Drug Criteria & Outcomes
Zoledronic acid (Zometa) drug evaluation
By Anna Morton, PharmD Candidate
Harrison School of Pharmacy Auburn (AL) University Clinical rotation at Huntsville (AL) HospitalIntravenous bisphosphonates
Zoledronic acid (Zometa) — Novartis
Pamidronate disodium (Aredia) — Novartis
Mechanism of action
Zoledronic acid inhibits osteoclast activity while inducing osteoclast apoptosis. By binding to the bone, osteoclastic resorption of mineralized bone and cartilage also is blocked. In addition, zoledronic acid inhibits the skeletal calcium release and osteoclast activity induced by tumors. Zoledronic acid is 100-850 times more potent than pamidronate at inhibiting bone resorption.
Indications
• Zoledronic acid:
— Hypercalcemia of malignancy
— Multiple myeloma and bone metastases of solid tumors
• Pamidronate:
— Hypercalcemia of malignancy
— Osteolytic bone metastases of breast cancer and myeloma
— Paget’s disease
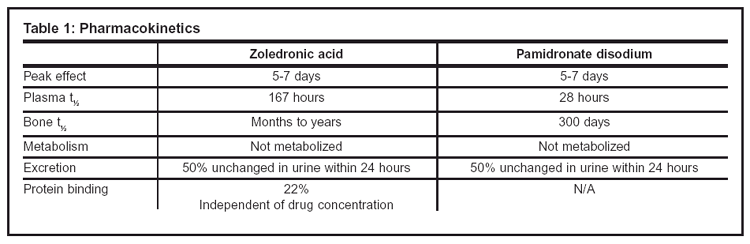
Pharmacokinetics
The pharmacokinetic parameters of zoledronic acid and pamidronate are similar (see Table 1). The pharmacokinetics have not been extensively studied in patients with hepatic or severe renal dysfunction.
Dosing
For hypercalcemia of malignancy, zoledronic acid is dosed 4 mg intravenously over no less than 15 minutes (see Table 2, for comparative dosing information). Doses are not recommended to exceed 4 mg. In clinical trials, patients receiving infusions shorter than 15 minutes were more likely to experience renal toxicity. When treating multiple myeloma and metastatic bone lesions, treatment should be administered every three to four weeks. Pamidro-nate has a dosage range from 30 mg to 90 mg depending on the indication. Pamidronate infusions times beyond two hours may reduce the risk of renal toxicity in hypercalcemic patients. The infusion rate is significantly longer than zoledronic acid with times ranging from two to 24 hours. At least seven days should be allowed to show a full response before retreatment with either agent is considered.
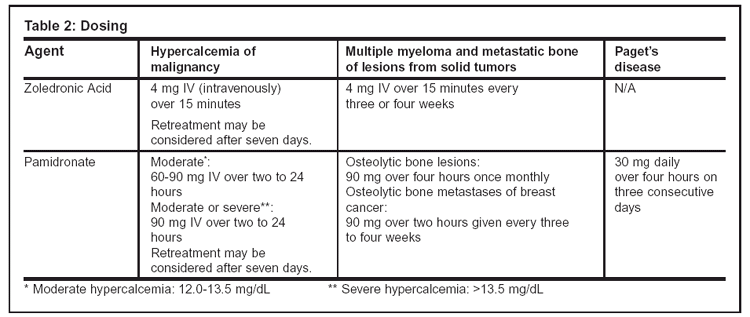
Table 2: Administration
Patients should be adequately rehydrated with saline hydration prior to every administration of either agent. An attempt should be made to restore urine output to approximately 2 L/day throughout the treatment period.
Caution should be taken to not overhydrate, particularly in cardiac failure patients. Diuretic therapy should be avoided until hypovolemia is corrected. Table 3, below, outlines administration guidelines for both agents.
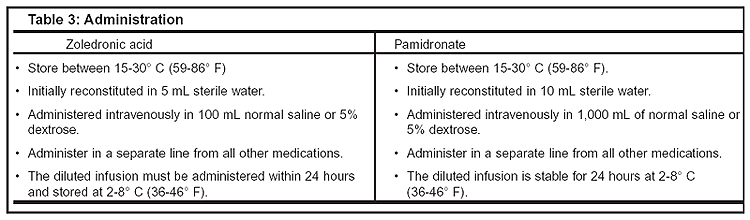
Table 3: Contraindications
Zoledronic acid and pamidronate are contraindicated in patients with clinically significant hypersensitivity to the agent, other bisphosphonates, or any excipients used in the formulation.
Warnings/precautions
Pharmacokinetic and safety data are limited in renally impaired patients. Zoledronic acid trials excluded patients with serum creatinine (SrCr) levels greater than 3 mg/dL, and pamidronate trials excluded patients with SrCr greater than 5 mg/dL. Zoledronic acid is not recommended for the treatment of bone metastases in renally impaired patients, and the risks and benefits must be evaluated before treating a hypercalcemic, renally impaired patient. Patients with renal impairment should be carefully monitored when treated with pamidronate. Neither drug should be used in pregnancy or in nursing women. Limited data are available regarding the use of zoledronic acid in patients with hepatic insufficiency. Although not observed in clinical trials, other bisphosphonates have been associated with bronchoconstriction in patients with aspirin-sensitive asthma. Table 4, provides a checklist of warnings and precautions.
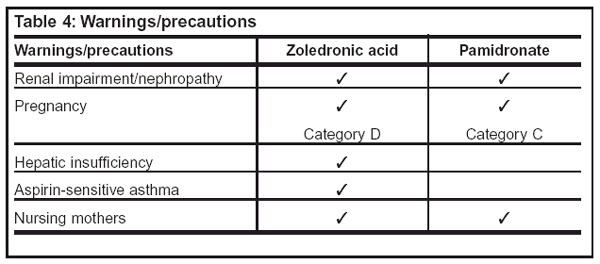
Table 4: Drug interactions
Few drug interactions have been identified with either agent (see Tables 5 and 6). Zoledronic acid appears to have more additive drug interactions when combined with aminoglycosides, loop diuretics, and thalidomide. Neither agent should be combined with calcium-containing IV fluids.
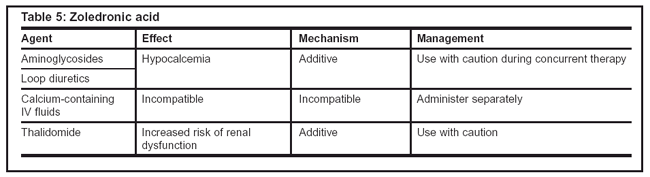
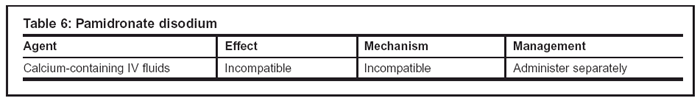
Table 6: Adverse effects
The adverse effect profiles of both agents are comparable (see Table 7). Renal toxicity is the most serious adverse event reported with both agents.
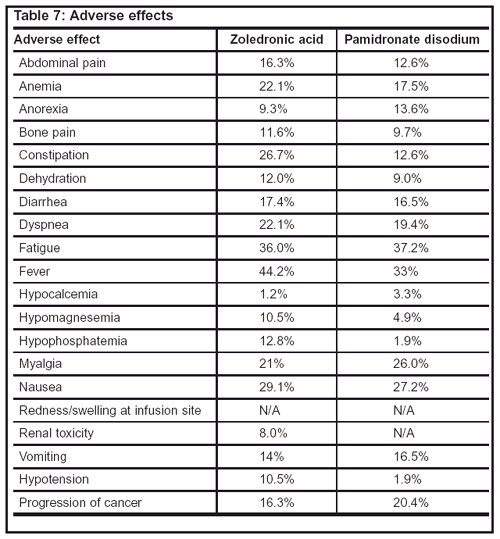
Table 7: Monitoring parameters
Patients treated with either zoledronic acid or pamidronate should be monitored prior to each treatment for renal function, serum calcium, serum phosphate, serum magnesium, serum electrolytes, complete blood count with differential, and hemoglobin and hematocrit.
Clinical Trials
Trial 1: Body JJ, Lortholary A, Romieu G, et al. A dose-finding study of zoledronate in hypercalcemic cancer patients. J Bone Miner Res 1999; 14: 1557-1561.
Objective: To determine the most effective dose of zoledronic acid that was nontoxic and could induce normocalcemia in at least 80% of patients with hypercalce-
mia of malignancy following rehydration.
Study design: An open-label, dose-finding, Phase I study consisting of 33 hypercalcemic cancer patients.
Intervention: Patients received doses of 0.002, 0.005, 0.01, 0.02, or 0.04 mg/kg zoledronate in a single dose infused over a median time of 30 minutes.
Patient population:
• Inclusion criteria:
— Persistence of tumor-induced hypercalcemia (TIH) after 24 hours of intravenous rehydration with 2-3 L of saline. TIH was defined as Ca++ > 11 mg/dL.
• Exclusion criteria:
— Serum creatinine greater than 1.5 times the upper limit of normal after rehydration.
— Treatment with bisphosphonates within the previous six months.
— Treatment with any agent capable of influencing Ca++ levels within previous one month.
— Hypercalcemia as a "flare" reaction within two weeks of initiating endocrine therapy.
— Chemotherapy within previous seven days.
Outcomes measured: The primary efficacy variable was the response to each dosage level.
Results: The two most effective doses were 0.02 mg/kg and 0.04 mg/kg.
- With a dose of 0.02 mg/kg, 100% (5/5) of patients became normocalcemic.
- With a dose of 0.04 mg/kg, 93% (14/15) of patients became normocalcemic.
- At the lower doses, only 30% (3/10) of patients became normocalcemic.
Strengths: Inclusion and exclusion criteria were appropriate and clearly defined.
Limitations:
- Open-label trial design allows for potential biases.
- No placebo control group.
- Small patient population.
- Financial support from Novartis.
Authors’ conclusions: Small doses of zoledro-nate (0.02 mg/kg and 0.04 mg/kg) administered over a short infusion were effective in treating patients with TIH. Zoledronate was well tolerated.
Trial 2: Major P, Lortholary A, Hon J, et al. Zoledronic acid is superior to pamidronate in the treatment of hypercalcemia of malignancy: A pooled analysis of two randomized, controlled clinical trials. J Clin Oncol 2001; 19:558-567.
Objective: To compare the safety and efficacy of pamidronate and zoledronic acid in treating hypercalcemia of malignancy.
Study design: Two concurrent, identical, parallel, randomized, double-blind, double-dummy, multicenter trials consisting of 185 patients.
Intervention: Subjects were randomized to receive either a single IV dose of zoledronic acid (4 mg or 8 mg) by five-minute infusion, or pamidronate disodium (90 mg) by two-hour infusion. The bisphosphonate was administered concomitantly with IV hydration. Refractory patients and patients that relapsed 56 days after initial treatment with either agent were retreated with a single 8 mg dose of zoledronic acid over five minutes and were followed for 28 days.
Patient population:
• Inclusion criteria:
— 18 years of age or older.
— Histologic or cytologic confirmation of cancer and severe hypercalcemia of malignancy (HCM) (corrected serum calcium [CSC]) > 3.00 mmol/L; 12.0 mg/dL.
— Written informed consent.
• Exclusion criteria:
— History of allergic reaction or sensitivity to a bisphosphonate.
— Treated with a bisphosphonate for hypercalcemia within 90 days.
— Treated with a bisphosphonate for other complication within 30 days.
— Serum creatinine greater than 4.5 mg/dL.
— Treated with calcitonin within 72 hours.
— Treated with mithramycin, antineoplastic cytotoxic chemotherapy, or hormone therapy within seven days.
— Treated with gallium nitrate within 14 days.
— Treated with any investigational drug within 30 days.
— Severely dehydrated.
— Could not tolerate IV hydration.
— Hyperparathyroidism.
— Adrenal insufficiency.
— Vitamin D intoxication.
— Milk alkali syndrome.
— Sarcoidosis.
— Granulomatous disease.
— Multiple endocrine neoplasia syndrome.
Outcomes measured:
• Primary:
— Patients were evaluated for complete response (CR) of normalization of CSC to < 10.8 mg/dL by day 10.
• Secondary:
— Time, in days, to relapse (CSC > 10.8 mg/dL) of HCM.
— Duration of CR (time from a CR to last CSC < 10.8 mg/dL).
— Duration of response (time from CR to last CSC < 2.9 mg/dL).
— Efficacy of zoledronic acid 8 mg used as re-treatment for relapse and refractory patients.
— Effects on the biochemical markers of bone resorption.
Results:
• Normalization of CSC occurred by day four in half of the zoledronic acid group and one-third of the pamidronate group.
• Mean CSC levels at days four, seven, and 10 were lower (P < 0.05) in patients treated with either dose of zoledronic acid when compared to pamidronate.
• Serum Calcium Normalization (CR rates, see Table 8).
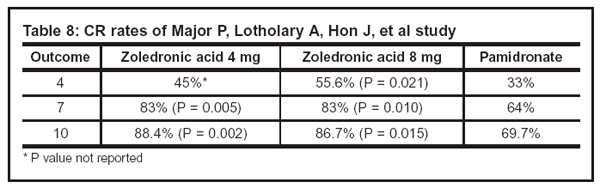
Table 8
• The median time to relapse was 30 days for the zoledronic acid 4 mg group (P = 0.001), 40 days for the 8 mg groups (P = 0.007), and 17 days for the pamidronate group.
• The median CR durations were 32 days for
the zoledronic acid 4 mg group, 43 days for the 8 mg groups, and 18 days
for the pamidronate group.
• Following retreatment with 8 mg zoledronic acid, the mean CSC decreased from 12.68 mg/dL to 10.84 mg/dL by day 10. The median time of CR was 10.5 days, the median time of response was 15 days, and the median time to relapse was eight days.
• The most common adverse events were fever, anemia, nausea, constipation, and dyspnea. The occurrences were similar between all groups. Renal adverse events occurred in two patients in the zoledronic acid 4 mg group, five patients in the zoledronic acid 8 mg group, and three patients in the pamidronate group.
Strengths:
• Randomized, double-blind, double-dummy, multicenter trials.
• Inclusion and exclusion criteria were appropriate and clearly stated.
• Large patient population.
• Patient demographics similar at baseline.
Limitations:
• Results reported as a combined analysis with no recording of individual trial data.
• A priori sample size analysis was reported, but the zoledronic acid 4 mg did not meet the 90-subject set point.
• Patient population predominantly male and Caucasian.
• Supported by Novartis.
Author’s conclusions: Intravenous zoledronic acid is superior and more convenient than pami-dronate when treating hypercalcemia of malignancy. The safety profile of both agents is similar.
Trial 3: Berenson J, Rosen L, Howell A, et al. Zoledronic acid reduces skeletal-related events in patients with osteolytic metastases. Cancer 2001; 91:1191-1200.
Objective: To compare pamidronate with three different doses of zoledronic acid regarding the need for radiotherapy to bone metastases.
Study design: A randomized, controlled, double-blind trial lasting 10 months and including 280 patients.
Intervention: The trial consisted of three doses of zoledronic acid infused over five minutes. The studied doses were 0.4 mg, 2 mg, and 4 mg. As an internal control for tolerability and efficacy, a two-hour infusion of 90 mg pamidronate was used.
Patient population:
• Inclusion criteria:
— A histological confirmed diagnosis consistent with metastatic breast cancer or multiple myeloma.
— Radiologic evidence of a minimum of one osteolytic lesion.
— Life expectancy of at least 10 months.
— Eastern Cooperative Oncology Group (ECOG) performance status of 0, 1, or 2.
• Exclusion criteria:
— Osteolytic lesions only present in areas of previous radiation.
— Previous bisphosphonate therapy.
— Participation in a previous pamidronate protocol.
— Other investigational drug use within 30 days.
— History of hypercalcemia.
— History of bisphosphonate allergy or sensitivity.
— Scheduled for or recently undergone orthopedic surgery or radiation therapy within two weeks of study entry.
Outcomes measured:
• Primary:
— Frequency of radiation to bone — less than 30% receiving radiation to bone.
• Secondary:
— Other skeletal related events.
— Bone mineral density.
— Safety.
— Bone markers.
The primary and secondary results of this study are summarized in Table 9, below.
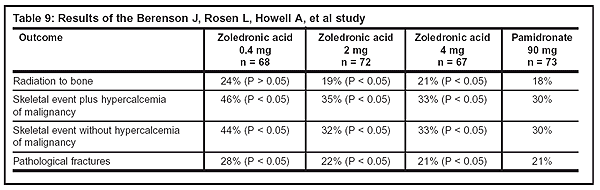
Strengths:
• A randomized, controlled, double-blind trial.
• Length of trial appropriate to assess outcome.
• Inclusion and exclusion criteria appropriate and clearly stated.
Limitations:
• Population predominantly female.
• Several patients dropped out of the study, but the data were not reported.
• Financial support from Novartis.
Authors’ conclusions: Zoledronic acid infusions of 2.0-4.0 mg given over a five-minute infusion are as effective as a two-hour infusion of pamidronate in the treatment of osteolytic bone metastases. Both agents were well tolerated.
Trial 4: Rosen L, Gordon D, Kaminski M, et al. Zoledronic acid versus pamidronate in the treatment of skeletal metastases in patients with breast cancer or osteolytic lesions of multiple myeloma: A Phase III, double-blind, comparative trial. Cancer J 2001; 7:377-387.
Objective: To compare zoledronic acid to pamidronate in the treatment of osteolytic or mixed bone metastases/lesions.
Study design: A Phase III, double-blind, randomized, double-dummy, multicenter, international, parallel-group study lasting 12 months. The trial included 1,648 patients.
Intervention: Patients were randomized to receive IV infusions of either zoledronic acid (4 mg or 8 mg) or pamidronate (90 mg). The infusions were given every three to four weeks for 12 months. Initially, the zoledronic acid was given over five minutes and diluted in 50 mL of hydration solution, but was changed to a 15-minute infusion with 100 mL of hydration solution due to concerns of renal safety. The concerns of renal safety at higher doses also prompted the 8 mg treatment group to be changed to 4 mg. All patients received a 500 mg calcium supplement daily and a multivitamin with 400-5,000 units of vitamin D.
Patient population:
• Inclusion criteria:
— 18 years of age or older.
— Durie-Salmon stage III multiple myeloma.
— At least one osteolytic bone lesion or breast cancer patients with at least one bone metastasis.
• Exclusion criteria:
— Received treatment with any bisphosphonate during the 12 months prior to screening.
— Hypercalcemia.
— Serum creatinine > 3 mg/dL.
— Bilirubin > 2.5 mg/dL.
— Pregnant or lactating females.
— History of noncompliance with medical regimens.
Outcomes measured:
• Primary efficacy variable:
— Proportion of patients that experienced at least one skeletal related event (SRE).
• Secondary efficacy variables:
— The proportion of patients that experienced any SRE.
— Time to the first SRE.
— Skeletal morbidity rate (SMR).
— Proportion of patients experiencing individual SREs.
— The time to progression of bone metastasis.
— Objective bone lesion response.
— The time to overall progression of disease.
— ECOG performance status.
— Analgesic and pain scores.
— Bone resorption and formation markers.
— Primary and secondary efficacy results are summarized in Tables 10 and 11.
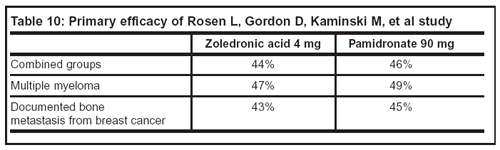
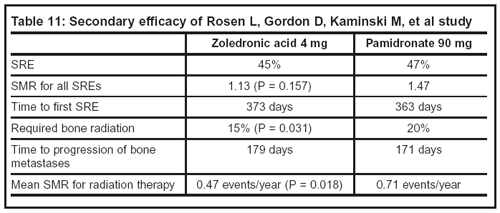
Limitations:
• Majority of patient population were female.
• Financial support from Novartis.
Strengths:
•A phase III, double-blind, randomized, double-dummy, multicenter, international, parallel-group study.
• A priori sample size analysis was reported and criteria were met.
• Patient demographics similar at baseline.
• Intention-to-treat analysis utilized.
Author’s conclusions: Zoledronic acid given as a 4 mg, 15-minute infusion was well tolerated and as effective as pamidronate 90 mg in the treatment of osteolytic lesions of multiple myeloma.
Further clinical trials: Zoledronic acid has a promising future in the treatment of osteolytic bone metastases, Paget’s disease, and osteoporosis, but further clinical studies are needed to define its place in therapy. There are ongoing trials evaluating zoledronic acid and pamidronate in metastatic bone disease due to breast cancer or multiple myeloma. Several larger studies have been proposed to determine the efficacy and safety of zoledronic acid in the prevention of disease recurrence in patients with node-positive breast cancer and the prevention of bone metastases in breast cancer and prostate cancer.
Two previous trials have shown zoledronic acid efficacy in Paget’s disease; however, a trial comparing the agent to pamidronate has yet to be performed. One study has evaluated zoledronic acid as a single annual injection for the treatment of postmenopausal osteoporosis. The study was a placebo-controlled trial enrolling 351 patients with T scores less than -2. Zoledronic acid was administered at does of 0.25 mg, 0.5 mg, and 1 mg every three months, 4 mg as a single annual dose, and 2 mg every six months. Increases of bone mineral density were shown at the hip and spine with all dosage regimens, but further clinical trials are warranted to define efficacy and place in therapy.
Summary and recommendations: Advantages of zoledronic acid over pamidronate include indications for metastasis of all solid tumors, a shorter and more convenient infusion duration, a longer therapeutic effect, and a higher response rate. However, the significantly higher cost of zoledronic acid compared to generic pamidronate is a disadvantage (see Table 12), which cannot be ignored.
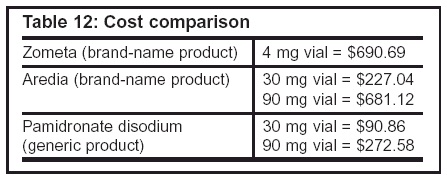
Table 12: Cost comparison:
• Hypercalcemia of malignancy:
— Zoledronic acid is effective and safe for the treatment of moderate-to-severe hypercalcemia of malignancy.
— Clinical trials have shown zoledronic acid to be superior to pamidronate in treating hypercalcemia by lowering serum calcium at a faster rate and by maintaining normalization for longer periods of time.
— Patients with hypercalcemia of malignancy do not receive scheduled infusions of bisphosphonates. Initiation of therapy is based on laboratory results and signs and symptoms of the disease.
— Most patients receive infusions as inpatient therapy so the cost and time savings associated with the shorter infusions may not be as pertinent when compared to outpatient therapy. However, the increased efficacy warrants the use in both settings.
— Patients with a SrCr greater than 1.8 mg/dL should not receive zoledronic acid infusions. Orders for zoledronic acid should be changed to pamidronate 90 mg infused over four hours.
• Multiple myeloma and metastatic bone lesions from solid tumors:
— Zoledronic acid has been proven equivalent to pamidronate for this indication. Superiority has not been shown at this point and therefore pamidronate should remain the formulary agent for this indication.
— Most infusions are scheduled and occur in the outpatient setting.
— Orders for zoledronic acid 4 mg over 15 minutes should be changed to pamidronate 90 mg over two to four hours given once monthly.
• Paget’s disease:
— There are ongoing trials studying zoledronic acid for this indication, but it has not received FDA approval at this time.
— Orders written for zoledronic acid 4 mg IV over 15 minutes should be changed to pamidronate 30 mg daily over four hours on three consecutive days.
• Postmenopausal osteoporosis:
— Neither zoledronic acid nor pamidronate have indications in postmenopausal osteoporosis.
— Zoledronic acid has shown efficacy for this indication but has not been approved at this point.
— Zoledronic acid should not be used as inpatient therapy for osteoporosis.
Resources
• Anon. Zoledronate (Zometa). Med Lett Drugs Ther 2001; 43:110-111.
• Berenson J, Rosen L, Howell A, et.al. Zoledronic acid reduces skeletal-related events in patients with osteolytic metastases. Cancer 2001; 91:1191-1200.
• Bisphosphonates. Drug Facts and Comparisons. St. Louis: Facts and Comparisons 2003; 279-286b.
• Body J, Lortholary A, Romieu G, et al. A dose-finding study of zoledronate in hypercalcemic cancer patients. J Bone Miner Res 1999; 14:1557-1561.
• Cada D, Levien T, Baker D. Zoledronic Acid for Injection. Hospital Pharmacy 2001;36:1-8.
• Major A, Lortholary A, Hon J, et al. Zoledronic acid is superior to pamidronate in the treatment of hypercalcemia of malignancy: A pooled analysis of two randomized, controlled clinical trials. J Clin Oncol 2001;19:558-567.
• Novartis Pharmaceutical Corp. Pamidronate disodium (Aredia) package literature. Cambridge, MA; 2002.
• Novartis Pharmaceutical Corp. Zoledronic acid (Zometa) package literature. Cambridge, MA; 2002.
• Pamidronate disodium. In: McEvoy GK, ed. AHFS 2002: Drug Information. Bethesda, MD: American Society of Health System Pharmacists; 2002:3697-3700.
• Personal communication. Cindy Hall, Buyer, Pharmacy Department. Huntsville (AL) Hospital System; January 2003.
• Rosen L, Gordon D, Kaminski M, et al. Zoledronic acid vs. pamidronate in the treatment of skeletal metastases in patients with breast cancer or osteolytic lesions of multiple myeloma: A Phase III, double-blind, comparative trial. Cancer J 2001; 7:377-387.
• Zoledronic acid. In: McEvoy GK, ed. AHFS 2002: Drug Information. Bethesda, MD: American Society of Health System Pharmacists; 2002:3739-3740.
Zoledronic acid (Zometa) drug evaluationSubscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
