Vaginal Bleeding in Pregnancy: Part II
Vaginal Bleeding in Pregnancy: Part II
Author:
Pamela Arsove, MD, FACEP, Associate Residency Director, Department of Emergency Medicine, Long Island Jewish Medical Center, New Hyde Park, NY; Assistant Professor of Emergency Medicine, Hofstra North Shore Long Island Jewish School of Medicine.
Peer Reviewer:
Richard S. Krause, MD, Department of Emergency Medicine, University of Buffalo, NY.
This issue is the second in a two-part series covering vaginal bleeding in pregnancy. Part I discussed spontaneous abortion and ectopic pregnancy. Part II will discuss other causes of vaginal bleeding that typically present later in pregnancy.
Gestational Trophoblastic Disease. Gestational trophoblastic disease (GTD) refers to a group of benign and malignant tumors that arise from trophoblastic cells of the placenta. These tumors secrete high levels of beta-hCG and therefore cause symptoms similar to early pregnancy. Because of this, the diagnosis is often not made on initial presentation to the ED. The most common clinical manifestation is vaginal bleeding.1 GTD is a rare complication of pregnancy found in 1 per 1,500 live births in the United States. The main risk factors are extremes of maternal age and a history of previous GTD.2 GTD is divided into 4 main categories: hydatiform mole (molar pregnancy), persistent or invasive mole, placental site trophoblastic tumor, and choriocarcinoma. Despite differences in management, this group of tumors has an excellent prognosis even when metastatic disease is present.
Hydatiform mole accounts for 90% of GTD and is the most likely of this group of diseases to be encountered in the emergency department.3 Molar pregnancies are caused by an abnormally fertilized ovum and overproliferation of trophoblastic tissue. They are benign tumors with a small but significant potential for malignant transformation. There are two genetically different types of molar pregnancies: complete and partial. A complete mole does not contain fetal tissue, while a partial mole does. Because fetal tissue is present with a partial mole, it is often indistinguishable from an incomplete or missed abortion by clinical examination and ultrasound.
Traditionally, a complete mole has been distinguished from other causes of GTD and vaginal bleeding in pregnancy by the following reported features:
Uterine enlargement greater than expected for dates;
Abnormally high beta-hCG. Levels often exceed 100,000 mIU/mL;
Preeclampsia before the 20th week of gestation;
Theca lutein cysts;
Hyperthyroidism;
Characteristic "snowstorm" pattern on ultrasound. (See Figure 1.)
Figure 1: "Grape-like" Appearance of Molar Pregnancy on TVS
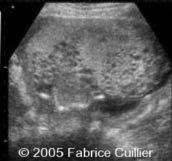
Reprinted with permission from TheFetus.net. © 2005 Fabrice Cullier.
These features are characteristic of "classic" disease in the second trimester and are uncommon in patients who seek medical attention earlier in their pregnancy.4 Patients typically now present with vaginal bleeding in their first trimester when sonography is routinely performed and the diagnosis is made before these classic findings are detected.
The typical "grape-like" appearance on ultrasound is due to histological changes causing edema of the chorionic villi. An ultrasound done in the first trimester often misses both types of molar pregnancy, even with a high index of suspicion. A study of more than 1,000 patients referred to a trophoblastic disease center suspected of having hydatiform mole compared pre-evacuation ultrasound results with postoperative histology. The sensitivity and specifity of ultrasound examination in detecting histologically confirmed hydatiform mole was 44% and 74%, respectively (LR+ = 1.8 and LR- = 0.8).5
Molar pregnancies must always be treated surgically. In suspected cases, surgical evacuation of the uterus provides histological confirmation. The minimal lab work needed prior to surgery is a complete blood count and blood typing. Some authors also recommend a CXR and liver function tests to assess for metastasis, and a baseline beta hCG. RhoGHAM should be given if indicated, as described in part I of this article. Fifteen to 20% of women with a complete hydatiform mole and 2% to 3% with a partial mole will develop malignant sequelae.6 For this reason, all patients with a molar pregnancy require close monitoring of beta hCG levels. A beta hCG that has plateaued or is rising should prompt the clinician to pursue a work-up for malignant GTD.
Malignant Gestational Trophoblastic Disease. Malignant GTD refers to the cancerous changes in gestational trophoblastic disease. There are three different types of malignant GTD: persistent or invasive mole, choriocarcinoma, and placental site trophoblastic tumors. Each of these may develop after a molar or nonmolar pregnancy and may present with local invasion or metastatic disease. The majority of malignant GTD, however, come from persistent disease after a complete molar pregnancy, and are not metastatic. Patients are usually asymptomatic, and the diagnosis is made when routine hCG monitoring shows a plateauing or increasing level of beta-hCG. Of patients who are symptomatic, vaginal bleeding is the most common complaint.7 Additional characteristics are associated with the possible development of GTD after a molar pregnancy. (See Table 1.)
Table 1: Characteristics Concerning for the Development of Malignant GTD After Molar Pregnancy
- Initial hCG > 100,000 mIU/mL
- Theca lutein cysts > 6 cm
- Excessively enlarged uterus
- Age > 40 years
- Histology revealing hyperplasia or atypia
Choriocarcinoma is a very aggressive form of GTD that is often metastatic at the time of diagnosis. The most common sites of metastasis are the lung and vagina. The liver and brain are less commonly involved.8 Choriocarcinoma is a very vascular tumor and typically presents with bleeding in the postpartum period. Choriocarcinoma follows a molar pregnancy in 1/40 women. It is uncommon after normal gestation or abortion, occurring in approximately 1/15,000-1/16,000 pregnancies.8 Placental site trophoblastic tumors are extremely rare, accounting for about 1% of malignant GTD.9
In the absence of a known molar pregnancy, the diagnosis of malignant GTD is difficult to make on clinical grounds. The diagnosis is made by histology of a surgical specimen or after investigation of a high or abnormally rising beta hCG when normal pregnancy is excluded. It is important, therefore, to maintain a high index of suspicion for this disease in any patient after an abortion, ectopic pregnancy, or normal delivery with a beta hCG level higher than expected. Ultrasound in both invasive mole and choriocarcinoma may show an abnormal uterine mass that often is hypervascular on color Doppler. The treatment of malignant GTD is chemotherapy. In general, single-agent chemotherapy with methotrexate or dactinomycin may be used for low-risk disease, with good results. Combination chemotherapy is used for high-risk cases.
Vaginal Bleeding in the Second Half of Pregnancy
Vaginal bleeding in the second half of pregnancy is also termed antepartum bleeding. It occurs in 4-5% of pregnancies and, by definition, is not related to labor and delivery.10 Although it is much less common than bleeding earlier in pregnancy, it is more likely to be life-threatening. Fetal viability can occur as early as 23 weeks gestation, so any significant bleeding after this gestational age threatens the health of both the mother and the fetus. Bleeding in the second half of pregnancy is associated with a number of perinatal complications, including preterm birth.11 The ED evaluation involves stabilizing the patient, implementing fetal monitoring if obstetrical consultation is delayed, and obtaining timely obstetrical evaluation. Bedside ultrasound examination should be performed if it does not interrupt patient management. No patient with bleeding in this time period should be discharged from the ED without evaluation by the obstetrical service. Placentia previa, placental abruption, uterine rupture, and vasa previa are the major causes of obstetrical bleeding in the second half of pregnancy, and will be discussed here.
Figure 2: Placenta Previa
The color Doppler shows vascularity within the placenta (arrow). Reprinted with permission from TheFetus.net. © Heron Werner.
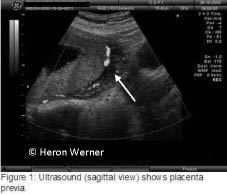
Placenta Previa. Placenta previa is a condition in which the placenta implants in the lower uterine segment near, or covering, the internal cervical os. (See Figure 2.) During labor, the dilation of the cervix can disrupt the vascular integrity of the placental attachment, and significant bleeding may occur. It occurs in approximately 1/300 deliveries in the United States and carries a maternal mortality rate of about 0.03%.12 The etiology of placenta previa is unknown, but several independent risk factors have been identified: advanced maternal age, prior C-section, and infertility treatment.12 In the first half of pregnancy, ultrasound shows placenta previa in up to 6% of patients. As the uterus grows, most of these migrate away from the internal os.13
Placenta previa characteristically presents with painless vaginal bleeding. A small percentage of patients also have uterine contractions. Bleeding results from shearing forces on the placental attachments as the lower uterine segment grows and the cervix dilates. A woman may experience an initial episode of bleeding that resolves, only to return later in pregnancy. Placenta previa is associated with significant complications to mother and fetus at delivery, including postpartum hemorrhage and preterm delivery.14 In the perinatal setting, recent research has found an association between short cervical length (< 30 mm) on transvaginal ultrasound and preterm delivery and hemorrhage in third-trimester patients with placenta previa.15
There are three types of placenta previa: total placenta previa, partial placenta previa, and marginal placenta previa. This classification is based on the proximity of the placenta to the internal os:
Total placenta previa: the placental completely covers the internal os.
Partial placenta previa: the placenta partly covers the internal os.
Marginal placenta previa: the placenta is next to the internal os.
A patient who presents in the second half of pregnancy with painless vaginal bleeding is considered to have placenta previa until proven otherwise. Pelvic examination is contraindicated in these patients because of the risk of life-threatening hemorrhage caused by insertion of the examiner's digit into the cervical canal, impacting the highly vascular placenta. Emergency obstetrical consultation should be obtained, along with bedside fetal monitoring and preoperative laboratory studies. Ultrasound is the only safe method for reliably diagnosing placenta previa. TVS provides a better image of the location of the placenta to the internal os than transabdominal ultrasound, and is safe to perform.16 Placenta previa is excluded if the placenta is greater than 2 cm from the internal os. There are several situations that make diagnosis of placentia previa difficult on ultrasound. An overdistended bladder may distort the anatomy to give the appearance of a placenta previa when it is not present. A fetal head low in the pelvis may obstruct visualization of the placenta, and it may be difficult to localize the placenta if uterine contraction occurs.16
Emergent obstetrical consultation is required in cases of placenta previa that present with vaginal bleeding. Indications for emergency cesarean section include hemodynamic instability, a stable patient who deteriorates, a mature gestation estimated by age greater than 36 weeks or weight greater than 2500 g, and evidence of fetal distress.17 In the ED, start aggressive fluid resuscitation, send preoperative labs, and begin fetal monitoring. In the stable patient, if bleeding has slowed or stopped and the fetus is not mature, inpatient observation is reasonable. There is currently no firm evidence to favor prolonged hospitalization over at-home observation for stable patients, and the decision should be made in conjunction with the patient and her obstetrician.18
Placental Abruption. Placental abruption refers to the premature separation of the placenta from its site of implantation in the uterus. It most commonly occurs around the 25th week of gestation, but this varies depending on the etiology of the abruption. Abruption occurs in approximately 1/100 births and is complicated by fetal death 15% of the time. The cause of abruption is unknown, but it has been associated with advanced maternal age, existing hypertension, cocaine use, trauma, smoking, and premature rupture of membranes.19 Bleeding occurs into the decidual basalis, which then separates from the placenta. The separation may be large or small, and may dissect along the uterine placental junction and present as vaginal bleeding. Up to 20% of the time, the bleeding does not escape the placental margins and is concealed.20 In this circumstance, vaginal bleeding is absent. The disruption of the maternal placental interface interferes with gas and nutrient exchange to the fetus with potentially devastating fetal compromise. If more than 50% of placental surface is disrupted, fetal death may occur.21 Disseminated intravascular coagulation (DIC) is a serious complication of placental abruption that occurs in about 10% of patients. It is thought to be caused by leakage of a thromboplastin-like material from the placental system and is more common in cases associated with fetal distress and death.22
The classic presentation in placental abruption is painful vaginal bleeding and uterine contractions. The amount of vaginal bleeding is variable and does not correlate well with the extent or severity of the abruption and potential fetal risk.23 The diagnosis is mainly a clinical one and should be suspected in any patient in the second half of pregnancy with painful vaginal bleeding, trauma, or preterm labor. If the placental separation is small, the physical findings may be minimal. In severe cases, hypotension and tachycardia, with uterine tenderness or palpable tetany are present on physical exam. Recent data from the New Jersey-Placental Abruption Study found a poor correlation between clinical predictors of abruption and a histopathological diagnosis.24 Unfortunately, ultrasound is also often inconclusive in diagnosing abruption. The classic finding is a retroplacental clot (see Figure 3), but a normal ultrasound cannot exclude the diagnosis. Laboratory studies may reveal anemia and, in severe cases, evidence of DIC.
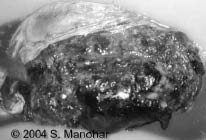
Figure 3: Placental Abruption with Retroplacental Clot
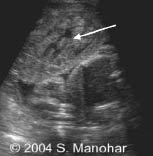
Ultrasound shows retroplacental clot (arrow) caused by a placental abruption. Fetal parts are visualized underneath the clot. The surgical specimen is seen on the right with clot visible. Reprinted with permission from TheFetus.net. © 2004 S. Manohar.
ED management of patients with placental abruption depends on the hemodynamic stability of the patient. In the unstable patient, the goal is to maintain hemodynamic stability and oxygenation of the patient. Aggressive volume resuscitation, supplemental oxygen, fetal monitoring, and emergent obstetric consultation should be instituted. Laboratory studies should include a complete blood count, blood type and crossmatch for two units of packed red blood cells, prothrombin time (PT), activated partial thromboplastin time (aPTT), and fibrinogen level. Transfuse blood emergently if bleeding is heavy and persistent, and administer fresh frozen plasma (FFP) if coagulopathy is present. If abruption is definitely diagnosed, and the fetus is alive and viable, preparation should be made for emergent C-section. If the fetus is dead, vaginal delivery should be attempted as soon as possible. In the stable patient, admit to the obstetrical service for continual fetal monitoring. Corticosteriods to accelerate fetal maturation and tocolytics to prevent labor are given at the discretion of the admitting service. In a minority of stable patients, vaginal delivery ultimately may be attempted.
Vasa Previa. Vasa previa is a rare condition in which the fetal blood vessels abnormally cross the internal os in the uterus. (See Figure 4.) Under normal circumstances the fetal vessels are housed in the umbilical cord, which inserts into the placenta. In vasa previa, the fetal vessels run through the fetal membranes without the protection of the cord and cross the internal os before they reach the placenta. Because they lie between the fetus and the cervix, there is no way to prevent trauma to these vessels when the membranes rupture or childbirth occurs. Since fetal circulation is involved, rupture of these vessels and loss of as little as 100 mL of blood can quickly result in fetal death.25 Risk factors associated with vasa previa include a low-lying placenta or placenta previa, velamentous insertion of the umbilical cord, multi-lobed placenta, multiple gestations, and in vitro fertilization.25 Vasa previa occurs in about 1/2,500 pregnancies, but may be underrepresented if missed at ultrasound. In cases of velamentous insertion of the umbilical cord, rates increase to as high as 1/50.26 Neonatal survival rates are greatly increased when the diagnosis is made antenatally.27,28 Recent guidelines for the management of vasa previa published by The Society of Obstetricians and Gynaecologists of Canada showed a survival rate of 97% in cases diagnosed antenatally compared to a rate of 44% in those not diagnosed before symptoms appear.29 Based on these findings, TVS screening should be performed with careful attention to the umbilical cord on all patients at risk.
Figure 4: Uterine Rupture

Transvaginal ultrasound of uterine rupture in the lower uterine wall. The tear is clearly visualized interoperatively. Reprinted with permission from TheFetus.net. © 2005 Francois Manson.

The most common clinical presentation is vaginal bleeding at the time of rupture of membranes. Because this is a rare diagnosis that may be unknown to the patient, it is most commonly confused with placenta previa and placenta abruption.
The diagnosis of vasa previa is made by ultrasound and color Doppler prior to delivery or by inspection of the placenta after delivery. (See Figure 3.)
The optimal management of vasa previa is prenatal diagnosis and close monitoring, with elective C-section at about 35 weeks gestation. These patients often are hospitalized weeks earlier, when corticosteroids are administered to induce lung maturity in the fetus. The diagnosis is rarely made in the ED, and fetal survival is unlikely. In the rare instance of pre-term rupture of membranes with a live fetus, immediate C-section should be performed.
Uterine Rupture. Uterine rupture is caused by a complete tear in the uterine wall and is usually associated with labor. In rare instances, it occurs spontaneously in the second half of pregnancy. Risk factors in this circumstance include a previous classic cesarean section scar, uterine overdistension as seen in multiple gestations, cornual ectopic pregnancy, gestational trophoblastic disease, trauma, or placenta increta or percreta.30 Most uterine ruptures studied occur in patients undergoing vaginal birth after cesarean section (VBAC). A recent review suggests the current at-risk population includes more women who have no history of previous uterine surgery and are not in labor than previously thought.31 The true incidence is not precisely understood because the literature often fails to distinguish between true uterine rupture and scar dehiscence, and statistics often are limited to patients undergoing VBAC. In a large prospective observation study of almost 18,000 women who underwent a trial of labor after prior C-section, the rate of uterine rupture was 0.7%.32
Symptoms of uterine rupture not associated with labor can be misleading. Abdominal pain and vaginal bleeding is often minimal or even absent. In extreme situations, pain and bleeding may be severe, and hypovolemic shock may ensue. The fetus may protrude into the abdominal cavity, allowing fetal parts to be palpable on abdominal exam.
Mortality data, like prevalence data, are largely limited to information from VBAC births complicated by uterine rupture. Factors influencing morbidity and mortality include time to delivery after rupture occurs, the amount of blood lost, and the degree of placental separation from the uterus. In developed countries, maternal mortality has been reported to be as low as 0.2%, and fetal mortality, 5-6%.33,34
Ultrasound in the ED may reveal the rupture. (See Figure 4.) Once uterine rupture is suspected or diagnosed, steps should be taken to stabilize the mother and fetus in preparation for emergency cesarean section. Unfortunately, if complete uterine rupture occurs prior to arrival to the ED, fetal death usually has already occurred.
References
1. Jauniaux E. Trophoblastic diseases and pregnancy. The Obstetrician & Gynaecologist 2003;5:130-135.
2. Tse KY, Chan KL, Tam KF. Gestational trophoblastic disease. Obstetrics, Gynaecology &Reproductive Medicine 2007;19:89-97.
3. Gerulath AH. Gestational trophoblastic disease. SOCG Clinical Practice Guidelines 2002;114:1-6.
4. Kirk E, Papageprgjiou A, Condous G. The accuracy of first trimester ultrasound in the diagnosis of hydatidiform mole. Ultrasound in Obstetrics and Gynecology 2007;29:70–75.
5. Fowler D, Lindsay I, Seckl M, et al. Routine pre-evacuation ultrasound diagnosis of hydatidiform mole: Experience of more than 1000 cases from a regional referral center. Ultrasound in Obstetrics and Gynecology 2006;27:56–60.
6. Gerulath AH. Gestational trophoblastic disease. SOCG Clinical Practice Guidelines 2002;114:1-6.
7. Berkowitz R, Goldstein D. Current management of gestational trophoblastic diseases. Gynecologic Oncology 2009;112:654-662.
8. Lurain J. Gestational trophoblastic disease I: Epidemiology, pathology, clinical presentation and diagnosis of gestational trophoblastic disease, and management of hydatidiform mole. Am J Obstet Gynecol 2010;doi:10.1016/j.ajog.2010.06.073.
9. Lan C, Li Y, He J, et al. Placental site trophoblastic tumor: Lymphatic spread and possible target markers. Gynecologic Oncology 2010;116:430-437.
10. Mukherjee S, Bhide A. Antepartum haemorrhage. Obstetrics, Gynaecology & Reproductive Medicine 2008;18:335-339.
11. Koifman A, Levy A, Zaulan Y, et al. The clinical significance of bleeding during the second trimester of pregnancy. Arch Gynecol Obstet 2008;278:47-51.
12. Oppenheimer L. SOGC Clinical Practice Guideline 2007;189:261-266.
13. Rosenberg T, Pariente G, Sergienko R, et al. Critical analysis of risk factors and outcome of placenta previa. Arch Gynecol Obstet 2010 [Epub ahead of print].
14. Zlatnik MG, Cheng YW, Norton ME, et al. Placenta previa and the risk of preterm delivery. J Maternal-Fetal and Neonatal Medicine 2007;20:719-723.
15. Stafford I, Dashe,J, Shivvers S, et al. Ultrasonographic cervical length and risk of hemorrhage in pregnancies with placenta previa. Obstetrics & Gynecology 2010;116:595-600.
16. Oppenheimer L. SOGC Clinical Practice Guideline 2007;189:261-266.
17. Dola CP, Longo SA. Diagnosis and safe management of placenta previa. J Family Practice 2006;18. http://www.jfponline.com/Pages.asp?AID=4488.
18. Neilson JP. Interventions for suspected placenta praevia. Cochrane Database Syst Rev 2003;(2):CD001998.
19. Oyelese, Y. Placental abruption. Obstetrics & Gynecology 2006;108:1005-1016.
20. Hladky K, Yankowitz J, Hansen WF. Placental abruption. Obstet Gynecol Surv 2002;57:299–305.
21. Oyelese, Y. Placental abruption. Obstetrics & Gynecology 2006;108:1005-1016.
22. Leunen K, Hall DR, Odendaal HJ, et al. The profile and complications of women with placental abruption and intrauterine death. J Trop Pediatr 2003;49:231-234.
23. Tikkanen M. Etiology, clinical manifestations, and prediction of placental abruption. Acta Obstetricia et Gynecologica Scandinavica 2010;89:732-740.
24. Elsasser DA, Ananth CV, Prasad V, et al. Diagnosis of placental abruption: Relationship between clinical and histopathological findings. Eur J Obstet Gynecol Reprod Biol 2010;148:125-130.
25. Gagnon R, Morin L, Bly S, et al. SOGC Clinical Practice Guideline: Guidelines for the management of vasa previa. Int J Gynaecol Obstet 2010;108:85-89.
26. Gagnon, R. Guidelines for the management of vasa previa. JOGC 2009;231:748-753.
27. Daly-Jones E, John A, Leahy A, et al. Vasa praevia: A preventable tragedy. Ultrasound 2008;16:8–14.
28. Oyelese Y, Catanzarite, V, Prefumo, F, et al. Vasa previa: The impact of prenatal diagnosis on outcomes. Obstetrics & Gynecology 2004;103:937-942.
29. Gagnona R, Morin L, Bly S, et al. Guidelines for the management of vasa previa. International Journal of Gynecology & Obstetrics 2010;108:85-89.
30. Vakim Z, Maymon R, Mendlovic, et al. Clinical, sonographic, and epidemiologic features of second- and early third-trimester spontaneous antepartum uterine rupture: a cohort study. Prenatal Diagnosis 2008;28:478–484.
31. Wuntakal R, Hollingworth T, Visvanathan D. Changing trends: Uterine rupture in the UK. BJOG: An International Journal of Obstetrics & Gynaecology 2010;117:1428-1429.
32. Landon M, Hauth J, Leveno K, et al. Maternal and perinatal outcomes associated with a trial of labor after prior cesarean delivery. N Engl J Med 2004;351:2581-2589.
33. Hofmeyr G, Say L, Gulmezogly AM. Systematic review: WHO systematic review of maternal mortality and morbidity: The prevalence of uterine rupture. BJOG: An International Journal of Obstetrics & Gynaecology 2005;112:1221-1228.
34. Lydon-Rochelle M, Holt V, Eastering T, et al. Risk of uterine rupture during labor among women with a prior cesarean delivery N Engl J Med 2001;345:3-8.
This issue is the second in a two-part series covering vaginal bleeding in pregnancy. Part I discussed spontaneous abortion and ectopic pregnancy. Part II will discuss other causes of vaginal bleeding that typically present later in pregnancy.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
