Noninvasive Ventilation in the Emergency Department
Authors: Larissa I. Velez, MD, Assistant Professor, Assistant Director of Emergency Medicine Residency Program, University of Texas Southwestern Medical Center, Dallas; Fernando L. Benitez, MD, Assistant Professor, Emergency Medicine, University of Texas Southwestern Medical Center, Dallas; and Shameem R. Nazeer, MD, Assistant Professor, Emergency Medicine, University of Texas Southwestern Medical Center, Dallas.
Peer Reviewers: Sandra Schneider, MD, FACEP, Professor and Chair, Department of Emergency Medicine, University of Rochester Medical Center, Rochester, NY; and O. John Ma, MD, Vice Chair, Department of Emergency Medicine, Truman Medical Center, and Associate Professor, University of Missouri—Kansas City School of Medicine.
The emergency physician is faced with a wide variety of acute respiratory emergencies in daily practice. Efforts to preserve the patient’s airway and breathing capabilities result in a barrage of therapies and diagnostics aimed at identifying and reversing the cause of the respiratory distress. The need for endotracheal intubation (ETI) is a question that is addressed early and repeatedly during the patient’s course in the emergency department (ED). ETI is invasive and not without complications. The benefit of intubation often must be weighed against substantial risks of a prolonged and complicated intubation, prolonged sedation and paralysis, and subsequent pneumonia and barotraumas during hospitalization. In the patient who is obviously becoming altered or agitated, little else can be done. Often, however, there is the patient who is in moderate to severe respiratory distress but may improve substantially, and avoid intubation, with respiratory assistance in addition to continued therapy. In the patient who is alert and cooperative with impending respiratory failure, non-invasive ventilation provides a hopeful alternative.
Noninvasive ventilation (NIV) is a means of delivering positive pressure ventilation without the use of an endotracheal tube. It offers the obvious benefit of preserving the patient’s ability to speak, eat, and cough. Its less invasive character reduces the need for sedation, potential airway trauma, and risk of nosocomial pneumonia. Recent interest in this mode of ventilation has generated some guidelines for selecting patients, predicting success, and weaning from NIV. Clearly, NIV is a powerful therapeutic tool in the hands of an informed physician.
This article will delineate the physiology of NIV. It will provide guidelines for initiation, weaning, and possible complications of NIV. It also will highlight current research in the topic.—The Editor
Introduction
NIV is defined as the delivery of airway support without the use of an artificial airway such as an endotracheal tube.1,2 It generally uses the patient’s upper airway and a mask for the delivery of positive pressure.3 It has some definitive benefits over the conventional ETI, such as the preservation of speech, feeding capabilities, and the cough reflex. It reduces the common problems associated with ETI, such as nosocomial pneumonia. In addition, the requirement for sedation is minimized or even completely avoided when NIV is used.4
The use of NIV slowly has been increasing in the United States, somewhat lagging behind the research efforts and more widespread use of this modality in Europe. Most of the early literature on NIV comes from Europe, where it has been used in the intensive care setting, in specialized respiratory wards, and in the emergency setting. In a 1998 United Kingdom survey on the availability of NIV, researchers found that this treatment modality was available in 48% of the hospitals surveyed. The main reasons given for not offering this service in the remaining institutions were cost and lack of training on the technique.5
The Basics of Noninvasive Ventilation
Pathophysiology. NIV affects the mechanics of respiration in many ways. It increases functional residual capacity and helps recruit underventilated or collapsed alveoli. This results in improved oxygenation. NIV also can increase the tidal volume, helping decrease the CO2. The net effect is a decreased work of breathing, which allows the respiratory muscles to rest.6-8 During increased work of breathing, large negative intrathoracic pressures are generated. Positive pressure ventilation reduces these large negative pressure swings. This is thought to decrease afterload, thus increasing cardiac output. However, in some cases, NIV can result in decreased cardiac output by virtue of decreasing venous return.9 Furthermore, positive pressure ventilation offsets the intrinsic positive end-expiratory pressure (PEEP), or "auto-PEEP," that occurs in chronic obstructive pulmonary disease (COPD) exacerbations. This offsetting of the intrinsic PEEP further decreases the work of breathing.10 In patients with pulmonary edema, NIV can help reduce ventilation perfusion mismatches.11
Modalities of Noninvasive Ventilation. There are three basic modalities for NIV. The first one is continuous positive airway pressure ventilation, or CPAP. It provides a continuous positive pressure during the complete respiratory cycle. CPAP mainly is used to improve oxygenation by recruiting underventilated or collapsed alveoli.12
The second modality is bilevel positive airway pressure ventilation (BiPAP). It provides different levels of pressure during inspiration and exhalation. BiPAP is a combination of CPAP throughout the respiratory cycle coupled with a boost of pressure during inspiration. The inspiratory pressure is called inspiratory positive airway pressure or IPAP, and the expiratory pressure is termed expiratory positive airway pressure or EPAP. The difference between the IPAP and the EPAP is the true pressure support. The IPAP assists in improving tidal volume, thus improving pCO2. The EPAP helps recruit more alveoli, similar to the PEEP in a mechanical ventilator, thus improving oxygenation.
There are two modes in BiPAP. In the spontaneous mode, the IPAP is triggered by the patient’s inspiratory effort. Once the airway pressures reach a limit, the machine starts delivering the EPAP. There is no spontaneous triggering by the machine unless the patient initiates a breath. In the spontaneous timed mode, the operator sets a time limit so the machine will initiate the delivery of IPAP if the patient has not initiated a breathing effort spontaneously. One difficulty with this mode is that it leads to patient-machine asynchrony, which limits tolerance to the NIV. The main benefit of BiPAP is that it is better tolerated than CPAP. This is because the differential in pressure between inspiration and expiration allows for better patient-ventilator synchrony and thus more comfort.
Pressure support ventilation (PSV) is a less commonly used modality where only inspiratory pressure is provided. It works by augmenting tidal volume. The pressure support is triggered when the patient makes an inspiratory effort and is terminated when the inspiratory flow falls below a predetermined rate. There is no flow (support) during exhalation.12 In this mode, the patient determines both the rate and the duration of the inspiration. Most of the studies on NIV have used either the CPAP or BiPAP modes. Thus, the information on the utility of PSV in the emergency setting is scarce. This article will focus discussion on CPAP and BiPAP.
Patient Interfaces. Many different interfaces are being designed and tested, but the two most frequently used are the nasal mask and the full face (oronasal) mask. All masks have a soft cushion that prevents skin necrosis and additional patient discomfort. They are attached to the patient by straps that should not be applied too tightly.10 Placing two fingers between the strap and the scalp is a good measure of an adequate fit.
The nasal mask usually results in more air leaks but is more comfortable for claustrophobic patients. This type of mask requires a cooperative patient who can keep a closed mouth.
The full face mask delivers higher ventilation pressures without leaks, allows for mouth breathing, and requires less patient cooperation. However, it is not as well tolerated by claustrophobic patients, is less comfortable, impedes speech and communication, and limits oral intake.1,3,13 In a recent study comparing the nasal to the oronasal mask for patients with acute respiratory failure, both masks performed well, but the oronasal mask was better tolerated.14 The full face mask generally is the first choice in the emergency setting.
Other less commonly used types of interfaces include the total face mask, which covers the whole face from the forehead to the chin. This results in the relief of pressure from sensitive facial structures such as the nasal bridge.10 Finally, the nasal "pillows" are rubber or silicone pledgets that are inserted in the nostrils. They can be used in patients who develop skin problems due to mask pressure at the nasal bridge.10
Patient Selection. Indications. Patients with respiratory distress requiring support first must be identified. Clinical parameters used to identify these patients include: respiratory rate (RR) > 24/minute, accessory muscle use, paradoxical breathing, hypercarbia (pCO2 > 45 mm Hg), acidemia (pH < 7.35), or hypoxemia (pO2/FiO2 < 200).10,15 NIV should not be used in patients who just have mild respiratory distress. In addition, the cause for the respiratory failure must be one amenable to treatment with NIV, such as acute exacerbations of COPD, asthma, decompensated heart failure, acute pulmonary edema, pneumonia, and pulmonary contusions.
Contraindications. Most of the contraindications for NIV are intuitive. They include hemodynamic instability, cardiac or respiratory arrest, severe acidosis, vomiting, obstructed bowel, absent gag reflex, coma, confusion, inability to tolerate the device, inability to handle secretions, facial surgery, facial trauma, facial deformity, upper airway obstruction, and high risk for aspiration.1,16 Other contraindications to NIV include severe epistaxis, pre-existing bullous lung disease, pneumomediastinum, pneumothorax, life-threatening arrhythmias, and evidence of cardiac ischemia (for BiPAP). However, these contraindications are not absolute, and they derive from the fact that most NIV trials excluded these patients.
Extremes of age are not a contraindication for NIV, and there are several reports of its successful use in both the pediatric and the elderly populations.17-19 In one of the studies, NIV using a nasal mask was applied to 30 patients with acute respiratory failure. The median age was 76 years. NIV was successful in 60% of these cases.19 Of importance, NIV requires patient cooperation, which may be relatively difficult in the very young or very old.
Table 1 summarizes the generally accepted contraindications for noninvasive ventilatory support.
|
Initiation of Therapy. Once a decision has been made to initiate NIV, the mode of ventilation should be selected. Use CPAP if the main problem is hypoxemia, and favor BiPAP if the main problem is hypercarbia. The use of PSV will not be discussed, since most of the available information focuses on either CPAP or BiPAP. Although conventional ventilators can be used, there are several ventilators on the market that specifically are designed to deliver CPAP, BiPAP, or both.
The second step involves selecting the interface. Have several types of masks available in different sizes so that an adequate fit can be achieved. Hold the mask in place without securing it at first. Do this until the patient is synchronized with the ventilator. Once this synchrony is achieved, secure the mask with straps, avoiding a tight fit.10 In the elderly, dentures can be left in place. They will allow for a better mask fit and less air leakage. Loose dentures, however, can pose a risk of aspiration and should be removed.3
The initial NIV parameters should provide a pressure support of 5 cm H2O for CPAP. Start BiPAP with an IPAP of 8-10 cm H2O and an EPAP of 3-5 cm H2O. Increase these parameters gradually, usually by 2 cm H2O at a time, until an exhaled tidal volume of 10-15 mL/kg is achieved and the respiratory rate falls below 25 breaths per minute.2 Increases in IPAP predominantly will affect pCO2, and increases in EPAP predominantly will affect pO2. One important concept to remember is that for BiPAP, the inspiratory component always must be higher that the expiratory component. If IPAP equals EPAP, then the machine just behaves like a CPAP, since the same pressure is provided throughout the entire respiratory cycle.
The FiO2 is titrated to maintain oxygen saturation above 90%. Humidification and heating generally are not required since the airway mechanisms remain intact.
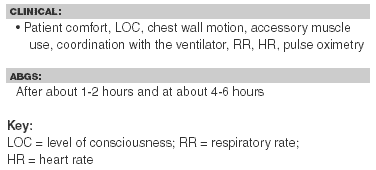
Adequate monitoring of patients receiving NIV includes closely following the vital signs, cardiac monitoring, and pulse oximetry. Perform serial clinical examinations that include patient comfort with the device, mental status, work of breathing, and handling of secretions. The arterial blood gases are a useful tool to assess the adequacy of treatment. Chest radiographs should be done daily or whenever there is a sudden change in the clinical status of the patient. Table 2 summarizes the monitoring parameters for patients being treated with NIV.
|
Patients who benefit from NIV have improvements of the dyspnea and of the signs of respiratory distress. These changes can be seen as early as one hour after the initiation of NIV. A rapid decrease in the respiratory rate is a very good indicator of successful treatment.20,21 Other signs of success include improvements in the level of consciousness, oxygen saturation, and the arterial blood gas parameters.22
The total duration of NIV depends on the cause for the respiratory failure. In some cases, it is only a few hours, but in most cases, the support needs to be delivered over several days. Many authors suggest the use of ventilator-free breaks.10 These breaks serve a dual purpose. Patients who do not have recurrence of the respiratory distress during the break can be weaned off the NIV. The breaks also allow for delivery of oral medications, patient feeding and drinking, and relief from the constant mask pressure on the face.
Several authors have tried to establish characteristics of patients that will predict success or failure of NIV. Items reflecting increased success include: younger age, good dentition, normal mental status, adequate cooperation, ability to achieve synchrony with the ventilator, lower acuity of illness, moderate hypercarbia, moderate hypoxemia, and improvement in clinical and laboratory parameters within two hours of treatment initiation.10
Discontinuation of Therapy. Although NIV is effective for many patients, it is important to know when to discontinue it and move to invasive airway management. Indicators of NIV failure include patient intolerance for NIV; deterioration of the vital signs; failure to improve after 1-2 hours in NIV; progressive confusion or sedation; inability to handle secretions; chest pain; arrhythmias; and apnea.20,23,24 In several studies, the degree of initial acidosis has been one of the best predictors for a failed NIV attempt.16,25 Increased illness severity scores also have been found to correlate with failure of NIV in two studies, but not in others.16,26,27 See Table 3 for failure criteria.
|
Resource Utilization. The use of NIV does not increase employee workload or increase hospital charges.26,28-30 Other studies have shown an actual reduction in costs with the use of NIV.31,32 NIV may consume more time during the initial hours of therapy, but this excess time requirement does not last for the duration of the therapy.26,29 Finally, familiarity with the equipment and the protocols helps decrease time spent at the bedside.
Complications of Noninvasive Ventilation. The most common complication associated with the use of NIV is irritation and pressure necrosis over the skin at the mask interface.13,33 Break periods from the noninvasive machine and proper cushioning will minimize this.1 Other problems include gastric distention due to aerophagia, vomiting, sinus or ear pain, and eye irritation. Claustrophobia may limit patient tolerance of NIV, especially when using the full face mask. Constant reassurance and a slow increase in the respiratory parameters will increase patient tolerance. Finally, barotrauma is possible, especially when using high inspiratory pressures.
Although positive pressure ventilation produces changes in the dynamics of respiration, only small changes in cardiac output have been demonstrated in patients after the initiation of NIV.34,35
The Evidence for Noninvasive Ventilation
Chronic Obstructive Pulmonary Disease. The use of NIV has been best studied in acute exacerbation of COPD. NIV seems to work by unloading the respiratory muscles, thereby improving alveolar ventilation and reducing pCO2.1 Since the early 1990s, there has been good evidence demonstrating that NIV reduces the rate of ETI and reduces mortality when compared to conventional treatments.36 Researchers studied patients with COPD and found that the use of NIV resulted in a decreased rate of ETI, length of stay (LOS), and mortality.25,26
A study from the United Kingdom suggests that up to 20% of patients with a COPD exacerbation are candidates for NIV.37 This suggestion is based on the percentage of patients with an acute decompensation of COPD who are acidotic.37,38 The mortality from a COPD admission is approximately 25-33%, and most of the mortality is from complications of ETI and mechanical ventilation.39,40 Furthermore, up to 20% of the mechanically ventilated COPD patients are difficult to wean.41 If ETI and mechanical ventilation is avoided in this group, a significant benefit to the patients will occur, with the added benefit of savings due to decreased complications.
The largest randomized controlled trial compared conventional treatment to NIV in a total of 232 COPD patients with acidosis and hypercarbia. In this study, 27.1% of patients required intubation in the standard group, vs 15.3% in the NIV group. In-hospital mortality was 20% for the standard group and 10% for the NIV group. NIV resulted in faster pH correction at one hour, faster correction of respiratory rate at four hours, and a trend for pCO2 improvement at four hours. All these values were statistically significant. Breathlessness also was relieved faster with NIV (4 vs 7 days). In subgroup analysis, the patients with an initial pH < 7.30 had higher failure of NIV and higher in-hospital mortality. The study concluded that NIV resulted in decreased costs and decreased mortality.42
The second largest report is a prospective descriptive study. The authors collected data on COPD patients admitted to a respiratory intensive care unit (ICU) from 1992 to 1999. The publication included 208 cases in which NIV was used. The failure rate for NIV was 17.4% throughout the study period, but the disease severity of the patients increased over the years. The patients who later failed NIV had lower initial pH, higher pCO2, and higher APACHE II scores. Two variables were associated with NIV success at 1 hour: improvement in pH and pCO2. A stepwise regression showed that the APACHE II score; change in pH and pCO2 after 1 hour; and the absolute pH after 1 hour were independent predictors of NIV success. All of these were statistically significant variables. The use of NIV did not increase nursing workload and resulted in savings to the patients.43 Other studies agree with these findings when trying to correlate clinical parameters with success of NIV.16,20,22,44
Plant and Owen followed up their clinical trial with a long-term survival and predictors of in-hospital outcomes analysis.44 A worse pH and pCO2 at enrollment were associated with treatment failure (p < 0.001). At 4 hours, improvement of acidosis (p = 0.035) and a fall in respiratory rate (RR) (p = 0.009) were predictors of success. Although their multivariate analysis had a poor discrimination, these clinical and laboratory parameters help assess patients and risk of NIV failure in a clinically meaningful fashion.44
A study of COPD exacerbations in the ICU demonstrated a decrease in the intubation rates (from 74% to 25%) and mortality (29% to 9%) with the use of NIV. NIV also resulted in a reduction in complications and length of stay.25 Other studies support these findings.36,45 In another randomized controlled trial with 31 patients, researchers found a marked reduction in the intubation rates for COPD patients (67% vs 9%, p < 0.05) when NIV was used. However, in this study, the use of NIV did not decrease length of stay and did not improve mortality.26
Two studies have initiated NIV in the ED. A study with 24 COPD patients randomized them into conventional treatment with or without NIV. The mean initial arterial pH in the group was 7.33. The authors found no difference between NIV and conventional treatment.46 Another researcher also studied patients (n = 27) with respiratory distress in the ED, with only six patients in the study group having COPD. There was a trend toward excess mortality in the NIV group, which the authors attributed to a delay in intubation and mechanical ventilation.47
Another study looked at sicker COPD patients, with a mean arterial pH of 7.2. The patients were randomized into NIV or immediate intubation. Although 52% of the patients in the NIV group later required intubation and mechanical ventilation, NIV was at least as good as ETI with mechanical ventilation.48
A Cochrane Systematic Review on the use of NIV for acute exacerbations of COPD recently was published. The authors evaluated 8 clinical trials totaling 529 patients. When used along with standard care, NIV is associated with lower mortality (RR 0.41); lower need for intubation (RR 0.42); lower likelihood of treatment failure (RR 0.51); and greater improvements in pH, pCO2, and respiratory rate at 1 hour. NIV also resulted in fewer complications (RR 0.32) and shorter LOS (by more than three days). The authors conclude that NIV clearly is effective as an adjunct to standard medical treatment for COPD exacerbations and that it should be instituted early. Furthermore, the rate of complications was significantly lower in the NIV group (risk reduction of 68%). The major benefit of NIV is due to the reduction in the rates of ETI and its subsequent complications.39 A second meta-analysis agrees with these conclusions.49
It is evident that the most appropriate use of NIV is in the patient with an acute exacerbation of COPD. NIV should be tried first and promptly, unless there is an obvious indication for immediate ETI and mechanical ventilation.
Congestive Heart Failure and Acute Pulmonary Edema. Several studies have looked at the use of NIV for congestive heart failure and acute pulmonary edema. A study group used pressure support ventilation in the ED for patients with acute cardiogenic pulmonary edema. Twenty patients received NIV. Of those, 18 improved, and NIV eventually was discontinued. There were statistically significant improvements in pH, pCO2, respiratory rate, and heart rate. Two patients required ETI. There were no reported complications from the use of NIV.12
Researchers studied NIV in patients with acute pulmonary edema. They showed improved vital signs and oxygenation in the NIV group. The need for ETI was reduced from 35% to 0% by the use of CPAP.50 Other researchers reported the use of CPAP in acute pulmonary edema. They also found decreased intubation rates in the NIV group.11 Other study groups agree with these results and also demonstrated a benefit of NIV in pulmonary edema.51,52
A meta analysis of three published trials of CPAP in congestive heart failure and pulmonary edema also showed a reduction in intubation rates by 26% and a trend toward improved mortality.53 In contradiction to this, a study comparing the use of BiPAP, CPAP, and historical controls in pulmonary edema (n = 27) showed a higher rate of acute myocardial infarction in the BiPAP group (71%, 31%, and 38%, respectively).54 This study received much attention, and other similar studies were halted in view of the adverse outcomes of this trial. However, the study enrolled more chest pain patients in the BiPAP group (p = 0.06), which makes the results difficult to interpret.
In a study using BiPAP in patients with severe CHF in the ED, patients were randomized to either BiPAP (n = 21) or oxygen by mask (n = 17) in addition to conventional treatment. The authors found no difference in acute myocardial infarction rates (19% in the BiPAP groups vs 29.4% in the mask group; 95% CI = -37.8 to 17%). However, the study was terminated early after the publication of Mehta’s study.55
Until more evidence arises showing the safety of BiPAP in acute pulmonary edema, CPAP should be the preferred mode of NIV for this group. BiPAP can be used if the patient has a component of hypercarbia. However, exclude patients who have chest pain, cardiac ischemia, or acute myocardial infarction if choosing BiPAP.10
Acute Asthma Exacerbations and Status Asthmaticus. There are a few uncontrolled studies that have used NIV for the treatment of status asthmaticus. In one of the studies, 22/33 eligible patients were treated with NIV. Only three patients required ETI in the NIV group. There were no complications associated with delaying the decision to intubate. There were no statistical differences in LOS and mortality among the groups.56 Another study reviewed the cases of asthma with hypercarbia admitted to an ICU. Seventeen patients who failed an initial trial with medical management were treated with NIV. Two patients in this group (12%) failed NIV and required ETI. NIV was well tolerated and resulted in improved pH, pCO2, and respiratory rate.57
A very recently published randomized placebo-controlled trial compared 15 patients using NIV and 15 patients using a sham device for three hours. The NIV group had a greater improvement in FEV1, with 80% of patients achieving a greater than 50% improvement in the FEV1. Only 20% of patients in the control group achieved the same degree of improvement (p < 0.004). The hospitalization rate was 17.6% in the NIV group and 62.5% in the control group (p = 0.0134).58 This study shows that NIV helps improve the mechanics of respiration in asthmatics, and that it may have a bronchodilatory effect. Ultimately, NIV resulted in a significantly lower admission rate.
In a randomized study, 60 patients with mild to moderate asthma were randomized to receive medications via NIV or a conventional nebulizer. The patients in the NIV group had more improvement of the peak expiratory flow rate. NIV was well tolerated.59
NIV is thought to improve the symptoms of acute asthma by several mechanisms. Positive inspiratory pressure produces bronchodilation and decreases airway resistance, expands atelectasis, and promotes removal of secretions. By improving alveolar ventilation, the respiratory muscles are unloaded and can rest.56,57 Finally, NIV decreases the large negative inspiratory pressures generated during an episode of bronchospasm.59 The use of NIV does not preclude the administration of aerosolized medications. These routinely are delivered through the circuit.
It seems reasonable to attempt NIV in asthmatic patients who are having moderate to severe respiratory distress. There seems to be no detrimental effect when delaying ETI in favor of a NIV trial. Although NIV is a useful tool for the delivery of aerosol medications, the costs probably offset any benefit in the mild cases of asthma exacerbation.
Acute Hypoxemic Respiratory Failure. There have been a few studies using NIV in acute respiratory failure of different etiologies. Most of the studies only show a benefit if there is a component of hypercarbia as part of the respiratory failure.60
One study looked at 41 patients with hypoxemic respiratory failure of varied etiologies. In this group, 34% of the patients failed NIV, requiring ETI. The reasons for intubation were non-improvement during NIV (50%), persistent dyspnea, and poor management of secretions. The mortality was not increased by the use of NIV.61 Another study examined 12 incidents in 10 patients where NIV was used for patients who had acute lung injury. The success rate for NIV in this small group was 50%, and the mortality rate was 30%.62 Another study of patients with acute respiratory failure found similar rates of intubation (70% vs 62%) in the conventional treatment group when compared to the conventional treatment plus NIV group. Length of stay and mortality were similar in both treatment groups. The authors found, however, that NIV was of benefit in a subgroup of patients who had hypercarbia (pCO2 > 45 mm Hg).60 Finally, 64 patients with hypoxemic respiratory failure were studied. In this study, 31% of the patients failed NIV and were intubated. The rate of serious complications was lower in the NIV group (38% vs 66%, p = 0.02); the LOS was shorter in the NIV group; and there was a trend for a lower mortality rate in the NIV group.63
BiPAP ventilation has been studied in the ED setting for a mixed population of patients with acute respiratory failure. It was successful in 86% of the cases, independent of the cause.61 In this case series, 52.5% of the patients were admitted to a lower level of care, which probably reduced the overall cost of admission. On the negative side, another ED-based study showed no reduction in intubation rates and a trend toward increased mortality with the use of NIV. The authors attributed the results to delays in ETI.47
Most of these studies are small in sample size, and thus may lack power to detect a benefit of NIV. In addition, the first two studies are observational and non-randomized. This limits the ability to make any conclusions about the possible benefit of NIV in the setting of severe hypoxemia. Finally, the groups included patients with varied etiologies for the hypoxemia. Sub-group analysis for specific diagnoses is needed to evaluate for any possible roles for NIV. Prospective, randomized studies are needed to provide more information as to the role, if any, of NIV in hypoxemic respiratory failure.47,64
Pneumonia. The few studies looking at NIV in patients with pneumonia have yielded conflicting results. In one of the COPD studies, pneumonia was one of the risk factors associated with NIV failure.16 A recent trial randomized 56 patients to receive conventional treatment with or without NIV. Patients treated with NIV had lower intubation rates (21% vs 50%, p < 0.03). The hospital lengths of stay and hospital mortality rates were similar in both groups. This benefit from NIV occurred in the patients with concomitant COPD and hypercarbia.28
One study of patients with AIDS and P. carinii pneumonia had a 70% success rate (n = 11).61 It seems that the avoidance of ETI decreases complication rates in immunocompromised patients with pneumonia, as shown by the positive results of NIV in the study by Hilbert and colleagues. They found decreased need for ETI, decreased complications, and decreased mortality in a group of immunocompromised patients with pneumonia.65 It is reasonable to do a trial of NIV in these pneumonia patients, but better studies are needed to establish who will benefit the most from this intervention.
Pulmonary Contusion. The use of NIV for pulmonary contusions was described in a retrospective study of 75 patients identified using a trauma registry in Australia. Twelve of those patients successfully were treated with NIV. Unfortunately, most of the other patients required ETI and mechanical ventilation for other related injuries.66 The study suggests that if the patients do not have other injuries that require intubation, pulmonary contusions can be managed using NIV without a worse outcome.
A second study used CPAP in 33 hypoxemic trauma patients. Twenty had pulmonary contusions as one of the diagnoses. The authors report an intubation rate of 6%. Thirty-two of the 33 patients had improvement of the hypoxemia while on CPAP. The study also supports the use of CPAP on trauma patients who have a decreased functional residual capacity resulting in hypoxemia.67
Patients Who Refuse Endotracheal Intubation. Uncontrolled studies have used NIV for patients who refuse ETI and have a reversible cause for acute respiratory failure.68,69 In these patients, NIV relieves dyspnea, preserves speech, and preserves patient autonomy.10 When used in elderly patients who refuse intubation, it has a good success rate, reported in one study at 60%.19 Some authors argue that this use of NIV is inappropriate.70 However, it should be stressed that NIV can be used in cases where the cause of the acute respiratory failure is deemed reversible by the treating physician.
Chronic and Acute Neuromuscular Diseases. NIV is being used for patients with amyotrophic lateral sclerosis when symptoms of ventilatory failure develop. Although clear standards for its initiation have not been developed, authors have suggested a forced vital capacity (FVC) below 70% as a reasonable clinical parameter.71
Others have used BiPAP in patients with myasthenic crises. In a case series of 11 episodes of myasthenic crises, BiPAP was successful in seven of these cases. The authors found that hypercarbia, defined as a pCO2 > 50 mmHg, was a predictor of NIV failure. This is probably a reflection of disease severity. There were no complications arising from the use of NIV.72
Noninvasive Ventilation in the Prehospital Setting. A study group recently introduced NIV in ambulances. Seventy-one patients with suspected congestive heart failure were enrolled. Paramedics reported that the technique was easy to use. Out-of-hospital treatment times were not prolonged by the use of NIV. Although pulse oximetry was improved (p < 0.05) with NIV, there were no statistical differences in LOS, intubation rates, or mortality rates. The authors conclude that paramedics can be trained to deliver NIV, find it easy to apply, and believe that it helps relieve dyspnea in patients with suspected CHF.73
Conclusions
NIV rapidly is emerging as a useful tool in the ED armamentarium. The best evidence on the benefit of NIV is found in the COPD patients. The benefit of NIV in this group seems to be related to the avoidance of the morbidity associated to ETI and mechanical ventilation. There is some evidence on the benefit of NIV for many other etiologies of respiratory and ventilatory failure, such as congestive heart failure, acute pulmonary edema, asthma, pneumonia, pulmonary contusions, and acute neuromuscular diseases.
When faced with a patient who may benefit from NIV, keep in mind that it works best when it is instituted early. (See NIV algorithm.) If the main problem is hypoxemia, then CPAP is the most appropriate modality. The positive pressures help recruit underventilated or collapsed alveoli. If the main problem is hypercarbia, then BiPAP is the most appropriate modality. The inspiratory pressure works by improving tidal volume, thus decreasing pCO2. The expiratory component works like adding PEEP to a ventilator, which results in improved oxygenation.
For COPD patients, most studies demonstrate that an improvement in the arterial blood gases, especially the pH and pCO2, are good predictors of the success of NIV. Most studies also show that the more acidemic patients tend to have worse outcomes and more failures with NIV. After initiation of NIV, have a clear plan on how to recognize a treatment failure and what to do for those patients who fail. There is no convincing evidence that a failed trial of NIV is harmful.74
Finally, more studies are needed to determine the optimal threshold for initiating NIV. Studies also are needed to better evaluate the patient populations and the diagnoses that likely will benefit from a trial of NIV. Although there is some evidence that suggests that NIV is a cost-effective intervention, more studies need to evaluate this further.41 Always remember that NIV should be viewed as a means of preventing, rather than an alternative to, ETI and mechanical ventilation.
References
1. Evans TW. International Consensus Conferences in Intensive Care Medicine: Non-invasive positive pressure ventilation in acute respiratory failure. Organised jointly by the American Thoracic Society, the European Respiratory Society, the European Society of Intensive Care Medicine, and the Societe de Reanimation de Langue Francaise, and approved by the ATS Board of Directors, December 2000. Intensive Care Med 2001;27:166-178.
2. Sharma S. Noninvasive ventilation. In: O.W. Hnatiuk, et al, eds. Emedicine 2003.
3. British Thoracic Society Standards Committee. Non-invasive ventilation in acute respiratory failure. Thorax 2002;57:192-211.
4. Pollack C Jr, Torres MT, Alexander L. Feasibility study of the use of bilevel positive airway pressure for respiratory support in the emergency department. Ann Emerg Med 1996;27:189-192.
5. Doherty MJ, Greenstone MA. Survey of non-invasive ventilation (NIPPV) in patients with acute exacerbations of chronic obstructive pulmonary disease (COPD) in the UK. Thorax 1998;53:863-866.
6. Katz JA, Marks JD. Inspiratory work with and without continuous positive airway pressure in patients with acute respiratory failure. Anesthesiology 1985;63:598-607.
7. Carrey Z, Gottfried SB, Levy RD. Ventilatory muscle support in respiratory failure with nasal positive pressure ventilation. Chest 1990; 97:150-158.
8. Belman MJ, Soo Hoo GW, Kuei JH, et al. Efficacy of positive vs negative pressure ventilation in unloading the respiratory muscles. Chest 1990;98:850-856.
9. Bradley TD, Holloway RM, McLaughlin PR, et al. Cardiac output response to continuous positive airway pressure in congestive heart failure. Am Rev Respir Dis 1992;145:377-382.
10. Mehta S, Hill NS. Noninvasive ventilation. Am J Respir Crit Care Med 2001;163:540-577.
11. Lin M, Yang YF, Chiang HT, et al. Reappraisal of continuous positive airway pressure therapy in acute cardiogenic pulmonary edema. Short-term results and long-term follow-up. Chest 1995;107: 1379-1386.
12. Wigder HN, Hoffmann P, Mazzolini D, et al. Pressure support noninvasive positive pressure ventilation treatment of acute cardiogenic pulmonary edema. Am J Emerg Med 2001;19:179-181.
13. Navalesi P, Fanfulla F, Frigerio P, et al. Physiologic evaluation of noninvasive mechanical ventilation delivered with three types of masks in patients with chronic hypercapnic respiratory failure. Crit Care Med 2000;28:1785-1790.
14. Kwok H, McCormack J, Cece R, et al. Controlled trial of oronasal versus nasal mask ventilation in the treatment of acute respiratory failure. Crit Care Med 2003;31:468-473.
15. Foundation ARC. Consensus Conference: Non-invasive positive pressure ventilation. Respir Care 1997;42:364-369.
16. Ambrosino N, Foglio K, Rubini F, et al. Non-invasive mechanical ventilation in acute respiratory failure due to chronic obstructive pulmonary disease: Correlates for success. Thorax 1995;50: 755-757.
17. Teague GW. Pediatric application of noninvasive ventilation. Respir Care 1997;42:414-423.
18. Padman R, Lawless ST, KettrickRG. Noninvasive ventilation via bilevel positive airway pressure support in pediatric practice. Crit Care Med 1998;26:169-173.
19. Benhamou D, Girault C, Faure C, et al. Nasal mask ventilation in acute respiratory failure. Experience in elderly patients. Chest 1992;102:912-917.
20. Soo Hoo GW, Santiago S, Williams AJ. Nasal mechanical ventilation for hypercapnic respiratory failure in chronic obstructive pulmonary disease: Determinants of success and failure. Crit Care Med 1994;22:1253-1261.
21. Poponick JM, Renston JP, Bennett RP, et al. Use of a ventilatory support system (BiPAP) for acute respiratory failure in the emergency department. Chest 1999;116:166-171.
22. Anton A, Guell R, Gomez J, et al. Predicting the result of noninvasive ventilation in severe acute exacerbations of patients with chronic airflow limitation. Chest 2000;117:828-833.
23. Meduri GU, Abou-Shala N, Fox RC, et al. Noninvasive face mask mechanical ventilation in patients with acute hypercapnic respiratory failure. Chest 1991;100:445-454.
24. Lightowler JV, Elliott MW. Predicting the outcome from NIV for acute exacerbations of COPD. Thorax 2000;55:815-816.
25. Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995;333:817-822.
26. Kramer N, Meyer TJ, Meharg J, et al. Randomized, prospective trial of noninvasive positive pressure ventilation in acute respiratory failure. Am J Respir Crit Care Med 1995;151:1799-1806.
27. Putinati S, Ballerin L, Piatella M, et al. Is it possible to predict the success of non-invasive positive pressure ventilation in acute respiratory failure due to COPD? Respir Med 2000;94:997-1001.
28. Confalonieri M, Potena A, Carbone M, et al. Acute respiratory failure in patients with severe community-acquired pneumonia. A prospective randomized evaluation of noninvasive ventilation. Am J Respir Crit Care Med 1999;160:1585-1591.
29. Nava S, Evangelisti I, Rampulla C, et al. Human and financial costs of noninvasive mechanical ventilation in patients affected by COPD and acute respiratory failure. Chest 1997;111:1631-1638.
30. Jolliet P, Babjo B, Pasquina P, et al. Non-invasive pressure support ventilation in severe community-acquired pneumonia. Intensive Care Med 2001;27:812-821.
31. Keenan SP, Gregor J, Sibbald WJ, et al. Noninvasive positive pressure ventilation in the setting of severe, acute exacerbations of chronic obstructive pulmonary disease: More effective and less expensive. Crit Care Med 2000;28:2094-2102.
32. Plant PK, Owen JL, Parrott S, et al. Cost effectiveness of ward based non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease: Economic analysis of randomised controlled trial. BMJ 2003;326:956.
33. Hill NS. Complications of noninvasive positive pressure ventilation. Respir Care 1997;42:432-442.
34. Ambrosino N, Nava S, Torbicki A, et al. Haemodynamic effects of pressure support and PEEP ventilation by nasal route in patients with stable chronic obstructive pulmonary disease. Thorax 1993;48: 523-528.
35. Confalonieri M, Gazzaniga P, Gandola L, et al. Haemodynamic response during initiation of non-invasive positive pressure ventilation in COPD patients with acute ventilatory failure. Respir Med 1998;92:331-337.
36. Bott J, Carroll MP, Conway JH, et al. Randomised controlled trial of nasal ventilation in acute ventilatory failure due to chronic obstructive airways disease. Lancet 1993;341:1555-1557.
37. Plant PK, Owen JL, Elliott MW. One year period prevalence study of respiratory acidosis in acute exacerbations of COPD: Implications for the provision of non-invasive ventilation and oxygen administration. Thorax 2000;55:550-554.
38. Jeffrey AA, Warren PM, Flenley DC. Acute hypercapnic respiratory failure in patients with chronic obstructive lung disease: Risk factors and use of guidelines for management. Thorax 1992;47:34-40.
39. Lightowler JV, Wedzicha JA, Elliott MW, et al. Non-invasive positive pressure ventilation to treat respiratory failure resulting from exacerbations of chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. BMJ 2003;326:185.
40. Hudson LD. Survival data in patients with acute and chronic lung disease requiring mechanical ventilation. Am Rev Respir Dis 1989; 140:S19-S24.
41. Plant PK, Elliott MW. Non-invasive ventilation in acute exacerbations of COPD. QJM 1998;91:657-660.
42. Plant PK, Owen JL, Elliott MW. Early use of non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease on general respiratory wards: A multicentre randomised controlled trial. Lancet 2000;355:1931-1935.
43. Carlucci A, Delmastro M, Rubini F, et al. Changes in the practice of non-invasive ventilation in treating COPD patients over 8 years. Intensive Care Med 2003;29:419-425.
44. Plant PK, Owen JL, Elliott MW. Non-invasive ventilation in acute exacerbations of chronic obstructive pulmonary disease: Long term survival and predictors of in-hospital outcome. Thorax 2001;56: 708-712.
45. Confalonieri M, Parigi P, Scartabellati A, et al. Noninvasive mechanical ventilation improves the immediate and long-term outcome of COPD patients with acute respiratory failure. Eur Respir J 1996;9:422-430.
46. Barbe F, Togores B, Rubi M, et al. Noninvasive ventilatory support does not facilitate recovery from acute respiratory failure in chronic obstructive pulmonary disease. Eur Respir J 1996;9:1240-1245.
47. Wood KA, Lewis L, Von Harz B, et al. The use of noninvasive positive pressure ventilation in the emergency department: Results of a randomized clinical trial. Chest 1998;113:1339-1346.
48. Conti G, Antonelli M, Navalesi P, et al. Noninvasive vs. conventional mechanical ventilation in patients with chronic obstructive pulmonary disease after failure of medical treatment in the ward: A randomized trial. Intensive Care Med 2002;28:1701-1707.
49. Peter JV, Moran JL, Phillips-Hughes J, et al. Noninvasive ventilation in acute respiratory failure—a meta-analysis update. Crit Care Med 2002;30:555-562.
50. Bersten AD, Holt AW, Vedig AE, et al. Treatment of severe cardiogenic pulmonary edema with continuous positive airway pressure delivered by face mask. N Engl J Med 1991;325:1825-1830.
51. Rasanen J, Heikkila J, Downs J, et al. Continuous positive airway pressure by face mask in acute cardiogenic pulmonary edema. Am J Cardiol 1985;55:296-300.
52. Kelly CA, Newby DE, McDonagh TA, et al. Randomised controlled trial of continuous positive airway pressure and standard oxygen therapy in acute pulmonary oedema: Effects on plasma brain natriuretic peptide concentrations. Eur Heart J 2002;23:1379-1386.
53. Pang D, Keenan SP, Cook DJ, et al. The effect of positive pressure airway support on mortality and the need for intubation in cardiogenic pulmonary edema: A systematic review. Chest 1998;114: 1185-1192.
54. Mehta S, Jay GD, Woolard RH, et al. Randomized, prospective trial of bilevel versus continuous positive airway pressure in acute pulmonary edema. Crit Care Med 1997;25:620-628.
55. Levitt MA. A prospective, randomized trial of BiPAP in severe acute congestive heart failure. J Emerg Med 2001;21:363-369.
56. Fernandez MM, Villagra A, Blanch L, et al. Non-invasive mechanical ventilation in status asthmaticus. Intensive Care Med 2001;27: 486-492.
57. Meduri GU, Cook TR, Turner RE, et al. Noninvasive positive pressure ventilation in status asthmaticus. Chest 1996;110:767-774.
58. Soroksky A, Stav D, Shpirer I. A pilot prospective, randomized, placebo-controlled trial of bilevel positive airway pressure in acute asthmatic attack. Chest 2003;123:1018-1025.
59. Pollack CV Jr, Fleisch KB, Dowsey K. Treatment of acute bronchospasm with beta-adrenergic agonist aerosols delivered by a nasal bilevel positive airway pressure circuit. Ann Emerg Med 1995;26: 552-557.
60. Wysocki M, Tric L, Wolff MA, et al. Noninvasive pressure support ventilation in patients with acute respiratory failure. A randomized comparison with conventional therapy. Chest 1995;107:761-768.
61. Meduri GU, Turner RE, Abou-Shala N, et al. Noninvasive positive pressure ventilation via face mask. First-line intervention in patients with acute hypercapnic and hypoxemic respiratory failure. Chest 1996;109:179-193.
62. Rocker GM, MacKenzie MG, Williams B, et al. Noninvasive positive pressure ventilation: Successful outcome in patients with acute lung injury/ARDS. Chest 1999;115:173-177.
63. Antonelli M, Conti G, Rocco M, et al. A comparison of noninvasive positive-pressure ventilation and conventional mechanical ventilation in patients with acute respiratory failure. N Engl J Med 1998; 339:429-435.
64. Miletin MS, Detsky AS, Lapinsky SE, et al. Non-invasive ventilation in acute hypoxemic respiratory failure. Intensive Care Med 2000;26:242-245.
65. Hilbert G, Gruson D, Vargas F, et al. Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. N Engl J Med 2001;344:481-487.
66. Vidhani K, Kause J, Parr M. Should we follow ATLS guidelines for the management of traumatic pulmonary contusion: The role of non-invasive ventilatory support. Resuscitation 2002;52:265-268.
67. Hurst JM, DeHaven CB, Branson RD. Use of CPAP mask as the sole mode of ventilatory support in trauma patients with mild to moderate respiratory insufficiency. J Trauma 1985;25:1065-1068.
68. Meduri GU, Fox RC, Abou-Shala N, et al. Noninvasive mechanical ventilation via face mask in patients with acute respiratory failure who refused endotracheal intubation. Crit Care Med 1994;22: 1584-1590.
69. Adnet F, Racine SX, Lapostolle F, et al. Full reversal of hypercapnic coma by noninvasive positive pressure ventilation. Am J Emerg Med 2001;19:244-246.
70. Clarke DE, Vaughan L, Raffin TA. Noninvasive positive pressure ventilation for patients with terminal respiratory failure: The ethical and economic costs of delaying the inevitable are too great. Am J Crit Care 1994;3:4-5.
71. Melo J, Homma A, Iturriaga E, et al. Pulmonary evaluation and prevalence of non-invasive ventilation in patients with amyotrophic lateral sclerosis: A multicenter survey and proposal of a pulmonary protocol. J Neurol Sci 1999;169:114-117.
72. Rabinstein A, Wijdicks EF. BiPAP in acute respiratory failure due to myasthenic crisis may prevent intubation. Neurology 2002;59: 1647-1649.
73. Craven RA, Singletary N, Bosken L, et al. Use of bilevel positive airway pressure in out-of-hospital patients. Acad Emerg Med 2000; 7:1065-1068.
74. Elliott MW. Non-invasive ventilation in acute exacerbations of chronic obstructive pulmonary disease: A new gold standard? Intensive Care Med 2002;28:1691-1694.
The emergency physician is faced with a wide variety of acute respiratory emergencies in daily practice. Noninvasive ventilation (NIV), a means of delivering positive pressure ventilation without the use of an endotracheal tube, is a powerful therapeutic tool in the hands of an informed physician. This article will delineate the physiology of NIV. It will provide guidelines for initiation, weaning, and possible complications of NIV. It also will highlight current research in the topic.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.