An Evidence-based Approach to Beta-Blocker and Calcium Channel Blocker Toxicity
An Evidence-based Approach to Beta-Blocker and Calcium Channel Blocker Toxicity
Authors:
Mark Su, MD, North Shore University Hospital, Department of Emergency Medicine, Medical Toxicology, Manhasset, NY.
Rachel S. Weiselberg, MD, North Shore University Hospital, Department of Emergency Medicine, Medical Toxicology, Manhasset, NY.
Peer Reviewer:
Frank LoVecchio, DO, Director, Banner Poison Center, Maricopa Medical Center, Phoenix, AZ.
Introduction
You have just taken sign-out when a nurse comes up to you and says that there is a 64-year-old man in the critical bay who took an overdose of his medications. The patient has a history of hypertension, atrial fibrillation, and depression. The patient is lethargic but arousable, and reports he took about 40 tablets of immediate-release metoprolol three hours ago in an attempt to "end it all." The nurses are starting IV lines, checking vitals, and putting the patient on the monitor. You wonder, "Is it too late for gastric decontamination? If he is symptomatic, which therapy will I try first, and what are my options?"
Meanwhile, the resident working with you pulls you aside to tell you about a 2-year-old child in the pediatric area who was found playing with grandma's bottle of verapamil controlled release 15 minutes ago. The grandmother thinks that at most there are three tablets missing. The resident says the child looks great and is playing in the room and then asks you, "Are three tablets a big deal? Can we just watch the child for a couple of hours? Do we need an IV and blood work? Since there is no pediatric unit here, should we consider transfer now?"
Beta-blockers and calcium channel blockers are very widely prescribed cardiovascular medications. In the emergency department, we are all aware of these medications for the treatment of hypertension, heart failure, atrial fibrillation, and a variety of other illnesses. When misused, either intentionally or unintentionally, the effects can be life-threatening. It is important for an emergency physician to be aware of the potential severity of an overdose. When signs or symptoms do develop, there are many therapeutic options.
Epidemiology
In 2008, the American Association of Poison Control Centers in the Toxic Exposure Surveillance System Database recorded 21,282 exposures to beta-blockers and 10,398 exposures to calcium channel blockers. Of these exposures, 707 beta-blocker and 423 calcium channel blocker cases had "moderate" to "severe" outcomes, and there were 6 beta-blocker and 12 calcium channel blocker deaths reported.1 Approximately 40% of each were unintentional exposures, and 14% involved children younger than age 6.1
Physiology
The Calcium Channel. There are many different types of calcium channels, but our focus here is the L-type channel. It is a voltage-gated channel found in cardiac myocytes, pacemaker cells, vascular smooth muscle, and on beta islet cells in the pancreas.2 When the cell is depolarized, it allows for calcium influx. Depending on the cell type, increased calcium initiates different intracellular pathways.
In myocytes, on entering the cell, calcium binds the ryanodine receptor on the sarcoplasmic reticulum, releasing calcium stores, which bind troponin, initiating contraction.2,3 (See Figure 1.) This corresponds to phase 2, also known as the plateau phase, of the myocyte action potential. (See Figure 2.)
Figure 1: The Ventricular Myocyte: The Calcium Channel
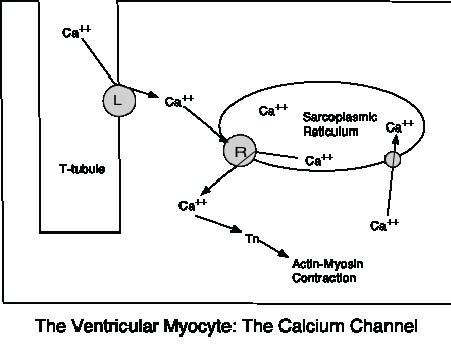
When the cell is depolarized, the L-type (L) calcium channel opens and calcium moves into the cytoplasm. Calcium then binds the ryanodine (R) receptor on the sarcoplasmic reticulum (SR), triggering a large calcium efflux from the SR into the cell. The calcium can then bind troponin, which leads to contraction of the actin and myosin filiments.
Figure 2: The Ventricular Myocyte Action Potential
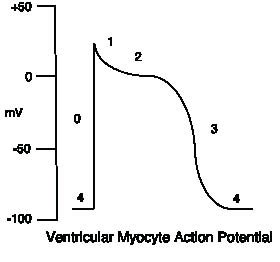
Phase 0 corresponds to the fast inward sodium current and depolarization of the myocyte. Phase 1 is a transient outward potassium current that begins repolarization. Phase 2, or the plateau phase, is the slow inward calcium current through the L-type channel. Phase 3 is further repolarization by another outward potassium current.
In the pacemaker cells of the heart, slow calcium influx begins in phase 4 and is also responsible for phase 0 of the pacemaker action potential, which is further depolarization of the cell. (See Figure 3.) In smooth muscle, intracellular calcium binds directly to calmodulin, which leads to contraction.3 In the pancreas, the calcium allows for exocytosis of insulin.
Figure 3: The Pacemaker Action Potential
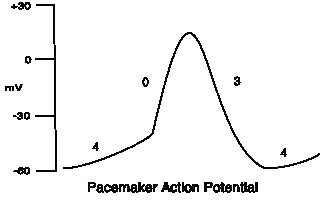
Phase 4 corresponds to the slow spontaneous depolarization of the pacemaker cell. It is mostly sodium influx. During this phase a transient calcium current flows inward, followed by the slow inward calcium current through the L-type channel. In phase 0, the L-type channel calcium current continues to depolarize the cell. Phase 3 is repolarization by an outward potassium current.
The Beta-Receptor. The beta-receptor is an adrenergic receptor, so its major ligands are epinephrine and norepinephrine. There are three types of beta-receptors.
The beta-1 receptor is mainly located in the heart and is the most important for this discussion. It is coupled to a stimulatory G protein and, when bound, activates adenylate cyclase, which converts ATP to cAMP.4-6 This stimulates protein kinase A (PKA) to phosphorylate, or activate, the L-type calcium channel, and leads to the pathways mentioned above.2,3,6 PKA also phosphorylates phospholamban, which leads to increased intracellular calcium.2 (See Figure 4.) Stimulation of the beta-1 receptor, therefore, leads to increased inotropy and chronotropy.
Figure 4: The Ventricular Myocyte: The Beta-Receptor
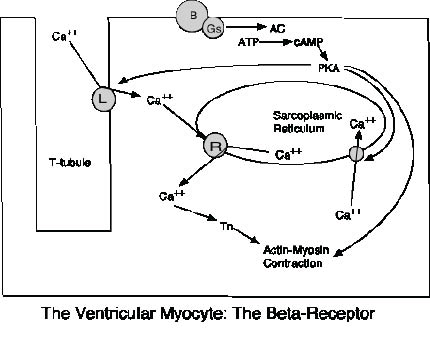
When the beta-receptor (B) is activated, the G-stimulatory protein activates adenylate cyclase (AC) to convert ATP to cAMP. This activates PKA to further activate other pathways; it activates the L-type receptor, the channel allowing calcium influx into the sarcoplasmic reticulum, and leads to contraction coupling.
The beta-2 receptor is found on vascular smooth muscle where stimulation causes vasodilation.4 In skeletal muscle it causes glycogenolysis and potassium uptake (hypokalemia).4,6 In the liver it leads to glycogenolysis and gluconeogenesis.4,6 In the lungs, stimulation results in bronchodilation. The beta-3 receptor is involved in lipolysis, and is not significant for this discussion.
Pharmacology
Calcium Channel Blockade. Understanding how calcium ion movement affects the body on a cellular level allows us to appreciate the mechanism of calcium channel blockers. Blocking the L-type calcium channels in the pacemaker cells leads to prolonged time for depolarization and therefore impulse propagation. The result is slowing of the heart rate.
Blockade in the ventricular myocytes leads to decreased strength of contraction. Smooth muscle calcium channel blockade leads to relaxation. In the pancreas, calcium channel blockade inhibits insulin release, resulting in hyperglycemia.7
Calcium Channel Blockers. There are three main types of calcium channel blockers currently prescribed in the United States. Verapamil is the only available phenylalkylamine. Of all the calcium channel blockers, it has the strongest effect on the heart, and a measurable effect on peripheral vasculature. The result is decreased inotropy, chronotropy, and peripheral vasodilation. Diltiazem is the only available benzothiazepine. It has a mechanism of action similar to verapamil, although it is less potent peripherally. This makes it a good choice for patients in the emergency department with atrial fibrillation with a rapid ventricular response, as it usually decreases inotropy without significant hypotension.
The last major category is the dihydropyridines. Examples include amlodipine, felodipine, nifedipine and nicardipine. These drugs primarily act on the peripheral vasculature, producing vasodilation. This is how they produce their anti-hypertensive effect. There is less direct effect on the heart in therapeutic doses compared to verapamil and diltiazem, and due to decreased preload, the patient may experience a reflex tachycardia.
Beta-Blockade. Beta-1 receptor blockade leads to decreased adenylyl cyclase activation. In turn, less cAMP is produced. In the pacemaker cells, this slows the action potential, leading to decrease in heart rate and can result in SA or AV nodal blocks.8 In the myocytes, decreased calcum influx disrupts the pathway to contraction, and so there is decreased inotropy.8 By decreasing heart rate and contractility, cardiac output is decreased.
Blockade of beta-2 receptors causes constriction of the peripheral vasculature, bronchospasm, hyperkalemia, and hypoglycemia.8
Beta-Blockers. While the focus of this article is the toxicity of beta-1 blockade, there are many different beta-blockers available that have different affinities for each receptor type. There are beta-blockers that are selective for the beta-1 receptor, which include atenolol, esmolol, and metoprolol. The main effects are decreased inotropy and chronotropy. This specificity, however, may be lost in overdose.
Other beta-blockers that are non-selective for any one type of beta-receptor include carvedilol, labetalol, propranolol, sotalol, and timolol. They all possess some beta-2 antagonist activity in addition to beta-1 antagonist activity. Carvedilol and labetalol also antagonize the alpha-receptor. Labetalol has some intrinsic sympathomimetic activity. Sotalol also blocks potassium channels and is considered a class III antidysrhythmic.
Pharmacokinetics/Pharmacodynamics
Calcium Channel Blockers. The pharmacokinetics and pharmacodynamics for the calcium channel blockers vary depending upon the specific class and type, so it is essential to know which drug the patient took. The exact formulation, thus, has significant impact on emergency department management and disposition.
Verapamil can be prescribed as immediate release (IR), sustained release (SR), and controlled release (CR). Diltiazem and the dihydropyridines come in IR and SR forms.
Immediate- and sustained-release formulations can take effect within 1 hour, whereas the controlled-release forms delay onset of effect for 4-5 hours. Peak plasma concentrations generally are achieved for IR preparations within 30 minutes to 2 hours, and can be delayed for 5-7 hours for sustained-release formulations. Half-lives for immediate-release formulations range from 2-7 hours after a single dose and 4-7 hours for sustained release. It can be longer with repetitive dosing or liver dysfunction. All calcium channel blockers are extensively metabolized by the liver, and many by the cytochrome P450 system, specifically CYP3A4.2 CYP3A4 is both inhibited and induced by a variety of other medications, and this may have a significant effect on kinetics.
Beta-Blockers. The pharmacokinetics among the beta-blockers are even more varied than the calcium channel blockers. Beta-blockers differ greatly in their lipophilicity. Propranalol, metoprolol, and labetalol are highly lipid-soluble and consequently have higher volumes of distribution. High lipophilicity enhances distribution into tissues, especially the central nervous system (CNS). Beta-blockers are also primarily metabolized in the liver.6,8 Some of the metabolites are active. One of the active metabolites of propanolol has a longer half-life than propranolol itself, and so its duration of effect is prolonged.8 Atenolol and sotalol are examples of beta-blockers that are hydrophilic, i.e., have low volumes of distribution, low protein binding, and are excreted unchanged by the kidneys.6,8 Half-lives vary greatly, from less than 10 minutes for esmolol, to up to 12 hours for sotalol, and even longer for some less commonly used beta-blockers.6
Toxic Dose and Toxicokinetics/Toxicodynamics.
In using the word "overdose," we refer to any amount of drug taken that exceeds the prescribed dose. There is no precise dose for any of these medications that we can clearly say is a "toxic" dose. In children, there are multiple case reports of toxicity, even death, following ingestion of only 1-2 pills of calcium channel blockers.3,9 Unfortunately, we cannot use the limited information from case reports to define a "safe" vs. "toxic" amount.
In overdose, many of the pharmacokinetic and pharmacodynamic parameters may be altered. We thus use the terms toxicokinetics and toxicodynamics to describe this difference. Following overdose, a drug may be absorbed over a longer period of time or its elimination half-life can increase. This is something to keep in mind during management of the patient. It may, for example, affect the decision to implement gastric decontamination and/or the choice of gastrointestinal decontamination (i.e., activated charcoal vs. whole bowel irrigation). These agents may also have different clinical effects in the setting of overdose. For example, the dihydropyridines typically cause reflex tachycardia when used therapeutically to lower blood pressure, but in overdose, significant bradycardia may occur.
Prehospital Care
Transport to an emergency department for evaluation is recommended for the following10,11:
any intentional overdose;
patients with comorbid conditions or taking other cardiovascular medications;
immediate release preparations if time since ingestion less than 6 hours;
sustained release preparations up to 8 hours for beta-blockers (12 for sotalol), 18 hours for calcium channel blockers (24 for verapamil). Remember that children can become symptomatic after ingestion of only 1-2 tablets.
En route to the hospital, depending on the capabilities of the emergency medical service10,11:
Activated charcoal can be given if unabsorbed drug is believed to still be present in the GI tract; there are no contraindications (i.e., the airway is protected).
For hypotension or bradycardia, calcium, glucagon, and epinephrine may be indicated (see doses below).
Clinical Course
Depending on the time since ingestion and the severity of the overdose, the patient may appear clinically well on presentation. Some patients will never become symptomatic; however, others may decompensate rapidly, and all patients should be monitored closely initially. Patients may complain of dizziness or nausea. Seizures or syncope may occur. In the more severe cases, a patient can present obtunded or in cardiac arrest.
The main clinical signs that will be encountered are cardiovascular. Hypotension and bradycardia are common and can be severe. They may occur simultaneously, or either can precede the other. Deterioration into cardiogenic shock is typical of severe toxicity. A variety of conduction disturbances may be seen, mainly in the form of a conduction delay. It can occur anywhere, and can be a mild interval prolongation or complete heart block. QT prolongation and torsades de pointes may be encountered with any of these medications, but most commonly are described with sotalol due to its effect on potassium channels.8
CNS findings, as mentioned above, include decreased or altered mental status, obtundation, and coma. Seizures have been reported following beta-blocker overdose, especially propranolol, because it blocks sodium channels.8,12
There are metabolic findings associated with overdose. Beta-blocker toxicity can cause hypoglycemia that seems to occur more commonly in children, and significant hyperkalemia. In contrast, calcium channel blocker toxicity typically causes hyperglycemia.
Differential Diagnosis
The differential includes toxicity from these agents:
digoxin
imidazoline (e.g., clonidine)
opiate
sedative hypnotic
cholinergic toxicity.
Emergency Department Evaluation
When obtaining the history, be as thorough as possible regarding the overdose. It is helpful to know the time of ingestion, the specific name of the medication (e. g., propranolol instead of just "beta-blocker"), the number of pills ingested, the formulation (i.e., immediate release vs. sustained release), and the dose per tablet. Ask about co-ingestants, which may include other tablets (e.g., prescribed, over-the-counter, herbals) taken in overdose, chronic medications taken as prescribed, alcohol, or illicit drugs. Patients who are on other cardioactive medication are at higher risk for morbidity overdose.8,13
With more severe intoxications, the patient may require intubation for airway protection. Place at least one peripheral IV line initially, place the patient on a cardiac monitor, and obtain a baseline ECG and a full set of vital signs. Ensure that the patient is in a room that is easily accessible and in an area where he or she can be monitored closely for decompensation. Repeat vital signs frequently. Order laboratory tests including serum electrolytes, creatinine, and a bedside fingerstick glucose level.
When the patient becomes symptomatic, management can be difficult, and it is often helpful to discuss the case with a toxicologist. Consider consulting the local poison control center early on, even if the patient is asymptomatic.
On a psychosocial level, try to elicit the cause of the overdose. Was it a medication error or a suicide attempt? In children, was the ingestion unintentional or the act of a caregiver? Psychiatry, social work, and child protective services may need to be involved.
Gastric Decontamination
Gastric decontamination may be indicated, depending on the parameters of the ingestion, with orogastric lavage, activated charcoal, and whole bowel irrigation. Good scientific evidence for the use of these techniques is currently lacking; however, depending upon the individual circumstance, they could theoretically be effective at mitigating toxicity. All of these modalities require a protected airway, either by an alert patient or placement of an endotracheal tube. Intubation may be necessary based on the patient's clinical status; however, intubation should not be done for the sole purpose of performing gastrointestinal decontamination.
Many emergency physicians believe that all forms of gastric decontamination must be done within a specific time frame, such as only performing gastric lavage within one hour of overdose. However, absorption from the gastrointestinal tract may be delayed in the overdose setting, and gastrointestinal decontamination should be considered in all potentially life-threatening overdoses. While there are a myriad of therapeutic options, there is no pure antidote for calcium channel or beta-blocker toxicity. With significant morbidity and mortality possible, early aggressive management is key. Consequently, if the patient has ingested a potentially life-threatening dose of these medications, GI decontamination can be performed, even more than 1 hour after ingestion.
In cases where orogastric lavage is believed to be ineffective or contraindicated, activated charcoal can be considered. In a human trial, volunteers took standard doses of verapamil and then took activated charcoal. The results showed that activated charcoal given immediately after ingestion decreased the amount absorbed (the area under the curve) for all formulations, while delayed activated charcoal was only able to decrease the area under the curve in a slow-release preparation.14 One case report details multiple diltiazem levels through treatment and chronicles multi-dose activated charcoal. There did not appear to be any significant reduction in the area under the curve.15 If the ingestion is serious and there are no contraindications, give activated charcoal. If the amount of drug ingested is known, the dose of activated charcoal is 10 times that dose up to a maximum of 50 g/dose. In adults, if the dose is unknown, give 1 g/kg; in children, the dose is 0.5 g/kg.
Whole bowel irrigation (WBI) is a method of washing out the gastrointestinal tract. One of the indications for WBI is the ingestion of sustained-release products. Volunteer trials and case reports have shown mixed results.16 Although it has never been proven effective, if the physician is concerned for severe toxicity, WBI is sometimes utilized when sustained-release beta-blocker or calcium channel blocker preparations have been ingested. To perform WBI, place a nasogastric tube and infuse polyethylene glycol electrolyte solution at 1-2 liters per hour. Continue until the stool is clear, and observe for pill fragments.
The decision to proceed with any type of gastric decontamination is not clear, and it is a controversial subject even among toxicologists. If there is a question, call the on-call medical toxicologist or local poison center to discuss the potential risks and benefits.
Treatment
Treatment is similar for both beta-blockers and calcium channel blockers. The focus of treatment is reversal of the cardiovascular effects. Table 1 summarizes currently recommending therapies and dosing. (See Table 1.)
Table 1: Summary of Therapeutic Options and Dosing
|
Therapy |
Dose |
|
Atropine18,19 |
Adult: 0.5-1 mg IV bolus Pediatric: 0.02 mg/kg IV bolus, min. 0.1 mg Max: 3 mg |
|
Calcium19,20 |
Adult: Calcium gluconate 10% 3 g (30 mL) IV bolus Calcium chloride 10% 1 g (10 mL) IV bolus Pediatric: Calcium gluconate 10% 60 mg/kg (0.6 mL/kg) Calcium chloride 10% 20 mg/kg (0.2 mL/kg) Max: at the adult dose Repeat as needed |
|
Glucagon19,36 |
Adult and Pediatric: Bolus: 50 mcg/kg IV over 1-2 min Drip: 1-5 mg/hour IV (generally the effective bolus dose/hour) Max: 10 mg (or more) |
|
Vasopressors19 |
Epinephrine: Adult: 1-10 mcg/min Pediatric: 0.1-1 mcg/min IV Norepinephrine: Adult: 2-20 mcg/min Pediatric: 0.1-2 mcg/kg/min IV |
|
High-dose Insulin Euglycemia39 |
Insulin bolus: 1 unit/kg IV Insulin drip: 0.5 unit/kg IV, titrate to effect Dextrose bolus: 0.5 g/kg IV Dextrose drip: 0.5 g/kg/hr IV |
|
Phosphodiesterase Inhibitors19 |
Adult and Pediatric: Milrinone: Bolus: 50 mcg/kg IV over 10 min Drip: 0.5 mcg/kg/min Inamrinone: Bolus: 0.75 mg/kg IV over 5 min Drip: 5-10 mcg/kg/min |
|
Intralipid6,57 |
Lipid emulsion 20% Bolus: 1.5 mL/kg IV, can be repeated Drip: 0.25 mL/kg/min Max: 8 mL/kg |
Intravenous Fluids and Atropine. It is standard practice to start with intravenous crystalloid fluid boluses for patients who are hypotensive, as long as no contraindication exists. In mild cases, this may be all that is needed to reverse toxicity.
Atropine is a standard agent used in the emergency department for patients with symptomatic bradycardia. By antagonism of muscarinic receptors, there may be less inhibition of adenylate cyclase in the heart, and therefore, more cAMP.8 Its efficacy is minimal, and it is unlikely to improve hemodynamics in patients with beta-blocker or calcium channel blocker overdose.17 Nevertheless, since it is readily available, there is little down side to its administration. The adult dose is 0.5-1 mg IV repeated to maximum of 3 mg;18 pediatric dose is 0.02 mg/kg IV with a minimum of 0.1 mg.19
Calcium Salts. Calcium can be given in an attempt to raise serum calcium levels to a point where it overcomes the blockade of the L-type channel by calcium channel blockers and the downstream effects of beta-blockade.20 Success of this therapy is inconsistent,15,21,22 but multiple case reports indicate that giving many boluses or prolonged infusion of calcium may improve hemodynamics.23-25 Animal studies of beta-blockers and verapamil toxicity show that calcium chloride infusion improves inotropy but heart rate is unaffected.26-28
As baseline serum calcium levels are not specifically altered by beta-blocker or calcium channel blocker toxicity, giving calcium, especially multiple boluses, will raise serum calcium levels. There are reports of giving 13 to 30 grams of calcium to patients without adverse effect.23,24 However, there are still risks associated with this treatment. There is a reported case of verapamil overdose in which the patient received 5 g of calcium gluconate, 3 g of calcium chloride, and then a calcium chloride drip at 4 g/hour. The patient later died, and the autopsy revealed that cause of death was multi-organ failure presumably secondary to calciphylaxis.29 There is also a report of extravasation of calcium chloride from a peripheral IV that resulted in skin necrosis requiring debridement and a skin graft.25
Currently, the accepted dosing for adults is an IV bolus of calcium gluconate 10% 3 g (30 mL) or calcium chloride 10% 1 g (10 mL), which can be repeated as needed.20 For children, the bolus for calcium gluconate 10% is 60 mg/kg (0.6 mL/kg) and for calcium chloride 10% it is 20 mg/kg (0.2 mL/kg), with a maximum equivalent to adult dosing.20
Glucagon. Glucagon activates adenylate cyclase, increasing cyclic AMP independently of the beta-adrenergic receptor.7,30 It increases inotropy and chronotropy in animal models.22,31,32 One animal study showed no significant difference between glucagon and vasopressin in beta-blocker overdose.33 There are no human studies on the effect of glucagon after beta-blocker or calcium channel blocker overdose; however, given the positive hemodynamic effects in animal studies, it is recommended as part of the treatment in overdose.8,34,35
Glucagon is usually dosed as an IV bolus of 50 mcg/kg (max 10 mg) pushed over 1-2 minutes followed by an infusion of that dose/hour.36 Massive doses are recommended by some. Adverse effects are mainly gastrointestinal (e.g., nausea, vomiting) and either hypo- or hyperglycemia.36
Vasopressors. If the patient is persistently hypotensive and unstable, vasopressors should be started. There are many to choose from, but unfortunately in both animal studies and human case reports, none have had high rates of success.2,6
Epinephrine and norepinephrine activate both beta- and alpha-receptors. They compete with beta-blockers, and in calcium channel blocked patients, beta activation can provide some activation of the L-type channels, and also activate downstream channels to produce contraction and increase heart rate. Also, peripheral alpha-1-receptor activation causes vasoconstriction.
Dopamine must be converted to epinephrine or norepinephrine to exert its clinical effects. When catecholamine stores have already been depleted by physiologic stress, this is less likely to be effective compared to giving epinephrine or norepinephrine.2 Invasive blood pressure monitoring, in addition to continuous telemetry, is important to help keep these effects in balance.
Isoproterenol is a non-specific beta-adrenergic agonist. Overcoming the beta-blockade may induce more peripheral vasodilation via the beta-2 receptor. Without alpha effects, this can exacerbate hypotenison. Animal studies have shown benefit over glucagon in beta-blocker toxicity, but this has not held true in human case reports.6
Dobutamine is specific for the beta-1 receptor and does not act peripherally. It may be more useful for beta-1-specific beta-blockers, like metoprolol, or in central acting calcium channel blockers, like diltiazem.
Selection of the best agent should be based on the mechanism of the agent involved in the overdose and the mechanisms of the available vasopressors. Often, more than one agent will be needed. Monitor hemodynamic parameters closely and titrate the medications accordingly.
Phosphodiesterase Inhibitors. Phosphodiesterase (PDE) inhibition leads to increased cAMP without activation of the beta-receptor.32 Animal studies show PDE inhibitors improve inotropy with little effect on heart rate.32,37 Glucagon provides similar inotropic effects with the added benefit of the chronotropic effects.37 Phosphodiesterase inhibitors also have a vasodilating effect, and this may counteract their inotropic effects on blood pressure.16 When all other therapies are failing, a trial of one of these medications (e.g., milrinone or inamrinone) may be considered.
High-Dose Insulin Euglycemia (HDIE) Therapy. High-dose insulin may be administered to increase inotropy.7,22,38 Its proposed mechanism of action is by improving calcium use in the myocytes, although the exact mechanism is unclear.7 Under normal circumstances, cardiac cells acquire energy via fatty acid oxidation, but need glucose in times of stress, such as toxin-induced cardiogenic shock.7 Insulin improves glucose utilization in the heart.7,22,38,39
There are no randomized controlled human trials. There are multiple case reports of the hemodynamic improvement after institution of HDIE.40,41 Animal studies of calcium channel blocker and beta-blocker toxicity have shown improved survival with insulin vs. placebo.7,22 Animal studies on beta-blocker toxicity have shown HDIE to be superior to both vasopressin and epinephrine.42 Human case reports of calcium channel blocker toxicity have shown HDIE improves survival, although in severe cases, other treatments should be used in conjunction.7,43
High-dose insulin administered as a 1 unit/kg bolus with a dextrose bolus of 0.5 g/kg has been described.39 This is followed by an insulin drip at 0.5 unit/kg/hr with a dextrose infusion of 0.5 g/kg/hr, which is titrated to effect.39 It can take up to 60 minutes to see improvement.39 It is given along with a dextrose infusion of 0.5 g/kg/hr.39 It is important to follow glucose and potassium levels closely during HDIE and avoid hypoglycemia and hypokalemia.
Invasive Procedures. When these treatments fail, even when the patient is in cardiac arrest, there are case reports of using an intra-aortic balloon pump44,45 or extracorporeal membrane oxygenation with success.46,47,48 If the patient is symptomatic following overdose, consult cardiology early, so that if the patient continues to deteriorate, these invasive procedures can be performed.
Controversies
Intravenous Fat Emulsion Rescue Therapy. Intravenous fat emulsion (IFE) rescue therapy has been primarily used by anesthesiologists for local anesthetic toxicity (e.g. bupivicaine), and more recently, its use has been reported in beta-blocker and calcium channel blocker toxicity. No human randomized control trials regarding treatment with IFE are available. A few animal studies of IFE in beta-blocker and calcium channel blocker toxicity show hemodynamic benefit.49-52 There are multiple human case reports in which IFE appears to have contributed to recovery from cardiogenic shock or arrest.41,49,51-54 As these are case reports, and there may exist a bias against reporting or publishing negative outcomes, human case-control studies are needed to further elucidate the efficacy of IFE in beta-blocker and calcium channel blocker overdose. At this time, given the potential benefit and despite no human randomized controlled trials performed to date, IFE may be considered for patients who are failing other modalities or during cardiac arrest.
Reported adverse effects include acute reactions such as an anaphylactoid reaction, and subacute reactions or the "fat overload syndrome" (i.e., coagulopathy, jaundice, lipid accumulation in the liver).55 One case report of iatrogenic IFE overdose describes interference with laboratory studies, but otherwise no other attributable adverse effects were noted.56
Based on previous use of intralipid for local anesthetic toxicity, the accepted dosing for IFE is a 20% lipid emulsion given as a 1.5 mL/kg bolus, followed by an infusion of 0.25 mL/kg/min for 30 minutes (not to exceed 8 mL/kg total initial dose).57
Risk Management Pitfalls for BB/CCB Toxicity
"The patient looked great on arrival. I thought he would be OK in one of the back rooms." These patients can look well on presentation and rapidly deteriorate. They should be in an area of the emergency department where they can be closely monitored.
"The child may have only ingested one pill." With this class of medications, one pill can cause serious toxicity. Extra attention needs to be paid to pediatric patients in this type of ingestion.
"The ingestion was over one hour ago. I didn't think I could do gastric decontamination." Medications taken in large overdose may have delayed or prolonged absorption, and given the risk of severe toxicity and lack of antidote, gastric decontamination can be considered after the first hour.
"One unit/kg of insulin seems too high. The patient was not diabetic." Euglycemia can be maintained when high-dose insulin is given along with dextrose and glucose if levels are closely monitored.
"I didn't know what formulation he took, but he was asymptomatic at 6 hours, so I sent him home." If the formulation is unknown, manage for the worst-case scenario and observe/admit for 18-24 hours.
Disposition
For symptomatic patients, disposition is simple. They should be admitted to a monitored unit, preferably some form of intensive/critical/cardiac care unit. The patients will require very close monitoring and may be on various cardioactive or vasoactive medications. Asymptomatic patients who have been medically cleared from the overdose may still require psychiatric admission if it was a suicide attempt. In the case of a child who is medically cleared, he or she may require a social admission if there is any concern that the medication was intentionally given to the child to cause harm.
Other than these instances, medically clearing a patient for discharge is generally based on an observation period dictated by the drug and its formulation. For most immediate-release preparations, if the patient is completely asymptomatic at 6-8 hours, it is safe to discharge him or her home.13 For these patients, the decision to hold them in the emergency department for observation or perhaps admit to an observation unit is at the discretion of the emergency physician. For sustained-release preparations, the general recommendation is a minimum observation period of 18 to 24 hours. It is best to admit these patients to a monitored setting, preferably one where the patient will be observed closely for hemodynamic decompensation.
Summary
Beta-blockers and calcium channel blockers are commonly prescribed cardiovascular medications. They are involved in a large number of overdoses every year and a number of fatalities. While no antidote exists, there are multiple therapeutic options that may be life saving. It is important for emergency physicians to recognize signs and symptoms of toxicity early and to treat patients aggressively.
Case Conclusions
Shortly after speaking with the patient who overdosed on metoprolol, the nurse informs you that his pulse was 44 and his blood pressure was 70/48. The patient becomes obtunded, and you intubate him for airway protection. You attempt gastric decontamination with orogastric lavage and activated charcoal. You order IV fluids and give atropine and calcium without improvement. After a 5 mg bolus of glucagon, vital signs improve. The patient is started on a glucagon drip and admitted to the ICU.
On your way to see the pediatric patient exposed to verapamil, you explain to the resident that the possible ingestion of 3 tablets is very serious. You tell the grandmother that while the child appears well, it is a controlled-release formulation, and the patient might not exhibit signs or symptoms of toxicity for hours to come and will require at least 24 hours of monitoring. You ask the nurse to start an IV, send basic labs, set up telemetry, and get an ECG. Then you call the nearby pediatric hospital to arrange for transfer.
References
1. Bronstein AC, Spyker DA, Cantilena LR, et al. 2008 Annual Report of the American Association of Poison Control Centers' National Poison Data System (NPDS): 26th Annual Report. Clin Toxicol 2009;47:911-1084.
2. DeRoos FJ. Chapter 60: Calcium Channel Blockers. In: Nelson LS, et al eds. Goldfrank's Toxicologic Emergencies 9th ed. New York: McGraw Hill; 2010:884-92.
3. Ranniger C, Roche C. Are one or two dangerous? Calcium channel blocker exposure in toddlers. J Emerg Med 2007;33:145-54.
4. Westfall TC, Westfall DP. Chapter 6. Neurotransmission: The Autonomic and Somatic Motor Nervous Systems. In: Brunton LL, Lazo JS, Parker KL. Goodman & Gilman's The Pharmacological Basis of Therapeutics 11e: http://www.accessmedicine.com/content.aspx?aID=954433.
5. Johnson M. The Beta-Adrenoreceptor. Am J Respir Crit Care Med 1998;158:S146-53.
6. Brubacher JR. Chapter 61: Beta-Adrenergic Antagonists. In: Nelson LS, et al eds. Goldfrank's Toxicologic Emergencies 9th ed. New York: McGraw Hill; 2010:896-909.
7. Megarbane B, Karyo S, Baud FJ. The role of insulin and glucose (hyperinsulinaemie/euglycaemia) therapy in acute calcium channel antagonist and beta-blocker poisoning. Toxicol Rev 2004;23:215-222.
8. Taboulet P, Cariou A, Berdeaux A, et al. Pathophysioogy and management of self-poisoning with beta-blockers. Clin Toxicol 1993;31:531-51.
9. Arroyo AM, Kao LW. Calcium channel blocker toxicity. Pediatr Emer Care 2009;25:532-41.
10. Olson KR, Erdman AR, Woolf AD, et al. Calcium channel blocker ingestion: An evidence-based consensus guideline for out-of-hospital management. Clin Toxicol 2005;43:797-822.
11. Wax PM, Erdman AR, Chyka PA, et al. Beta-blocker ingestion: An evidence-based consensus guideline for out-of-hospital management. Clin Toxicol 2005;43:131-146.
12. Reith DM, Dawson AH, Epid D, et al. Relative toxicity of beta blockers in overdose. Clin Toxicol 1996;34:273-8.
13. Love JN, Howell JM, Litovitz TL, et al. Acute beta blocker overdose: Factors associated with the development of cardiovascular morbidity. Clin Tox 2000;38:275-81.
14. Laine K, Kivisto KT, Neuvonen PJ. Effect of delayed administration of activated charcoal on the absorption of conventional and slow-release verapamil. Clin Toxcol 1997;35:263-8.
15. Roberts D, Honcharik N, Sitar DS, et al. Diltiazem overdose: Pharmacokinetics of diltiazem and its metabolites and effect of multiple dose charcoal therapy. Clin Toxicol 1991;29:45-52.
16. American Academy of Clinical Toxicology, European Association of Poison Centres and Clinical Toxicologists. Position Paper: Whole Bowel Irrigation. J Toxicol Clin Toxicol 2004;42:843-54.
17. Howland MA. Antidotes in depth: Atropine. In: Nelson LS, et al eds. Goldfrank's Toxicologic Emergencies 9th ed. New York: McGraw Hill; 2010:1473-6.
18. Neumar RW, Oto CW, Link MS. Part 8: Adult Advanced Cardiovascular Life Support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122:S729-67.
19. Kleinman ME, Chameides L, Schexnayder SM. Part 14: Pediatric Advanced Life Support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122:S876-908.
20. Howland MA. Antidotes in depth: Calcium. In: Nelson LS, et al eds. Goldfrank's Toxicologic Emergencies 9th ed. New York: McGraw Hill; 2010:1381-5.
21. Patel NP, Pugh ME, Goldberg S, et al. Hyperinsulinemic euglycemia therapy for verapamil poisoning: A review. Am J Crit Care 2007;16:498-503.
22. Kline JA, Leonova E, Raymond R. Beneficial myocardial metabolic effects of insuin during verapamil toxicity in the anesthetized canine. Crit Care Med 1995; 3:1251-63.
23. Hung Y-M, Olson KR. Acute amlodipine overodse treated by high dose intravenous calcium in a patient with severe renal insufficiency. Clin Toxicol 2007;45:301-3.
24. Isbister GK. Delayed asystolic cardiac arrest after diltiazem overdose; resuscitation with high dose intravenous calcium. Emerg Med J 2002;19:355-7.
25. Lam Y-M, Lau C-P. Continuous calcium chloride infusion for massive nifedipine overdose. Chest 2001;119:1280-1282.
26. Love JN, Hanfling D, Howell JM. Hemodynamic effects of calcium chloride in a canine model of acute propranolol intoxication. Ann Emerg Med 1996:28:1-6.
27. Hariman RJ, Mangiardi LM, McAllister RG. Reversal of thecardiovascular effects of verapamil by calcium and sodium: Differences between electrophysiologic and hemodynamic responses. Circulation 1979;59:797-804.
28. deWildt D, deGroot G, Sangster B. Calcium interferes with the cardiodepressive effects of beta blocker overdose in isolated rat hearts. Clin Toxicol 1986;24:111-33.
29. Sim MT, Stevenson FT. A fatal case of iatrogenic hypercalcemia after calcium channel blocker overdose. J Med Toxicol 2008;4:25-29.
30. Levey GS, Epstein SE. Activation of adenyl cyclase by glucagon in cat and human hearts. Circ Res 1969;24:151-6.
31. Farah A, Tuttle R. Studies on the pharmacology of glucagon. J Pharm Exp Ther 1960;129:49-55.
32. Sat S, Tsuji MH, Okubo N, et al. Milrinone versus glucagon: Comparative hemodynamic effects in canine propranolol poisoning. Clin Toxicol 1994;32:277-289.
33. Holger JS, Engebretsen KM, Obetz CL, et al. A comparison of vasopressin and glucagon in beta-blocker induced toxicity. Clin Toxicol 2006;44:45-51.
34. Bailey B. Glucagon in beta-blocker and calcium channel blocker overdoses: A systemaitc review. Clin Toxicol 2003;41:595-602.
35. Lucchesi BR. Cardiac actions of glucagon. Circ Res 1968;22:777-87.
36. Howland MA. Antidotes in depth: Glucagon. In: Nelson LS, et al eds. Goldfrank's Toxicologic Emergencies 9th ed. New York: McGraw Hill; 2010:910-913.
37. LoveJN, Leasure JA, Mundt DJ, et al. A comparison of amrinone and glucagon therapy for cardiovascular depression associated with propranalol toxicity in a canine model. Clin Toxicol 1992;30:399-412.
38. Tune JD, Mallet RT, Downey HF. Insulin improves contractile function during moderate ischemia in canine left ventricle. Am J Physiol 1998;274:1574-81.
39. Kerns W. Antidotes in Depth: Insulin-Euglycemia Therapy. In: Nelson LS, et al eds. Goldfrank's Toxicologic Emergencies 9th ed. New York: McGraw Hill; 2010:893-5.
40. Nickson CP, Little M. Early use of high-dose insulin euglycaemic therapy for verapamil toxicity. Med J Aust 2009;191:350-2.
41. Stellpflug SJ, Harris CR, Engebretsen KM, et al. Intentional overdose with cardiac arrest treated with intravenous fat emulsion and high-dose insulin. Clin Toxicol 2010;48:227-229.42.
42. Holger JS, Engebretsen KM, Fritzlar SJ, et al. Insulin versus vasopressin and epinephrine to treat beta-blocker toxicity. Clin Toxicol 2007;45:396-401.
43. Yuan TH, Kerns WP, Tomaszewski CA, et al. Insulin-glucose as adjunctive therapy for severe calcium channel antagonist poisoning. Clin Toxicol 1999;37:463-74.
44. Janion M, Stepien A, Sielski J, et al. Is the intra-aortic balloon pump a method of brain protection during cardiogenic shock after drug intoxication? J Emerg Med 2010;38:162-7.
45. Lane AS, Woodward AC, Goldman MR. Massive propranolol overdose poorly responsive to pharmacologic therapy: Use of the intra-aortic balloon pump. Ann Emerg Med 1987;16:1381-3.
46. Holzer M, Sterz F, Schoerkhuber W. Successful resuscitation of a verapamil-intoxicated patient with percutaneous cardiopulmonary bypass. Crit Care Med 1999;27:2818-23.
47. Kolcz J, Pietrzyk J, Januszewska K, et al. Extracorporeal life support in severe propranolol and verapamil intoxication. J Intensive Care Med 2007;22:381-5.
48. Rygnestad T, Moen S, Wahba A, et al. Severe poisoning with sotalol andverapamil. Recovery after 4 h of normothermic CPR followed by extra corporeal heart lung assist. Acta Anaesthesiol Scand 2005;49:1378-80.
49. Jamaty C, Bailey B, Laroque A, et al. Lipid emulsions in the treatment of acute poisoning: A systematic review of human and animal studies. Clin Toxicol 2010;48:1-27.
50. Tebbutt S, Harvey M, Nicholson T, et al. Intralipid prolongssurvival in a rat model of Verapamil Toxicity. Acad Emerg Med 2006;13:134-139.
51. Cave, G, Harvey M. Intravenous lipid emulsion as antidote beyond local anesthetic toxicity: A systematic review. Acad Emerg Med 2009;16:815-24.
52. Cave G, Harvey M, Castle C. The role of fat emulsion therapy in a rodent model of propranolol toxicity: A preliminary study. J Med Toxicol 2006;2:4-7.
53. Dolcourt BA, Aaron CK. Intravenous fat emulsion for refractory verapamil and atenolol induced shock: A human case report. Clin Toxicol 2008;46:619.
54. Young AC, Velez LI, Kleinschmidt KC. Intravenous fat emulsion therapy for intentional sustained-release verapamil overdose. Resuscitation 2009;80:591-3.
55. Driscoll DF. Lipid injectable emulsions: Pharmacopeial and safety issues. Pharm Res 2006;23:1959–69.
56. West P, McKeown NJ, Hendrickson RG. Iatrogenic lipid emulsion overdose in a case of amlodipine poisoning. Clin Toxicol 2010;48:393-6.
57. Weinberg GL. Treatment of Local Anesthetic Systemic Toxicity (LAST). Reg Anesth Pain Med 2010;35:188-93.
You have just taken sign-out when a nurse comes up to you and says that there is a 64-year-old man in the critical bay who took an overdose of his medications. The patient has a history of hypertension, atrial fibrillation, and depression. The patient is lethargic but arousable, and reports he took about 40 tablets of immediate-release metoprolol three hours ago in an attempt to "end it all." The nurses are starting IV lines, checking vitals, and putting the patient on the monitor. You wonder, "Is it too late for gastric decontamination? If he is symptomatic, which therapy will I try first, and what are my options?"Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
