2003 Salary Survey Results
Salary increases are flat, but growth outlook is good
Research compliance is serious business, particularly when your studies involve humans. So serious, in fact that federal regulations exist, review boards have been developed to oversee the process, and organizations have sprung up to offer education and support to research professionals.
This summer, IRB Advisor took a focused look at the professionals who on a daily basis oversee compliance efforts. We are pleased to present the results of the 2003 Salary Survey for IRB Professionals.
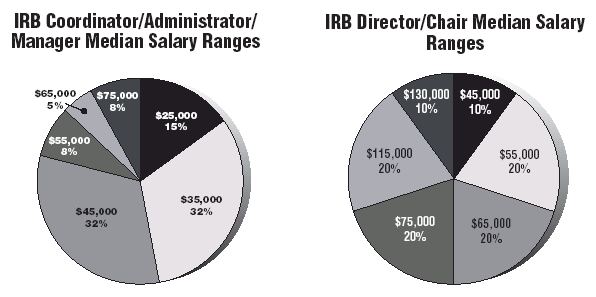
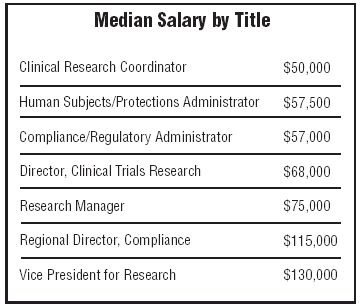
Respondents were diverse — titles ran the gamut from IRB Coordinator to Regulatory/ Compliance Officer to Vice President of Research Development. There were Chaplains among respondents — median annual income $70,000; Research and Clinical Trial directors — median income $53,000, though the salaries ranged from $40,000 to $130,000+; Senior Investigators — median income $95,000; and Medical Ethics Directors — median income $75,000.
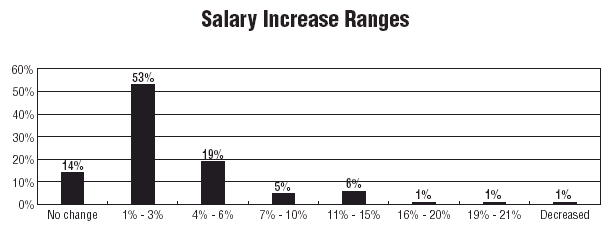
One factor was common, however, across title categories: The average wage increase percentage was 1% to 3%. One respondent in the Research Development area received a 21% increase; and a handful of Compliance Officers received increases in the 11% to 15% range. But 53% of respondents received 1% to 3% wage increases, and 14% reported no annual increase.
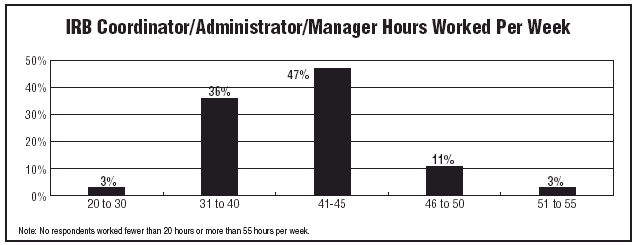
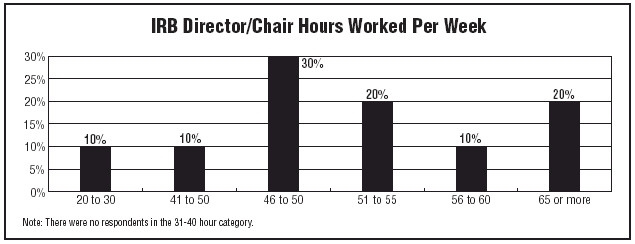
Data also revealed what many of you already know: IRB-related work is not a 9-to-5 effort. The majority of respondents are working long hours. Though 10% of respondents reported working 20-30 hours per week, four out of five professionals — 80% — worked 46 or more hours per week.
"I work 50 to 60 hours per week," says Lynn Bevan, administrative director for research regulatory affairs at The Children’s Hospital of Philadelphia. She’s not alone. In fact, 20% reported working 65 or more hours.
Growth looks promising
If the number of clinical trials being conducted is any indication, the job outlook for IRB professionals is good. The National Institutes of Health (NIH) alone currently has more than 7,000 clinical trials listed on its ClinicalTrials.gov web site, and its 2004 proposed research budget is an estimated $67 billion, up $3 billion over the 2003 budget.
The Pharmaceutical Research and Manufacturers of America reported in its 2002 Annual Survey that pharmaceutical companies spent $30 billion on research and development in 2001, and had more than 1,000 medicines in development going into 2002.
Additionally, genomic discoveries and resulting products being developed surely will increase the need for qualified professionals who can oversee human subjects protections programs.
"Go to IRB web discussion groups, and you can see there is a large need and market for IRB professionals," says Susan Kornetsky, MPH, CIP, director of Clinical Research Compliance at Children’s Hospital in Boston. "IRBs have been around more than 20 years; but as recently as five to seven years ago, organizations and institutions didn’t realize that IRBs needed to be supported. In the past, maybe one or two people supported the IRB. Now, at larger institutions, 15-20 people are on the IRB staff."
Many still are trying to fill slots, Kornetsky says. A quick Internet search supports her assertion. The IRB Forum (www.irbforum.com) job board has posted 86 research-associated ads since January. Of those, 31 were IRB-related positions, including listings for administrators, assistants, coordinators, managers, specialists, and a regulatory auditor. A recent search on the employment web site Monster.com yielded more than 1,000 responses to a "medical research compliance" job query.
"In the past, no one know what an IRB administrator did," says Kornetsky, who says she is beginning to see more qualified applicants.
"The IRB administrator is a liaison between the IRB and the research faculty," Kornetsky points out.
"Of course they have to understand the regulations, but they must also have good communications skills. They must understand what it means to be an investigator trying to do research," she adds.
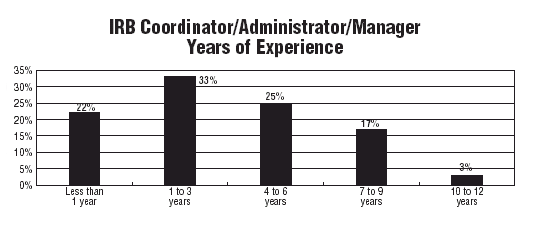
Kornetsky looks for people who have good people skills, as well as some experience with research or human subject protections. She says she received 10-15 resumes the last time she had an IRB administrator position open, and at least seven were qualified applicants.
Helen McGough, MA, CIP, director of the Human Subjects Division at the University of Washington in Seattle, lists the following as required skills:
- a thorough understanding of the regulations and policies (international, national, state, and local) protecting human subjects in both biomedical and social science and behavioral research;
- the ability to manage staff in an environment of limited resources;
- the ability to communicate complex regulatory issues to a variety of audiences including research subjects, researchers, research staff, institutional administrators, and sponsors;
- a good understanding of ethics and a willingness to stand firm in their defense.
"The pool of skilled IRB professionals is small, but growing," McGough says. "Five years ago, it was very difficult to recruit anyone with direct IRB experience. As institutions have expanded their staffing for this function, it has become easier to find people with some IRB experience. In another five years, I expect that we will have a wider and more experienced pool of professionals."
Certification may up increases
In October of 2000, the first certification test for IRB professionals was administered but it’s not clear yet whether CIP (certified IRB professional) designation will result in higher wage increases going forward. "I think if someone is certified, it may hold more water when requesting a salary increase," says Bevan, "but years on the job play more of a role in my mind."
Janine Beal, IRB coordinator, HIPAA, Children’s Hospital of Philadelphia, agrees. "Certification may play a small role, however, I believe job performance plays a bigger role," she says.
Kornetsky notes, however, that some of the job descriptions she’s seen posted are requiring CIP certification, and she thinks that’s a good thing. "CIP certification ensures that IRB professionals share a common body of knowledge," she says. Kornetsky serves on the Council for Certification of IRB Professionals, which is a part of ARENA/ PRIM&R (Applied Research Ethics National Association and Public Responsibility in Medi-cine and Research), and helped develop the certification test. "It’s our hope that certification will bring value to the individual and institution," she says. "It’s new, so only time will tell."
Kornetsky says she knows of at least two institutions that offer a salary increase to those passing the CIP certification exam.
Regardless of the CIP status, there seem to be more IRB-related jobs than people to fill them, Kornetsky points out.
"Employers are looking for IRB professionals who will assure that research conducted within the institution is ethical and regulatorily unassailable," says McGough. "I expect that there will continue to be growth in the field over the next five years as institutions become accredited and as research dollars continue to expand. I think institutions, both academic and nonacademic, have come to realize that although compliance is expensive, noncompliance is even more expensive."
Research compliance is serious business, particularly when your studies involve humans. So serious, in fact that federal regulations exist, review boards have been developed to oversee the process, and organizations have sprung up to offer education and support to research professionals.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
