Cardiac Biomarkers: How to Use Them Wisely
Cardiac Biomarkers: How to Use Them Wisely
How the use of cardiac biomarkers has changed. I remember ordering serum SGOT and LDH as an intern to detect heart attacks for my patients. Back then, the typical "rule out chest pain" admission was two to three days, primarily because the LDH isoenzymes were run once per day by the clinical laboratory and the norm was three sets, usually obtained over three days. Also, it was well recognized that these tests were not sensitive or very specific for myocardial infarction.
We now have cardiac biomarker tests that are highly sensitive, highly specific, available at all times from the clinical laboratory, and often available as bedside point-of-care testing. You would think with all this, that physicians would be more confident in their assessment of chest pain patients and excluding myocardial ischemia. I would venture the observation that the opposite has occurred. These highly sensitive biomarkers have enabled us to detect more myocardial ischemia and infarction in patients with atypical clinical presentations and nondiagnostic electrocardiograms; patients who in previous years would not have been identified.
So, we have a bounty of riches when it comes to cardiac biomarkers. For me, the key is to risk stratify the patient based on clinical and ECG criteria and then use the biomarker tests according to the clinical circumstance. As the authors of this review note, the utility of any test is only as good as the user.
J. Stephan Stapczynski, MD, Editor
Chest Pain in the Emergency Department
In 2005, more than 1.6 million visits to the emergency department (ED) were for a chief complaint of chest pain. Only one-fifth of these patients were admitted for further observation and treatment. For the remainder of these patients, the chief complaint of chest pain was attributed to diagnoses other than an acute myocardial infarction.1
As overall visits to EDs continue to increase, the accuracy of assessment of patients presenting with chest pain becomes even more important. In the initial triage process, the first electrocardiogram (ECG) can be nondiagnostic for acute coronary syndrome (ACS); patient symptoms may be atypical for the classic presentation of acute myocardial infarction (AMI), or the patient may be an unreliable historian.2 Not surprisingly, missed diagnosis of AMI accounts for the largest portion of settled lawsuits in the clinical practice of emergency medicine.
Thus, timely and accurate diagnosis of patients presenting with chest pain cannot be understated; yet, the timely and efficient diagnosis of acute coronary syndromes can still pose a significant challenge for the emergency physician.
The Utility of a Biomarker
The adage, "A tool is only as useful as its user," holds somewhat true when describing the use of biochemical markers in evaluating chest pain. In selecting a cardiac biomarker, it is important to assess its clinical utility and role in the etiologic diagnosis of chest pain. An ideal biomarker may have the following qualities: a profile that quickly and accurately identifies patients with acute myocardial infarction (AMI) or myocardial ischemia; be present in high concentrations in cardiac tissue; be absent from other tissues; and be undetectable in the blood stream of healthy patients.3
This ideal biomarker also should be rapidly released from injured tissues and should maintain elevated serum levels, with elevated levels corresponding to the degree of myocardial injury. Furthermore, a perfect biomarker should also be inexpensive, rapidly available, and biochemically simple enough to be run by any laboratory at any time of day. To date, no such biomarker exists.
The utility of a biomarker can be objectively evaluated by its sensitivity, specificity, and the calculation of the likelihood ratios. In brief, the more sensitive a marker, the higher the chance that it will correctly detect patients with myocardial injury. However, a marker with a higher specificity will more correctly aid in identifying those patients who truly do not have myocardial injury. This added specificity may allow for improved throughput flow and disposition of these patients, limiting unnecessary or otherwise inconclusive workups of cardiac chest pain.
Biochemistry of Serum Markers in Myocardial Ischemia
The process of myocardial ischemia results in multiple pathophysiologic events. In the earliest phase, myocardial ischemia leads to cellular acidosis. This, in turn, creates increased permeability of the cell wall with resultant leakage or spilling of intracellularly contained chemical molecules. As ischemia evolves into infarction, the cell wall becomes more unstable, with eventual breakdown and further release of cytoplasmic structures into the circulatory system. Markers that are smaller are released first and are detected sooner in serum analyses. Markers that do not bind to other structures in the circulatory system also have a higher chance of being detected in the serum. Rate of clearance by kidney and liver, as well as kinetic properties of regional blood flow and lymphatic systemics will affect the presence of markers in the serum.4 (See Figure 1.)
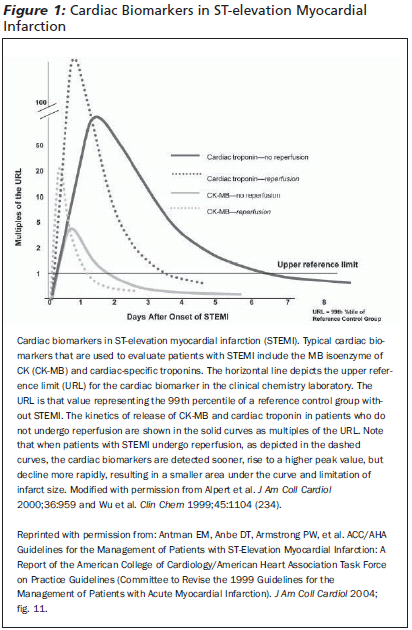 |
Cardiac Biomarkers
Historically, the first biomarkers used in the detection of AMI included lactate dehydrogenase (LDH) and aspartate aminotransferase (AST). However, these two markers are found in a variety of body tissues and are elevated in many different disease processes, making them less sensitive or specific, and they have long been abandoned as useful markers of cardiac disease in the acute setting.5,6
Myoglobin. Myoglobin is a small protein found in the cytoplasm of both skeletal and cardiac muscle tissue. This oxygen-carrying molecule tends to be released early during infarction because of its small size, and thus serum levels of myoglobin tend to be elevated far earlier than other cardiac markers.7 Myoglobin tends to rise rapidly in blood (usually within two hours of onset of symptoms), reaching peak levels by nine hours. The early increase in myoglobin levels would suggest a significant advantage for the early detection of myocardial infarction. However, there are two notable disadvantages to myoglobin as a biomarker. It is relatively nonspecific to myocardium, and clearance depends heavily upon renal function. Studies have shown that when measured within three hours of the onset of chest pain, myoglobin assays have substantial ranges in sensitivities and specificities (24-81% and 76-96%, respectively) with increases in both sensitivity and specificity when measured at 6 hours (55-100% and 76-98%, respectively).3,8
Serial myoglobin determinations have demonstrated many times to be the most useful diagnostic strategy in detecting the presence of myocardial injury. Tucker et al. found that doubling of the serum values of this biomarker within two hours of the onset of symptoms yielded a sensitivity and specificity for acute coronary ischemia of 95%.8 Brogan et al. concluded that in the first hour of presentation to the ED, the rapid quantitative assay for myoglobin was statistically more sensitive than CK-MB at 91% and had a specificity of 96% and a negative predictive value of 99% for ruling out acute myocardial infarction in patients presenting to the ED.9
The disadvantages of myoglobin as a single biomarker should be emphasized. The rapidity of the rise and fall in serum allows for a limited diagnostic window. Several other reasons exist for elevated levels of myoglobin, including but not limited to renal failure, muscle injury or inflammation, and many other chronic disease states, all of which substantially increases false-positive opportunities. In general, myoglobin may have little utility as a single biomarker for acute coronary syndrome or myocardial disease more than six hours after onset of symptoms.
Multi-marker approaches utilizing myoglobin have shown superior sensitivities to biomarkers used alone, especially when deployed very early in the diagnostic period. Kontos et al. reported sensitivities of 85% when using CK-MB and myoglobin in combination on initial evaluation and 100% at four hours.10 Maisel et al. demonstrated 88% sensitivity and a negative predictive value of 99% when the combination of myoglobin and cardiac troponin I (cTnI) were measured at two hours after presentation.11 This adds to the body of existing evidence that myoglobin is best used in a panel of biomarkers and in the context of serial or delta marker panels.
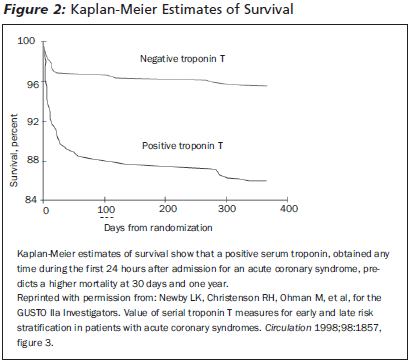 |
Creatine Kinase. Creatine kinase (CK) is an enzyme expressed by various body tissues but found in highest quantities in cardiac and skeletal muscle tissues. It is a large molecule that is released by injured cardiac muscle, travels in the lymphatic system, and is not detectable in serum until three to eight hours after myocardial injury. The serum levels of enzyme are highest at 12-24 hours after cardiac injury and subsequently return to baseline levels in three to four days.12
Because of the ubiquitous nature of CK, the isoenzyme CK-MB traditionally is measured when discussing cardiac evaluation. There are two subunits of CK: M, found predominantly in muscle tissue, and B, found predominantly in the brain. CK that is released by cardiac tissue is roughly 15% CK-MB and 85% CK-MM. However, cardiac cells remain the most abundant sources of CK-MB in the body; so an elevation of CK-MB usually is associated with cardiac injury. The CK contained within skeletal muscle is approximately 98% CK-MM and 1% CK-MB. However, because of the large amount of skeletal muscle within the human body, relatively small amounts of muscle injury can release enough CK-MB to result in elevated levels. Therefore, abnormal CK-MB elevations can occur with vigorous exercise, rhabdomyolysis, trauma, myositis, and muscular dystrophy.13 CK-BB is found predominantly in the brain and CK-MM in the skeletal muscle.
In the clinical setting of AMI, CK-MB, like CK, is released by cardiac tissue into the cardiac lymphatic system. It is initially detected in serum at four to six hours after injury, peaking at 12-24 hours, with normalization at two to three days.12 While the peak serum level of CK-MB does not correlate well with the size of infarcted tissue, any elevation of CK-MB in the clinical setting of myocardial infarction is suggestive of increased morbidity.14,15
How useful is the CK-MB serum measurement? The sensitivity in diagnosis of AMI relies heavily on elapsed time since symptom (chest pain) onset. If measured within four hours of symptom onset, the sensitivity is as low as 25-50%.16,17 Sensitivity continues to improve to 60-100% as the time from symptom onset reaches four to 12 hours. Therefore, significant variability in utility exists in part due to different types of assays, differences in diagnostic thresholds, and the heterogeneous enrollment criteria in the literature. However, the majority of studies do agree that the sensitivity of CK-MB does improve after four hours of symptom onset and that one measurement of CK-MB alone is not enough to rule out AMI.
The specificity of CK-MB ranges from 85-100%. This range tends to decrease as time from symptom onset extends beyond 12 hours. In conditions that increase total CK values, concomitant elevations in CK-MB values also can increase false-positive rates. In such cases, the specificity of the test can be improved by including the relative index that requires that the CK-MB mass be at least 5% of the total CK mass.17
In the ED setting, a single CK-MB measurement cannot provide confirmation of an acute coronary syndrome or other myocardial event. There is general consensus that CK-MB measurements made within 12-24 hours of symptom onset provide greater sensitivity and specificity in diagnosis of AMI.3,18 Additionally, a positive increase in paired CK-MB measurements separated by 90 to 120 minutes (termed the "delta") increases the sensitivity and specificity of diagnosing AMI.19 CK-MB is used in many ED and in-hospital settings as part of a biomarker panel, often along with myoglobin, to provide serial evaluations for change in values over time from initial symptoms onset. It is currently not recommended for solitary use to rule in or rule out AMI or ACS.
Troponins. Cardiac troponins T and I (cTnT and cTnI) are the regulatory proteins of the actin filaments of cardiac muscle tissue. The American College of Cardiology, the American Heart Association, and the European Society of Cardiology have jointly agreed that serum troponin measurement is the accepted standard biomarker for diagnosis of acute myocardial infarction and for the diagnosis of acute coronary syndrome.20 The cardiac isoforms are very specific for myocardial tissue: cardiac troponin I (cTnI) has not been identified outside cardiac muscle, and cardiac troponin T (cTnT) is only found in small amounts in skeletal muscle.21,22
Testing of monoclonal antibodies is the technique used to detect cardiac troponins in serum. The established first-generation cTnT enzyme-linked immunosorbent assay (ELISA) has 1-2% cross reactivity with skeletal muscle TnT.23 These skeletal isoforms are not detected in the second- or third-generation assays for cTnT.24 Several assays have been developed for CTnI, all with differing sensitivities and cutoff values, making standardization a problematic issue. These assays are composed of different antibody configurations that recognize different isotopes, thereby providing differences in coefficients of variability as well as differences with calibration materials. The American Association of Clinical Chemistry is attempting to standardize these assays for cTnI.25
The European Society of Cardiology (ESC) and American College of Cardiology (ACC) have determined that individual laboratories should use a cutoff point for each test at the 99th percentile of normal with less than 10% coefficient of variation. The 99th percentile reference limits provide a basis for establishing appropriate cutoff concentrations. What this means is that cTnI values indicative of myocardial damage range from 0.03 mcg/L to 1.1 mcg/L.20,26 The cutoff concentration, at the 99th percentile, for cTnT is 0.01 mcg/L.27 (See Table 1.) A review of stored plasma samples from 3557 participants in the population-based Dallas Heart Study evaluated the prevalence of cTnT elevations in the general population using contemporary assays. This study found that 99.3% of normal individuals have cTnT levels below the detectable limit (0.01 mcg/L).28
 |
The sensitivity of cTnT as an initial diagnostic marker of AMI has been noted to be as high as 87% when measured at least four hours after initial onset of symptoms. The specificity of this same marker has been documented to be as high as 94%. More importantly, a value of cTnT below the cutoff values for detection of AMI confers a negative predictive value (NPV) of 96%.24 In general, serum troponin levels remain elevated for 10-14 days after myocardial infarction.
In the ongoing search for the rapid and reliable diagnosis of AMI, there have been several investigations of newer, more sensitive troponin assays. Reichlin et al. looked at the performance of four ultra-sensitive troponins compared with the standard troponin assay in assessing biochemical changes of AMI. Their multicenter analysis showed that the diagnostic accuracy at patient ED presentation was significantly higher with each of the ultra-sensitive troponin assays as compared to the standard assay. When patients presented within three hours of onset of chest pain, the diagnostic accuracies ranged from 93-94% for the newer assays, as compared to 76% for the standard assay.29 These findings suggest an improved method for more rapid detection of AMI in patients presenting to the ED with chest pain. However, as with any potential change in evidence, it is important to consider all aspects of that evidence. In one case report series, the use of highly sensitive cardiac troponin (cTnI) increased both the true-positive and the false-negative rates of patients suspected to have AMI at presentation. In two patients who later ruled in for AMI, the initial cTnI was actually nondiagnostic. In another patient with blunt cardiac trauma, the initial cTnI was positive but later showed that this finding was not related to changes of myocardial ischemia.30 This particular report highlights the importance of utilizing any biochemical marker in the context of patient presentation and adjunctive laboratory tests. Most importantly, it stresses that we have yet to find a single biomarker that can be used unilaterally or in one isolated testing to conclusively detect AMI or ACS.
Elevated troponin values have important diagnostic and prognostic implications for patients. Therefore, it is important to recognize that a wide variety of conditions are associated with elevated serum troponin, so that diagnostic confusion and treatment dilemmas are reduced.
Patients with renal disease may have elevated cardiac troponin (cTn) measurements in the absence of clinically suspected myocardial ischemia or infarction. There are several theories behind this finding. One hypothesis suggests that uremia-induced skeletal myopathy leads to increased troponin in renal failure.31 However, other findings point to an increased likelihood that elevations of serum troponin in this patient population may be due to small events of clinically insignificant myocardial necrosis.32 Patients with renal disease often have left ventricular hypertrophy, which is highly correlated with increased cardiac troponin T (cTnT). What is least likely is that elevated serum troponin is due to decreased clearance of this molecule by a diseased kidney.33
Regardless of the underlying mechanism of elevated serum troponin, it is important for the clinical practitioner to understand the significance of these values. In suspected ACS with underlying renal failure, assessment of elevated cardiac troponins can be problematic. Sequential serum troponins should be obtained in this situation (delta troponin). An elevated but nonvarying troponin level indicates no new myocardial injury or infarction.34
With the use of the most current assays, cTnT is elevated more frequently that cTnI in patients with renal disease or failure. Dialysis also may affect levels of cTnT and cTnI, with a roughly 85% decrease in TnI in pre- and post-dialysis analyses.35 Cardiac disease is the most common cause of death in patients with end-stage renal disease. Apple et al. studied over 700 patients with renal disease and showed that elevated cTnT and cTnI were associated with increased risk of death in this population. Furthermore, individual assessments of serum values of cTnT and cTnI at various cutoff points determined by 99th percentile, ROC, and 10% CV showed a more prominent role for cardiac risk assessment when cTnT was elevated in patients with ESRD.36
BNP and NT-Pro BNP. During the past several years, B-type natriuretic peptide has been recognized as a useful marker in detecting the presence of left ventricular dysfunction.37 B-type natriuretic peptide (BNP) is 32 amino acid polypeptide that is secreted from the ventricular myocardium in response to stretching of cardiac myocytes under ventricular wall stress and overload. This is more evident in the presence of systolic dysfunction. BNP has both vasodilative and diuretic as well as natriuretic properties and is released by the ventricles as a neurohormonal response to ventricular wall stress.38
Pro-BNP is the precursor of BNP, which is enzymatically cleaved into an inactive amino terminal fragment known as NT-pro BNP and the biologically active carboxyl terminal fragment (BNP). NT-pro BNP has a longer half-life (one to two hours vs. 22 minutes) compared to BNP, with substantially more stability in EDTA blood samples. Concentrations of NT-Pro BNP tend to be about 20 times greater than circulating levels of BNP, and this ratio tends to increase with renal failure, as renal excretion has far greater differential effect on NT-pro BNP.27 Assays for both BNP and NT-Pro BNP exist; a meta-analysis performed by Clerico et al. found that assays for both fragments of pro BNP have equivalent diagnostic accuracy.39
There is recent evidence that BNP measurements may be useful in providing prognostic information in patients with ACS.40 The theory behind the value of BNP measurement in acute coronary syndrome is the finding that acute regional ventricular dysfunction, whether systolic, diastolic, or both, can occur in the early stages of the cascade of acute coronary ischemia that eventually leads to myocardial cell necrosis and cell death. The expression of BNP is independent of myocardial injury and cell death, and this alternative mechanism has the potential of identifying patients who are at risk but may not have necessarily crossed the threshold of cell necrosis.
Given that BNP is clearly associated with ventricular dysfunction, the logical hypothesis is that the setting of acute coronary occlusion and prolonged ischemia will lead to ventricular dysfunction and the expression of BNP. Studies have illustrated that elevations in plasma BNP levels in patients with AMI may be biphasic in those with large infarctions with systolic dysfunction.41
Other studies seem to demonstrate that BNP is a predictor of early and late cardiac events in patients who present with ST-segment elevation as well as non ST-segment elevation acute coronary syndrome.42,43 In the Orbofiban in Patients with Unstable Coronary Syndromes (OPUS)-TIMI 16 trial, BNP levels were measured at approximately 40 hours after the onset of symptoms, with high levels shown to correlate with higher rates of death and heart failure during 10 months of follow-up.44
BNP also has been shown to increase acutely after ischemia and to normalize after reversible ischemia. In patients undergoing stress testing, BNP seems to rise proportional to the size of the ischemic territory after exercise.45,46 More intriguing is the impact of adding NT-Pro BNP to other cardiac markers to increase the sensitivity, and positive and negative predictive values in the diagnosis of coronary ischemia in patients presenting to the emergency department with chest pain. (See Table 2.)
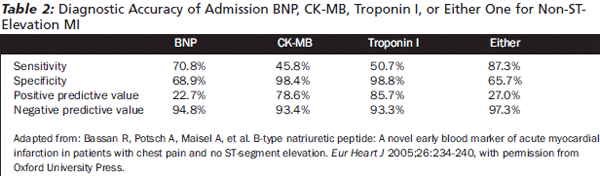 |
In a prospective study of 631 patients, Bassan et al. found that of the 72 patients with a final diagnosis of non-ST-segment elevation MI, only 37 had met diagnostic criteria via CK-MB and/or troponin I (cTnI) on admission.47 Elevated levels of BNP beyond the confirmed 100 pg/mL cutoff allowed for the identification of 22 more patients. It was noted that BNP had a significantly higher sensitivity for the independent diagnosis of AMI when compared to CK-MB and cTnI, but with lower specificities and positive predictive values, although negative predictive values were equivalent to CK-MB and cTnI. The authors concluded that the sensitivity of BNP was superior to the other cardiac markers in detecting emergency department chest pain patients presenting with non-ST-segment elevation myocardial infarctions without clinical left ventricular dysfunction.
The weight of evidence suggests that increased BNP levels cannot be used as a proxy for the degree of myocardial necrosis, but rather, can be used to assess the impact of the ischemic event on ventricular dysfunction and thus may be used as an additional predictive tool in the setting of chest pain with nondiagnostic ECGs and other biomarkers. There is no evidence to show that BNP or NT-Pro BNP is useful as a single biomarker in the identification of patients with acute coronary artery disease, but there is clear evidence for its use in a multivariate model as a screen for rapidly identifying patients who may be at risk for coronary artery disease. Furthermore, the use of BNP for risk stratification in patients who present with chest pain and its prognostic value in the short term for patients with acute coronary syndrome, holds significant promise.
Which Biomarker Is Best?
Accepted standards of practice in diagnosing AMI and ACS utilized serial measurements of CK-MB to evaluate patients presenting with chest pain. As newer studies emerge, the evidence seems to suggest that other biomarkers may be more useful in the risk assessment of patients with chest pain. Collinson et al. showed that serial CK-MB measurements were less powerful than cTnT measurement for diagnosis of myocardial infarction in their population of nearly 800 patients admitted to a chest pain observation unit (CPOU).48 In this study, cTnT measurements at six hours after symptom onset had high diagnostic sensitivity and were superior to CK-MB for detection of AMI.
The emergency physician often is challenged by patients who present with chest pain but who have an atypical story, unreliable history, or clinically uncertain diagnosis. This population was closely studied by McErlean and colleagues. Their study showed that cTnT and CK-MB were comparable in identifying patients at high risk for in-hospital adverse events. However, they also showed that patients with intermediate cTnT values had a higher risk of suffering an early adverse event. Importantly, the authors found that in patients with clinically suspected ACS admitted to the hospital, there was a 27% incidence of early adverse events even if the CK-MB and cTnT levels were not abnormal on serial measurements over the initial 16 hours. Thus, in patients deemed as having ACS by other criteria, a negative cTnT did not reduce the risk to enable discharge from the emergency department.49
Although the evidence can be contradictory, there is general consensus that an initial cTnT has higher specificity and positive predictive value for underlying cardiac ischemia or infarction than traditional serial CK-MB measurements.50 There is evidence to suggest that a single biomarker can be preferentially used to rule out the presence of acute coronary syndrome. James et al. conducted a retrospective review of patients from Fragmin and fast Revascularization during InStability in Coronary artery disease II (FRISC-II) trial and examined a prospective evaluation of 7115 patients in Global Utilization of Strategies To open Occluded arteries IV (GUSTO-IV) trial to identify a cutoff value for TnT in patients with ACS at low risk for mortality. They concluded that a cTnT value of < 0.01 mcg/L is useful in identifying patients with ACS at low risk for subsequent mortality.51 At present, the National Association of Clinical Biochemistry does not recommend ruling out ACS based on single biomarker assay.
Cameron et al. suggested that a multi-marker approach be used to facilitate prediction of adverse events. In their study, cTnT had greater prognostic value for determining adverse events in patients with chest pain when compared to NT-proBNP, myoglobin, and CK-MB. Importantly, the combination of cTnT and NT-proBNP outperformed the other markers in predicting adverse events in patients with suspected ACS.52
Morita el al. was one of the first investigators to demonstrate significant elevations in BNP on patients with ST-segment elevation myocardial infarction compared to controls.41 Bassan et al. noted that adding BNP to other biomarkers increased both the sensitivity and negative predictive value for non-ST-segment myocardial infarction.47
Brown et al. looked at the impact to sensitivity and negative predictive values of adding BNP to cTnI, CK-MB, and myoglobin in the evaluation of chest pain patients with acute myocardial infarction or acute coronary syndrome.53 The study conclusions noted that more than one BNP measurement (at presentation and 90 minutes later) had very limited potential in identifying patients with acute ischemia. However, this study did indicate that adding a BNP to other cardiac markers increased sensitivity, and negative predictive value of myocardial infarctions was greater than 99% in patients with no evidence of congestive heart failure. The overall sensitivities and negative predictive values did decrease slightly for all comers with chest pain and the potential for acute coronary syndrome, but remained higher when the combination of myoglobin, CK-MB, and cTnI were used alone even though the authors could not effectively conclude that the incremental value of adding BNP differed significantly with biomarker panels that included myoglobin. The authors also suggest that measuring BNP identified an additional patient with acute myocardial infarction for every 14 false positives.
Biomarker Panels
Recommendations on the use of cardiac biomarkers in ED triage of chest pain patients come from the National Academy of Clinical Biochemistry (NACB). The Academy supports an accelerated protocol for cardiac marker use that includes: an early marker that is reliably increased in the blood within six hours of symptoms and a definitive marker that is increased in the blood after six to nine hours.55 Furthermore, the definitive marker should have high sensitivity and specificity for myocardial necrosis/injury and should remain elevated for several days after onset of symptoms. The two markers best fitting these criteria are CK-MB (recommended as the early detection marker) and cardiac troponin (recommended as the definitive marker). (See Table 3.)
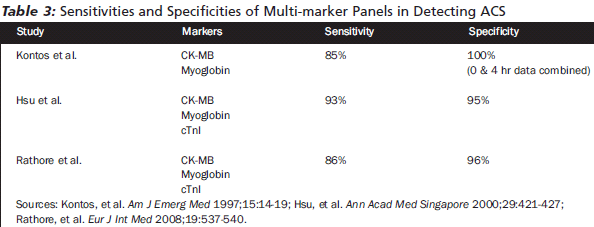 |
The natural process of myocardial ischemia, injury, or necrosis is a dynamic cellular event. This pathophysiology strongly supports the diagnostic strategy of obtaining serial biomarker measurements, ideally over a six- to nine-hour period. Whereas a single cardiac marker determined at time of ED presentation has low sensitivity for detection of AMI, serial biomarker evaluation has high sensitivity for diagnosing AMI.55 Rathore et al. showed that the specificity of paired triple cardiac marker panel (CK-MB, myoglobin, and cTnI) was as high as 96% in a population of ED patients at low risk for ACS. The use of the paired panel (initially upon arrival and 120 minutes later) allowed them to rapidly exclude ACS in 30% of the study population and discharge them out of the ED. They also found that the re-admission rate for ACS during a six-month follow-up period was 1%, and there were no deaths.56
The ED physician evaluating a patient with chest pain faces the challenges of inaccurate historical time of symptom onset and nondiagnostic ECGs. The utility of serial biomarker measurements is extremely beneficial in these situations. Hamm et al. suggest that a finding of two negative values on serial cardiac biomarker testing, the second test being obtained at least six hours after initial symptom onset, is adequate to allow safe ED discharge.57 A similar conclusion about six-hour serial sampling in a low-risk chest pain population was reached by Collinson et al.48 Eggers et al. studied cumulative sensitivities and specificities of triple biomarker panels (cTnI, CK-MB, and myoglobin) and showed an increase in sensitivities at two hours from initial presentation, with further increased sensitivities at six hours. This group also looked at individual marker sensitivities and concluded that even in patients presenting with less than four hours of chest pain, cTnI demonstrated the highest early sensitivities for detecting AMI.58
Other Biomarkers
Although not routinely used in the ED setting, there are a host of other cardiac biomarkers that have been studied in the setting of acute coronary syndromes. Heart-type fatty acid binding protein and ischemia modified albumin are two of the more prominent markers for potential development in ED use.
Heart-type Fatty Acid-binding Protein (H-FABP). Fatty acid-binding protein is ubiquitous in the cytoplasm of fatty acid-utilizing cells and functions as a cellular transporter of fatty acids. Heart-type fatty acid-binding protein (H-FABP) also is specific to cardiac tissue. In the setting of myocardial ischemia or infarction, the loss of sarcolemmal integrity in injured myocytes leads to rapid release of H-FABP into the circulation. This immediate release significantly precedes the release of CK-MB and cardiac troponins, and so H-FABP has been suggested as the ideal marker for the early detection of ischemic myocardial necrosis.59,60
The study by Nakata et al. suggests that H-FABP can be an early diagnostic and prognostic biomarker in patients presenting with chest pain to the ED within the first six hours of symptom onset. However, the authors noted that an important limitation in their study was the prolonged time (up to 24 hours) to obtain a quantitative result of H-FABP.61 An immunochromatography test for H-FABP has been developed, with qualitative results available in 15 minutes. Initial results show high sensitivity, near 93%, but low specificity. Further validation and comparison of qualitative versus quantitative results are required.62
Ischemia Modified Albumin. The cellular changes seen in myocardial ischemia result in structural changes in the N-terminus of serum albumin, which reduces its binding capacity for cobalt cations. These changes are likely due to production of reactive oxygen species during hypoxic events, acidosis, ischemia, and/or reperfusion.63 Ischemia Modified Albumin (IMA) is an emerging, highly sensitive marker for the diagnosis of myocardial ischemia. At acute chest pain presentation, elevated levels of IMA have been shown to be an early indication of subsequent elevations of cardiac troponin levels. The correlate is that normal IMA levels at chest pain presentation predict subsequent negative troponin elevations, suggesting that IMA has a useful role in early rule out of ACS, with high sensitivity and negative predictive values.64,65
Recently, Roy and colleagues showed that, in patients with acute ED presentation of chest pain, normal ECG, and negative initial cardiac troponin values, elevation of initial IMA was an early and independent predictor of ACS.66 Despite these supportive studies, IMA is not routinely available in most hospitals for use as a cardiac biomarker for ED patients.
Conclusion
Emergency physicians continue to be challenged by the evaluation of patients presenting with chest pain. However, with more accurate and dependable diagnostic tools, emergency physicians are better able to exclude coronary artery syndromes and acute myocardial infarctions from these patient populations. Accurate historical time frames are important elements in aiding the interpretation of serum biomarker values, with certain cTnI values showing diagnostic sensitivity as early as four hours from the time of symptom onset.
While biomarker panels continue to evolve, the most sensitive and specific tools currently available are CK-MB and cTnI or cTnT. Although single use of any individual biomarker cannot decisively exclude the presence of ACS, there is a preponderance of evidence to support best practices in accurate biomarker panel use and interpretation. When used in the correct frame of evaluation, these marker panels provide a wealth of information regarding patient prognosis in the setting of ACS.
References
1. Chest Pain a Leading Reason for Hospital Emergency Department Visits. AHRQ News and Numbers, February 27, 2008. Agency for Healthcare Research and Quality, Rockville, MD. http://www.ahrq.gov/news/nn/nn022708.htm)
2. McQueen MJ, Holder D, El-Maraghi NR. Assessment of the accuracy of serial electrocardiograms in the diagnosis of myocardial infarction. Am Heart J 1983;105:258-261.
3. Bakker AJ, Koelemay MJW, Gorgels JPMC, et al. Failure of new biochemical markers to exclude acute myocardial infarction at admission. Lancet 1993;342:1220.
4. Bhayana V, Cohoe S, Leung FY, et al. Diagnostic evaluation of creatinine kinase-2 mass and kinase-3 and -2 isoform ratios in early diagnosis of acute myocardial infarction. Clin Chem 1993;39:488.
5. Jaffe AS, Landt Y, Parvin C, et al. Comparative sensitivity of cardiac troponin I and lactate dehydrogenase isoenzymes for diagnosing acute myocardial infarction. Clin Chem 1996;42:1770–1776.
6. Apple FS, Murakami M, Panteghini M, et al. International survey on the use of cardiac markers. Clin Chem 2001;47:587-588.
7. Gibler WB, Gibler CD, Weinshenker E, et al: Myoglobin as an early indicator of acute myocardial infarction. Ann Emerg Med 1987;16:851-856
8. Tucker JF, Collins RA, Anderson AJ, et al. Value of serial myoglobin levels in the early diagnosis of patients admitted for acute myocardial infarction. Ann Emerg Med 1994;24:704-708.
9. Brogan GX, Friedman S, McCuskey C, et al. Evaluation of a new rapid quantitative immunoassay for serum myoglobin versus CK-MB for ruling out acute myocardial infarction in the emergency department. Ann Emerg Med 1994;24:665–671.
10. Kontos MC, Anderson FP, Hanbury CM, et al. Use of the combination of myoglobin and CK-MB mass for the rapid diagnosis of acute myocardial infarction. Am J Emerg Med 1997;15:14–19.
11. Maisel AS, Templin K, Love M, et al. A prospective study of an algorithm using cardiac troponin I and myoglobin as adjuncts in the diagnosis of AMI and intermediate coronary syndromes in a veteran's hospital. Clin Cardiol 2000;23:915–920.
12. Lee TH, Goldman L. Serum enzyme assays in the diagnosis of acute myocardial infarction. Ann Intern Med 1986;105:221.
13. Lee TH, Weisberg MC, Cook EF, et al. Evaluation of creatine kinase and creatine kinase-MB for diagnosis myocardial infarction. Arch Int Med 1987;147:115
14. Alexander JH, Sparapani RA, Mahaffey KW, et al. Association between minor elevations of creatine kinase-MB level and mortality in patients with acute coronary syndromes without ST-segment elevation. Eur Heart J 2004;25:313-321.
15. Fiolet JW, ter Welle HF, van Capelle FJ, et al. Infarct size estimation from serial CK-MB determination: Peak activity and predictability. Br Heart J 1983;49:373-380.
16. Collinson PO, Rosalki SB, Kuwana T, et al. Early diagnosis of acute myocardial infarction by CK-MB mass measurements. Ann Clin Biochem 1992;29:43-47.
17. Collinson PO, Rosalki SB, Kuwana T, et al. Early diagnosis of acute myocardial infarction by CK-MB mass measurements. Ann Clin Biochem 1992;29:43-47.
18. Laurino JP, Bender EW, Kessimian N, et al. Comparative sensitivities and specificities of the mass measurements of CK-MB2, CK-MB, and myoglobin for diagnosing acute myocardial infarction. Clin Chem 1996;42:1454-1459.
19. Bakker AJ, Koelemay MJW, Gorgels JP et al. Failure of new biochemical markers to exclude acute myocardial infarction at admission. Lancet 1993;342:1220.
20. Fesmire FM, Percy RF, Bardoner JB, et al. Serial creatinine kinase MB testing during the emergency department evaluation of chest pain: Utility of a 2-hour delta CK-MB of +1.6 ng/mL. Am Heart J 1998;136:237-244.
21. Ammann P, Pfisterer M, Fehr T, et al. Raised cardiac troponins. BMJ 2004;328:1028-1029.
22. Bodor GS, Porterfield D, Voss EM, et al. Cardiac troponin I is not expressed in fetal and healthy or disease adult human skeletal muscle tissue. Clin Chem 1995;41:1710.
23. Ricchiuti V, Voss EM, Ney A, et al. Cardiac troponin T isoforms expressed in renal diseased skeletal muscle will not cause false-positive results by the second generation cardiac troponin T assay by Boehringer Mannheim. Clin Chem 1998;44:1919.
24. Myocardial infarction redefineda consensus document of the joint European Society of Cardiology/American College of Cardiology committee for the redefinition of myocardial infarction. J Am Coll Cardiol 2000;36:959.
25. Jaffe AS, Ravkilde J, Roberts R, et al. It's time for a change to a troponin standard. Circulation 2000;102:1216-1220.
26. Apple FS, Murakami MM. Serum 99th percentile reference cutoffs for seven cardiac troponin assays. Clin Chem 2004;50: 1477-1479.
27. Apple FS, Quist HE, Doyle PJ, et al. Plasma 99th percentile reference limits for cardiac troponin and creatine kinase mb mass for use with European society of cardiology/American society of cardiology consensus recommendations. Circulation 2003;49:1331-1336.
28. Wallace TW, Abdullah SM, Drazner MH, et al. Prevalence and determinants of troponin T elevation in the general population. Circulation 2006;113:1958-1965.
29. Reichlin T, Hochholzer W, Bassetti S, et al. Early diagnosis of myocardial infarction with sensitive cardiac troponin assays. N Engl J Med 2009;361:913-915.
30. Wu AH. Interpretation of high sensitivity cardiac troponin I results: Reference to biological variability in patients who present to the emergency room with chest pain: Case report series. Clin Chim Acta 2009;401: 170-174.
31. Diesel W, Emms M, Knight BK, et al. Morphologic features of the pathology associated with chronic renal failure. Am J Kid Dis 1993;22:677-684.
32. Ooi DS, Isotalo PA, Veinot JP. Correlation of antemortem serum creatine kinase, creatine kinase MB, troponin I and troponin T with cardiac pathology. Clin Chem 2000; 46:338-344.
33. Fredricks S, Chang R, Gregson H, et al. Circulating cardiac troponin T in patients before and after renal transplantation. Clin Chim Acta 2001;310:199-203.
34. Freda BJ, Tang W, Peacock WF, et al. Cardiac troponins in renal insufficiency: Review and clinical implications. J Am Coll Cardiol 2002;40:2065-2071.
35. Wayand D, Baum H, Schatzle G, et al. Cardiac troponin T and I in end-stage renal failure. Clin Chem 2000:46:1345-1350.
36. Apple FS, Murakami MM, Pearce LA, et al. Predictive value of cardiac troponin T and I for subsequent death in end stage renal disease. Circulation 2002;106:2941-2945.
37. Bassan R, Potsch A, Maisel A, et al. B type natriuretic peptide: A novel early blood marker of acute myocardial infarction in patients with chest pain and no ST-segment elevation. Eur Heart J 2005;26:234-240.
38. Maeda K, Tsutamoto T, Wada A, et al. Plasma brain natriuretic peptide as a biochemical marker of high left ventricular end-diastolic pressure in patients with symptomatic left ventricular dysfunction. Am Heart J 1998;135:825-832.
39. Yasue H, Yoshimura M, Sumida H, et al. Localization and mechanism of secretion of B-type natriuretic peptide in comparison with those of A-type natriuretic peptide in normal subjects and patients with heart failure. Circulation 1994;90:195-203.
40. Clerico A, Fontana M, Zyw L, et al. Comparison of the diagnostic accuracy of brain natriuretic peptide (BNP) and the N-terminal part of the propeptide of BNP immunoassays in chronic and acute heart failure: A systematic review. Clin Chem 2007;53:813-822.
41. Morita E, Yasue H, Yoshimura M, et al. Increased plasma levels of brain natriuretic peptide in patients with acute myocardial infarction. Circulation 1993;88:82-91.
42. Hall C, Cannon CP, Forman S, et al. Prognostic value of N-terminal proatrial natriuretic factor plasma levels measured within the first 12 hours after a myocardial infarction (TIMI) II Investigators. J Am Coll Cardiol 1995;26:1452-1456.
43. Omland T, Aakvaag A, Bonarjee VV, et al. Plasma brain natriuretic peptide as an indicator of left ventricular systolic function and long term survival after acute myocardial infarction. Comparison with plasma atrial natriuretic peptide and N-terminal proatrial natriuretic peptide. Circulation 1996;93:1963-1969.
44. Cannon CP, McCabe CH, Langer A, et al. Oral glycoprotein IIb/IIIa inhibition with orbofiban in patients with unstable coronary syndromes (OPUS TIMI 16 Trial). Circulation 2000;102:149-156.
45. Sabatine MS, Morrow DA, de Lemos JA, et al. Acute changes in circulating natriuretic peptide levels in relation to myocardial ischemia. J Am Coll Cardiol 2004;44: 1988-1995.
46. Marumoto K, Hamada M, Hiwada K. Increased secretion of atrial and brain natriuretic peptides during acute myocardial ischemia induced by dynamic exercise in patients with angina pectoris. Clin Sci 1995;88:551-556.
47. Bassan R, Potsch A, Maisel A, et al. B-type natriuretic peptide: A novel early blood marker of acute myocardial infarction in Patients with chest pain and no ST-segment elevation. Eur Heart J 2005;26,234-240.
48. Collinson PO, Gaze C, Morris F, et al. Comparison of biomarker strategies for rapid rule out of myocardial infarction in the emergency department using ACC/ESC diagnostic criteria. Ann Clin Biochem 2006;43:273-280.
49. McErlean S, Deluca SA, van Lente F, et al. Comparison of troponin T versus creatine kinase-MB in suspected acute coronary syndromes. Am J Cardiol 2000;85:421-426.
50. Zarich SW, Qamar AU, Werdmann MJ, et al. Value of a single troponin T at the time of presentation as compared to serial CK-MB determinations in patients with suspected myocardial ischemia. Clin Chim Acta 2002;326:185-192.
51. James S, Armstrong P, Califf R, et la. Troponin T levels and risk of 30-day outcomes in patients with the acute coronary syndrome: Prospective verification in the GUSTO-IV trial. Am J Med 2003;115: 178-184.
52. Cameron SJ, Sokoll LJ, Laterza OF, et al. A multi-marker approach for the prediction of adverse events in patients with acute coronary syndromes. Clin Chim Acta 2007;376:168-173.
53. Brown AM, Sease,KL, Robey JL, et al. The Impact of B-type natriuretic peptide in addition to troponin I, creatinine kinase-MB, and myoglobin on the risk stratification of emergency department chest pain patients with potential acute coronary syndrome. Ann Emerg Med 2007;49:153-163.
54. Wu AHB, Apple FS, Gibler WB, et al. National Academy of Clinical Biochemistry Standards of Laboratory Practice: Recommendations for the use of cardiac markers in coronary artery disease. Clin Chem 1999;45:1104-1121.
55. Storrow AB, Gibler WB. Chest pain centers: Diagnosis of acute coronary syndromes. Ann Emerg Med 2000;35: 449-461.
56. Rathore S, Knowles P, Mann A, et al. Is it safe to discharge patients from accident and emergency using a rapid point of care triple cardiac marker test to rule out acute coronary syndrome in low to intermediate risk patients presenting with chest pain? Eur J Int Med 2008;19:537-540.
57. Hamm CW, Goldmann GU, Heeshcen C, et al. Emergency room triage of patients with acute chest pain by means of rapid testing for cardiac troponin T or troponin I. N Engl J Med 1997;337:1648-1653.
58. Eggers KM, Oldgren J, Nordenskjold A, et al. Diagnostic value or serial measurement of cardiac markers in patients with chest pain: Limited value of adding myoglobin to troponin I for exclusion of myocardial infarction. Am Heart J 2004;148:574-578.
59. Tanaka T, Hirota Y, Sohmiya K, et al. Serum and urinary human heart fatty acid-binding protein in acute myocardial infarction. Clin Biochem 1991;24:195-201.
60. Okamoto F, Sohmiya K, Ohkaru Y, et al. Human heart-type cytoplasmic fatty acid-binding protein (H-FABP) for the diagnosis of acute myocardial infarction. Clinical evaluation of H-FABP in comparison with myoglobin and creatine kinase isoenzyme MB. Clin Chem Lab Med 2000;38: 231-238.
61. Nakata T, Hashimoto A, Hase M, et al. Human heart-type fatty acid-binding protein as an early diagnostic and prognostic marker in acute coronary syndromes. Cardiology 2003;99:96-104.
62. Takeshita H, Nishida H, Horinouchi K, et al. Clinical usefulness of a whole-blood panel test for rapid detection of human heart-type fatty acid-binding protein. Jpn J Med Technol 2001;50:91-96.
63. Bar-Or D, Curtis G, Rao N, et al. Characterization of the Co (2+) and Ni(2+) binding amino-acid residues of the N-terminus of human albumin. An insight into the mechanism of a new assay for myocardial ischemia. Eur J Biochem 2001;268: 42-47.
64. Sinha MK, Gaze DC, Tippins JR, et al. Ischemia modified albumin is a sensitive marker of myocardial ischemia after percutaneous coronary intervention. Circulation 2003;107:2403-2405.
65. Sinha MK, Roy D, Gaze DC, et al. Role of ischemia modified albumin, a new biochemical marker of myocardial ischaemia, in the early diagnosis of acute coronary syndromes. Emerg Med J 2004;21:29-34.
66. Roy D, Quiles J, Aldama G, et al. Ischemia modified albumin for the assessment of patients presenting to the emergency department with acute chest pain but normal or non-diagnostic 12-lead electrocardiograms and negative cardiac troponin T. Int J Cardiol 2004;97:297-301.
How the use of cardiac biomarkers has changed.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
