Current Approaches in the Diagnosis and Treatment of Peripheral Arterial Disease
Current Approaches in the Diagnosis and Treatment of Peripheral Arterial Disease
Author:
Sula Mazimba, MD, MPH, Cardiology Fellow, Kettering Medical Center, Kettering, OH.
Peer Reviewer:
Bruce H. Rank, DO, Cardiovascular Thoracic, Vascular, and Endovascular Surgery, Grandview Medical Center, Dayton, OH.
Introduction
Peripheral arterial disease (PAD) is a pathological condition characterized by atherosclerotic occlusion of arterial blood supply to the lower or upper extremities. In a broader sense, it may be defined as a clinical entity that includes a diverse group of disorders that lead to progressive occlusion and/or aneurysmal dilation of the aorta and the noncoronary arteries, including the carotid, upper-extremity, visceral, and lower-extremity arterial branches. From a historical perspective, peripheral vascular disease (PVD), on the other hand, has been used to include all the non-cardiac diseases that affect the circulation, including those conditions that affect not only the arterial tree but also the venous and lymphatic circulation as well. For the purpose of this review, PAD will refer to the arterial disease condition affecting the lower and upper extremities.
Patients with PAD have increased risk of developing cardiovascular and cerebrovascular diseases.1 Patients with PAD, even in the absence of a history of prior myocardial infarction or ischemic stroke, have similar relative risk of death from cardiovascular death as do patients with a history of coronary or cerebrovascular disease.2
Epidemiology
The prevalence of PAD rises with advancing age. In the National Health and Nutrition Examination Survey (NHANES), the prevalence of PAD was 0.9% between the ages of 40-49, 2.5% between the ages of 50-59, 4.7% between the ages of 60-69, and 14.5% older than age 70.3 The prevalence varies depending on the population studied as well as the type of diagnostic tool, and presence or absence of symptoms. The most common diagnostic tool used for epidemiological studies is the ankle/brachial index (ABI). The ABI is the ratio of the ankle to brachial systolic blood pressure. Using the ABI as the measure of disease, the prevalence of PAD ranges from 3.6% to 29% of the general population.3-5 There is gender variation in the prevalence of PAD, with men being affected more than women. There is also ethnic variation in the disease distribution of PAD. The prevalence of PAD is greater in blacks than in non-Hispanic whites.6 Blacks were 1.47 times more likely to develop PAD than non-Hispanic whites, while the odds for Asians of Chinese decent and Hispanics for developing the disease is less than 0.5.7
Patients with PAD usually present with intermittent claudication. Intermittent claudication is defined as a reproducible discomfort of a defined group of muscles that is induced by exercise and relieved with rest. Prevalence of intermittent claudication varies depending on the age and gender of the population studied. In general, prevalence of claudication ranges from 1 to 4.5% of population groups 40 years and older.8,9
Prevalence
Patients with PAD have been shown to have increased prevalence of coronary artery disease and cerebrovascular events.1
PAD is a growing problem in developed countries due to the increasing elderly population. The modifiable risk factors that are associated with the development of coronary artery disease (CAD) are similar to those responsible for the development of PAD. Tobacco use, diabetes, dyslipidemia, and hypertension are the main risk factors that contribute to the development of PAD.10 There is a two- to three-fold increase in risk of developing PAD in patients who smoke.10 About 90% of patients with intermittent claudication are either active smokers or have a smoking history.11 There is a dose-dependent correlation of smoking status and the risk of developing PAD in both male and female patients. It has also been shown that this risk of PAD decreases with smoking cessation.12 Patients with diabetes mellitus have been shown to be at an increased risk of developing PAD compared with their non diabetic counterparts.13 The risk for diabetic patients can be up to 4 times more than that of non-diabetic patients.14 In addition, diabetic patients tend to have worse arterial disease and worse outcomes than non-diabetic patients.15 The Framingham study demonstrated that the odds ratio of developing intermittent claudication was 2.6 for diabetes mellitus, and 1.2 for every 40 mg/dL elevation in the serum cholesterol concentration.10 Hypertriglyceridemia is positively correlated with the risk of developing PAD.16 Hypertension also has a positive correlation to developing PAD.17 There is evidence to suggest that inflammation plays a role in the development of PAD. Inflammation may be the link between the traditional risk factors for atherosclerosis and the pathological processes associated with PAD.18 Markers of inflammation such as C-reative protein (CRP) and fibrinogen leukocyte adhesion molecules correlate with the development of PAD and future complications.19,20 There are some emerging new risk factors that are also associated with PAD, such as hyperhomocysteinemia, which have been shown to confer increased risk of developing PAD.21 (See Table 1.)
Table 1: Risk of Developing PAD Estimated from Epidemiological Studies
|
Risk Factor |
Relative Risk |
|
Data from: Dormandy JA, Rutherford RB. Management of peripheral arterial disease (PAD). TASC Working Group. TransAtlantic Inter-Society Consensus (TASC). J Vasc Surg 2000;31 (1 Pt 2):S1-S196. |
|
|
Smoking |
3-4 |
|
Diabetes |
3-4 |
|
Hypercholesterolemia |
1-2 |
|
Hyperhomocysteinemia |
2-3 |
|
C-reactive protein |
2 |
Clinical Presentation
The majority of patients with PAD are asymptomatic. Approximately one-third of patients with PAD will present with the typical intermittent claudication symptoms. Intermittent claudication refers to pain or discomfort in the lower extremity precipitated by walking or exercise and relieved with rest.2 The location of the pain depends on the site of occlusion of the artery. The groups of muscles distal to the area of arterial stenosis manifest with ischemic symptoms. Gluteal, hip, or thigh claudication usually points to diseased aorta or iliac arteries. Calf claudication characterizes femoral or popliteal artery occlusion. In general, the most commonly affected muscle group is the gastrocnemius muscle. This is the one muscle that uses the greatest amount of oxygen during exercise. Upper-extremity muscles also can be affected. Subclavian artery disease affects the shoulders, axillary artery disease manifests with symptoms in the biceps, and brachial artery disease causes symptoms in the forearm. Patients with PAD are usually limited by these symptoms and will usually walk shorter distances. (See Table 2.)
Table 2: Classification of PAD Based on Symptoms
|
Stage |
Fontaine Classification of PAD |
|
I |
Asymptomatic |
|
II |
Intermittent claudication |
|
IIa |
Pain-free, claudication walking > 200 m |
|
IIb |
Pain-free, claudication walking < 200 m |
|
III |
Rest and nocturnal pain |
|
IV |
Necrosis and gangrene |
Intermittent claudication should be distinguished from other mimickers. Symptoms resembling claudication may result from non-atherosclerotic causes of arterial disease. (See Table 3.) Lumbosacral radiculopathy can cause pain in the buttock, hip, thigh, or calf with walking. Nerve pain is "electric" and shooting in nature and usually is relieved by sitting down or bending forward. The entire leg may be affected. Patients with chronic venous insufficiency may also report intermittent claudication with exertion. Venous pooling of blood during exercise leads to elevation of venous pressures that also causes an increase in the arterial resistance. This leads to diminished blood flow, leading to tissue ischemia.
Table 3: Differential Diagnosis of Intermittent Claudication
|
Characteristic |
Intermittent Claudication |
Venous Claudication |
Nerve Root Pain |
|
Quality |
Cramping |
Pressure-like |
Electric |
|
Onset |
Gradual, predictable |
Usually gradual |
Immediate, unpredictable |
|
Relieved by |
Rest |
Elevation of the leg |
Sitting down/bending |
|
Location |
Localized |
Entire leg |
Diffuse/whole leg |
|
Limbs affected |
Single limb |
Single leg |
Both legs |
Signs and Symptoms
Diagnosis of PAD is based on symptoms. The cardinal symptom of PAD is intermittent claudication. The history obtained from patients reporting claudication should note the walking distance, speed, and incline that precipitated claudication. This baseline information helps to determine progression or deterioration of symptoms. It is important to distinguish symptoms resulting from other causes that may mimic PAD. Limb pain at rest is suggestive of critical limb ischemia (CLI), a result of total arterial occlusion. This condition must be urgently recognized so that there is no delay in referring the patient for anticoagulation or revascularization therapies in order to salvage the limb. CLI can result from atherosclerosis or thrombo-embolic events. Other conditions that can cause CLI disease include vasospasms, thromboangiitis obliterans, mechanical occlusions of vascular conduits, and traumatic disruption of vessels. From the history, it is important to obtain information about the cluster of conditions that serve as risk factors.
The physical examination should include palpation of pulses and auscultation for bruits. Diminished pulses and bruits are strongly correlated with PAD.23 The presence of the bruit may indicate the site of arterial occlusion. Elicit for the integrity of the connective tissue and skin. PAD can lead to skin ulcers, muscle atrophy from immobility, brittle nails, and loss of hair on the skin.
Investigations
In patients with suspected PAD, evaluation of the presence and severity of the disease can be done by non-invasive modalities.
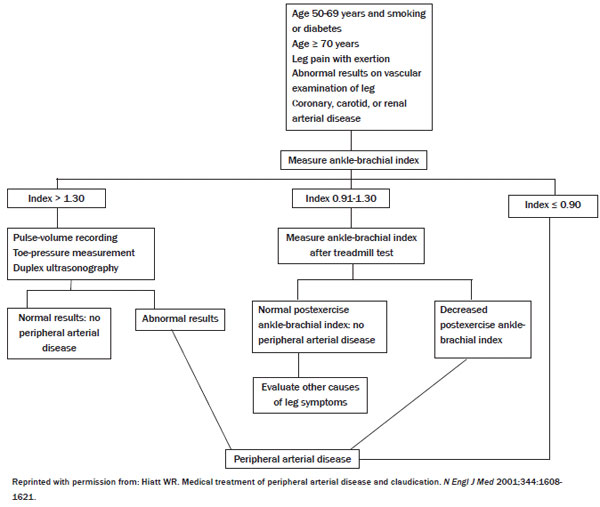
The initial screening tool for the presence of PAD is ABI measurements, which involve the measuring pressure using inflatable pneumatic cuffs to suprasystolic levels. The cuffs are then deflated to allow for reflow. Flow of blood distal to the cuffs in the legs is detected with a Doppler probe placed at the dorsalis pedis and posterior tibial arteries.24 The brachial blood pressure is measured in the usual manner by auscultation of Korotikof sounds. The ratio of the two measurements is taken to calculate the ABI. The highest of the four measurements in the lower extremity is divided into the higher of the two measurements in the upper extremity to obtain the ABI.
The normal ABI is between 1.0 and 1.3. Values above 1.3 indicate that the artery may be calcified and is therefore non-compressible. A value below 0.9 has 95% sensitivity and about 100% specificity for detecting angiogram-positive PAD.24 ABI also is used to assess the severity of disease. Most patients with intermittent claudication have ABI between 0.5 and 0.8. Values below 0.5 indicate severe critical limb ischemia and will present with resting ischemia.
In cases where the ABI is greater than 1.3 and the arteries are non-compressible, the specially designed toe cuffs can be used to measure the pressure in the toes and the toe-brachial index (TBI) can be calculated.
Treadmill Exercise ABI
Resting ABI may be normal, but this does not preclude presence of obstructive arterial disease. A stenosis of less than 70% does not result in significant flow gradient that would result in abnormal ABI. During exercise, however, there is a reduction in the peripheral resistance and an increase in blood flow, which further creates a pressure gradient across the stenotic segment. This causes a decrease in the ABI because the increase in the blood pressure in the legs is not matched by the increase in the arms. Exercise ABI is a sensitive measure for documenting PAD in subjects with normal resting ABIs.25
Measuring the volume of blood flow in the legs assesses disease severity. This is done through the use of segmental volume plethysmography. This done by using pressure pneumatic cuffs, applying some pressure to the segment of the blood vessel of interest, and then measuring the degree of flow distal to the blood vessel using an ultrasound Doppler probe that displays the contour and amplitude of the wave form.
Duplex Ultrasound Imaging
Ultrasound imaging provides a direct measurement of flow velocities of blood in the blood vessel. With the addition of color flow imaging, stenotic areas can be localized. Ultrasound has up to 90% sensitivity and about 95% specificity for detecting PAD.26
The other testing modalities include magnetic resonance angiography, computer tomography angiography (CTA), magnetic resonance imaging angiography (MRA), and the contrast angiography. MRA and CTA are used in assessing patients for endovascular and surgical intervention.
Contrast angiography is the "gold standard" test for evaluation of PAD. However, it is invasive and has attendant risk. It is still used in defining vascular anatomy, particularly in patients undergoing endovascular repair.
Clinical investigations should also be directed at assessing the global risk factors of the patient. These include assessing for hyperlipidemia, diabetes, hypertension, and renal function, and, in some cases, inflammatory markers such as C-reactive protein.
Management
Management of PAD involves aggressive risk factor and lifestyle modification, exercise rehabilitation, pharmacological therapy, and percutaneous intervention or surgical procedures. Initial treatment should include risk reduction, ASA, statins, exercise, and strict diabetic control. Early evaluation with a vascular/endovascular surgeon is key for those with positive ABI as well as those with lifestyle-limiting disease.
The goals of treatment of PAD include reduction in the cardiovascular morbidity and mortality, improvement in the quality of life by improving the level of functional status, and decreasing symptoms.
Risk Factor Modification
All patients with PAD should undergo aggressive risk factor and lifestyle modification. The principle factors for the development of PAD are smoking, diabetes, hypertension, and hyperlipidemia, which account for about 69% of patients.27 Quitting cigarette smoking reduces the progression of disease and lowers the incidence of rest ischemia among patients who quit.28 Smokers with PAD have a higher mortality rate from myocardial infarction than those who do not smoke. Additionally, PAD patients who quit smoking have twice the five-year survival as those PAD patients who continue to smoke.11 Lipid-lowering therapies with diet or pharmacotherapy reduce the progression of PAD.29 In one trial, artovastatin therapy improved pain-free walking distance by 60% compared to placebo.30 There are limited data on the effect of antidiabetic therapy on the natural history of PAD. However, aggressive control of blood sugar reduces the risk of microvascular complications.31 Hypertension control reduces the risk of cardiovascular and cerebrovascular events. There are still no data that address the role of antihypertensive medication on the progression of PAD. Hypertension should be treated to reduce cardiovascular and cerebrovascular events. Angiotensin-converting enzymes inhibitors (ACEIs) have been shown to reduce cardiovascular events in patients with atherosclerosis. In the Heart Outcome Prevention Evaluation, PAD patients treated with ramipril reduced cardiovascular events to levels comparable to those in patients without PAD.32 A target goal of treating hypertension for patients with PAD is less than 140/90 mm Hg for non-diabetics and less than 130/80 mm Hg for patients with diabetes.33
Exercise Rehabilitation
Supervised exercise rehabilitation programs have been shown to increase functional capacity in patients with PAD symptoms. In some studies, exercise programs increased the average distance walked to the onset of claudication by 180%.34 The greatest benefit comes when patients exercise for 30 minutes three times per week for 6 months. The exercises include an initial warming-up phase, which involves intermittent walking, which is graduated to a few minutes of increased activity. The benefits of the exercise program are thought to be due to formation of collateral blood supply, endothelium-related vasodilatation, muscle metabolism, and improvements in the efficiency of walking.35 It also has been suggested that exercise decreases red blood cell aggregation and improves the filterability of the blood.2
Pharmacotherapy
Besides pharmacological agents that are used to treat risk factors in patients with PAD, there are some pharmacological agents that are used primarily to reduce symptoms and slow the progression of the disease.
Antiplatelet therapy has been shown to reduce the risk of nonfatal myocardial infarction, ischemic stroke, and death from a vascular cause.36
Figure 2: Evaluation of Patients in Whom PAD Is Suspected
Aspirin is the most commonly prescribed drug in PAD. Aspirin has been associated with significant reduction in the risk of development of stroke and myocardial infarction.37 The combination of ASA and dipyridamole (Persantine) was found to increase pain-free walking distance and resting blood flow in patients with PAD.37 Clopidogrel (Plavix) was found to be more effective than aspirin in reducing the combined risk of ischemic stroke, myocardial infarction, or vascular death.38 Cilostazol (Pletal) is a phosphodiesterase inhibitor that can be used in patients with PAD with intermittent claudication. It has both antiplatelet and vasodilator properties. It was shown to be more effective than pentoxifylline in the treatment of intermittent claudication.39 Cilostazol, like other phosphodiesterase inhibitors, should not be used in patients with heart failure because of an increase in mortality in these patients. Pentoxifylline (Trental) is a rheological modifier used in the treatment of intermittent claudication. Pentoxifylline was found to increase walking distance by 29 meters compared to placebo, although the benefits are less than the benefits derived from a supervised exercise program.40 There are other classes of drugs that are being considered or are currently under investigation for the treatment of PAD. These include statins, ACE inhibitors, serotonin antagonists, calcium channel blockers, carnitine derivatives, vasodilator prostaglandins, and angiogenic growth factors. ACEIs have been shown to increase the claudication distance.41 A serotonin antagonist, such as naftidrofuryl, has been shown to be effective in reducing intermittent claudication.42,43 L-carnitine, which is a co-factor in fatty metabolism, has been shown to improve symptoms in PAD, particularly those patients who have markedly diminished walking distance.44 Prostaglandin administrations in patients with symptomatic PAD have not shown consistent results.45-47 Angiogenic growth factors increase collateral blood supply in animal models but have failed to show improvement in clinical trials.48 Immune modulators have recently been shown to be effective in the treatment of severely incapacitated patients with short walking distance claudication.49 Other therapies that have not been shown to be effective include estrogen replacement, ginkgo biloba50, vitamin E, and chelating therapy.51-53
Surgery/Percutaneous Intervention
Peripheral catheter-based interventions for PAD are indicated for patients with severe lifestyle limiting symptoms even after a trial of exercise rehabilitation or pharmacotherapy.33 Endovascular interventions are also indicated in patients with critical limb ischemia. Long-term success depends on the anatomy of the blood vessel involved. Larger caliber vessels have better long-term outcomes than smaller vessels (aortoiliac disease).
Surgical revascularization is indicated in patients with disabling claudication on maximal medical therapy who are not candidates for percutanous intervention. The surgical risk profile must be favorable and must not outweigh the benefits. Patients who benefit the most are those younger than age 70, non-diabetics, and those with little disease distal to the primary lesion.54 Early evaluation with a vascular/endovascular surgeon is important for those patients with positive ABIs as well as lifestyle-limiting disease.
Conclusion
Peripheral arterial disease is a prevalent condition that is associated with high morbidity and mortality. Unfortunately, PAD often is under-diagnosed and under-treated. Prevalence will continue to rise because of the increasing segment of the elderly population. Early recognition and diagnosis is key to early adoption of those evidence-based practices that have been shown to improve the quality of life of patients and reduce the adverse outcomes associated with the disease. Initial treatment should include risk reduction, ASA, statins, exercise, and strict diabetic control. Early evaluation with a vascular/endovascular surgeon is key for those with positive ABI as well as those with lifestyle-limiting disease.
The secondary prevention strategies such as aggressive risk factor modifications, exercise programs, and pharmacotherapy should be routinely tailored to patients with PAD, just as much as these measures are employed in patients with CAD. Primary care providers are key in translating the body of knowledge we have on PAD into everyday clinical care of PAD patients.
References
1. Criqui MH, Denenberg JO, Langer RD, et al. The epidemiology of peripheral arterial disease: Importance of identifying the population at risk. Vasc Med 1997;2:221-226.
2. Ernst EE, Matrai A. Intermittent claudication, exercise, and blood rheology. Circulation 1987;76:1110-1114.
3. Selvin E, Erlinger TP. Prevalence of and risk factors for peripheral arterial disease in the United States: Results from the National Health and Nutrition Examination Survey, 1999-2000. Circulation 2004;110:738-743.
4. Criqui MH. Peripheral arterial disease epidemiological aspects. Vasc Med 2001;6(3 Suppl):3-7.
5. Diehm C, Kareem S, Lawall H. Epidemiology of peripheral arterial disease. Vasa 2004;33:183-189.
6. Criqui MH, Vargas V, Denenberg JO, et al. Ethnicity and peripheral arterial disease: the San Diego Population Study. Circulation. Oct 25 2005;112(17):2703-2707.
7. Allison MA, Criqui MH, McClelland RL, et al. The effect of novel cardiovascular risk factors on the ethnic-specific odds for peripheral arterial disease in the Multi-Ethnic Study of Atherosclerosis (MESA). J Am Coll Cardiol 2006;48:1190-1197.
8. Norgren L, Hiatt WR, Dormandy JA, et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg 2007;45: Suppl S:S5-67.
9. Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease) endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. J Am Coll Cardiol 2006;47:1239-1312.
10. Murabito JM, D'Agostino RB, Silbershatz H, et al. Intermittent claudication. A risk profile from The Framingham Heart Study. Circulation 1997;96:44-49.
11. Lu JT, Creager MA. The relationship of cigarette smoking to peripheral arterial disease. Rev Cardiovasc Med 2004;5:189-193.
12. He Y, Jiang Y, Wang J, et al. Prevalence of peripheral arterial disease and its association with smoking in a population-based study in Beijing, China. J Vasc Surg 2006;44:333-338.
13. Marso SP, Hiatt WR. Peripheral arterial disease in patients with diabetes. J Am Coll Cardiol 2006;47:921-929.
14. Doyle J, Creager MA. Pharmacotherapy and behavioral intervention for peripheral arterial disease. Rev Cardiovasc Med 2003;4:18-24.
15. Jude EB, Oyibo SO, Chalmers N, et al. Peripheral arterial disease in diabetic and nondiabetic patients: A comparison of severity and outcome. Diabetes Care 2001;24:1433-1437.
16. Cheng SW, Ting AC, Wong J. Lipoprotein (a) and its relationship to risk factors and severity of atherosclerotic peripheral vascular disease. Eur J Vasc Endovasc Surg 1997;14:17-23.
17. Olin JW. Hypertension and peripheral arterial disease. Vasc Med 2005;10:241-246.
18. Wildman RP, Muntner P, Chen J, et al. Relation of inflammation to peripheral arterial disease in the national health and nutrition examination survey, 1999-2002. Am J Cardiol 2005;96:1579-1583.
19. Barani J, Nilsson JA, Mattiasson I, et al. Inflammatory mediators are associated with 1-year mortality in critical limb ischemia. J Vasc Surg 2005;42:75-80.
20. Brevetti G, Schiano V, Chiariello M. Cellular adhesion molecules and peripheral arterial disease. Vasc Med 2006;11:39-47.
21. Taylor LM, Jr., Moneta GL, Sexton GJ, et al. Prospective blinded study of the relationship between plasma homocysteine and progression of symptomatic peripheral arterial disease. J Vasc Surg 1999;29:8-19; discussion 19-21.
22. Rutherford RB, Baker JD, Ernst C, et al. Recommended standards for reports dealing with lower extremity ischemia: Revised version. J Vasc Surg 1997;26:517-538.
23. Khan NA, Rahim SA, Anand SS, et al. Does the clinical examination predict lower extremity peripheral arterial disease? JAMA 2006;295:536-546.
24. Belch JJ, Topol EJ, Agnelli G, et al. Critical issues in peripheral arterial disease detection and management: A call to action. Arch Intern Med 2003;163:884-892.
25. Hiatt WR, Hirsch AT, Regensteiner JG, et al. Clinical trials for claudication. Assessment of exercise performance, functional status, and clinical end points. Vascular Clinical Trialists. Circulation 1995;92:614-621.
26. Pemberton M, London NJ. Colour flow duplex imaging of occlusive arterial disease of the lower limb. Br J Surg 1997;84:912-919.
27. Meijer WT, Grobbee DE, Hunink MG, et al. Determinants of peripheral arterial disease in the elderly: The Rotterdam study. Arch Intern Med 2000;160:2934-2938.
28. Jonason T, Bergstrom R. Cessation of smoking in patients with intermittent claudication. Effects on the risk of peripheral vascular complications, myocardial infarction and mortality. Acta Med Scand 1987;221:253-260.
29. Buchwald H, Bourdages HR, Campos CT, et al. Impact of cholesterol reduction on peripheral arterial disease in the Program on the Surgical Control of the Hyperlipidemias (POSCH). Surgery 1996;120:672-679.
30. Mohler ER, 3rd, Hiatt WR, Creager MA. Cholesterol reduction with atorvastatin improves walking distance in patients with peripheral arterial disease. Circulation 2003;108:1481-1486.
31. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;352:837-853.
32. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med 2000;342:145-153.
33. Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA Guidelines for the Management of Patients with Peripheral Arterial Disease (lower extremity, renal, mesenteric, and abdominal aortic): A collaborative report from the American Associations for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (writing committee to develop guidelines for the management of patients with peripheral arterial disease) summary of recommendations. J Vasc Interv Radiol 2006;17:1383-1397; quiz 1398.
34. Gardner AW, Katzel LI, Sorkin JD, et al. Improved functional outcomes following exercise rehabilitation in patients with intermittent claudication. J Gerontol Biol Sci Med Sci 2000;55:M570-577.
35. Stewart KJ, Hiatt WR, Regensteiner JG, et al. Exercise training for claudication. N Engl J Med 2002;347:1941-1951.
36. Collaborative overview of randomised trials of antiplatelet therapy III: Reduction in venous thrombosis and pulmonary embolism by antiplatelet prophylaxis among surgical and medical patients. Antiplatelet Trialists' Collaboration. BMJ 1994;308:235-246.
37. Libretti A, Catalano M. Treatment of claudication with dipyridamole and aspirin. Int J Clin Pharmacol Res 1986;6:59-60.
38. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee. Lancet 1996;348:1329-1339.
39. Dawson DL, Cutler BS, Hiatt WR, et al. A comparison of cilostazol and pentoxifylline for treating intermittent claudication. Am J Med 2000;109:523-530.
40. Hiatt WR, Regensteiner JG, Hargarten ME, et al. Benefit of exercise conditioning for patients with peripheral arterial disease. Circulation 1990;81:602-609.
41. Ahimastos AA, Lawler A, Reid CM, et al. Brief communication: ramipril markedly improves walking ability in patients with peripheral arterial disease: A randomized trial. Ann Intern Med 2006;144:660-664.
42. Barradell LB, Brogden RN. Oral naftidrofuryl. A review of its pharmacology and therapeutic use in the management of peripheral occlusive arterial disease. Drugs Aging 1996;8:299-322.
43. Kieffer E, Bahnini A, Mouren X, et al. A new study demonstrates the efficacy of naftidrofuryl in the treatment of intermittent claudication. Findings of the Naftidrofuryl Clinical Ischemia Study (NCIS). Int Angiol 2001;20:58-65.
44. Brevetti G, Diehm C, Lambert D. European multicenter study on propionyl-L-carnitine in intermittent claudication. J Am Coll Cardiol 1999;34:1618-1624.
45. Boger RH, Bode-Boger SM, Thiele W, et al. Restoring vascular nitric oxide formation by L-arginine improves the symptoms of intermittent claudication in patients with peripheral arterial occlusive disease. J Am Coll Cardiol 1998;32:1336-1344.
46. Mohler ER, 3rd, Hiatt WR, Olin JW, et al. Treatment of intermittent claudication with beraprost sodium, an orally active prostaglandin I2 analogue: A double-blinded, randomized, controlled trial. J Am Coll Cardiol 2003;41:1679-1686.
47. Lievre M, Morand S, Besse B, et al. Oral Beraprost sodium, a prostaglandin I(2) analogue, for intermittent claudication: A double-blind, randomized, multicenter controlled trial. Beraprost et Claudication Intermittente (BERCI) Research Group. Circulation 2000;102:426-431.
48. Tateishi-Yuyama E, Matsubara H, Murohara T, et al. Therapeutic angiogenesis for patients with limb ischaemia by autologous transplantation of bone-marrow cells: A pilot study and a randomised controlled trial. Lancet 2002;360:427-435.
49. McGrath C, Robb R, Lucas AJ, et al. A randomised, double blind, placebo-controlled study to determine the efficacy of immune modulation therapy in the treatment of patients suffering from peripheral arterial occlusive disease with intermittent claudication. Eur J Vasc Endovasc Surg 2002;23:381-387.
50. van Rij AM, Solomon C, Packer SG, et al. Chelation therapy for intermittent claudication. A double-blind, randomized, controlled trial. Circulation 1994;90:1194-1199.
51. Hsia J, Simon JA, Lin F, et al. Peripheral arterial disease in randomized trial of estrogen with progestin in women with coronary heart disease: The Heart and Estrogen/Progestin Replacement Study. Circulation 2000;102:2228-2232.
52. Pittler MH, Ernst E. Ginkgo biloba extract for the treatment of intermittent claudication: A meta-analysis of randomized trials. Am J Med 2000;108:276-281.
53. Kleijnen J, Mackerras D. Vitamin E for intermittent claudication. Cochrane Database Syst Rev 2000(2):CD000987.
54. Zannetti S, L'Italien GJ, Cambria RP. Functional outcome after surgical treatment for intermittent claudication. J Vasc Surg 1996;24:65-73.
Peripheral arterial disease (PAD) is a pathological condition characterized by atherosclerotic occlusion of arterial blood supply to the lower or upper extremities.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
