Special Feature: Acetaminophen Toxicity
Special Feature
Acetaminophen Toxicity
By Saadia R. Akhtar MD MSc, St. Luke's Idaho Pulmonary Associates, Boise, is Associate Editor for Critical Care Alert.
Dr. Akhtar reports no financial relationship to this field of study.
Acetaminophen (known as paracetamol outside the United States) is the most commonly used analgesic in the world, usually considered to be safe and benign. In 2008, however, according to the American Association of Poison Control Centers, acetaminophen overdose occurred in 27,790 cases, resulting in 13,650 hospitalizations and 43 deaths.1 Acetaminophen overdose may lead to hepatotoxicity, acute tubular necrosis and, less likely, pancreatitis.2 It is the most common cause of acute liver failure in many Western countries, accounting for 40%-50% of cases in the United States.3,4 (Interestingly, of those cases with acute liver failure from acetaminophen, half or more are unintentional overdoses.) Fortunately, with current treatment more than 90% of patients with acetaminophen toxicity recover completely. Of those who develop acute liver failure, 70% survive and recover fully with supportive care. Histologically, normal liver architecture is restored within about 3 months from initial ingestion/overdose.2
Factors that may slightly increase susceptibility to acetaminophen-related hepatotoxicity include age > 40 years, tobacco use, malnutrition or starvation, and anything else that may impact the way acetaminophen is metabolized in the liver: presence of certain polymorphisms of cytochrome isoenzymes, chronic alcohol ingestion, or chronic use of medications that induce P450 isoenzymes such as some anticonvulsants and isoniazid. Chronic liver disease is not associated with increased risk. Age < 5 years and acute alcohol ingestion seem to decrease susceptibility to acetaminophen toxicity.2,5
Pathophysiology
Acetaminophen is quickly absorbed from the GI tract and reaches peak serum levels in 1-4 hours. Normally, at recommended therapeutic doses (≤ 4 g in adults or 80 mg/kg in children, daily), more than 90% of ingested acetaminophen is converted in the liver by sulfation or glucuronidation to inactive metabolites, which are then excreted in the urine or bile. About 2% of acetaminophen is directly excreted in the urine; the remaining 5%-8% is metabolized in the liver by cytochrome P450 isoenzymes to the extremely toxic substance N-acetyl-p-benzoquinoneimine (NAPQI). Usually, glutathione inactivates NAPQI, ultimately forming mercapturic acid and cysteine conjugates, which are nontoxic and cleared in the urine.
When excess amounts of acetaminophen are ingested, however, the usual glucuronide and sulfate pathways are saturated and more drug passes through the P450 system; once glutathione stores are consumed, NAPQI accumulates and leads to direct injury of hepatocytes and to centrilobular necrosis. As previously noted, factors that deplete glutathione (such as starvation) or stimulate P450 isoenzymes (like chronic use of certain anticonvulsants) predispose patients to hepatic injury in the setting of acetaminophen ingestion.2,5
N-acetylcysteine (NAC) has been used as an antidote for acetaminophen toxicity since the mid-1970s. It works primarily by restoring glutathione, thus averting NAPQI buildup and hepatic injury. (Methionine and cysteamine have similar effects and were the first agents utilized for acetaminophen toxicity; they fell out of use because of significant side effects including severe flushing and vomiting.) NAC also stimulates sulfation and appears to have some anti-inflammatory, anti-oxidant, and vasodilating effects.5,6
Clinical Presentation
The clinical presentation of acute acetaminophen toxicity is fairly predictable and can be divided into four stages, based on time from ingestion. During stage 1 (0-24 hours), patients experience anorexia, nausea, vomiting, abdominal pain, and general malaise. Laboratory studies (liver function tests, chemistries) may be normal or only minimally abnormal. In stage 2 (24-72 hours), hepatic injury becomes apparent; symptoms improve transiently but patients develop transaminitis, elevated bilirubin, and rising prothrombin time. Right upper quadrant pain and jaundice begin to appear late in this stage.
Stage 3 (72-96 hours or longer) represents the peak of hepatotoxicity and it is during this stage that frank acute liver failure may manifest. Gastrointestinal (GI) symptoms return or worsen and jaundice, general malaise, and hepatic encephalopathy (confusion, somnolence, or coma, sometimes with cerebral edema) occur. Liver function tests reach their maximum levels.2,5 Coagulopathy may be severe, so bleeding risk is high. Acute renal failure (due to acute tubular necrosis and dehydration) will occur in 25% of patients with severe hepatotoxicity and in more than 50% of those with acute liver failure.7 Lactic acidosis, hypoglycemia, and hypophosphatemia are also commonly noted. Finally, recovery occurs in stage 4 (96 hours to 14 days), with resolution of symptoms and gradual normalization of liver function studies.2,5,6
Diagnosis
Diagnosis of acute acetaminophen overdose relies on a detailed history and high suspicion in patients presenting with GI symptoms without another clear etiology, altered mental status, drug ingestion, or suicide attempt. It is important to remember that acetaminophen is present in many combination medications (analgesics, antipyretics, allergy and sinus medications, etc). The history should elicit information about all available drugs, dosage, route, and time of ingestion. A single ingestion of ≥ 150 mg/kg or ≥ 10-15 g of acetaminophen will result in hepatotoxicity. Chronic daily ingestions at lower doses (4-10 g) may also lead to hepatotoxicity.2,5
Serum acetaminophen level should be measured at 4 and 24 hours after ingestion or at presentation if > 4 hours have passed since ingestion. (A false-positive acetaminophen level may be found in the setting of severe hyperbilirubinemia, > 10 mg/mL, from another cause.) In the case of a single ingestion at a known time, serum acetaminophen level at 4-24 hours post-ingestion can be used to predict the risk of hepatotoxicity using the Rumack-Matthew nomogram (see Figure below).5
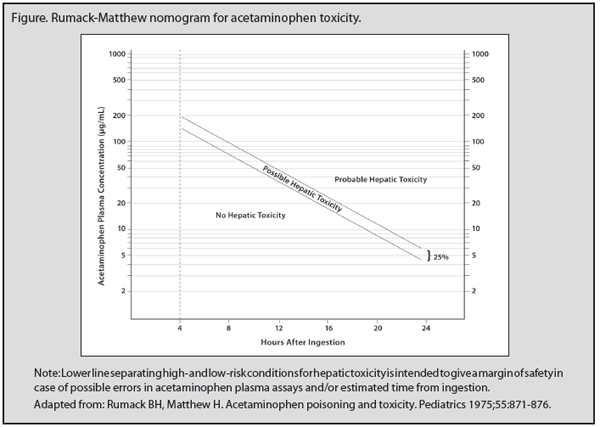
The diagnosis of acetaminophen toxicity in the setting of chronic ingestion is more difficult. A detailed drug history is essential. An acetaminophen level simply confirms ongoing ingestion but is not useful for prediction risk of hepatotoxicity.2
Measurement of the NAPQI-protein adducts released following hepatocellular injury has been evaluated as a more reliable way to identify acetaminophen toxicity in acute or chronic ingestions. This test is not widely available or part of standard clinical practice at this time.5
Management
Treatment of acute acetaminophen toxicity begins with gastric lavage and administration of activated charcoal within 1-4 hours of ingestion, in the setting of a protected airway (in order to avoid aspiration).2
Early initiation of NAC is the "gold standard" for those patients at risk of hepatotoxicity. Again, for single ingestions with clear timing, this risk is measured and predicted by the Rumack-Matthew nomogram (see Figure 1). Most experts and poison control centers also recommend giving NAC for patients with acetaminophen level > 10-20 mg/mL and undeterminable time of significant (> 150 mg/kg or > 10 g) ingestion, history of significant ingestion with laboratory evidence of hepatotoxicity, or history of repeated supratherapeutic ingestions with elevated liver function studies or associated GI symptoms (abdominal tenderness, jaundice, malaise).5
When indicated, NAC should be started as soon as possible, ideally within 8-10 hours of ingestion; serious hepatotoxicity is rare if NAC is started in this interval. Although some benefit has been documented even if NAC is started up to 24-48 hours after ingestion, the effect is decremental. In the largest prospective study of NAC for acetaminophen toxicity including more than 11,000 subjects with suspected acetaminophen overdose and about 2500 that met criteria for NAC, severe hepatotoxicity occurred in 2.9% of patients treated within 8 hours, 6.1% treated within 10 hours, and in 26.4% of patients treated at 10-24 hours.9
NAC may be dosed orally (140 mg/kg x 1, best tolerated as a 5% solution in cola or juice, followed by 70 mg/kg every 4 hours for 17 doses) or intravenously (150 mg/kg followed by 50 mg/kg over 4 hours and then 100 mg/kg over 16 hours), with equivalent response. The duration of therapy should be extended if patients still have measurable acetaminophen levels, elevated transaminases, or coagulopathy (INR > 1.5-2.0) at the end of the usual course of treatment.5
Common side effects of oral NAC include nausea, vomiting, diarrhea, and rash. IV NAC is generally well-tolerated but 10%-20% of patients may have anaphylactoid reactions; after usual supportive treatments (diphenhydramine, corticosteroids, and bronchodilators) and transient (1-2 hours) cessation of the infusion, the remaining course of NAC can generally be completed without difficulty at a slower infusion rate.2,6
Although outpatient management with self-administration of NAC has been suggested for selected patients with acetaminophen toxicity and no symptoms or laboratory abnormalities, most patients will require hospital admission.
Patients with frank acute liver failure will need supportive care in an ICU setting. Common complications are encephalopathy with inability to protect airway; thus, intubation and mechanical ventilatory support may be needed. Cerebral edema may occur and consideration must be given to ICP monitoring in selected patients. Severe coagulopathy will be present, but, in general, transfusion of fresh frozen plasma should be avoided unless necessary (for bleeding complications or procedures) as the INR may be one of the most useful indicators for tracking recovery of hepatic function.3 Dialysis support for acute renal failure may be necessary. IV NAC is indicated in patients with acetaminophen overdose and acute liver failure; IV NAC appears to decrease the risk of disease progression, cerebral edema, and mortality in this population.10 Furthermore, consideration should be given to transferring such patients to liver transplantation centers early in the course of their acute liver failure. Poor prognosis without rapid liver transplantation is seen in patients with persistent arterial pH < 7.30 after resuscitation or prothrombin time > 100 seconds, serum creatinine > 3.4 mg/dL, and advanced grade (III or IV) encephalopathy (King's College criteria).5
Conclusion
Acetaminophen is a commonly used and generally safe analgesic, but overdose has high morbidity and is potentially lethal. It is important to maintain a high suspicion for acetaminophen toxicity in the right clinical setting because prompt identification and early treatment with NAC can avert hepatotoxicity, prevent death, and result in essentially full recovery for the majority of affected patients. For those with acute hepatic failure due to acetaminophen overdose, aggressive supportive care and management at a liver transplantation center should be considered.
References
- Bronstein AC, et al. 2008 Annual Report of the American Association of Poison Control Centers' National Poison Data System (NPDS): 26th Annual Report. Clin Toxicol 2009;47:911-1084.
- Chun LJ, et al. Acetaminophen hepatotoxicity and acute liver failure. J Clin Gastroenterol 2009;43:342-349.
- Trotter JF. Practical management of acute liver failure in the intensive care unit. Curr Opin Crit Care 2009;15:163-167.
- Daly F, et al. Guidelines for the management of paracetamol poisoning in Australia and New Zealand. MJA 2008;188: 296-301.
- Larson AM. Acetaminophen hepatotoxicity. Clin Liver Dis 2007;11:525-548.
- Heard KJ. Acetylcysteine for acetaminophen poisoning. N Engl J Med 2008;359:285-292.
- Mazer M, Perrone J. Acetaminophen-induced nephro-toxicity: Pathophysiology, clinical manifestations and management. J Med Toxicol 2008;4:2-6.
- The Merck Manuals. Acetaminophen Poisoning. Available at: www.merck.com/mmpe/sec21/ch326/ch326c.html. Accessed Oct. 31, 2010.
- Smilkstein MJ, et al. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976 to 1985). N Engl J Med 1988;319:1557-1562.
- Keays R, et al. Intravenous acetylcysteine in paracetamol induced fulminant hepatic failure: A prospective controlled trial. BMJ 1991;303:1026-1029.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
