Special Feature: Tranexamic Acid: Nonhormonal Treatment for Heavy Menstrual Bleeding
Special Feature
Tranexamic Acid: Nonhormonal Treatment for Heavy Menstrual Bleeding
Without fanfare, the FDA approved oral tranexamic acid tablets (Lysteda), the first non-hormonal product cleared to treat heavy menstrual bleeding in the United States. Tranexamic acid (TA) was initially approved by the FDA in 1986 as an injection (Cyklokapron®) to reduce or prevent bleeding during and following tooth extraction in patients with hemophilia.
Tranexamic acid [4-(aminomethyl)cyclohexane-1-carboxylic acid] is an antifibrinolytic that competitively inhibits activation of plasminogen, thereby reducing conversion of plasminogen to plasmin (fibrinolysin), an enzyme that degrades fibrin clots, fibrinogen, and other plasma proteins, including the procoagulant factors V and VIII. Although tranexamic acid also directly inhibits plasmin activity, this requires higher doses than are needed to reduce plasmin formation. While clinicians in the United Kingdom, Europe, and many other countries have had oral forms of TA available for the indication of heavy menstrual bleeding for many years, gynecologists in the United States have no direct experience with this drug. Since this novel and interesting new treatment option is now available for your patients, I thought a short review could provide context for its use.
Do women with HMB require treatment?
The terms used to describe abnormal uterine bleeding are poorly defined. To improve communication, research, and patient care, a consensus group of leading academic experts led by Ian Fraser of the University of Sydney has proposed eliminating the terms menorrhagia, metrorrhagia, and dysfunctional uterine bleeding, and replacing these with clear descriptive terminology easily translated into any language.1 The term heavy menstrual bleeding (HMB) is now the preferred term for excessive bleeding. The normal volume of flow is defined as measured menstrual blood loss of 5-80 mL. The 80 mL threshold comes from detailed studies that determined that women become anemic when blood loss exceeds this amount.2 While the 80 mL definition makes sense for research, it offers little guidance for clinicians. Not all women that complain of HMB will become anemic and a woman's perception of her own menstrual loss is the key determinant in her presentation to the clinic for evaluation and therapy, a position endorsed by the National Institute for Clinical Excellence (NICE) in the United Kingdom.3
That said, menstrual disorders account for more than 12% of total gynecologic emergency room visits4 and estimates of the economic costs due to lost productivity as a result of HMB vary between $1692 and $3600 per patient per year.5,6 Compared to women with normal or light bleeding, women with HMB are significantly less likely to be employed6 and more likely to use health care services.7 About 17% of the roughly 600,000 hysterectomies performed annually in the United States are done to manage menstrual disorders.8,9 While surgical options such as hysterectomy and endometrial ablation are effective treatments for heavy menstrual bleeding, both approaches have significant risks and costs, and health authorities in New Zealand and the United Kingdom have recommended that medical therapies be considered before surgery.
How effective are hormonal therapies for HMB?
Hormonal therapies are useful in alleviating menstrual symptoms in women with excessively heavy or symptomatic menstrual periods. Progestogen-only methods can be used in women with contraindications to estrogen therapy. Until the October 2009 approval of the LNG IUS in the United States, the only approved therapies for "abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology" had been cyclic oral medroxy progesterone acetate (MPA) and norethindrone (NET), but neither of these is indicated for contraception. A Cochrane review found that no randomized placebo-controlled trials of oral progestogens for HMB have been performed.10 The LNG IUS provides a high local level of levonorgestrel directly to the endometrium. While bleeding is unpredictable during the early months of use, it is typically light and improves such that by 6 months most users report either light regular cycles or amenorrhea. A number of observational11,12 and randomized13-15 studies have demonstrated the superiority of LNG IUS to all other medical options (see Figure, below), with an effect comparable to that achieved with endometrial ablation.5,16,17
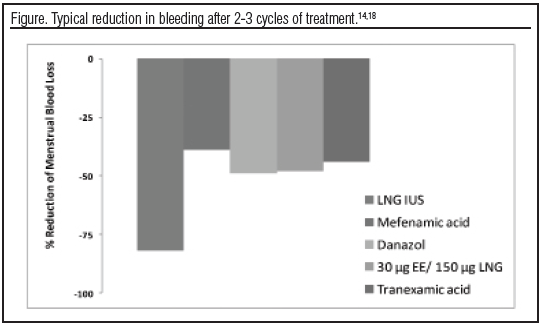
Combined oral contraceptives (COC) reduce the duration and intensity of menstrual bleeding, and are widely used to manage abnormal menstruation despite few objective data and no labeling indications to support this practice. Fraser and McCarron used a crossover design to study women with ovulatory HMB and found that the reduction in bleeding achieved with a COC (30 µg EE/150 µg LNG) was similar to that observed with danazol and the nonsteroidal agent mefenamic acid (approximately 30%-50%).18 New data presented at the 2009 meetings of the International Federation of Obstetrics and Gynecology19 and the American Society of Reproductive Medicine20 provided evidence that a novel extended oral contraceptive regimen containing estradiol valerate and dienogest (E2V/DNG) may be more effective than other COC, with a reduction in measured blood loss of more than 60%. The E2V/DNG pill has been approved in Europe for over a year, and was recently approved for contraception in the United States (Natazia, Bayer Healthcare). A second indication for the treatment of HMB is under review.
How effective are other medical therapies for HMB?
NSAIDs are widely available and easy to use, but not all will be equally effective. The randomized study by Fraser demonstrated that mefenamic acid reduced measured blood loss by up to 39%; this was not significantly different than the reduction seen with a COC (43%) and danazol (49%), but was better than naproxen (12%). Unfortunately, mefenamic acid is not available without a prescription, so cost is higher than with over-the-counter NSAIDs such as ibuprofen. Gastrointestinal tolerance also limits the acceptability of NSAIDs. Many women object to the androgenic side effects of danazol, and since this drug also carries a black box warning for DVTs, there is no real advantages to it over a COC.
What does tranexamic acid offer?
Tranexamic acid reduces menstrual blood loss by about one-third. The pivotal U.S. trials (one 3-cycle treatment and one 6-cycle treatment) that led to marketing approval are described in the package insert but are not published. HMB was defined as an average menstrual blood loss of ≥ 80 mL as assessed by the alkaline hematin method, so the results are comparable to existing literature including the recently completed LNG IUS and E2V/DNG OC studies. A dose of 3900 mg (two 650 mg tablets TID) resulted in a 39% reduction in mean menstrual blood loss from baseline. These results are similar to other published randomized studies with TA. While impressive, they fall short of the results observed in U.S. women treated with the LNG IUS (71%) and the E2V/DNG COC (62%).20
Who are good candidates for tranexamic acid?
Women with idiopathic heavy menstrual bleeding that would prefer not to use a hormonal therapy or NSAID will benefit from TA. One particular group will be women seeking pregnancy. I frequently see patients complaining of heavy bleeding after stopping hormonal contraceptive therapy in order to get pregnant. We have had little to offer these women except NSAIDs, iron, and good wishes for a speedy conception. Since TA is Category B for pregnancy and is used only during menstruation (well before ovulation and fertilization), even the most risk-averse preconception patients should find it acceptable.
Users of the copper IUD represent another group that could benefit from TA. A randomized study demonstrated this reduced bleeding by about 50%, but side effects were greater than with a NSAID.21
What are the side effects and contraindications?
Among the side effects reported during clinical trials of TA, only sinus and nasal symptoms (25% vs 17% placebo), back pain (21% vs 15%), and musculoskeletal pain (11% vs 3%) appear to be related to use of the drug (data from package insert).
Women with risk factors for thrombosis should not use TA due to an increased chance of arterial and venous clots. Therefore, any women with a medical condition that would represent a contraindication to hormonal therapy should probably not use TA. While use of TA in a woman already using a combined hormonal method is not listed as contraindication in the labeling, there is a specific warning that co-administration with a combined method may increase the risk of clot formation. Therefore, risks should be considered along with potential benefits. Caution should also be observed in women taking either Factor IX complex concentrates or anti-inhibitor coagulant concentrates, or oral tretinoin as the risk of clots may increase. Tretinoin (a treatment for acute promyelocytic leukemia) should not be confused with isotretinoin, the treatment for acne.
Many women with HMB also suffer from dysmenorrhea. TA will not improve these symptoms, but NSAIDs can be safely co-administered.
When will tranexamic acid be available to clinicians, and how should it be prescribed?
Tranexamic acid will be marketed under the brand name of Lysteda (Ferring Pharmaceuticals). While Ferring is well known as a leading supplier of gonadotropins for use in infertility, this will be its first product for use in general gynecology patients, so a large sales force is not currently in place. The product was introduced at the 2010 ACOG meeting in San Francisco, and should be available from pharmacies.
TA is supplied as a 650 mg tablet. The recommended dose of TA is two tablets (1300 mg) three times a day (3900 mg/day) for a maximum of 5 days during monthly menstruation. Dose adjustment is needed in women with renal failure.
Bottom line
Oral tranexamic acid provides an important new nonhormonal option to manage idiopathic heavy menstrual bleeding. Clinicians in the United States need to recognize that this therapy has been available around the world for a long time and is not a panacea. Similar to hormonal therapy, TA should not be used in women at high risk for thrombosis. The reduction in bleeding is similar to that observed with traditional oral contraceptives and NSAIDs, but less than that reported with the LNG IUS or the new E2V/DNG pill. Despite these limitations, TA presents an excellent new treatment choice for women with HMB preferring to manage symptoms episodically and in those seeking pregnancy or simply wishing to avoid hormonal therapy. I expect the drug will have a meaningful impact in the care of our patients.
References
- Fraser IS, et al. A process designed to lead to international agreement on terminologies and definitions used to describe abnormalities of menstrual bleeding. Fertil Steril 2007;87:466-476.
- Hallberg L, et al. Menstrual blood loss and iron deficiency. Acta Med Scand 1966;180:639-650.
- National Collaborating Centre for Women's and Children's Health. Heavy menstrual bleeding Commissioned by the National Institute for Health and Clinical Excellence. Available at: www.nice.org.uk/nicemedia/live/11002/30404/30404.pdf. Accessed July 23, 2010.
- Curtis KM, et al. Visits to emergency departments for gynecologic disorders in the United States, 1992-1994. Obstet Gynecol 1998;91:1007-1012.
- Hurskainen R, et al. Clinical outcomes and costs with the levonorgestrel-releasing intrauterine system or hysterectomy for treatment of menorrhagia: Randomized trial 5-year follow-up. JAMA 2004;291:1456-1463.
- Cote I, et al. Work loss associated with increased menstrual loss in the United States. Obstet Gynecol 2002;100:683-687.
- Cote I, et al. Use of health services associated with increased menstrual loss in the United States. Am J Obstet Gynecol 2003;188:343-348.
- Wu JM, et al. Hysterectomy rates in the United States, 2003. Obstet Gynecol 2007;110:1091-1095.
- Whiteman MK, et al. Inpatient hysterectomy surveillance in the United States, 2000-2004. Am J Obstet Gynecol 2008;198:34.e1-7.
- Lethaby A, et al. Cyclical progestogens for heavy menstrual bleeding. Cochrane Database Syst Rev 2008;(1):CD001016.
- Andersson JK, Rybo G. Levonorgestrel-releasing intrauterine device in the treatment of menorrhagia. Br J Obstet Gynaecol 1990;97:690-694.
- Xiao B, et al. Therapeutic effects of the levonorgestrel-releasing intrauterine system in the treatment of idiopathic menorrhagia. Fertil Steril 2003;79:963-969.
- Reid PC, Virtanen-Kari S. Randomised comparative trial of the levonorgestrel intrauterine system and mefenamic acid for the treatment of idiopathic menorrhagia: A multiple analysis using total menstrual fluid loss, menstrual blood loss and pictorial blood loss assessment charts. BJOG 2005;112:1121-1125.
- Milsom I, et al. A comparison of flurbiprofen, tranexamic acid, and a levonorgestrel-releasing intrauterine contraceptive device in the treatment of idiopathic menorrhagia. Am J Obstet Gynecol 1991;164:879-883.
- Endrikat J, et al. A Canadian, multicentre study comparing the efficacy of a levonorgestrel-releasing intrauterine system to an oral contraceptive in women with idiopathic menorrhagia. J Obstet Gynaecol Can 2009;31:340-347.
- Hurskainen R, et al. Quality of life and cost-effectiveness of levonorgestrel-releasing intrauterine system versus hysterectomy for treatment of menorrhagia: A randomised trial. Lancet 2001;357:273-277.
- Kaunitz AM, et al. The levonorgestrel-releasing intrauterine system and endometrial ablation in heavy menstrual bleeding: A systematic review and meta-analysis. Obstet Gynecol 2009;113:1104-1116.
- Fraser IS, McCarron G. Randomized trial of 2 hormonal and 2 prostaglandin-inhibiting agents in women with a complaint of menorrhagia. Aust N Z J Obstet Gynaecol1991;31:66-70.
- Fraser IS, et al. A novel oral contraceptive comprising estradiol valerate/dienogest for the treatment of heavy and/or prolonged menstrual bleeding without organic cause: A double-blind, randomised, placebo-controlled trial. Int J Gynecol Obstet 2009;107:S183.
- Jensen J, et al. A multicenter, double-blind, randomized, placebo-controlled study of oral estradiol valerate/dienogest for the treatment of heavy and/or prolonged menstrual bleeding. Fertil Steril 2009;92:S32.
- Ylikorkala O, Viinikka L. Comparison between antifibrinolytic and antiprostaglandin treatment in the reduction of increased menstrual blood loss in women with intrauterine contraceptive devices. Br J Obstet Gynaecol 1983;90:78-83.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
