Drug Criteria & Outcomes: Moxifloxacin IV formulary evaluation
Drug Criteria & Outcomes: Moxifloxacin IV formulary evaluation
By Karisa S. Wilks
PharmD candidate
Harrison School of Pharmacy
Auburn (AL) University
Moxifloxacin (Avelox), a member of the fluoroquinolone class of antibacterial agents, possesses a broad spectrum of bacterial coverage, including both gram-positive and gram-negative microorganisms.1 Moxifloxacin also offers some coverage against anaerobic bacteria. The methoxy group at the C-8 position enhances the drug’s anaerobic activity and allows it to bind to both DNA gyrase and topoisomerase, which is thought to give the drug a low propensity of selective resistance. Gatifloxacin (Tequin), another methoxyquinolone, also offers the above benefits.2 Moxifloxacin is available as 400 mg film-coated tablets and ready-to-use 250 mL flexible bags containing 400 mg of the drug; both are manufactured by Bayer Pharmaceuticals.
Mechanism of action
Moxifloxacin is an 8-methoxy fluoroquinolone; it is a synthetic, broad-spectrum antibacterial agent for oral and intravenous (IV) administration. The quinolones are the only direct inhibitors of DNA synthesis. These bactericidal antimicrobial agents exert their effects by binding to DNA gyrase (bacterial topoisomerase II), which is required for bacterial DNA replication, transcription, repair, and recombination.
Indications1
Moxifloxacin is approved by the Food and Drug Administration (FDA) for the following indications:
- acute bacterial sinusitis caused by Streptococcus pneumoniae, Haemophilus influenzae, or Moraxella catarrhalis;
- acute bacterial exacerbation of chronic bronchitis caused by S. pneumoniae, H. influenzae, H. parainfluenzae, Klebsiella pneumoniae, Staphylococcus aureus, or M. catarrhalis;
- community-acquired pneumonia (of mild- to-moderate severity) caused by S. pneumoniae, H. influenzae, Mycoplasma pneumoniae, Chlamydia pneumoniae, or M. catarrhalis;
- uncomplicated skin and skin-structure infections caused by S. aureus or Streptococcus pyogenes.
Table 1, below, provides a list of the indications currently approved for moxifloxacin, levofloxacin, and gatifloxacin.
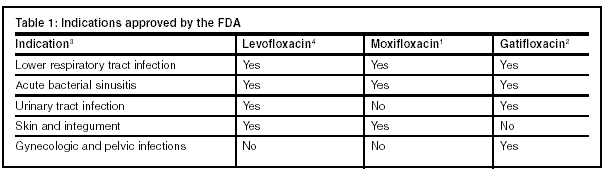
Moxifloxacin has lower minimum inhibitory concentration (MIC) values than gatifloxacin or levofloxacin against some gram-positive organisms, such as Enterococcus faecalis, S. aureus, and S. pneumoniae.
Moxifloxacin does not show susceptibility to methicillin-resistant S. aureus (MRSA).
The safety and efficacy of moxifloxacin in treating clinical infections due to the following organisms has not been established in adequate and well-controlled trials:1
Aerobic gram-positive microorganisms
- Staphylococcus epidermidis (methicillin-susceptible strains only)
- Streptococcus agalactiae
- S. pneumoniae (penicillin-resistant strains)
- Streptococcus viridans
Aerobic gram-negative microorganisms
- Citrobacter freundii
- Enterobacter cloacae
- Escherichia coli
- Klebsiella oxytoca
- Legionella pneumophila
- Proteus mirabilis
Anaerobic microorganisms
- Fusobacterium species
- Peptostreptococcus species
- Prevotella species
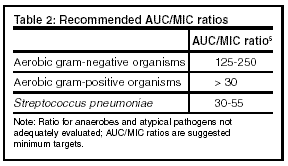
The recommended area under the curve (AUC)/MIC ratios to predict clinical and microbiological response to quinolones are listed in Table 2, above. Table 3, below, presents the AUC/MIC ratios of levofloxacin, gatifloxacin, and moxifloxacin for selected pathogens.
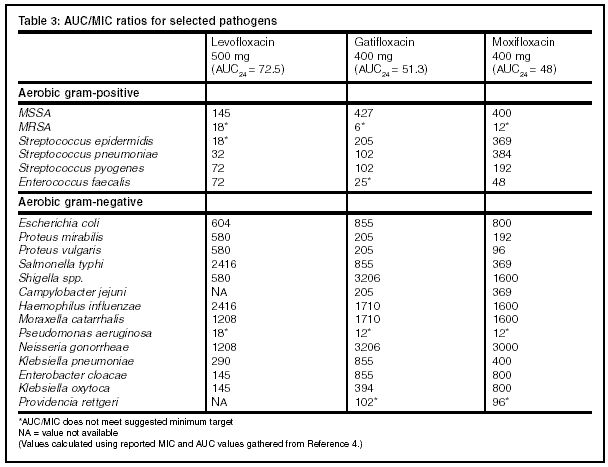
An AUC/MIC ratio less than the suggested range predicts clinical failure, as with all the compared agents in regard to P. aeruginosa and MRSA. A ratio within the suggested range denotes the highest likelihood of a clinical cure; however, a ratio well above the suggested range results in no further improvement in clinical cure rate.
The fluoroquinolones are well-distributed throughout various body tissues. Tissue concentrations often exceed serum values, with the exception of low concentrations of cerebrospinal fluid and aqueous humor. An important characteristic shared by all fluoroquinolones is a high uptake into human phagocytes, which facilitates continued activity against intracellular pathogens.
Contraindications/warnings
Hypersensitivity to moxifloxacin or any other quinolone antimicrobial agent is a contraindication to therapy with this agent. There are several precautions, including the following:
- Pregnancy: The quinolones are pregnancy category C. No adequate and well-controlled studies of these agents have been conducted in pregnant women, and quinolones should be used in pregnancy only if the potential benefit outweighs the potential risk to the fetus.
- Breast-feeding: There is not a sufficient amount of human data available.
- Severe hepatic insufficiency: More pharmacokinetic data are needed before dosage recommendations can be determined in these patients.
- QT interval prolongation: The FDA recommends that the package insert contain a statement suggesting that the risk of arrhythmias may be reduced by avoiding their use or administering them with caution in patients with known underlying cardiac conditions, those with known QTc interval prolongation or history of significant cardiac arrhythmia, those with uncorrected hypokalemia, and those receiving concomitant therapy with agents known to increase the QTc interval or to cause bradycardia (metoclopramide, cisapride, erythromycin, classes Ia and III antiarrhythmics, and tricyclic antidepressants).
Each of the listed precautions applies to levofloxacin, gatifloxacin, ciprofloxacin, and moxifloxacin, with these exceptions:
- It is thought that levofloxacin should not require dosage adjustment in hepatic insufficiency.
- Each of the other drugs besides moxifloxacin requires dosage adjustment in renal insufficiency.
- The labeling in the patient package insert differs in regard to the recommendations on the QT interval prolongation. Currently, moxifloxacin has the strongest warning of these four drugs regarding this potential side effect.6
Average QT prolongation for moxifloxacin and comparators:7
- Moxifloxacin: 6 ± 26 msec
- Amoxicillin: 4 ± 30 msec
- Doxycycline: 2 ± 34 msec
- Cefuroxime axetil: 2 ± 20 msec
- Clarithromycin: 2 ± 23 msec
- Ofloxacin: 0 ± 81 msec
- Cephalexin: 3 ± 16 msec
No comparative trials have taken place to compare the effects of moxifloxacin, gatifloxacin, and levofloxacin on QT prolongation. The QT prolongation effects of both levofloxacin and gatifloxacin are thought to be slightly less than that of moxifloxacin. A large population has been exposed to levofloxacin, with more than 15 million prescriptions written for the drug in the United States from January 1997 to March 2000, and reporting rates of cardiovascular adverse events are low. Less than one case of QT prolongation or torsades de pointes per million prescriptions has been reported, regardless of whether the condition was attributed to underlying disease or concomitant therapy.
Pharmacokinetics4,8,9
Oral moxifloxacin is rapidly absorbed with an average Tmax of 0.75-3.5 hours. The mean Cmax range is 2.50-4.38 mg/L after the usual therapeutic dose of 400 mg in subjects with normal renal and hepatic function. According to the package insert, plasma concentrations increase proportionately with doses up to the highest tested (1,200 mg single oral dose). Moxifloxacin has excellent oral bioavailability, ranging anywhere from 86-100%. The half-life of oral moxifloxacin ranges from 11-14 hours, and it has a volume of distribution ranging from 2.0-3.5 L/kg. Moxifloxacin is approximately 50% protein-bound. The rate of renal clearance is 12.7-15.2 L/h. Linear increases in Cmax and AUC also are seen with intravenous infusions of moxifloxacin 100-400 mg. After 400 mg IV moxifloxacin, the Cmax has been found to be 6.1 mg/L compared to 4.5 mg/L for the equivalent oral dose.
A slight reduction in Cmax is seen after coadministration of 400 mg moxifloxacin and a standard high-fat meal (mean 2.5 mg/L fed, and 2.8 mg/L fasting). Also, the Tmax was slightly prolonged (2.5 vs. 1.0 hours). The AUC remains the same for both fed and fasting states. When the standard dose of moxifloxacin is given with yogurt, a modest delay in the Tmax has been observed (0.88 vs. 2.75 hours). Reductions in mean Cmax (2.87 vs. 2.44 mg/L, 15.3% decrease) and AUC (33.9 vs. 31.8 mg hr/L, 6.2% decrease) also are seen. These results conclude that moxifloxacin may be administered regardless of food intake.
Information regarding the steady state concentrations of moxifloxacin is limited. A pharmacokinetics study of the drug that involved a 10-day treatment period with 400 mg QD observed a steady state Cmax and Cmin of 4.52 mg/L and 0.95 mg/L, respectively. Another multiple-dose study involving a five-day treatment regimen found Cmax and Cmin steady states to be 3.24 mg/L and 0.47 mg/L, respectively.
Moxifloxacin undergoes hepatic metabolism by means of sulfate and glucuronide conjugation. Approximately 38% of the dose is converted to the sulfate conjugate, and 14% to the glucuronide conjugate. After single oral and intravenous doses of 400 mg were administered, 96.3% and 98.4% were recovered in the urine and feces respectively as unchanged parent or metabolite. Most of the sulfate conjugate is excreted in the feces, whereas all of the glucuronide conjugate is excreted in the urine.
The major clinical difference in the pharmacokinetics of moxifloxacin, levofloxacin, and gatifloxacin is the elimination profile. Both gatifloxacin and levofloxacin are excreted primarily by the kidneys, whereas moxifloxacin undergoes a high percentage of hepatic elimination. This difference would account for the different recommendations for hepatic and renal dosage adjustment among the agents. This difference may account for moxifloxacin not achieving renal/urine concentrations as high as those achieved by gatifloxacin and levofloxacin, but no clinical studies of moxifloxacin for treatment of urinary tract infection (UTI) have yet been done to confirm this.
Adverse effects
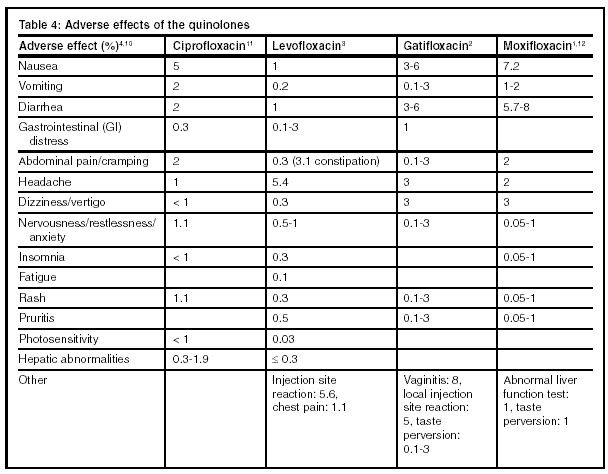
The side-effect profiles of the above agents do not have any major differences. Moxifloxacin has a slightly greater incidence of gastrointestinal side effects than levofloxacin and gatifloxacin. Moxifloxacin also has a stronger warning regarding QTc interval prolongation than the other two agents. Table 4 below lists the more common adverse effects association with quinolones.
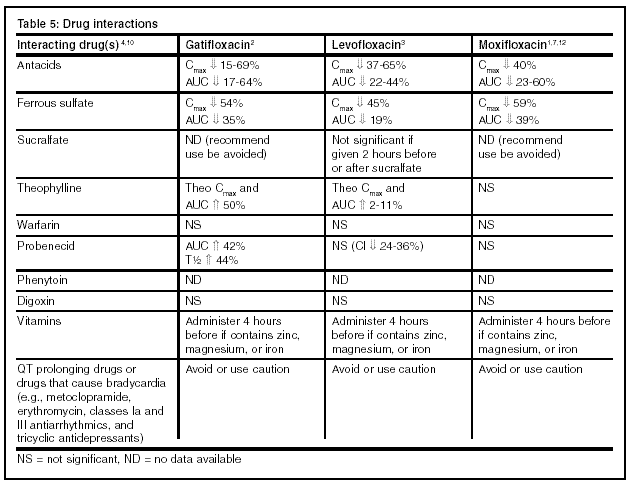
Drug interactions
Table 5 below presents the interaction profiles of gatifloxacin, levofloxacin, and moxifloxacin.
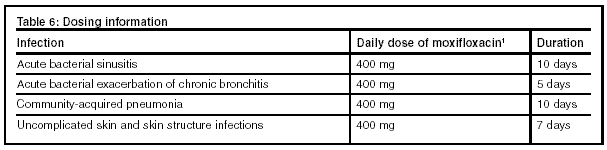
Dosage
Table 6 below lists the doses of moxifloxacin and the duration of therapy for the given indications.
Moxifloxacin IV should be administered by intravenous infusion over a period of 60 minutes by direct infusion or through a Y-type intravenous infusion set that may already be in place.
The IV form is compatible with the following intravenous solutions at ratios from 1:10 to 10:1:
- 0.9% NaCl injection, USP;
- 1M NaCl injection;
- 5% dextrose injection, USP;
- sterile water for injection;
- 10% dextrose for injection;
- lactated Ringer’s for injection.
Clinical trials
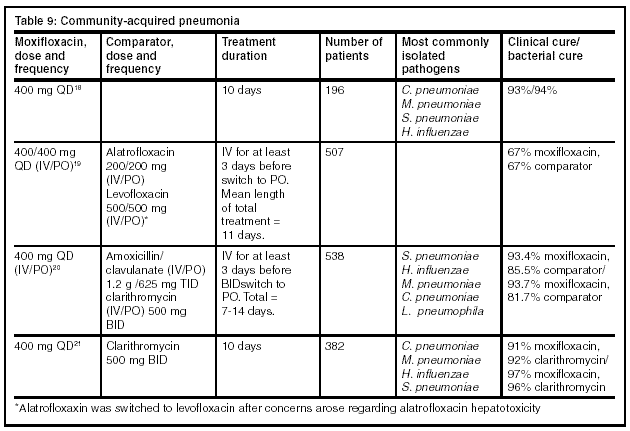
Few head-to-head trials among the fluoroquinolones have taken place. The trials presented in Tables 7-9 below show results of the major trials that have been completed to date. Each of these trials were well-structured, with relatively large numbers of patients. The inclusion and exclusion criteria were similar among the different trials and the comparator drug was given at a dose comparable to that of moxifloxacin for the given indication.
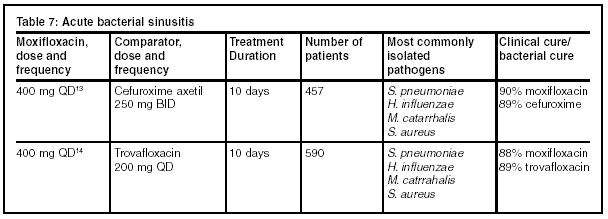
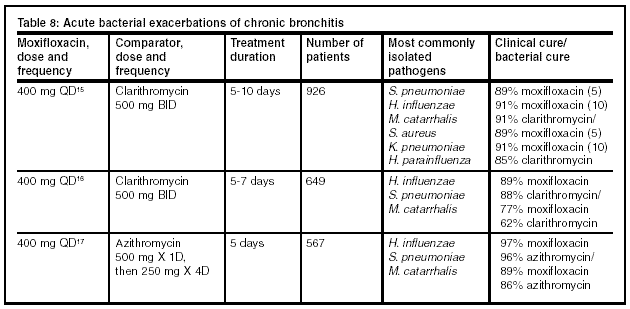
There also has been a randomized, double-blind, controlled clinical trial involving 401 patients conducted in the United States comparing the efficacy of moxifloxacin 400 mg once daily for 7 days with cephalexin 500 mg three times a day for 7 days for the treatment of uncomplicated skin and skin-structure infections.22 The clinical success rates in evaluable patients were 89% (108/122) for moxifloxacin and 91% (110/121) for cephalexin.
Economic issues
Bayer Corp. recently introduced a new pricing structure for acute care hospitals that includes both oral and IV formulations of ciprofloxacin and moxifloxacin. Pricing structures also are being developed for those hospitals that do not place the full lines of ciprofloxacin and moxifloxacin on the formulary.
Bayer Corp. is marketing moxifloxacin for the treatment of gram-positive infections and ciprofloxacin for the treatment of gram-negative infections. The probability of reaching tier 1 (90-100% of quinolone market share with ciprofloxacin/ moxifloxacin) for lowest pricing with ciprofloxacin IV is low, and even at that point the daily cost of ciprofloxacin would be significantly more than gatifloxacin. Therefore, the cost savings benefit of this pricing structure would not surpass the current savings for many hospitals. Currently, the cost savings resulting from a switch from levofloxacin to gatifloxacin would be approximately $30,000-40,000 per year.
Summary
Gatifloxacin should continue to be the primary formulary quinolone. Gatifloxacin has been studied in several areas that moxifloxacin has not, such as UTIs and bone and joint infections. Both of these infections are seen regularly in the hospital, and information regarding the use of moxifloxacin in these infections is lacking. Several studies currently are in progress concerning the use of IV/PO moxifloxacin in the areas of complicated skin and soft-tissue infections, complicated hospital-acquired pneumonia, community-acquired pneumonia, and intra-abdominal infections. After the results of these studies are available, it may be necessary to review the use of moxifloxacin once again.
References
1. Bayer Corp. Avelox package insert. West Haven, CT; 2001.
2. Bristol-Myers Squibb Co. Tequin prescribing information. Princeton, NJ; December 1999.
3. UHC Drug Monograph. Fluoroquinolones. May 2001.
4. Ortho-McNeil Pharmaceuticals. Levaquin package insert. Raritan, NJ; March 1999.
5. Ament P. Predicting fluoroquinolone therapy outcomes potential role of the AUC/MIC ratio. Formulary 1999; 34: 1033-1040.
6. Frothingham R. Rates of torsades de pointes associated with ciprofloxacin, ofloxacin, levofloxacin, gatifloxacin and moxifloxacin. Pharmacother 2001; 21:1468-1472.
7. Church D, Haverstock D, Andriole V. Moxifloxacin: A review of its safety profile based on worldwide clinical trials. Todays Ther Trends 2000; 18:205-232.
8. Rodvold K, Neyhauser M. Pharmacokinetics and pharmacodynamics of fluoroquinolones. Pharmacother 2002; 21:233S-252S.
9. Bayer Corp. Avelox slide presentation. Moxifloxacin pharmacokinetic parameters with 400 mg once daily dosing. West Haven, CT; 2001.
10. Fish D. Fluoroquinolone adverse effects and drug interactions. Pharmacother 2001; 21:253S-272S.
11. Bayer Corp. Cipro package insert. West Haven, CT; 2000.
12. Culley C, Lacy M, Klutman N, et al. Moxifloxacin: clinical efficacy and safety. Am J Health-Syst Pharm 2001; 58:379-388.
13. Burke T, Villanueva C, Mariano H, et al. Comparison of moxifloxacin and cefuroxime axetil in the treatment of acute maxillary sinusitis. Clin Ther 1999; 21:1664-1677.
14. Baz M, Janetti W, Villaneuva, et al. The efficacy and tolerability of moxifloxacin compared to trovafloxacin in the treatment of acute sinusitis. Todays Ther Trends 1999; 17:303-319.
15. Chodosh S, DeAbate C, Haverstock D, et al. Short-course moxifloxacin therapy for treatment of acute bacterial exacerbations of chronic bronchitis. Respir Med 2000; 94:18-27.
16. Wilson R, Kubin R, Ballin I, et al. Five-day moxifloxacin therapy compared with 7-day clarithromycin therapy for the treatment of acute exacerbations of chronic bronchitis. J Antimicrob Chemother 1999; 44:501-513.
17. Mathew C, Warner J, Heyd A, et al. Short course moxifloxacin versus azithromycin in the treatment of acute exacerbation of chronic bronchitis. Paper presented at American Thoracic Society International Meeting. Toronto; May 2000. Abstract
18. Patel T, Pearl J, Williams J, et al. Efficacy and safety of 10-day moxifloxacin 400 mg once daily in the treatment of patients with community-acquired pneumonia. Respir Med 2000; 94:97-105.
19. Choudhri S, Hollister A, Haverstock D, et al. Safety and efficacy of sequential (IV to PO) moxifloxacin for the treatment of community-acquired pneumonia in hospitalized patients [abstract]. Bayer Corporation. West Haven, CT; 2001.
20. Finch R, Collins O, Kubin R, et al. IV/PO moxifloxacin compared with IV/PO amoxicillin/clavulanate ± clarithromycin in the treatment of community-acquired pneumonia admitted to the hospital. The City Hospital, Nottingham, UK, and Bayer Corp., Newbury, UK, and Wuppertal, Germany.
21. Fogarty C, Grossman C, Williams J, et al. Efficacy and safety of moxifloxacin vs. clarithromycin for community-acquired pneumonia. Infect Med 1999; 16:748-763.
22. Parish L, Routh H, Miskin B, et al. Moxifloxacin versus cephalexin in the treatment of uncomplicated skin infection. Int J Clin Pract 2000; 54:497-503.
Moxifloxacin (Avelox), a member of the fluoroquinolone class of antibacterial agents, possesses a broad spectrum of bacterial coverage, including both gram-positive and gram-negative microorganisms.1 Moxifloxacin also offers some coverage against anaerobic bacteria.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
