Common Diagnoses Become Difficult Diagnoses When Geriatric Patients Visit the Emergency Department: Part I
Common Diagnoses Become Difficult Diagnoses When Geriatric Patients Visit the Emergency Department: Part I
It is another busy night in the emergency department (ED), but you are managing to keep up with the tempo. However, the next chart in the rack is bound to slow your pace: It is an 83-year-old man with the chief complaint of "not feeling well." While younger patients who present with vague complaints may not be diagnosed with anything more serious than needing a work excuse, elderly patients are at much higher risk for significant pathology. In addition, elderly patients often present atypically and thus are more likely to have important diagnoses overlooked. Further, elderly patients naturally require more time to evaluate as they may provide histories more slowly, often have extensive old records to review, and may have extensive laboratory studies.
Introduction
Definitions. The term "elderly" has arbitrarily been defined in the past as age greater than 65 years.1 Most authors refer to those between 65–74 years as young old, and those greater than 85 years as the oldest old. People older than 100 years are designated as centenarians.
Demographics. The elderly population has been growing more rapidly than the general population for the past 100 years. From 1900 to 2000, the percentage of elderly grew from 4.1% of the population to 12.4%.2 While the population of the United States increased 3.7 times during that time, the number of elderly grew 11-fold.2
But now the U.S. population is on the cusp of an unprecedented change, largely due to the aging of the baby-boomer generation. Beginning in 2011, the first of the baby boomers will turn 65 years old, and over the next 19 years the population of elderly people in the United States is projected to double, while the overall population will grow only 18%.2 In 2003, there were roughly 36 million people in the United States 65 years or older, making up 12% of the population. By 2030, those older than 65 years will number 72 million, or 20% of the U.S. population.2 By 2050, the rate of growth will have slowed, but there still will be 86.7 million elderly, or 22% of the population.2
The fastest growing subset of elderly people is the oldest old (greater than 85 years). By 2050, the number of oldest old will have increased from 4.3 million (1.6% of the population) in 2000 to 19.3 million (4.8% of the population).3
Utilization of ED Resources by the Elderly
These population changes have powerful implications for cost and utilization of resources in the ED. While elderly patients currently comprise only 12% of the population, they consume a much larger percentage of health care resources. Overall, care of the elderly consumes roughly 33% of all health care expenditures, a number that has remained stable since the 1980s.4 Utilization of ED/hospital resources is even higher: elderly patients represent 15% of all ED patients but 36% of ambulance transports, 40% of all patients admitted, and nearly 50% of critical care admissions.5,6
Age-Related Changes in Physiology
Vital Signs. As we age, the normal oral temperature actually falls to 97.4° F and the ability to mount a fever (> 100.5° F) diminishes.7 Thus, up to 20% of elderly patients with bacterial infection do not have a fever, and up to 47% may have a blunted fever response (< 101° F).8
Unlike baseline temperature, the normal resting respiratory rate and heart rate do not change significantly with age, but many common disease processes will affect these values (i.e., sick sinus syndrome, emphysema). Blood pressure in the healthy older adult naturally increases with time but should not reach abnormal levels (> 140 mmHg) without the addition of a disease process (hypertension, obesity, etc). Further, normal age-related changes do increase the risk for orthostatic hypotension in the elderly.
Pain Perception. The concept of pain perception and subsequent pain management in elderly patients is multifaceted. Debate still occurs as to whether elderly people experience pain similarly to younger individuals, with articles suggesting both that elderly patients have higher pain thresholds and lower ones.
While some authors suggest that perceived differences in pain sensation in geriatric patients stem from the presence of coexisting diseases (dementia, cancer) or analgesic/sedative medications, several documented changes in the peripheral nervous system occur with aging. Nerve conduction is reduced due to decreased blood flow, the number of both myelinated and unmyelinated nerve fibers decreases with age, and levels of neurotransmitters decreases. All these factors contribute to alter the geriatric patient's response to acute pain.9
These types of findings could explain why elderly patients tend to present later with abdominal problems such as bowel obstruction or have increased rates of silent myocardial infarction (MI). For example, a review of 212 cases of older adults with peritonitis found that 45% did not report any pain.10 Likewise, a study of more than 4100 elderly patients with unstable angina found that only 14% reported chest pain.11 These atypical presentations are even more common in the oldest patients. Another study found that while the rate of atypical MI was near 50% in 75-85 year olds, the rate was 75% in those older than 85 years.12 Instead of pain complaints, the elderly may only present with vague behavioral changes such as confusion, restlessness, aggression, or fatigue.
Treatment of pain in the elderly patient also can be challenging. Older patients often require lower doses of pain medications due to decreased metabolism or lower renal clearance. They also are more likely to have cross-reactions with other medications or to suffer from side effects (decreased cognition).
Laboratory Values. Many of the standard ED laboratory values are affected by the natural aging process. One of the more important values for the ED physician is creatinine clearance, as it affects drug dosing and risk of acute renal failure with contrasted CT scans. Creatinine clearance naturally decreases with age, falling about 6.5 mL/min/1.73 m2 every decade. Loss of renal mass (30% by age 90) causes this decline and results in a loss of GFR of up to 50% by age 90. Serum creatinine does not naturally rise, though, as the loss in renal mass is offset by a decline in muscle mass.
D-dimer levels rise with normal aging, with one study finding D-dimer levels in patients older than 80 years were 4.5 times higher than levels for people aged 16-40 years without any evidence of a reason for elevated D-dimer levels.13 Arterial PO2 levels also decline with age, falling roughly 3 mmHg per decade. This occurs as alveoli lose elasticity over time and gradually increase in size, thus lowering the overall lung surface area for gas exchange. Since pH and PCO2 levels do not change with age, many elderly patients naturally have an increased A-a gradient.
Immune System. Decline in immune system function with age (termed immunosenescence) is well documented, but the decrease appears to be more dramatic in cellular rather than humoral immunity. T cells are particularly important for aiding in immune system control of intracellular pathogens that can survive inside macrophages, such as Listeria, Salmonella, Legionella, and Mycobacteria spp.14 Loss of T cell function likely explains the increased risk of infection by these organisms in the elderly.
Humoral (antibody-based) immunity also is affected by aging. However, many of the declines in B cell antibody production can be traced back to loss of helper T cells. Many components of host defense against infection not directly part of the immune system also are reduced with aging, such as reduced circulation or thinning of skin. Malnutrition, more frequent hospitalization, chronic corticosteroid use, and co-existing diseases (renal disease, diabetes, HIV) also contribute to increase the elderly person's risk of infection.
Common Presentations in Geriatric Patients
Mental Status Change. Mental status change is perhaps the most generic presenting complaint for elderly patients. Behavior changes can represent anything from infection (pneumonia, urosepsis) or acute MI to medication reaction or simple depression. At times the change in mental status will be obvious, i.e. a patient can no longer walk who was walking without assistance the day before. Other times, the change may be more subtle. For example, the patient may be more confused than normal or may have stopped eating. Besides loss of activities, mental status changes also can manifest as increased aggression, restlessness, or agitation.
Delirium. Distinguishing acute delirium from dementia-related changes in mental status in elderly patients is critical, as conditions that produce delirium can be life-threatening. Unfortunately, delirious patients are both common and easily missed. Previous studies have documented the incidence of delirium in elderly inpatients to be as high as 25-40%.15 Estimates of delirious elders in ED populations are lower but consistently are in the 10-12% range.16,17 Despite the relative regularity with which delirious elders present to the ED, the diagnosis is easily missed by treating physicians, with studies estimating the diagnosis is not made in 32-67% of delirious elders.18 Failing to recognize the presence of delirium in elderly patients is a significant problem, as the presence of delirium greatly increases risk of death. Mortality rates during hospitalization range from 22-76%, giving a 14-fold increase in death with the diagnosis of delirium.19
Table 1: Diagnostic Criteria for Acute Delirium
- Acute onset with fluctuating course
AND
- Inattention (difficulty following a conversation, perseveration)
WITH EITHER
- Disorganized thinking
OR
- Altered level of consciousness (can be less active, i.e., lethargy or coma, or more active, i.e., hypervigilance or hyperactive)
Delirium has been defined as an acute, reversible and global disruption of cognition that manifests as altered thinking, memory, and perception. (See Table 1.) The requirement of an acute onset (change in hours to days) with fluctuating course is not surprising, but the other required criterion is inattention. Inattention can be subtle but is described as an important clue in the diagnosis of delirium.20 Inattention refers to not being able to hold a conversation, being easily distracted, or the presence of perseveration. In many cases, one can only determine if mental status changes are new by speaking with relatives or nursing home caregivers who know the patient's baseline behavior.
Besides these first two criteria, an altered level of consciousness or disorganized thinking needs to be present to diagnose delirium. Disorganized thinking can be easily dismissed as caused by underlying dementia. Verbose but illogical answers to questions make disorganized thinking easier to detect, but patients who answer most questions with few words (OK, yes, no, sometimes) are especially difficult to recognize. Again, one must speak with caregivers who can attest to the patient's baseline behaviors. When unable to obtain this information, one may need to assume that being a "poor historian" possibly is not the patient's baseline. Similarly, altered level of consciousness is easy to perceive in lethargic or comatose patients, but patients with "hypoactive delirium" often are missed.18 These patients are not unresponsive, and neither are they pulling their IVs out and trying to get out of bed repeatedly. They may just sit quietly without appearing obviously altered. While one classically thinks of a hyperactive patient as delirious, these represent only 25% of all delirious patients compared to 50% being diagnosed as hypoactive.21
Although infection/sepsis is a common cause of delirium in elderly ED patients, multiple other causes also can be responsible. It is important to keep alcohol-related conditions (intoxication, withdrawal) in mind when treating elderly patients. Alcoholism often is overlooked in treating elderly patients. While some studies find a problem-drinking rate of 1-3% in the elderly U.S. population, problem drinking has been found in up to 14% of elderly ED patients.22 Other causes of delirium include drugs (especially benzodiazepines, opiates), dehydration, acute renal failure, myocardial infarction, arrhythmia, congestive heart failure, and subdural hematoma.
Polypharmacy. It seems rare these days to see an elderly patient who is not taking multiple mediations. When the evaluation for delirium has not yielded a cause, the patient's medications may be the source of the problem. A review of 300 elderly ED patients found that 10% of ED visits and 10-17% of hospital admissions were due to adverse drug events in the elderly.23 All of this drug use naturally leads to increased risk of adverse drug reactions, many of which can be serious. One article suggested that if fatal adverse drug reactions were classed as a distinct cause of death, they would rank as the 5th leading cause of death, above pneumonia and diabetes.24
Common medications alone that have been documented to cause delirium in elderly patients include: digoxin, amiodarone, amitriptyline, phenytoin, carbamazepine, diphenhydramine, ranitidine, haloperidol, cyclobenzaprine, diazepam, prednisone, indomethacin, lithium, meperidine, proproxyphene, and codeine.18 While it may be impossible to determine in some cases, relatives/caregivers should be asked if the patient has started taking any new medications or changed his or her medication doses recently. Another clue to recent medication changes can be that the patient was recently in the office or admitted to the hospital before the onset of delirium. If no other cause for delirium is found, withholding medications may be the key to recovery and is useful information to pass on to the admitting physician.
Occult Traumatic Brain Injury. As expected, injured elderly patients do not fare as well as their younger counterparts, and thus trauma is the fifth leading cause of death in patients older than 65 years. Falls from a standing height are the most common mechanism of injury in those older than 70 years of age, with 60.7% of injuries caused by falls and only 21% of injuries due to motor vehicle accidents.25 People older than 65 years fall frequently, with a 30% annual incidence of falls. This number rises to 50% for those older than 80 years.26
When the elderly fall, they also tend to be injured more frequently. One study found that simple falls resulted in serious injury in 30% of elderly patients compared to only 4% of young patients.26 The risk of death from simple falls was 10-fold higher in the elderly compared to younger patients (25% vs. 2.5%).26 Contrary to what one might think, the most common area of the body injured in these low-level falls is the head/face and not the pelvis or femur.27
Multiple factors contribute to the increased risk of brain injury in the elderly. Loss of brain volume averages about 10% (140 grams) between ages of 30 and 70 years, resulting in an enlarged subdural space. This increases the stretch of bridging veins and makes them more vulnerable to shear stresses, but also allows the subdural space to accommodate larger blood volumes before cerebral compression occurs. Thus, elderly patients can present with relatively large subdural fluid collections but with little or no clinical evidence of their existence. (See Figure 1.) The same process applies to alcoholics who experience advanced brain atrophy for their age.28
Figures 1A-C: Subacute Subdural Hematoma
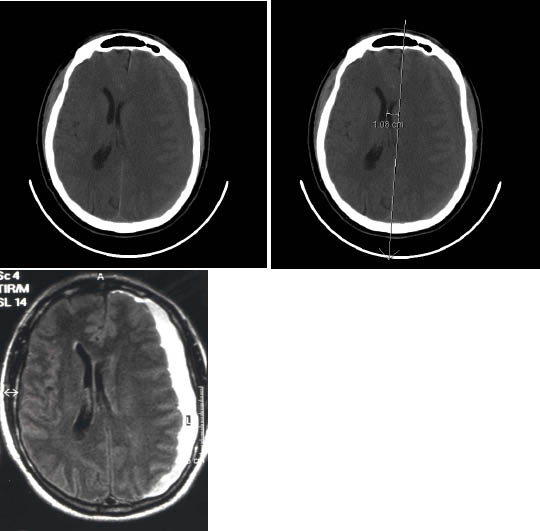
Subacute Subdural. Patient presented two weeks after head trauma with loss of consciousness and subsequent headaches, but few other symptoms. 1A shows that blood is isodense with the brain. 1B shows that midline shift and loss of sulci are the important findings to indicate subdural hemorrhage. 1C shows MRI image of the subdural hemorrhage.
Use of anticoagulants is another significant risk factor for development of intracranial hemorrhage in the elderly. Anticoagulation therapy is most often used to prevent ischemic stroke and does lower the risk an estimated 68% in patients with chronic atrial fibrillation.29 However, this reduction is balanced by a 40% increased risk of subdural hematoma in those elderly at risk for falls.30 In addition, use of warfarin increases risk of spontaneous intracranial hemorrhage up to 5% per year,31 with a 1% mortality per year.32
As expected in patients on warfarin, the higher the average INR (international normalized ratio), the greater the risk. One study of elderly patients on warfarin with traumatic brain injury found that INR levels greater than 6.0 were associated with a mortality of 91%.33 For an average INR of 4.4, the mortality was still 80%. More importantly, there was a sizeable subset of patients (56 of 77) with average INR of 4.4 who presented with a Glasgow Coma Scale (GCS) score between 13 and 15 with normal initial CT scans. They experienced deterioration of their GCS in only 10 hours after injury and had mortality rates of 84%.33 Due to the increased capacity of the subdural space in elderly patients, as many as 14% of patients on warfarin who suffer blunt head trauma will have significant intracranial hemorrhage but little or no clinical symptoms.34 Thus, at least one author recommends observation for 12-24 hours for any elderly patient with supratherapeutic INR after head trauma, even with a normal head CT.35
Subdural hematoma in elderly patients can be very challenging to detect on clinical symptoms alone, and misdiagnosis rates are as high as 40%.36 Presenting symptoms have been described as falling into three main categories.37 First, some patients with focal neurologic deficits, such as hemiparesis or aphasia, can mimic stroke. Second, others present with confusion, personality changes, memory loss, or impaired judgment, which are symptoms that can be associated with dementia and depression. Third, patients can present with signs of elevated intracranial pressure, such as headache, vomiting, vision changes, and altered consciousness. Those presenting with only subtle cognitive/personality changes usually experience the longest delay in diagnosis.
When first detected on CT scan, many elderly patients with subdural hematoma have evidence of both acute and chronic bleeding. Extra care should be taken when one is interpreting CT scans in the ED, as subacute subdural hemorrhage can appear isodense to the adjacent brain. (See Figure 1.) Secondary mass effects of ventricular displacement, cortical sulci effacement, or narrowing of white matter may be the only clues that subdural hemorrhage is present. Patients with bilateral isodense subdural hemorrhage can be even harder to recognize, as the asymmetric shift of ventricles or sulci effacement are absent.
Lastly, it is important to recognize the risk of occult cervical spine injury when discussing brain injury in the elderly population. Blunt trauma causes cervical spine injury in elderly patients nearly twice as often as in younger people.40 Injury to C-1 and C-2 is especially common, with odontoid fractures representing nearly 20% of geriatric fractures in a study of nearly 3000 elderly patients.40 Thus, some centers recommend CT scan of at least the upper cervical spine in elderly patients with head trauma regardless of the indication for cervical spine imaging.27 (See Table 2.)
Table 2: Critical Points for Elderly Patients with Head Trauma33,37-39
- Head injury is the most common result of falls in elderly more common than hip fracture.
- Subdural hematoma is seen twice as often in elderly compared to younger patients.
- From 25-50% of those with chronic subdural hematoma do not recall history of any trauma.
- Mild, generalized headache is seen in up to 90% of patients with chronic subdural hematoma.
- Symptoms of subdural hematoma in elderly patients easily mimic acute stroke or dementia/depression.
- Consider C spine imaging with CT scan, as C1 and C2 fractures are more common in elderly patients with head trauma.
- Patients with supratherapeutic INR (> 4.0) are at high risk for delayed intracranial bleeding.
Infection Without Fever. Elderly patients are at higher risk for infection because of several factors. Immune system function naturally declines with aging (immunosenescence). Co-morbid conditions common in the elderly, such as diabetes, end-stage-renal disease, and vascular disease, decrease resistance to infection. Poor nutritional status, thinning skin, stasis ulcers, reduced cough reflex, nursing home living, and decreased mobility are just a few of the other factors that can increase the elderly person's risk of infection.
While elderly patients often present to the ED with active infections, unfortunately these infections frequently are difficult to diagnose compared to younger patients with the same conditions. Ultimately, one of the most important features of the atypical presentation of elderly patients with infection is the absence of fever.
Fever is a classic hallmark of infection regardless of the patient's age. In contrast to younger patients, the presence of fever in elderly patients is much more likely to be associated with significant bacterial infection rather than viral sources. It has been estimated that the source of fever in elderly ED patients is viral in only 5% of cases.41
While fever is defined as 38.0° C (100.5° F) in young patients, the definition of fever in elderly patients has been suggested as low as 37.2° C (98.9° F).42 Indeed, one study found that lowering the definition of a fever to 37.2° C (98.9° F) improved the sensitivity/specificity of detection of clinically significant bacterial infection to 83% and 89%, respectively.8
The question then arises, which infections are most likely to be present in geriatric patients who are presenting without fever? The answer is the same infections that are common in patients who present with fever. Respiratory sources are most common, causing up to 58% of cases, followed by urinary infections, which produce up to 34% of cases.43 Abdominal infections are the next most common, causing roughly 20%, followed by cellulitis, meningitis, endocarditis, and HIV.43
Pneumonia. Elderly patients with pneumonia are known to present without the typical signs and symptoms seen in younger patients, including fever, chills, productive cough, and shortness of breath. Instead, they often present with vague symptoms of delirium, worsening of chronic confusion, general weakness/lethargy, and increased falls.7 The oldest old (> 85 years) and nursing home residents often are the most likely to present atypically. (See Table 3.)
Table 3: Frequency in Percentage of Signs and Symptoms of Pneumonia in the Elderly*
|
Age of Patient in years |
||||
|
18-44 |
45-64 |
65-74 |
> 74 |
|
|
Symptom: |
||||
|
Cough |
90% |
71-84% |
66-80% |
46-84% |
|
Dyspnea |
75% |
52-72% |
32-71% |
23-66% |
|
Sputum |
64% |
62-79% |
63-65% |
42-64% |
|
Altered MS/delirium |
10% |
36% |
31-50% |
|
|
Pleuritic CP |
60% |
38-42% |
2-80% |
13-84% |
|
Weakness |
93% |
45-93% |
49-88% |
52-84% |
|
Physical Sign: |
||||
|
Fever |
85% |
75% |
60% |
53% |
|
Tachypnea |
36% |
44% |
68% |
65% |
|
Tachycardia |
41% |
43% |
40% |
37% |
|
* All numbers are in percentage of the total study population, total sample size 1960 patients with pneumonia. Missing numbers represent data not available from studies. |
||||
Data in Table 3 does show that altered mental status/delirium becomes a dominant feature in the oldest old and that this same group lacks fever in almost 50% of cases. Another key point was made by Metlay et al. in their large study of elders with community-acquired pneumonia. Even though the elder patients appeared less affected, their mortality was just as high as those with classic symptoms.
Thus the bottom line, unfortunately, is that clinical judgment can be of little help in diagnosing these patients. One must liberally screen for pneumonia in older ED patients when they present with vague, nonspecific complaints. High-risk patients may be more easily detected, but efforts to find criteria identifying subgroups of elderly patients who can forgo chest radiography have been unsuccessful.44
Implications of Atypical Presentations of Pneumonia on Pay for Performance. The fact that elderly patients with pneumonia can be hard for the ED physician to identify has implications in terms of recent pay-for-performance Medicare initiatives based on core measures of quality care. Two performance measures for CAP specifically affect ED patients: CAP patients are to have blood cultures drawn before they are given antibiotics, and their initial antibiotic dose for pneumonia should be given within 6 hours of hospital arrival.45
These recommendations are based on research that showed improved outcomes when antibiotics were given in fewer than 6 hours, although the improvements were modest. In contrast, the use of blood cultures has not been shown to be beneficial in the treatment of ED patients.46
Concern has grown that patients who ultimately don't need antibiotics may receive them as ED physicians rush to meet time windows for treatment. Indeed, one study found 28.5% of patients admitted for CAP were given antibiotics without any radiographic abnormalities.47 Nearly 40% of these patients did not have CAP as a diagnosis on discharge from the hospital, and most did not have any diagnosed infection.47 The risks of increased costs, increased antibiotic resistance, and potential side effects in this setting are clear. In addition, pressure is building to reduce antibiotic use in patients with viral respiratory infections,48 resulting in a "damned if you do, damned if you don't" feeling among some ED physicians.
Ultimately, one should remember several points. First, elderly patients with fever are more likely to have a bacterial infection, with some estimating a viral source is present in only 5% of cases.41 Second, patients who are critically ill with an infection clearly benefit from early administration of antibiotics, and this needs to be done before the patient leaves the ED. Third, elderly patients with pneumonia are much more likely to present atypically, and altered mental status is the most likely confounding factor in these patients.49 Fourth, this will naturally lead to elderly patients who present atypically being most at risk to "slip under the radar" for CMS core measures. Thus, with current performance measures in place, one may need to liberally treat elderly patients with fever or altered mental status with antibiotics, even if the diagnosis of pneumonia is not yet clear.
Urinary Tract Infection (UTI). UTIs are the second most common cause of infections leading to hospitalization in those over 65 years, behind only pneumonia.50 Mortality is relatively high as well, with an in-hospital mortality of 6% on average for patients admitted from the ED with UTI.51 There are multiple reasons why UTI frequency increases with age. Even in healthy adults, asymptomatic bacteriuria rates rise with age. While bacteriuria is relatively rare in men younger than 65 years, rates are between 5-10% for men older than 80 years and 20% in women older than 80 years.52 Nursing home patients have much higher rates of asymptomatic bacteriuria up to 40% of men and 50% of women.53 Other factors related to aging that increase risk of UTI include: co-morbid diseases (diabetes, renal calculi), anatomic changes that decrease bladder emptying (prostatic hypertrophy, bladder diverticuli, urethral strictures, neurogenic bladder, lax pelvic floor ligaments), strokes and Alzheimer's disease that remove voluntary control of voiding, and changes in vaginal flora after menopause that permit urethral colonization. In addition, people with chronic indwelling catheters are almost always bacteriuric. In fact, bacteriuria rates rise as high as 10% per day while catheters are in place, and a single in-out catheterization may result in bacteriuria in up to 20% of patients.54
While the majority of elderly patients will present with classic symptoms of frequency, urgency, dysuria, suprapubic pain, and incontinence, a large minority of geriatric patients do not present with fever or localizing symptoms. As many as 30% of elders with proven pyleonephritis do not present with fever.55 Studies also have shown that these patients can present instead with respiratory or gastrointestinal symptoms. Acute confusion was present in 11% of these patients.56 A study of 284 elderly ED patients found equal numbers presented with urinary symptoms as mental status change (26% each); only 17% had abnormal temperatures (> 37.7° C [99.9° F] or < 35° C [95° F]).51 However, 30% of these patients were tachycardic, providing at least some clue to illness.
Diagnosis of UTI relies on clinical symptoms as much as urinalysis in most patients. Unfortunately, when clinical symptoms are not present, laboratory tests alone are not as reliable as one would hope. Nitrate (bacteriuria) and leukocytes esterase tests (pyuria) correlate better with presence of UTI in young adults but not as much in geriatric patients. One study found that 50% of cultures were still positive in 1993 patients aged 11-70 years with both negative nitrite and leukocyte esterase tests.57 One explanation for these findings is that only E. coli are able to convert urinary nitrate to nitrite, and thus the absence of nitrites on the urine dipstick does not exclude the presence of other urinary pathogens.58
How, then, should one diagnose acute UTI in a patient with a chronic indwelling catheter if clinical findings are so variable and bacteriuria consistently present in these patients? Ultimately, consensus guidelines were published in 1991 by McGeer59 and remain the accepted criteria by several national organizations,60 including CMS.61 (See Table 4.) Two other key points are to remember that asymptomatic bacteriuria is not an indication for treatment and that one should replace existing catheters in patients before initiating antibiotic treatment.
Table 4: McGeer Consensus Guidelines for Treatment of Nursing Home Residents With or Without Indwelling Urinary Catheters59
Nursing Home WITHOUT Catheter:
(Any 3 of these 5)
- Fever ≥ 38°C (100.4°F)
- New or increased dysuria, frequency, or urgency
- New flank pain, suprapubic pain, or tenderness
- Change in character of urine
- Change or worsening mental status
Nursing Home WITH Catheter > 30 Days:**
(Any 2 of these 4)
- Fever ≥ 38°C (100.4°F)
- New flank pain, suprapubic pain, or tenderness
- Change in character of urine
- Change or worsening mental status
** Remember to replace chronic catheters before starting antibiotics for UTI/pyelonephritis.
Bacterial Meningitis. Bacterial meningitis can strike any patient, but the immunocompromised and those at extremes of age are at highest risk. When an older adult is diagnosed with meningitis, it is much more likely to be bacterial in origin than viral.62 S. pneumoniae is still the most common pathogen, but in older adults, gram-negative species (E. coli, Klebsiella pneumoniae) are much more common than in young adults. These gram-negative bacteria likely can be traced to hematogenous spread from urinary tract or abdominal infections. In addition, concomitant pneumonia was found more commonly in older patients (50 years or older) with bacterial meningitis in one study: 41% vs. only 6% of younger patients (15-49 years).62
As with other infections, geriatric meningitis can present atypically, and classic signs of fever, headache, nuchal rigidity, or altered mental status can be blunted or absent. A study of 138 adults with meningitis found only 59% of elderly patients with bacterial meningitis had fever.62 Two larger series on hundreds of adults with bacterial meningitis both found that the classic triad of fever, nuchal rigidity, and altered mental status was present in only 44%63 to 66% of cases.64
One meta-analysis of 11 studies and 845 patients concluded that the absence of all three signs (fever, stiff neck, and changed mental status) effectively rules out the diagnosis of meningitis.65 Note, however, that these same authors state that "high-risk patients" still will need lumbar puncture to rule out meningitis, "given the seriousness of this infection."65 One could argue that elderly patients with moderate dementia may be high risk, as it may not be possible to identify mental status changes as easily as in young, healthy patients.
CT Scan Before LP in Older Patients? Physicians have been concerned that lumbar puncture (LP) could precipitate herniation in patients with intracranial mass lesions nearly since the first LP was performed in the early 1900s.66 Early reports exist of patients who deteriorated and died soon after LP was performed, but actual cause and effect in these situations is difficult to determine.67 Multiple papers have addressed this issue over the years, but no definitive conclusions have been reached. Much indirect evidence exists that in most cases little potential for harm exists. For example, one review of 200 cases where LP was performed in patients with known increased intracranial pressure found no complications.68 In addition, the majority of patients with bacterial meningitis have elevated opening pressures when measured (> 400 mm of water in 39% of 216 patients), but no connection with complications can be found.63 Finally, using papilloedema as a marker of significantly increased intracranial pressure shows that a small minority (2-4%) of patients have this problem.64
While older patients may be more likely to have intracranial mass lesions than younger adults, it appears from retrospective analysis of the data available that the risk for complications from LP is very small. Several authors have produced recommendations on which patients should undergo CT scan before LP. LP without CT scan in suspected meningitis is considered safe unless any of the following findings are present: new-onset seizures, immunocompromised state, signs of space-occupying lesions (papilloedema or focal neurologic deficit, excluding cranial nerve palsy), rapid decline in mental status, or moderate/severe mental status changes.67,69
A very important point to remember when CT scan is performed before LP in patients with suspected bacterial meningitis is to start antibiotic therapy before the patient undergoes CT scanning. While it is debatable if an LP can trigger herniation in these patients, no debate exists on the fact that delay of antibiotics is associated with a worse outcome.69 One study found this effect is most noticeable in patients who are in the later stages of disease, as evidenced by seizures, hypotension, or altered mental status where delay of antibiotics was defined as more than 6 hours after ED arrival.70
In general, one also should not be concerned about changing the results of the LP if it is done soon after antibiotics have been given. Reviews have shown that LP results are not affected if performed within 1-2 hours of initiation of antibiotics.71 Finally, it is important to realize that past reviews of large numbers of patients have found that most physicians do not initiate antibiotics before sending the patient to CT scan.
Indications for Steroids in Bacterial Meningitis? The question of whether to use adjunctive treatment with steroids (dexamethasone) in bacterial meningitis has been debated since they showed promise in animal models of the disease. While initial data suggested reduced mortality and complications (hearing loss),71 later studies did not show an unequivocal answer. Some authors suggested only using steroids in cases of H. influenzae meningitis,69 while other meta-analyses recommended using steroids in all adults.72 The 2004 IDSA guidelines for treatment of bacterial meningitis recommend giving dexamethasone (0.15 mg/kg) 10-20 minutes before the first antibiotic dose in children and adults, and continuing doses every 6 hours for 2-4 days.73 A prospective, randomized, double-blind study of 301 patients in 2002 found rather striking effects.74 This study found steroids decreased risk of unfavorable outcome from 25% to 15% in all patients and reduced mortality from 15% to 7%.74
While it would appear from the above data that a consensus is forming for use of steroids in bacterial meningitis, the most recent meta-analysis of 2029 patients from five randomized, double-blind, placebo-controlled studies found no beneficial effect of steroid use.75 This meta-analysis was performed by the same group that has published many of the previous studies, but they concluded that benefits of steroids in bacterial meningitis remain unproven and do not support routine use.75 While this may leave physicians unsure of the proper course to take today, it is comforting to know that use of dexamethasone in meningitis patients has been associated with very few side effects. (See Table 5.)
Table 5: Key Points in Evaluation of Elderly Patients with Suspected Meningitis63,64,67,69,71,73,75
- Bacterial meningitis is now primarily a disease of adults as a result of H. influenzae vaccination.
- Large reviews find that the classic triad of fever, nuchal rigidity, and altered mental status are present in 44-66% of cases. These same studies find that at least one of these features is present in nearly all cases.
- Kernig and Brudzinski signs are unreliable in elder patients due to underlying arthritis.
- CT scan before LP is not indicated unless one of the following is present:
- New-onset seizures
- Immunocompromised state
- Signs of space-occupying lesions: papilloedema or focal neurologic deficit, NOT including cranial nerve palsy
- Rapid decline in mental status or moderate/severe mental status changes
- Presence of papilloedema
- Remember to begin antibiotic treatment BEFORE sending patient to CT scan.
- Delay of 1-2 hours in obtaining LP results after antibiotics have been given will not change LP findings.
- Adjunctive dexamethasone treatment before antibiotics remains controversial.
The majority of randomized, double-blind, placebo-controlled trials show beneficial effects and recommend their use in adults. However, the most recent meta-analysis of these studies show, NO benefit and does not recommend their routine use.
References
1. Duthie EH. General issues in geriatric practice. In: Duthie EH, Katz PR, Malaone ML, eds. Practice of Geriatrics, 4th ed. Philadelphia, PA: Saunders Elsevier; 2007: 3-4.
2. Wan H, Sengupta M, Velkoff VA, et al. U.S. Census Bureau, Current Population Reports, 65+ in the United States: 2005, U.S. Government Printing Office, Washington, DC, Dec 2005: P23-209.
3. Wiener JM, Tilly J. Population aging in the U.S.: Implications for public programmes. Int J Epidemiol 2002;4:776–781.
4. Batchelor WB, Jollis JG, Friesinger GC. The challenge of health care delivery to the elderly patient with cardiovascular disease. Cardiol Clinics 1999;17:1–15.
5. Aminzadeh F, Dalziel WB. Older adults in the emergency department: A systematic review of patterns of use, adverse outcomes, and effectiveness of interventions. Ann Emerg Med 2002;39:238–247.
6. Pitts SR, Niska RW, Xu J, et al. National Hospital Ambulatory Medical Care Survey: 2006 emergency department summary. National health statistics reports, Hyattsville, MD: National Center for Health Statistics 2008;7:1–39.
7. Htwe TH, Mushtag A, Robinson SB, et al. Infection in the elderly. Infect Dis Clin North Am 2007;21:711–743.
8. Castle SC, Norman DC, Yeh M, et al. Fever response in elderly nursing home residents: Are the older truly colder? J Am Geriatr Soc 1991;39:853–857.
9. McCleane G. Pain perception in the elderly patient. Clin Geriatr Med 2008;24:203–211.
10. Wroblewski M, Mikulowski P. Peritonitis in geriatric patients. Age Ageing 1991;20: 90–94.
11. Canto J, Fincher C, Kiefe C, et al. Atypical presentations among Medicare beneficiaries with unstable angina pectoris. Am J Cardiol 2003;91:118–119.
12. Muller RT, Gould LA, Betzu R, et al. Painless myocardial infarction in the elderly. Am Heart J 1990;119:202–204.
13. Harper PL, Theakston E, J Ahmed, et al. D-dimer concentration increases with age reducing the clinical value of the D-dimer assay in the elderly. Intern Med J 2007;37:607–613.
14. Castle SC. Clinical relevance of age-related immune dysfunction. Clin Infect Dis 2000;31:578–585.
15. Inouye SK. The dilemma of delirium: Clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med 1994;97:278–288.
16. Elie M, Rousseau F, Cole M, et al. Prevalence and detection of delirium in elderly emergency department patients. CMAJ 2000;163:977–981.
17. Hustey FM, Meldon SW. The prevalence and documentation of impaired mental status in elderly emergency department patients. Ann Emerg Med 2002;39:248–253.
18. Rudolph JL, Marcantonio ER. Delirium. In: Duthie EH, Katz PR, Malaone ML, eds. Practice of Geriatrics, 4th ed. Philadelphia, PA: Saunders Elsevier; 2007: 279–285.
19. Wilber ST. Altered mental status in older emergency department patients. Emerg Med Clin North Am 2006;24:299–316.
20. American Psychiatric Association. Practice guideline for the treatment of patients with delirium. Am J Psychiatry 1999;156:1–20.
21. Lipowski ZJ. Delirium in the elderly patient. N Engl J Med 1989;320:578–582.
22. Finnell JT, McMicken DB. The alcoholic and substance abuse patient: Alcohol-Related Disease. In: Marx, JA ed. Rosen's Emergency Medicine: Concepts and Clinical Practice, 7th ed. St. Louis: Mosby;2009:2858–2881.
23. Hohl CM, Dankoff J, Colacone A, et al. Polypharmacy, adverse drug-related events, and potential adverse drug interaction in elderly patients presenting to an emergency department. Ann Emerg Med 2001;38:666–671.
24. Blanda MP. Pharmacologic issues in geriatric emergency medicine. Emerg Med Clin North Am 2006;24:449–465.
25. Spaite DW, Criss EA, Valenzuela TD, et al. Geriatric injury: An analysis of pre-hospital demographics, mechanisms, and patterns. Ann Emerg Med 1990;19:1418–1421.
26. Sterling DA, O'Connor JA, Bonadies J. Geriatric falls: Injury severity is high and disproportionate to mechanism. J Trauma 2001;50:116–119
27. Aschkenasy MT, Rothenhous TC. Trauma and falls in the elderly. Emerg Med Clin NorthAm 2006;24:413–432.
28. O'Brien DF, Basu S, O'Donnell JR, et al. The impact of aspirin therapy and anticoagulation on the prevalence of spontaneous subdural heamatoma. Ir Med J 2000;93:244–246.
29. Man-Son-Hing M, Nichol G, Lau A, et al. Choosing antithrombotic therapy for elderly patients with atrial fibrillation who are at risk for falls. Arch Intern Med 1999;159:677–685.
30. Karnath B. Subdural hematoma: Presentation and management in older adults. Geriatrics 2004;59:18–23.
31. Shireman TI, Mahnken JD, Howard PA, et al. Development of a contemporary bleeding risk model for elderly warfarin recipients. Chest 2006;130:1390–1396.
32. The Stroke Prevention in Reversible Ischemia Trial (SPIRIT) Study Group. A randomized trial of anticoagulants versus aspirin after cerebral ischemia of presumed arterial origin. Ann Neurol 1997;42:857–865.
33. Cohen DB, Rinker C, Wilberger JE. Traumatic brain injury in anticoagulated patients. J Trauma 2006;60:553–557.
34. Li J, Brown J, Levine M. Mild head injury, anticoagulants, and risk of intracranial injury. Lancet 2001;357:771–772.
35. Itshayek E, Rosenthal G, Fraifield S, et al. Delayed posttraumatic acute subdural hematoma in elderly patients on anticoagulation. Neurosurg 2006;58:E851–E856.
36. Kostanian V, Choi JC, Liker MA, et al. Computed tomographic characteristics of chronic subdural hematomas. Neurosurg Clin North Am 2000;11:479–489.
37. Iantosca MR, Simon RH. Chronic subdural hematoma in adult and elderly patients. Neurosurg Clin North Am 2000;11:447–454.
38. Walker RA, Wadman MC. Headache in the elderly. Clin Geriatr Med 2007;23:291–305.
39. Mayer SA, Brun NC, Begtrup K, et al. Recombinant activated factor VII for acute intracerebral hemorrhage. N Engl J Med 2005;352:777–785.
40. Touger M, Gennis P, Nathanson N, et al. Validity of a decision rule to reduce cervical spine radiography in elderly patients with blunt trauma. Ann Emerg Med 2002;40:287–293.
41. Marco CA, Schoenfeld CN, Hansen KN, et al. Fever in geriatric emergency patients: clinical features associated with serious illness. Ann Emerg Med 1995;26:18–24.
42. Norman DC. Fever in the elderly. Clin Infect Dis 2000;31:148–151.
43. Woolard RH, Becker B, Haronian TJ. Geriatric considerations. In: Harwood-Nuss A, Wolfson AB, eds. The Clinical Practice of Emergency Medicine, 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2001;1769–1777.
44. Mehr DR, Binder EF, Kruse RL, et al. Clinical findings associated with radiographic pneumonia in nursing home residents. J Fam Pract 2001;50:931–937.
45. A comprehensive review of development and testing for national implementation of hospital core measures. The Joint Commission web site. Available at: http://www.jointcommission.org/NR/rdonlyres/48DFC95A-9C05-4A44-AB05-1769D5253014/0/acomprehensiveReviewofDevelopmentforCoreMeasures.pdf
46. Kennedy M, Bates DW, Wright SB, et al. Do emergency department blood cultures change practice in patients with pneumonia? Ann Emerg Med 2005;46:393–400.
47. Kanwar M, Brar N, Khatib R, et al. Misdiagnosis of community-acquired pneumonia and inappropriate utilization of antibiotics. Side effects of the 4-h antibiotic administration rule. Chest 2007;131:1965–1869.
48. Pines JM, Hollander JE, Datner EM, et al. Pay for performance for antibiotic timing in pneumonia: caveat emptor. J Comm J Qual Patient Saf 2006;32:531–535.
49. Waterer GW, Kessler LA, Wunderink RG. Delayed administration of antibiotics and atypical presentation in community-acquired pneumonia. Chest 2006;130:11–15.
50. Curns AT, Holman RC, Sejvar JJ, et al. Infectious disease hospitalizations among older adults in the United States from 1990 through 2002. Arch Intern Med 2005;165:2514–2520.
51. Ginde AA, Rhee SH, Katz ED. Predictors of outcome in geriatric patients with urinary tract infections. J Emerg Med 2004;27:101–108.
52. Hedin K, Peterson C, Wideback K, et al. Asymptomatic bacteriuria in a population of elderly in municipal; institutional care. Scand J Prim Health Care 2002;20:166–169.
53. Rodhe N, Lofgren S, Matussek A, et al. Asymptomatic bacteriuria in the elderly: high prevalence and high turnover of strains. Scand J Infect Dis 2008;40:804–810.
54. Nicolle LE. Catheter-related urinary tract infection. Drugs Aging 2005;22:627–639.
55. Ramakrishnan K, Scheid DC. Diagnosis and management of acute pyelonephritis in adults. Am Fam Physician 2005;71:933–942.
56. Barkham TMS, Martin FC, Eykyn SJ. Delay in the diagnosis of bacteraemic urinary tract infection in elderly patients. Age Ageing 1996;25:130–132.
57. Nys S, van Merode T, Bartelds AI, et al. Urinary tract infections in general practice patients: Diagnostic tests versus bacteriological culture. J Antimicrob Chemother 2006;57:955–958.
58. Norris DL, Young JD. Urinary tract infections: Diagnosis and management in the emergency department. Emerg Med Clin North Am 2008;26:413–430.
59. McGeer A, Campbell B, Emori TG, et al. Definitions of infection for surveillance in long term care facilities. Am J Infect Control 1991;19:1–7.
60. Loeb M, Bentley DW, Bradely S, et al. Development of minimum criteria for the initiation of antibiotics in residents of long-term-care facilities: Results of a consensus conference. Infec Control Hosp Epidemiol 2001;22:120–124.
61. Centers for Medicare and Medicaid (CMS) Manual System, State Operations Manual. Appendix PP. Section 483.25(d) vol; 2005:183–184.
62. Gorse GJ, Thrupp LD, Nudelman KL, et al. Bacterial meningitis in the elderly. Arch Intern Med 1984;144:1603–1607.
63. Van de Beek D, de Gans J, Spanjaard L, et al. Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med 2004;351:1849–1859.
64. Durand ML, Calderwood SB, Weber DJ, et al. Acute bacterial meningitis in adults A review of 493 episodes. N Engl J Med 1993;328:21–28.
65. Attia J, Hatala R, Cook DJ, et al. Does this adult patient have acute meningitis? JAMA 1999;282:175–181.
66. Cushing H. Some aspects of pathological physiology of intracranial tumors. Boston Med Surg J 1909;141:71.
67. Fitch MT, Abrahamian FM, Moran GJ, et al. Emergency department management of meningitis and encephalitis. Infect Dis Clin North Am 2008;22:33–52.
68. Masson CB. The dangers of diagnostic lumbar puncture in increased intracranial pressure due to brain tumor, with a review of 200 cases in which lumbar puncture was done. Res Nerv Ment Dis Proc 1927;8:422–426.
69. Choi C. Bacterial meningitis in aging adults. Clin Infect Dis 2001;33:1380–1385.
70. Aronin SI, Peduzzi P, Quagliarello VJ. Community-acquired bacterial meningitis: risk stratification for adverse clinical outcome and effect of antibiotic timing. Ann Intern Med 1998;129:862–869.
71. Quagliarello VJ, Scheld WM. Treatment of bacterial meningitis. N Engl J Med 1997;336:708–716.
72. Fitch MT, van de Beek D. Emergency diagnosis and treatment of adult meningitis. Lancet Infect Dis 2007;7:191-200.
73. Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 2004;39:1267–1284.
74. de Gans J, van de Beek D. Dexamethasone in adults with bacterial meningitis. N Engl J Med 2002;347:1549-1556.
75. Van de Beek D, Farrar JJ, de Gans J, et al. Adjunctive dexamethasone in bacterial meningitis: A meta-analysis of individual patient data. Lancet Neurol 2010;9:254–263.
It is another busy night in the emergency department (ED), but you are managing to keep up with the tempo. However, the next chart in the rack is bound to slow your pace: It is an 83-year-old man with the chief complaint of "not feeling well."Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
