Prevention of Breast Cancer with Tamoxifen
Prevention of Breast Cancer with Tamoxifen
Abstract & Commentary
Synopsis: Only 5% of white women and 0.6% of black women are potential candidates for tamoxifen chemoprevention.
Source: Freedman AN, et al. J Natl Cancer Inst. 2003; 95:526-532.
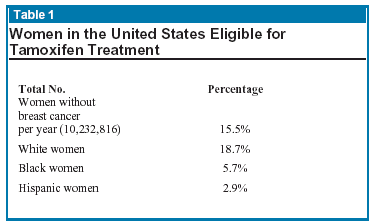
Freedman and colleagues from the National Cancer Institute estimated the total number of US women in the year 2000 eligible for tamoxifen treatment to prevent or defer breast cancer. Of this number, applying a benefit/risk ratio substantially reduced the number of women who could expect a benefit from treatment (see Tables 1 & 2).

Comment by Leon Speroff, MD
I reviewed the combined results of breast cancer prevention trials in the April 2003 issue of OB/GYN Clinical Alert. The important numbers include the following: Tamoxifen 20 mg daily for 5 years produced a 48% reduction in estrogen receptor-positive cancers, no effect on estrogen receptor-negative cancers, an increase in the relative risk of endometrial cancer to 2.4, and an increase in the relative risk of venous thrombosis to 1.9. There is still insufficient follow-up to answer 2 important questions: 1) will tamoxifen treatment yield a difference in breast cancer mortality and; 2) does tamoxifen treatment prevent or defer the diagnosis of breast cancers. It is estimated that treatment of 1000 high-risk women would produce an 18% reduction in breast cancer mortality over the ensuing 10 years.
This recent report from the National Cancer Institute serves to emphasize that because of the side effects, only a small percentage of eligible US women stand to benefit from this treatment. However, this still adds up to about 2 million women per year.
The decision to take 5 years of tamoxifen chemoprevention, therefore, is not easy. The individual patient that can anticipate an expected benefit must be carefully chosen. The patient has to balance potential benefit against the side effects and cost. Ultimately, this difficulty means that we need a better method for prophylactic treatment, and we must continue to emphasize early detection by exam and mammography. Whether raloxifene will provide a better benefit/risk ratio than tamoxifen awaits the outcome of the STAR (Study of Tamoxifen and Roloxifene) clinical trial. The problem with aromatase inhibitors is an increase in fractures and possibly coronary heart disease. The best chemoprevention method awaits future development.
Dr. Speroff is Professor of Obstetrics and Gynecology Oregon Health Sciences University Portland.
Only 5% of white women and 0.6% of black women are potential candidates for tamoxifen chemoprevention.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
