An Overview of Fetal Alcohol Spectrum Disorders for Physicians
An Overview of Fetal Alcohol Spectrum Disorders for Physicians
Authors: Sara J. Paton, PhD, Epidemiologist, Boonshoft School of Medicine, Wright State University, Dayton, OH; Public Health Dayton & Montgomery County, OH; and Christopher S. Croom, MD, Boonshoft School of Medicine, Wright State University, Dayton, OH.
Peer Reviewer: Judith Eckerle Kang, MD, Assistant Professor of Pediatrics, University of Minnesota, Minneapolis.
The U. S. Surgeon General has advised women not to drink if they become pregnant, plan to become pregnant, or are at risk of becoming pregnant. Innocent children suffer the consequences of physical, mental, and behavioral manifestations that can extend into adulthood. Since fetal alcohol syndrome is 100% preventable, the primary care physician needs to be vigilant and diligent in the education of female adolescents and women of childbearing age. This issue highlights the spectrum of fetal alcohol syndrome, its recognition, and treatment.
The Editor
Introduction
The idea that a fetus could be harmed by exposure to alcohol during pregnancy has been around for a long time. Perhaps the first written record is in the Bible, Judges 13:4, in which the birth of Samson is being discussed. The angel of the Lord appears to Samson's mother and says "Now see to it that you drink no wine or other fermented drink and that you do not eat anything unclean, because you will conceive and give birth to a son." Experimental data regarding the effects of alcohol on a fetus have been conducted since near the beginning of the 20th century with the use of animal models.1 The first journal article was published in 1968, when Lemoine et al.2 reported the common problems of 127 children born to mothers who drank large amounts of alcohol during pregnancy. This report was largely ignored until 5 years later, when Jones and Smith in 19733 described children of similar symptoms and coined the term "fetal alcohol syndrome." This term was used to describe a group of children, born to alcoholic mothers, who had growth retardation, characteristic facial features, and central nervous system involvement. It is now well known that prenatal alcohol exposure may lead to fetal alcohol spectrum disorders (FASD), an umbrella term describing the range of effects that can occur in an individual whose mother drank alcohol during pregnancy, including fetal alcohol syndrome. Since then, there have been four decades of research on the damaging, and potentially irreversible, effects of prenatal alcohol exposure to the child.
FASD is widespread within the United States and occurs in any population where women drink alcohol during pregnancy.4 Consequently, FAS has been identified in all racial and ethnic groups.5 In addition, FASDs are the most common form of developmental disabilities and birth defects in the western world, more prevalent than Down syndrome and autism.6 Furthermore, FASD is 100% preventable, since the development of FASD depends primarily on exposure of the fetus to alcohol.
This article addresses FASD with regard to the needs of the primary physician. FASD goes largely under-diagnosed in the United States, and primary care physicians may be unprepared to deal with the ramifications this diagnosis may bring.7 Children with even full-blown FAS often go undetected at birth and later in life.8 Children with a less severe form of FASD are even more problematic to diagnose because the physical signs often are more subtle.9 A review of FASD is necessary in order to provide the primary physician with more knowledge and tools to deal with this significant problem.
Fetal Alcohol Spectrum Disorders
The range of adverse effects of alcohol on human development during pregnancy is termed fetal alcohol spectrum disorders. The term FASD is not a diagnostic term, but rather a term for the broad continuum of effects, ranging from physical, mental, or behavioral effects, that develop in the fetus as a consequence of maternal consumption of alcohol. Often these effects have lifelong implications. There have been a variety of terms previously used to address this issue, including fetal alcohol effects, alcohol-related neurodevelopmental disorders, and alcohol-related birth defects.
Alcohol intake during pregnancy can have a large range of outcomes on a fetus. Alcohol is a teratogen that affects the central nervous system, and there is no known "safe" level defined during pregnancy.10-12 The teratogenic effects of alcohol can induce fetal malformations at any stage of pregnancy, as well as at low or high levels of alcohol intake, affecting the developing fetus to varying degrees, in both extent and severity. This depends on numerous factors, including the dosage and timing of alcohol intake in the pregnancy, genetics, age of the mother, parity, and nutritional factors.4 Thus, it is not surprising that not all individuals exposed to similar amounts of alcohol during pregnancy have the same outcomes. Some children may be severely affected in multiple areas, and other children may have no apparent effects.
Fetal alcohol syndrome (FAS) is the most severe form of fetal damage from alcohol and refers to a distinct disorder with clinical features. Three distinct areas are required to be diagnosed as having FAS, including growth retardation, facial appearance, and central nervous system (CNS) problems.4 Each component of the FAS diagnosis can range in severity according to the patient's age, environmental variables, and the quantity and quality of prenatal alcohol exposure.
In addition to FAS, the Institute of Medicine has identified three other diagnoses that are considered part of the FASD spectrum14:
Partial FAS. This describes people with facial anomalies and other symptoms of FAS without all the signs of FAS.
Alcohol-Related Neurodevelopmental Disorder (ARND). This refers to neurological defects, such as problems with communication skills, memory, learning ability, visual and spatial skills, intelligence, and motor skills. A child with ARND will not have all the physical features of FAS. Examples of ARND include speech delays, attention deficits, poor visual focus, and hyperactivity.
Alcohol-Related Birth Defects (ARBD). These are defects to organs, bones, or muscles. They may include abnormalities of the heart, eyes, ears, kidneys, and skeleton. Examples include holes in the heart, fused bones, and underdeveloped kidneys.
Only trained professionals can make a diagnosis. Teachers or relatives may identify a problem, but they cannot make the diagnosis of FASD.
Epidemiology
FAS. Estimates of the incidence of FAS vary significantly in different populations and with different types of research designs. Some of this variation is a valid reflection of actual differences in FAS rates between populations.15 This is especially true in populations with large differences in maternal drinking behavior.15 However, variance in FAS incidence between research studies also can be a function of the different research methods used to study the problem. Passive surveillance systems, which typically use hospital discharge data, tend to give lower estimates of FAS than clinic-based studies or active case ascertainment methods. A summary of various studies has reported that the overall prevalence of FAS in the United States is likely to be between 0.5 and 2.0 per 1,000 births.15
Disparities exist within FAS. "Heavy"-drinking women have the highest reported incidence of FAS. Heavy drinking is defined here as those consuming an average of 2 or more drinks per day, 5 to 6 drinks per occasion, a positive MAST score, or a clinical diagnosis of alcohol abuse. Children of women who fall in this category have an FAS rate of 4.3%.5
There is also a disparity in FAS with regard to race. The United States incidence of FAS in primarily Caucasian populations is estimated to be around 0.26 per 1,000 births.5 The African-American incidence of FAS in the United States is much higher, at 2.29 per 1,000 live births. Thus the ratio of African-American to Caucasian incidence of FAS is 8.8:1, meaning that for every Caucasian baby born with FAS, almost 9 African-American babies are born with FAS. An additional disparity was reported in Alaska natives, with an incidence of FAS estimated between 3.00-5.20 per 1,000.16
Another high disparity in FAS occurs with regard to maternal socioeconomic status. Abel, 1995, estimated that the incidence of FAS in women in the United States with incomes of $10,000 or less for a family of three was 2.29 per 1,000 births.5 Women of middle or high income were estimated to have an incidence of only 0.26 per 1,000 births. Children with FAS are frequently characterized by low socio-economic status.2,17 Low socioeconomic status and heavy alcohol consumption are both associated with poor nutrition, stress, poor health, smoking, and the use of other substances15,18-20 Although none of these factors is a cause of FAS, it is possible that they may exacerbate the effects of heavy alcohol intake, which may result in FAS.5
FASD. The true epidemiology of FASD is not known. Many cases of FAS go undetected at birth and even later in life.14 Children with less severe phenotypes in the FASD continuum present an even greater diagnostic challenge because often the phenotypic characteristics are even more subtle. In addition many children are misdiagnosed with a multitude of other disorders. Attention deficit hyperactivity disorder, autism, major depressive disorder, or bipolar disorder are examples of common misdiagnoses.21 Co-morbidities with other mental health problems are common in children with FASD and thus make diagnosis even more of a challenge. These less-severe types of FASD are much more common than FAS and may affect 1% or more of all children born in the United States.22,23
Alcohol Consumption During Pregnancy
The Centers for Disease Control and Prevention (CDC) estimates that more than half (53.7%) of nonpregnant women of childbearing age (18-44) have reported alcohol consumption in the past month, and approximately one in eight (12.1%) report binge drinking.24 Furthermore, in the United States, almost half of pregnancies are unplanned.25 For pregnant women, the CDC reported that the average annual percentage of any alcohol use was 12.2%, and of binge drinking was 1.9%. The highest percentages of pregnant women reporting any alcohol use were aged 35-44 years (17.7%), college graduates (14.4%), employed (13.7%), and unmarried (13.4%).24
The number of women who drink alcohol during pregnancy far surpasses the total number of children diagnosed with either FAS or FASD.26 This means that not every child who is exposed to alcohol during pregnancy will have FAS or FASD. In addition, the degree of severity of FAS or FASD in an individual varies greatly from person to person. Several factors may contribute to this variation in severity of alcohol exposure to a child. These factors include, but are not limited to, the following26,27:
Maternal drinking pattern
Maternal age
Parity
Maternal metabolism differences
Genetic differences in alcohol metabolism
Timing of the alcohol exposure to the fetus
Differences in the vulnerability of various brain regions
Polydrug use.
Diagnosis of FASD
FASD is an umbrella term describing the range of effects that can occur in an individual whose mother drank during pregnancy. These effects may include physical, mental, behavioral, and/or learning disabilities with possible lifelong implications.6 In most cases of FASD, the pathognomonic physical features are not present. This makes the diagnosis challenging at best and impossible at worst if a maternal drinking history is unknown. Although many articles discuss the behavioral phenotype of FASD, the clinical diagnosis remains problematic.28 For these reasons, the discussion of diagnosis in this article will be limited to FAS. No specific and uniformly accepted diagnostic criteria are available for FAS. Several standards have been suggested over the past 20 years.9,14,29 Although they have been widely used, their practical clinical applications are less than optimal. In 2004, a national task force published a set of suggested guidelines for the diagnosis of FAS.7 These criteria are discussed below.
Fetal Alcohol Syndrome (FAS)
Dysmorphia. Clear insight into the recognition of alcohol as a teratogen was not described until 1968.2 However, descriptions of the children of alcoholics noted as "born weak and silly ... shriveled and old, as though they had many numbered years" appeared in the medical literature as early as the 1700s.30 For the first time, in 1973, several investigators3 described in detail the consistent pattern of malformations observed in the children of mothers who drank heavily during their pregnancies. These observations provided the criteria for a condition they termed FAS.
Their early studies focused on short palpebral fissures, maxillary hypoplasia, and the presence of epicanthal folds. Over the course of the next 30 years, additional features that were described included cardiac anomalies, hypoplastic nails, microcephaly, short nose, smooth philtrum with thin vermillion border, cleft lip, micrognathia, webbed neck, meningomyelocele, hydrocephalus, and hypoplastic labia majora.13
Because of the heterogeneity of expression for dysmorphic features related to prenatal exposure to alcohol, the general consensus is that diagnostic criteria should be limited to core facial dysmorphia.31 This is based on human observational studies as well as experimental animal studies that have demonstrated fetal alcohol exposure disturbs cellular migration during organogenesis along the midline of the face.
The following dysmorphic facial features meet the dysmorphic criteria essential for FAS (based on racial norms):
smooth philtrum (measured as 4 or 5 on lip-philtrum guide) (see Figure 1);
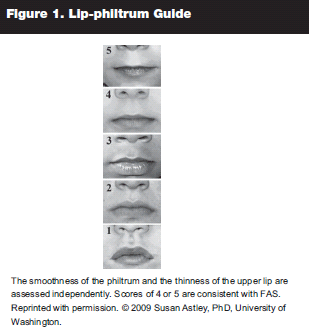 |
thin vermillion border (measured as 4 or 5 on lip-philtrum guide);
small palpebral fissures (measured as < 10th percentile) (see Figure 2).
 |
After puberty, these characteristic facial features can become more difficult to detect. However, recent findings indicate that these three features remain for the majority of individuals with FAS.
Growth Deficiency. Growth deficiency historically has been regarded as a major feature of FAS. The initial cases of FAS initially were assessed for failing to thrive. Abundant research in both humans and animals has demonstrated that alcohol can have an adverse impact on length and weight both prenatally and postnatally.32-34 Unlike the facial anomalies, fetal growth is most significantly impacted by alcohol exposure in the last half of pregnancy. The parameters (height, weight, head perimeter) and the severity (2nd to 25th percentile) for assessing growth deficiency vary greatly in the literature. Current criteria for growth deficiency is the following: Confirmed prenatal or postnatal height and/or weight at or below the 10th percentile at any one point in time. These parameters should be adjusted for age, sex, gestational age, and race or ethnicity.31
Central Nervous System (CNS) Abnormalities. Multiple publications documenting the teratogenic effects of alcohol on the CNS have been in the medical literature over the past 30 years.33-35 These congenital anomalies have been shown to result in a wide range of short-term and long-term behavioral and cognitive deficits. These neurobehavioral effects persist through affected individuals' lifetimes and have rendered many of them unable to live independently.275 The CNS abnormalities included in the criteria for the diagnosis of FAS are categorized as structural, neurological, functional, or combinations of any or all.
Structural. Structural CNS abnormalities associated with the diagnosis of FAS consist of two general criteria. First is a diminished occipitofrontal circumference (OFC). This is defined as < 10th percentile, adjusted for age and gender. In children with growth deficiency (height and weight < 10th percentile), the OFC should be disproportionately small: i.e., < 3rd percentile. The second criterion is clinically significant brain abnormalities that are detected through imaging techniques. Previously documented abnormal findings include reduction in the size and shape of the corpus callosum, cerebellum, or basal ganglia.36,37
Neurological. Numerous abnormal neurological findings have been associated with prenatal alcohol exposure.13 These include seizures, visual motor difficulties, nystagmus, coordination problems, and problems with motor control. However, the etiologies of intra-partum or postnatal insult and fever must be ruled out prior to assigning alcohol as the cause.
Functional. The functional abilities affected by prenatal alcohol exposure vary greatly from person to person, depending on the amount of alcohol, timing of exposure, and the pattern of exposure (i.e., chronic vs. binge). Despite the inherent variation, the observed functional deficits correspond to multiple locations in the brain affected by exposure. To address this issue, functional effects may be assessed in two ways: global deficits or deficits in functional domains (secondary disabilities). To meet the diagnostic criteria for FAS, the individual must have one or the other.
Global cognitive deficit is defined as decreased IQ (below the 3rd percentile) or significant developmental delay in children too young for IQ assessment. A decrease in performance in these assessments assumes multiple functional domains have been affected.
Deficits (one standard deviation below the mean for standardized testing) in three or more specific functional domains. These are described below:
Cognitive or developmental discrepancies: learning disabilities (particularly in math and/or visual-spatial deficits); uneven profile of cognitive skills; poor academic achievement; discrepancy between verbal and non-verbal skills; and slowed movements or reaction to people and stimuli.
Executive functioning deficits: poor organization, planning, or strategy use; difficulty grasping cause and effect; difficulty following multistep directions; difficulty changing strategies or thinking of things in a different way; poor judgment; and inability to apply knowledge in a new way.
Motor functioning delays or deficits: delayed motor milestones; difficulty writing or drawing; clumsiness; balance problems; tremors; poor dexterity.
Attention and hyperactivity problems: easily distracted; difficulty in calming down; overly active; difficulty completing tasks; trouble with transition. Of note, many children with FAS often receive the diagnosis of attention deficit hyperactivity disorder (ADHD). Although this diagnosis can be applied, research has shown that the attention problems of children with FAS do not fit the classic pattern of ADHD.38 Individuals with FAS tend to have difficulty with the encoding of information and flexibility aspects of attention, whereas children with ADHD typically display problems with focusing and sustaining attention.38 A recent publication provides a screening tool for distinguishing the behavioral characteristics between the two conditions.28
Social skills problems: lack of stranger fear; naivete and gullibility; inappropriate choice of friends; often scapegoated; inappropriate sexual behaviors; difficulty understanding the perspective of others; and clinically significant inappropriate initiations or interactions.
Other potential domains: sensory problems (tactile and oral); pragmatic language problems; memory deficits; and difficulty responding to normal parenting practices (not understanding cause and effect).
Ideally, the above described deficits should be established by a trained professional using appropriate standardized neuropsychological testing. In many cases, this type of testing is not readily available. In these situations, clinicians are encouraged to supplement their observations with standardized testing offered in early intervention programs and public schools. It must be emphasized that, when evaluating an individual for FAS, using clinical judgment without the benefit of psychometric testing can result in an incorrect diagnosis or an inappropriate treatment plan.
Maternal Alcohol Exposure
Documentation and confirmation of prenatal alcohol exposure is perhaps the most challenging of the criteria to obtain in making the diagnosis of FAS. Admission of alcohol use is stigmatizing to the mother. This information must be collected in a non-threatening, non-judgmental fashion. In the case of the mother actively drinking during pregnancy, obtaining accurate information about the amount and frequency of consumption is frequently not possible. The most difficult clinical scenario is when an individual is being evaluated and there is no information about the index pregnancy. This frequently occurs in foster care or adoptive homes.
In situations where it can be accurately determined that no alcohol had been consumed from conception to birth, the diagnosis of FAS is ruled out.
Categories for qualifying as prenatal alcohol exposure are as follows:
Confirmed prenatal alcohol exposure: requires a history of alcohol exposure by the birth mother during the index pregnancy based on clinical observation; self reports; report by a reliable informant; medical records documenting positive blood alcohol levels or alcohol treatment; or other social, legal, or medical problems related to alcohol consumption during the index pregnancy.
Unknown prenatal alcohol exposure: where there is neither confirmed presence of exposure nor confirmed absence of exposure.
In summary, the diagnosis of FAS requires the presence of all three of the following findings:
documentation of all three facial abnormalities (smooth philtrum, thin vermillion border, small palpebral fissures;
documentation of growth deficits;
documentation of CNS abnormalities (structural, neurological or functional, or combination thereof).
The confirmed or unknown status of alcohol exposure is not required for the diagnosis of FAS to be entertained. As stated above, in the confirmed absence of prenatal alcohol exposure, the diagnosis of FAS is ruled out.
Treatment
Unfortunately, because the alcohol-induced CNS abnormalities create permanent disabilities, there is no cure for FAS. However, effective treatment is possible. Since the CNS damage, symptoms, secondary disabilities, and needs vary greatly from one individual to the next, not one form of treatment works for everyone. Additionally, because research in the area of FAS treatment is in its infancy, relatively speaking, well-defined therapeutic modalities are still evolving. Nevertheless, human studies, clinical observations, and family experiences concur that the common features that most often result in successful treatment include early diagnosis; a loving, nurturing, stable environment; involvement of special education and social services; and absence of violence in the individual's life.
When describing the treatment of FAS, it can be helpful to divide the discussion into four general areas: early intervention; child-focused; adolescents; and adults.
Early Intervention. Current research indicates that early intervention has the potential to ameliorate at least some deficits found in individuals with FAS.39 If the diagnosis of FAS is suspected antenatally, the infant can qualify for early intervention at the time of birth. Early interventionists can be the first to confirm the diagnosis, alert caregivers to emerging learning and behavior problems, and connect families with needed services. Specific services for the child include direct intervention with evidence-based early education curricula focusing on math and handwriting skills and supportive services from occupational therapists and speech pathologists. Families are taught skills to support and participate in the child's learning, encouraged to join established support/advocacy networks, and provided alcohol treatment, if needed.
Child-Focused. Child-focused treatment addresses the specific functional deficits that have been diagnosed in the affected individual. As stated earlier, these can include deficits in attention and executive function, social skills, and behavioral regulation. Modalities that have been suggested to remediate these deficits are cognitive control techniques, friendship skill-building groups, calming techniques, social skills groups, and neuro-behavioral feedback.40 A recent study has demonstrated the potential for computer training in older children as a powerful resource for training in life and social skills.41
Adolescents. The life phases of adolescence are an especially difficult time for those with FAS, given the complex but often subtle neurodevelopmental disabilities they possess. Unfortunately, this critical period in their lives often is omitted in the continuum of FAS intervention. Despite this reality, there are specific treatment options suggested for this unique population of individuals.
Group therapy for teens and young adults with FAS is a new and promising direction in treatment. These groups have a central focus on learning and practicing social skills. Teens are taught how to appropriately establish intimacy and build trusting, positive peer relationships. These groups also provide appropriate and literal education on topics such as substance use, dating, and sexuality.
Support and resources for adolescents and younger adults are clearly important and necessary. In many communities, these are not readily available. The option of regionally sponsored retreats for teens with FAS is now a growing trend in some states. Generally, these retreats require that the diagnosed individual bring along a support person. As the numbers of teens diagnosed with FAS increases, so the opportunities for them to gather together will become more warranted and hopefully more available.
Adults. The functional deficits that are diagnostic of FAS are also described as secondary disabilities, with the primary disability being the CNS birth defect caused by prenatal alcohol exposure. These disabilities manifest clinically as mental health problems. The most common presentations are depression, attention deficit problems, panic attacks, auditory and visual hallucinations, and suicide threats and attempts. With the exception of attention deficit problems, all of these increase with age.27 Whereas much of the treatment focus in childhood is on addressing the developmental disability, in the adult the approach should be treating the patient as an individual with a dual diagnosis.
In the adult, treatment is usually multimodal. Many different types of therapies are applied over time as they are needed. These may include individual counseling, family therapy, special education, and psychopharmaco-therapy.
In regard to medical intervention, since this group of patients has deficits in both neuroanatomy and neuro-chemistry, their responses to medications can be quite unpredictable. It is wiser to identify areas of stress in the individual's environment and make changes there, as opposed to immediately treating the symptoms with medication.
Another approach to treatment of the adult with FAS is using an advocacy model.27 This method assumes that someone is needed to actively mediate between the environment and the person with FAS. The advocate (family member, friend, case manager, therapist) functions in three basic areas for the individual: interpreting FAS and the disabilities that arise from it and explaining it to the environment in which the patient operates; engendering change or accommodation on behalf of the patient; assisting the patient in developing and reaching attainable goals.
The ultimate goal of this model is that, with the help of a caring advocate, the individual with FAS negotiates life tasks and learns necessary skills.
Prevention
Fetal alcohol spectrum disorders are 100% preventable, as they do not occur among children born to women who abstain from alcohol during pregnancy. Prevention of FASD, therefore, is the most effective strategy for decreasing the impact of this health problem. There is no known safe level of alcohol consumption during pregnancy, so it is prudent for women to completely avoid alcohol while pregnant. In addition, if a woman is planning to get pregnant or is sexually active and not using effective birth control, she should avoid alcohol consumption because she could be pregnant and not know for several weeks or more.
Prevention of FAS in the United States primarily began in 1981 when the Office of the Surgeon General issued an advisory that pregnant women or women planning a pregnancy should abstain from drinking alcohol.42 In 2005, the U.S. Surgeon General issued an updated advisory that advises women not to drink alcohol if they are pregnant, planning to become pregnant, or at risk of becoming pregnant. In addition, warning labels have been required on alcoholic beverage containers sold in this country since 1990. Unfortunately, the warning labels have primarily resulted in a reduction of alcohol consumption in those women who are considered light drinkers.43 Despite these efforts, alcohol use among pregnant women has remained relatively unchanged from 1991-2005.24
To prevent FASD, no single group, organization, community, ministry, or level of government can deal effectively with the problem on its own. Broad-based efforts are required, with policy interventions that affect everyone in addition to more specific community-level interventions that may target specific populations. Prevention strategies is this paper, however, will focus on those that a physician or other health care professional can engage in.
Prevention is the first line of defense against the effects of alcohol in pregnancy and should include the following:
Primary prevention: These are interventions that avert a health problem prior to its occurrence. For FASD, an example of this could be informing the public, and particularly women of childbearing age, about the dangers of drinking during pregnancy.
Secondary prevention: These include interventions that seek to reduce the severity and/or duration of the health problem. For FASD, an example would include alcohol screening and early intervention programs and services for pregnant women who would be at risk for having a child with FASD.
Tertiary prevention: These are interventions that would prevent the recurrence of the health problem and attempt to lessen the impact of the health problem. For FASD, an example would be an intervention targeted at women who have already had a child with a FASD. Another example could include a therapy designed specifically for a child with FASD.
Table 1 provides FASD prevention strategies adapted from the Canadian Child Adolescent Psychiatric Review that a physician's or health provider's practice could use to assist in the prevention of FASD.44
 |
Conclusion
Physicians and other health care providers can have a large impact on how well fetal alcohol spectrum disorders are diagnosed and treated, as well as play a crucial part in prevention efforts. Improvement in these areas could reduce the incidence of FASD and increase the quality of life for those who have an FASD.
References
1. Srockard CR. The influence of alcohol and other anaesthetics on embryonic development. Am J Anat 1910;10:369-392.
2. Lemoine P, Harrousseau H, Borreyro JP, et al. Les enfants de parents alcoholiques. Ouest Med 1968;21:476-492.
3. Jones KL, Smith DW. Recognition of the fetal alcohol syndrome in early infancy. Lancet 1973;302:999-1001.
4. Riley EP, McGee CL. Fetal alcohol spectrum disorders: An overview with emphasis on changes in brain and behavior. Exp Biol Med (Maywood) 2005;230:357-365.
5. Abel EL. An update on incidence of FAS: FAS is not an equal opportunity birth defect. Neurotoxicol Teratol 1995;17:437-443.
6. Clarke ME, Gibbard WB. Overview of fetal alcohol spectrum disorders for mental health professionals. Can Child Adolesc Psychiatr Rev 2003;12:57-63.
7. Morse BA, Idelson RK, Sachs WH, et al. Pediatricians' perspectives on fetal alcohol syndrome. J Subst Abuse 1992;4:187-195.
8. Little BB, Snell LM, Rosenfeld CR, et al. Failure to recognize fetal alcohol syndrome in newborn infants. Am J Dis Child 1990;144:1142-1146.
9. Astley SJ, Clarren SK. Diagnosing the full spectrum of fetal alcohol-exposed individuals: Introducing the 4-digit diagnostic code. Alcohol Alcohol 2000;35:400-410.
10. Adams J, Bittner P, Buttar HS, et al. Statement of the Public Affairs Committee of the Teratology Society on the fetal alcohol syndrome. Teratology 2002;66:344-347.
11. Smith DW. The fetal alcohol syndrome. Hosp Pract 1979;14:121-128.
12. Streissguth AP, Landesman-Dwyer S, Martin JC, et al. Teratogenic effects of alcohol in humans and laboratory animals. Science 1980;209:353-361.
13. Jones KL. Smith's Recognizable Patterns of Human Malformation. 5th ed. Philadelphia: Saunders; 1997:857.
14. Hoyme HE, May PA, Kalberg WO, et al. A practical clinical approach to diagnosis of fetal alcohol spectrum disorders: Clarification of the 1996 institute of medicine criteria. Pediatrics 2005;115:39-47.
15. May PA, Gossage JP. Estimating the prevalence of fetal alcohol syndrome. A summary. Alcohol Res Health 2001;25:159-167.
16. Egeland GM, Perham-Hester KA, Gessner BD, et al. Fetal alcohol syndrome in Alaska, 1977 through 1992: An administrative prevalence derived from multiple data sources. Am J Public Health 1998;88:781-786.
17. Lesure JF. L'embryo foeto pathie alcoolique a' l'lle de la reunion: Un drame social. La Rev Pediatr 1988;24:265-271.
18. Hingson R, Alpert JJ, Day N, et al. Effects of maternal drinking and marijuana use on fetal growth and development. Pediatrics 1982;70:539-546.
19. Leichter J. Growth of fetuses of rats exposed to ethanol and cigarette smoke during gestation. Growth Dev Aging 1989;53:129-134.
20. Caley LM, Kramer C, Robinson LK. Fetal alcohol spectrum disorder. J Sch Nurs 2005;21:139-146.
21. Chudley AE, Kilgour AR, Cranston M, et al. Challenges of diagnosis in fetal alcohol syndrome and fetal alcohol spectrum disorder in the adult. Am J Med Genet C Semin Med Genet 2007;145C:261-272.
22. Abel EL, Kruger M. What do physicians know and say about fetal alcohol syndrome: A survey of obstetricians, pediatricians, and family medicine physicians. Alcohol Clin Exp Res 1998;22:1951-1954.
23. Sampson PD, Streissguth AP, Bookstein FL, et al. Incidence of fetal alcohol syndrome and prevalence of alcohol-related neurodevelopmental disorder. Teratology 1997;56:317-326.
24. Centers for Disease Control and Prevention (CDC). Alcohol use among pregnant and nonpregnant women of childbearing age United States, 1991-2005. MMWR Morb Mortal Wkly Rep 2009;58:529-532.
25. Henshaw SK. Unintended pregnancy in the United States. Fam Plann Perspect 1998;30:24-9, 46.
26. Maier SE, West JR. Drinking patterns and alcohol-related birth defects. Alcohol Res Health 2001;25:168-174.
27. West JR. Fetal alcohol-induced brain damage and the problem of determining temporal vulnerability: A review. Alcohol Drug Res 1987;7:423-441.
28. Kodituwakku PW. Defining the behavioral phenotype in children with fetal alcohol spectrum disorders: A review. Neurosci Biobehav Rev 2007;31:192-201.
29. Stratton K, Howe C, Battaglia F. Fetal alcohol syndrome: Diagnosis, epidemiology, prevention, and treatment. Washington, D.C.: National Academy Press; 1996.
30. Royal College of Physicians of London: Annals. London, England: Royal College of Physicians; 1726:253.
31. Bertrand J, Floyd LL, Weber MK, Fetal Alcohol Syndrome Prevention Team, Division of Birth Defects and Developmental Disabilities, National Center on Birth Defects and Developmental Disabilities, Centers for Disease Control and Prevention (CDC). Guidelines for identifying and referring persons with fetal alcohol syndrome. MMWR Recomm Rep 2005;54(RR-11):1-14.
32. Jacobson JL, Jacobson SW. Effects of prenatal alcohol exposure on child development. Alcohol Res Health 2002;26:282-286.
33. Samson HH. Microcephaly and fetal alcohol syndrome: Human and animal studies. In: West JR, ed. Alcohol and Brain Development. New York, N.Y.: Oxford University Press; 1986:163-167.
34. Streissguth AP, O'Malley K. Neuropsychiatric implications and long-term consequences of fetal alcohol spectrum disorders. Semin Clin Neuropsychiatry 2000;5:177-190.
35. Little RE, Graham JM, Samson HH. Fetal alcohol effects in humans and animals. Adv Alcohol Subst Abuse 1982;1:103-125.
36. Riley EP, Mattson SN, Sowell ER, et al. Abnormalities of the corpus callosum in children prenatally exposed to alcohol. Alcohol Clin Exp Res 1995;19:1198-1202.
37. Mattson SN, Riley EP, Sowell ER, et al. A decrease in the size of the basal ganglia in children with fetal alcohol syndrome. Alcohol Clin Exp Res 1996;20:1088-1093.
38. O'Malley KD, Nanson J. Clinical implications of a link between fetal alcohol spectrum disorder and attention-deficit hyperactivity disorder. Can J Psychiatry 2002;47:349-354.
39. Carmichael Olson H, Jirikowic T, Kartin D, et al. Responding to the challenge of early intervention of alcohol spectrum disorders. Infants and Young Children 2007;20:172-189.
40. O'Connor MJ, Frankel F, Paley B, et al. A controlled social skills training for children with fetal alcohol spectrum disorders. J Consult Clin Psychol 2006;74:639-648.
41. Coles CD, Strickland DC, Padgett L, et al. Games that "work": Using computer games to teach alcohol-affected children about fire and street safety. Res Dev Disabil 2007;28:518-530.
42. [No authors listed.] Surgeon general's advisory on alcohol and pregnancy. FDA Drug Bull 1981;11:9-10.
43. Hankin JR. Fetal alcohol syndrome prevention research. Alcohol Res Health 2002;26:58-65.
44. [No authors listed.] Prevention of fetal alcohol syndrome (FAS). Can Child Adolesc Psychiatr Rev 2003;12:87-91.
The U. S. Surgeon General has advised women not to drink if they become pregnant, plan to become pregnant, or are at risk of becoming pregnant. Innocent children suffer the consequences of physical, mental, and behavioral manifestations that can extend into adulthood. Since fetal alcohol syndrome is 100% preventable, the primary care physician needs to be vigilant and diligent in the education of female adolescents and women of childbearing age.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
