Drug Criteria & Outcomes: Sustained-release morphine: An evaluation of three products
Drug Criteria & Outcomes: Sustained-release morphine: An evaluation of three products
By Sharon W. Cooper,
PharmD candidate
Harrison School of Pharmacy,
Auburn (AL) University
Morphine sulfate is the most widely used opioid analgesic for the treatment of moderate to severe pain. The U.S. market for sustained-release opioids has shown an annual growth of 46% since 1996. However, nearly 85% of patients suffering from chronic pain may still be undertreated. There are various morphine products available to treat moderate-to-severe chronic pain. The following facts are provided to differentiate among three sustained-release products that are currently available.
MS Contin
MS Contin is Huntsville (AL) Hospital’s current formulary agent. It is available as 15, 30, 60, 100, and 200 mg tablets. In addition, MS Contin:
- is dosed every 8-12 hours. The 200 mg tablets are reserved for patients who are opioid-tolerant.
- can be titrated to provide appropriate doses for optimum pain control.
- can have decreased peak plasma concentration if administered with a high-fat meal.
- has a CYP2D6 enzyme substrate; half-life of 2-4 hours.
- is associated with adverse reactions, including palpitations, hypotension, bradycardia, nausea/vomiting, constipation, restlessness, and headache.
Kadian
Kadian is a sustained-release morphine formulation for the treatment of moderate-to-severe chronic pain. It is available as 20, 30, 50, 60, and 100 mg capsules. In addition, Kadian:
- is designed for BID to QD dosing, but most often is administered once daily.
- is not bioequivalent to other controlled-release formulations. Conversion from Kadian to the same total daily dose of another product may lead to excess sedation at peak or inadequate analgesia at trough.
- releases morphine significantly more slowly than sustained-release tablets.
Note: Do not chew, crush, or dissolve pellets in the capsules. The contents may be sprinkled on a small amount of applesauce prior to ingestion. Also, do not attempt to administer the pellets through a nasogastric tube.
Avinza
Avinza is the newest sustained-release morphine formulation. It is made of two components: The IR component (10%) rapidly achieves plateau morphine concentrations in plasma, and the ER component (90%) maintains plasma concentrations throughout a 24-hour period. Avinza is available as 30, 60, 90, and 120 mg capsules. The 60, 90, and 120 mg capsules are for use in opioid-tolerant patients. In addition, Avinza:
- is administered once daily, with a maximum daily dose of 1,600 mg.
- is not intended for use as a PRN analgesic.
- is not easily titrated due to IR and ER components.
- has plasma concentrations not affected by food.
The pharmacokinetics of Avinza have not been studied in patients younger than 18 years of age or older than 65 years of age. The capsules should be swallowed whole; the contents should not be chewed, crushed, or dissolved. The contents may be sprinkled on applesauce prior to ingestion.
Conversion from immediate-release morphine to sustained-release morphine:
- Give half the total daily oral morphine dose every 12 hours.
- Give one-third the total daily oral morphine dose every 8 hours.
- Give the full daily oral morphine dose every 24 hours (for Kadian and Avinza only).
There are no differences in the adverse reaction or drug interaction profiles of MS Contin, Kadian, and Avinza. A randomized, open-label, multicenter, cross-over study comparing Kadian (administered once daily) and MS Contin (administered twice daily) was conducted in cancer patients. Primary efficacy measures that were analyzed were pain scores and patient preference. One hundred thirty-four of 178 patients were randomized to receive at least one dose of study drug and were included in the tolerability analysis. One hundred fourteen patients were randomized to receive at least one dose of Kadian and one dose of MS Contin. These patients were included in the efficacy analysis.
At day one of the study, the median total daily doses were Kadian 100 mg and MS Contin 90 mg. Daily doses at day 10 were Kadian 120 mg and MS Contin 100 mg. The study indicated that Kadian and MS Contin were equally effective in achieving pain control. However, 55% of patients preferred Kadian because it was dosed once daily. Because pain control and patient preference were analyzed at the end of 10 days, the long-term outcomes cannot be assessed. Two other double-blind studies comparing Kadian and MS Contin recently have been conducted. Both medications in these studies were administered twice daily to maintain blinding. Kadian and MS Contin exhibited similar pain control and adverse reaction profiles.
A double-blind, placebo-controlled trial of patients receiving doses of Avinza, MS Contin, or placebo was conducted in 295 patients with osteoarthritis. A total of 184 patients completed the four-week study. Physical functioning was measured weekly during the double-blind portion. An additional open-label portion of this study was conducted for 26 weeks. Eighty-six of 181 patients completed the open-label portion of the study.
At the end of 30 weeks, the study results indicated that there were statistically significant improvements in physical function for patients in the Avinza group. However, Avinza is not easily titrated, and initial effective doses would be difficult to achieve. MS Contin administered in the hospital environment can be titrated to provide optimum pain control. Although the first four weeks of the study were double-blinded and placebo-controlled, the 26-week portion can create open-label associated biases. Another note of interest is that this trial studied the use of Avinza in patients with osteoarthritis. This is a potential weakness because morphine products generally are reserved for patients with more severe pain, such as that experienced by cancer patients.
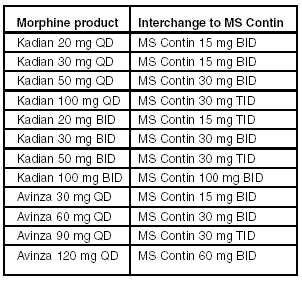
Although Avinza has proven effective in clinical trials, it is significantly more expensive than MS Contin. Kadian is dosed less frequently than MS Contin, but it is no more effective. Kadian also carries a higher price tag. Because the disadvantages of these two drugs outweigh their advantages, MS Contin should remain on the formulary as the drug of choice in this category. Physician orders for Kadian or Avinza should be interchanged to MS Contin according to the values in the table, below.
Resources
• Broomhead A, Kerr R, Tester W, et al. Comparison of a once-a-day sustained-release morphine formulation with standard oral morphine treatment for cancer pain. J Pain Symptom Manage 1997;14:63-73.
• Kadian. Physicians’ Desk Reference. 55th ed. Montvale, NJ: Medical Economics Co.; 2001:1227.
• Kerr RO, Tester WJ. A patient preference study comparing two extended-release morphine sulfate formulations for cancer pain. Clin Drug Invest 2000;19:25-32.
• Morphine Sulfate. Drug Facts and Comparisons. St. Louis: Facts and Comparisons; 2001:798-799.
• Morphine Sulfate. Physicians’ Desk Reference. 55th ed. Montvale, NJ: Medical Economics Co.; 2001:2680-2681.
• Personal communication. Cindy Hall, pharmacy buyer, Huntsville (AL) Hospital System Pharmacy. October 2002.
There are various morphine products available to treat moderate-to-severe chronic pain. The following facts are provided to differentiate among three sustained-release products that are currently available.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
