60% of hospitalizations are in adults older than age 65, and fewer than 10% are in children younger than 5 years of age.67 In contrast, adults older than age 65 accounted for fewer than 10% of hospitalizations due to 2009 (H1N1), while children accounted for more than 30%.
Deaths due to 2009 (H1N1) have been well below the 36,000 estimated to occur during a typical influenza season.41 In a normal influenza season, adults older than 65 years of age account for more than 90% of all deaths, and children account for fewer than 1%. Deaths due to 2009 (H1N1) were primarily concentrated in younger adults age 18–64 years (77%), while 9% of deaths occurred in children and another 12% in adults older than age 65.
Conclusion
The year 2009 will be remembered as the year of the influenza pandemic due to novel swine-origin A(H1N1) virus. After the first wave appeared in late spring and summer, some experts predicted that the subsequent wave would infect 30%–50% of the population, lead to 1.8 million hospitalizations, and cause 90,000 deaths.68 While this virus did prove to be unusually lethal for children and young adults compared with seasonal influenza, it was quite mild in its effect on older adults, and the overall attack rate was substantially lower than predicted. As a result, the overall number of infections, hospitalizations, and deaths was far below early estimates, and the strain on our health care system was far less than was initially feared.
As of February 2010, activity of 2009 H1N1 influenza has dropped below the epidemic threshold, and seasonal flu activity remains negligible. It is possible that there will be no winter outbreak of influenza in the Northern Hemisphere this year. Despite concerns about successive waves of pandemic influenza due to this new virus, the historical record suggests that subsequent waves are not inevitable.21 This virus has demonstrated only modest transmissibility compared to other influenza viruses. In addition, a large segment of the population appears to have at least some pre-existing protection against infection with 2009 (H1N1). Finally, a large proportion of the susceptible population has already been infected, leaving little "fuel" for further flare-ups. At this point, we should stay vigilant for the possible return of 2009 A(H1N1) swine flu, the influenza virus of the young.
References
1. Centers for Disease Control and Prevention. Swine Influenza A (H1N1) infection in two children–Southern California, March–April 2009. MMWR 2009;58:400-402.
2. Centers for Disease Control and Prevention. Avian influenza: Current H5N1 situation. Available online at http://www.cdc.gov/flu/avian/outbreaks/current.htm.
3. World Health Organization. Cumulative number of confirmed human cases of Avian Influenza A/(H5N1) reported to WHO. Available online at http://www.who.int/csr/disease/avian_influenza/country/cases_table_2010_01_28/en/index.html.
4. Centers for Disease Control and Prevention. Update: Swine influenza A (H1N1) infections–California and Texas, April 2009. MMWR 2009;58:435-437.
5. Centers for Disease Control and Prevention Press Briefing, April 27 2009. Available online at http://www.cdc.gov/media/transcripts/2009/t090427.htm.
6. World Health Organization situation update number 4, April 28, 2009. Available online at http://www.who.int/csr/don/2009_04_28/en/index.html.
7. BBC News online report, April 28, 2009. Five new UK cases confirmed. Available online at http://news.bbc.co.uk/2/hi/americas/8021827.stm.
8. CNN online report April 30, 2009. WHO raises pandemic alert to second highest level. Available online at http://www.cnn.com/2009/HEALTH/04/29/swine.flu/.
9. Centers for Disease Control and Prevention. Update: Drug susceptibility of Swine origin influenza A (H1N1) viruses, April 2009. MMWR 2009;58:433-434.
10. Centers for Disease Control and Prevention. Emergency use authorization of Tamiflu. Available online at http://www.cdc.gov/h1n1flu/eua/tamiflu.htm.
11. Centers for Disease Control and Prevention press briefing, May 28, 2009. Available online at http://www.cdc.gov/media/transcripts/2009/t090528.htm.
12. World Health Organization (WHO). Situation updates (H1N1) 2009. Available online at http://www.who.int/csr/disease/swineflu/updates/en/index.html.
13. Centers for Disease Control and Prevention. Prevention and control of influenza. MMWR 2008;57(RR-7).
14. CNN online report June 11 2009. Swine flu 'not stoppable,' World Health Organization says. Available online at http://www.cnn.com/2009/HEALTH/06/11/swine.flu.who/.
15. Centers for Disease Control and Prevention. H1N1 flu (swine flu): Past situation updates. Available online at http://www.cdc.gov/h1n1flu/updates/.
16. Centers for Disease Control and Prevention. Assessment of the effectiveness of the 2003-2004 influenza vaccine among children and adults–Colorado, 2003. MMWR 2004;53:707-710.
17. Department of Health and Human Services. Assessment of the 2009 A(H1N1) pandemic on selected countries in the southern hemisphere: Argentina, Australia, Chile, New Zealand, and Uruguay. Aug 26, 2009. Available online at http://www.flu.gov/professional/global/southhemisphere.html.
18. Centers for Disease Control and Prevention. Update: Influenza activity, United States, April to August 2009. MMWR 2009;58:1009-1012.
19. Centers for Disease Control and Prevention. H1N1 flu (Swine flu): Past situation updates, Sept. 4, 2009. Available online at http://www.cdc.gov/h1n1flu/updates/090409.htm.
20. Garten R, Davis CT, Russell CA, et al. Antigenic and genetic characteristics of Swine-Origin 2009 A(H1N1) influenza viruses circulating in humans. Science 2009;325:197-201.
21. Morens DM, Taubenberger JK, Fauci AS. The persistent legacy of the 1918 influenza virus. N Engl J Med 2009;361:225-229.
22. Zimmer SM, Burke DS. Historical perspective–Emergence of influenza A(H1N1) viruses. N Engl J Med 2009;361:279-284.
23. Coburn BJ, Wagner BG, Blower S. Modeling influenza epidemics and pandemics: Insights into the future of swine flu (H1N1). BMC Medicine 2009, 7:30. Available at: http://www.biomedcentral.com/1741-7015/7/30.
24. Fraser C, Donnelly C, Cauchemez S, et al. Pandemic potential of a strain of influenza A(H1N1): Early findings. Science 2009;324:1557-1561.
25. Munayco CV, Gomez J, Laguna-Torres VA, et al. Epidemiological and transmissibility analysis of influenza A(H1N1) virus in a southern hemisphere setting: Peru. Eurosurveillance 2009;14:1-5.
26. Cuachemez S, Donnelly CA, Reed C, et al. Household transmission of 2009 pandemic influenza A(H1N1) virus in the United States. N Engl J Med 2009;361:2619-2627.
27. Lessler J, Reich NG, Cummings DT, et al. Outbreak of 2009 pandemic influenza A(H1N1) at a New York City school. N Engl J Med 2009;361:2628-2636.
28. Update: novel influenza A(H1N1) virus infections–Worldwide, May 6, 2009. MMWR 2009;58:453-458.
29. 2009 pandemic influenza A(H1N1) virus infections–Chicago, Illinois April-July 2009. MMWR 2009;58:913-918.
30. Hancock K, Veguilla V, Lu X, et al. Cross-reactive antibody responses to the 2009 Pandemic H1N1 influenza virus. N Engl J Med 2009; 361:1945-1952.
31. Centers for Disease Control and Prevention. Swine-Origin Influenza A (H1N1) virus infection in a school–New York City, April 2009. MMWR 2009;58(Dispatch):1-3.
32. Nicholson KG. Clinical features of influenza. Semin Respir Infect 1992;7:26-37.
33. Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients with 2009 H1N1 influenza in the United States, April-June 2009. N Engl J Med 2009;361:1991-1993.
34. Louie JK, Acosta M, Winter K, et al. Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA 2009;302:1896-1901.
35. Monto AS. The risk of seasonal and pandemic influenza: Prospects for control. Clin Infect Diseases 2009;48:S20-S25.
36. Libster R, Bugna J, Coviello S, et al. Pediatric hospitalizations associated with 2009 pandemic influenza A(H1N1) in Argentina. N Engl J Med 2010;362:45-55.
37. The ANZIC influenza investigators. Critical care services and 2009 H1N1 influenza in Australia and New Zealand. N Engl J Med 2009;361:1925-1934.
38. Kumar A, Zarychanski R, Pinto R, et al. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA 2009;302:1872-1879.
39. Centers for Disease Control and Prevention. Update: Novel influenza A(H1N1) virus infections–Worldwide, May 6 2009. MMWR 2009;58:453-458.
40. Dominguez-Cherit G, Lapinsky S, Macias A, et al. Critically ill patients with 2009 influenza A(H1N1) in Mexico. JAMA 2009;302:1880-1887.
41. Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncitial virus in the United States. JAMA 2003;289:179-186.
42. Bhat N, Wright JG, Broder KR, et al. Influenza-associated deaths among children in the United States, 2003-2004. N Engl J Med 2005;353:2559-2567.
43. Centers for Disease Control and Prevention. Surveillance for pediatric deaths associated with 2009 pandemic influenza A (H1N1) virus infection–United States, April-August 2009. MMWR 2009;58:941-947.
44. Wilde JA. Rapid diagnostic testing for the identification of respiratory agents in the emergency department. Clin Ped Emerg Med 2002;3: 181-190.
45. Centers for Disease Control and Prevention. Evaluation of rapid influenza diagnostic tests for detection of novel influenza A (H1N1) virus–United States, 2009. MMWR 2009;58:826-829.
46. Centers for Disease Control and Prevention. Interim recommendations for the clinical use of influenza diagnostic tests during the 2009-2010 influenza season. Available online at http://www.cdc.gov/h1n1flu/guidance/diagnostic_tests.htm.
47. Jefferson T, Jones M, Doshi P, et al. Neruaminidase inhibitors for preventing and treating influenza in healthy adults: Systematic review and meta-analysis. BMJ 2009;339:b5106. Available online.
48. Shun-Sin M, Thompson M, Heneghan C, et al. Neruaminidase inhibitors for treatment and prophylaxis of influenza in children: Systematic review and meta-analysis of randomized trials. BMJ 2009;339:b3172.
49. McGeer A, Green KA, Plevneshi A, et al. Antiviral therapy and outcomes of influenza requiring hospitalization in Ontario, Canada. Clin Infect Dis 2007;45:1568-1575.
50. Centers for Disease Control and Prevention. Updated interim recommendations for the use of antiviral medications in the treatment and prevention of influenza for the 2009-2010 season. Available online at http://www.cdc.gov/h1n1flu/recommendations.htm.
51. Centers for Disease Control and Prevention. Emergency Use Authorization of Peramivir IV. Available online at http://www.cdc.gov/h1n1flu/eua/peramivir.htm
52. Greenberg ME, Lai MH, Hartel GF, et al. Response after one dose of a monovalent 2009 influenza A (H1N1) vaccine-preliminary report. N Engl J Med 2009;361:2405-2413.
53. Neuzil KM. Pandemic influenza vaccine policy–Considering the early evidence. N Engl J Med 2009;361:e59.
54. Nolan T, McVernon J, Skeljo M, et al. Immunogenicity of a monovalent 2009 influenza A(H1N1) vaccine in infants and children. JAMA 2010;303:37-46.
55. Fiore AE, Neuzil KM. 2009 influenza A(H1N1) monovalent vaccines for children. JAMA 2010;303:73-74.
56. Food and Drug Administration. FDA approves vaccines for 2009 H1N1 influenza virus. Available at www.fda.gov/newsevents/newsroom/pressannouncements/ucm182399.htm.
57. CS Mott Children's Hospital. National Poll on Children's Health: Parents may underestimate the risks of H1N1 flu for their children. Available at http://med.umich.edu/mott/npch/reports/h1n1.htm.
58. Associated Press. A third of US parents oppose swine flu vaccine. Available online at www.msnbc.msn.com/id/33212378.
59. Bodewes R, Kreijtz J, Rimmelzwaan G. Yearly influenza vaccinations: A double edged sword? Lancet Infect Dis 2009;9:784-788.
60. Centers for Disease Control and Prevention. CDC advisors make recommendations for use of vaccine against novel H1N1. Available online at www.cdc.gov/media/pressrel/2009/r090729b.htm.
61. Centers for Disease Control and Prevention. Interim results: Influenza A (H1N1) 2009 monovalent vaccination coverage–United States, October–December 2009. MMWR 2010;59:44-48.
62. Centers for Disease Control and Prevention. 2009 H1N1 vaccine doses allocated, ordered, and shipped by project area. Available online at http://www.cdc.gov/h1n1flu/vaccination/vaccinesupply.htm.
63. Centers for Disease Control and Prevention. Flu activity and surveillance. Available online at http://www.cdc.gov/flu/weekly/fluactivity.htm.
64. Centers for Disease Control and Prevention. Update: Influenza activity–United States, August 30, 2009–January 9, 2010. MMWR 2010; 59:38-43.
65. Reed C, Angulo FJ, Swerdlow DL, et al. Estimates of the prevalence of pandemic (H1N1) 2009, United States, April-July 2009. Emerg Infect Dis [serial on the Internet]. 2009 Dec. Available online at http://www.cdc.gov/EID/content/15/12/2004.htm
66. Centers for Disease Control and Prevention. CDC estimates for 2009 (H1N1) influenza cases, hospitalizations, and deaths in the United States, April to Dec 12 2009. Available online at http://www.cdc.gov/h1n1flu/estimates_2009_h1n1.htm#UnderCounting#UnderCounting.
67. Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004;292:1333-1340.
68. President's Council of Advisors on Science and Technology. Report to the President on US preparations for 2009 H1N1 influenza. August 7, 2009. Available online at http://www.cdc.gov/h1n1flu/estimates_2009_h1n1.htm#UnderCounting#UnderCounting,
In the past nine months, the world has witnessed the outbreak of not one but two waves of pandemic influenza due to a new virus of swine origin. World public health authorities moved quickly to contain what appeared initially to be the severe pandemic that had been anticipated for so long.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
