Special Feature: Blood Culture Dilemmas in the ICU
By Charles G. Durbin, Jr., MD
Sepsis and its consequences are common causes of death in the United States.1 Detection of infection and its proper treatment are essential for survival in all patients, but especially those in the ICU. Indiscriminate use of antibiotics will not improve clinical outcome and will increase risk of resistance and pathogenic overgrowth of fungal species in the GI tract and elsewhere. Cost of care will be adversely affected as well. While these general principles are agreed to, actual diagnostic and treatment approaches to infection and sepsis in the critically ill are not uniform or consistent with these principles. Much of our understanding of infection is derived from caring for hospitalized but not critically ill patients.
The Role of the Clinical Examination
Clinicians suspecting infection in the ambulatory patient "go for the gold" by obtaining information from the most clinically obvious place. A patient with a productive cough will have a sputum specimen sent for culture and perhaps a chest radiograph to rule in or out a lower respiratory tract infection. Symptoms of flank pain or painful urination result in urine cultures with Gram stain or chemical analysis for excessive leukocyte activity in the lower (or upper) urinary tract. Occasionally a patient will present with a fever and no specific organ system or physical symptoms suggestive of a locus of infection. This may lead to blind collection of blood cultures to rule out occult endocarditis or other source of persistent bacteremia as a marker of infection and a (very rare) cause of fever. If the cultures are negative, other causes of fever are considered (ie, occult tumors, vasculitis, venous thrombosis, inflammatory disorders, unusual infectious agents, etc) and the workup continues and expands. Negative cultures have a high negative predictive value, ie, that there is no occult infection or endocarditis. However, positive blood cultures in these asymptomatic patients create problems.
Blood Cultures
Suggestions for the use and interpretation of blood cultures in noncritically ill patients have been proposed.2 Large numbers of patients have been studied. These studies indicate that if a positive culture is obtained, there is about a 50% chance that it will be a "false-positive" (contaminant), rather than a "true-positive" culture. On examination of the published data, it appears that a positive blood culture is predictive of infection only in a small subset of patients—specifically, those with signs of infection other than fever.3 The added cost of finding "false-positive" cultures in patients without signs of infection (other than fever) was found to be much more than just the cost of the cultures.
The Table, below compares the effects on the length of stay and several components of hospital charges (a surrogate for hospital costs) for patients with false-positive blood cultures, compared to those with "true-negative" cultures. These data suggest a 20% increase in unnecessary laboratory charges and a 39% increase in pharmacy charges, mainly from unnecessary intravenous antibiotics. Bates and Lee encourage limiting the use of blood cultures to those patients in which there is a suspicion of infection (such as leukocytosis and clinical symptoms referable to an organ system), and not obtaining a blood culture when fever is the only sign present.
|
Table. |
||||
|
Increases in Length of Stay and Resource Use are Associated with False-Positive Blood Cultures |
||||
| Group | Length | Total | Pharmacy | Laboratory |
| of stay | Charges | Charges | Charges | |
| After Blood | following | following | ||
| Culture | Blood | Blood | ||
| Culture | Culture | |||
|
|
||||
| True Negative Blood Cultures (n = 1097) | 8 days | $8731.00 | $798.00 | $1426.00 |
|
|
||||
| False Positive Blood Cultures (n = 94) | 12.5 | $13,116.00 | $1456.00 | $2057.00 |
|
Adapted from: Bates DW, Lee TH. Rapid classification of positive blood cultures. Prospective validation of a multivariate algorithm. JAMA. 1992; 267(14):1962-1966. |
||||
This approach to limiting blood cultures has not been applied widely to hospitalized patients, and frequent "screening" blood cultures are obtained in patients in whom fever is the only sign suggesting infection. This situation continues to create diagnostic problems for clinicians and increases costs for hospital systems and patients. As an extension of these findings in noncritically ill patients, I believe that blood cultures are used in the critically ill. Although there is less published information in the critically ill patient, it seems reasonable to apply a similar diagnostic and treatment approach to critically ill patients.
Sepsis is Not Synonymous With Infection
Severe sepsis, sometimes called SIRS (systemic inflammatory response syndrome) or the systemic inflammatory/anti-inflammatory syndrome, is the major problem in the ICU today. This condition is responsible for most ICU-related deaths and is the leading cause of multiple organ failure. Estimates are that more than 800,000 patients develop sepsis and at least 215,000 die from uncontrolled inflammation due to sepsis during critical illness each year in the United States.1 Enormous energy and dollars are being spent researching and treating this condition. "Surviving Sepsis" is an international campaign spearheaded by the International Sepsis Forum to raise understanding and to reduce mortality from this condition. Information for patients and clinicians is provided at the organization’s web sites (www.survivingsepsis.org; www.sepsisforum.org).
The understanding of the basic mechanisms of this disease (or collection of diseases) is evolving, and a recent review suggests that immune suppression and organ "hibernation" may be key elements governing its poor outcome.4 Outcome from sepsis is not specifically related to the presence of a pathogenic organism in the blood or its antibiotic susceptibility but the degree of shock and organ dysfunction that occurs. Specific organisms and their byproducts seem to be important markers of the severity but not usually the primary precipitating process for sepsis in most ICU patients with this syndrome. Antibiotics are supportive (and essential) but not curative in severe sepsis. Antibiotic coverage should not be directed only to the organism, which grows from blood cultures, but should be broad enough to cover organisms residing in the intestinal tract and on body surfaces.
With the understanding that sepsis is not synonymous with "infection" I would like to propose a logical way of investigating "infection" in the critically ill. While repeated negative blood cultures (up to 6 sets) are helpful in ruling out continuing bloodstream invasion in the ambulatory (healthy?) patient, positive and negative blood cultures are more problematic in the septic ICU patient.
Bacteremia is Not Sepsis
Many studies have demonstrated that bacteremia is a common occurrence in healthy individuals and not associated with systemic manifestations of sepsis. Even 30 years ago, asymptomatic bacteremia was recognized as occurring during normal clinical interventions. In one study of intubation techniques, 16% of children had positive blood cultures during nasotracheal intubation.5 In the entire group studied, 65% of children had positive blood cultures during or following dental extractions.6 No child had any signs of sepsis or developed a perioperative infection, and none received antibiotics despite having pathogenic organisms cultured from their blood. Just manipulating the gums while brushing the teeth results in bacteremia.
The conclusion I would propose is that bacteremia is a normal occurrence, and thus the presence of a positive culture by itself is not sufficient evidence of sepsis, nor is it a reason to treat with antibiotics. If blood cultures are drawn from healthy individuals, many will be positive but none are pathologic. The same should be true in the critically ill. Blood culture positivity (asymptomatic bacteremia) must be occurring frequently during routine ICU manipulations and to separate out what is "normal" bacteremia from infection that should be treated is not possible based solely on the organism identified in a single set of blood cultures. If multiple sets of cultures contain the same organism, this probably does represent the need to treat; however, antibiotic treatment should remain broad, and not just directed toward the cultured organism.
Septic Patients May be Bacteremic
When we thought sepsis was "infection," a long, exhaustive search for the source of the infection was the diagnostic mantra. Some clinicians still believe this erroneous idea. Clearly, undrained pus and undiagnosed invasive infections must be rigorously searched for, but the role of obtaining frequent blood cultures in this search is less certain. If random and nonpathogenic bacteremia are assumed to be at least as common in the critically ill as in healthy individuals, positive blood cultures not representing disease would often be found. The remarkably low rate of culture positivity in the critically ill is probably related to the widespread use of broad spectrum antibiotics, suppressing bacterial growth. Using antibiotic-inactivating culture materials and larger amounts of blood per sample increases the positivity only marginally with no improvement in diagnosis. The point is that even a positive blood culture is unlikely to mean the patient is actually "infected" with that particular organism; specific antibiotic treatment directed only against the organism identified is inappropriate for several other reasons.
Bacteremia as a Marker of Severity of Sepsis?
Many studies of sepsis suggest that bacteremia may be an "epi-phenomenon," possibly related to the severity of sepsis but not its cause. In the early studies testing anti-endotoxin (AE) and antitumor necrosis factor (ATNF) antibodies, several things become clear. First, there was no overall survival benefit from the use of these expensive therapies (although several subgroups did appear to show improved outcome). Second, since patients were entered before culture results were available, many culture-negative septic patients were administered the test antibodies. Of the patients entered in these studies, only about 40% subsequently had positive blood cultures. The effect of the trial treatment in these culture-negative patients was not different than in those with positive cultures. The primary determinant of outcome was the degree of shock and organ failure, not whether a blood culture was positive or negative. There also was little relationship of survival to whether "appropriate, specific antibiotic" treatment was given. Patients not receiving appropriate antibiotics for the specific organism that eventually grew in the blood had similar survival to those patients in whom initial antibiotic coverage included the cultured organism. This suggests that the detected bacteremia was not usually pathogenic in septic patients.
Reports from the first human trial of monoclonal antibody, HA-1A (EA), suggested a survival benefit for those patents presenting in shock with positive blood cultures for Gram-negative organisms.7 However, only 200 of the 534 patients (37%) entered into the study had positive cultures. Of these, 16% had isolated bacteremia without identified infection. When all patients were considered, there was no benefit to receiving HA-1A treatment. In analyzing the treatment and control groups’ severity of illness, it became apparent that renal and hepatic failure occurred much more frequently in the control group. This failure of randomization invalidated the conclusions, and the therapy was not considered effective based on this trial. In a second large trial of HA-1A (EA), 1578 out of 2199 patients had culture-negative septic shock while only 621 (28%) had positive blood cultures.8 Treatment with HA-1A in this large trial was not effective in either group (blood or other positive culture, or no culture positive) at improving survival. Mortality of patients in the blood culture negative group was slightly worse at 39% than those in the blood culture positive group, 33%. Thus, in this study, bacteremia seemed to confer some survival benefit; it certainly did not correlate with a worsened outcome.
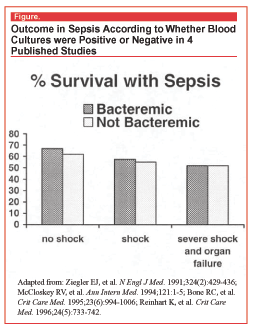
I present these data to suggest that bacteremia is incidental and not pathologic in most patients with sepsis. The determining factor in sepsis survival is the severity of the syndrome. As shown in the Figure (data extracted and combined from 4 studies), survival is reduced as the degree of shock and organ dysfunction increases, not whether cultures are positive.
When to Obtain Blood Cultures in the Critically Ill
The Society of Critical Care Medicine has developed and published Guidelines on fever workup in the critically ill.9 These Guidelines state: "A new onset of temperature equal to or above 38.3°C is a reasonable trigger for a clinical assessment, but not necessarily a laboratory (blood cultures) or radiologic evaluation for infection." I added the italics and comments in parentheses for emphasis and clarification. This is consistent with the recommendations described above for the workup of a noncritically ill hospitalized patient with a fever. These SCCM Guidelines suggest obtaining 2 independent sets of blood cultures if the clinical examination suggests infection within the first 24 hours after a new fever unless there is unequivocal evidence of a non-infectious source of the fever. Repeated blood cultures should be based on the clinical evaluation of the patient, not on persistent or recurrent fevers alone. Cultures of other potential infected sites (such as sputum and urine) are probably more important and significant for guiding therapy than the blood cultures.
Broad Spectrum Antibiotics
I have recommended not tailoring the antibiotic coverage to a single organism in the critically ill, especially if the organism is only identified in a blood culture. Antibiotic therapy is supportive in sepsis. Immune system failure (dysfunction) is the basis of sepsis. Barrier function of the gut and mucous membranes fails, resulting in a greater amount of "normal" leak of bacteria from these sources. It seems logical that supportive therapy should be directed against all the organisms usually contained within these locations. This means antibiotic coverage should include Gram-positive, Gram-negative, aerobic, and anaerobic organisms, and possibly fungus as well. The importance of antibiotic treatment increases with the severity of the sepsis.
Guidelines from many organizations suggest that when an identified infection is associated with positive blood cultures for the same organism, treatment should include coverage with 2 antibiotics instead of only 1 (double coverage doctrine). This has the practical effect of maintaining a "broad spectrum" of antibiotic treatment even if 1 specific organism is targeted. If signs of sepsis and septic shock are also present (or the patient fails to improve), then the broadest coverage is recommended despite there being only a single organism identified, for fear of missing an "uncultured" pathogen. While not explicitly stated, the result of this escalating therapy is the same as my recommendation of not narrowing the antibiotic spectrum when positive blood cultures are found in the critically ill.
Central Lines and the Risk of Bloodstream Infections (BSI)
The most important source of nosocomial blood stream infection (BSI) in the critically ill is colonization of a central venous catheters (CVC) leading to BSI. In the setting of a critically ill patient with a CVC, blood cultures have a significant role in diagnosis and therapy. While some clinicians assume that arterial lines are an important nosocomial source, this does not appear to be true. Likewise, peripheral intravenous lines are unlikely to be sources of "true" BSIs. Careful, aseptic, sterile placement techniques and meticulous line care are essential to reduce this problem. Subcutaneous tunneling of central line catheters should reduce the BSI risk but will increase the incidence of mechanical line complications. The use of elemental silver- and antibiotic-impregnated CVCs is a promising development to reduce this hazard, although risk reduction and cost increase must be balanced. Methods of sterilization without removal of the catheter have been developed to reduce the morbidity of catheter replacement where the replacement is difficult.10 An emerging risk for BSI may be needle-less connectors, which are mandated by Federal regulation and are almost impossible to sterilize. The use of many of these devices results in contamination and bacterial seeding during the injection process.
Diagnosis of Catheter-Related BSI
Removal and culture of the indwelling catheter is the standard for diagnosis and treatment of catheter colonization. If simultaneous peripheral blood cultures are positive, then catheter-related BSI is likely. If the line is suspected as the source of a fever and it is easy to replace, it should be removed and the tip cultured. The most widely used laboratory technique for the clinical diagnosis of catheter-related infection is the semi-quantitative method, in which the catheter segment is rolled across the surface of an agar plate and colony-forming units (CFU) are counted.11 Quantitative culture of the catheter segment requires either flushing the segment with broth or sonicating it in broth, followed by serial dilutions and surface plating on blood agar.12-14 A yield of ³ 15 CFU from a catheter, by means of semi-quantitative culture, or a yield of ³ 100 CFU from a catheter, with accompanying signs of local or systemic infection, is indicative of catheter-related infection.
In a prospective study that compared the sonication, flush culture, and roll plate methods, the sonication method was 20% more sensitive for the diagnosis of catheter infection than was the roll plate method, and it was > 20% more sensitive than was the method of flushing the individual catheter lumens.15 If only catheter-related BSI are considered, the sensitivities of the 3 methods are: sonication, 80%; roll plate method, 60%; and flush culture, 40-50%. Specificity could not be determined in this study since all patients had peripheral positive cultures to the organism found on the catheter by 1 or more of the test methods. Differentiation of bacteremia from another source causing catheter colonization rather than a primary CVC infection leading to bacteremia is a difficult problem to sort out.
Quantitative CVC aspiration blood culturing techniques have been developed as an alternative for the diagnosis of catheter-related BSI in patients for whom catheter removal is undesirable because of limited vascular access. Attempts have been made to improve the diagnosis of catheter-related infection by drawing samples for culture through the line and peripheral veins comparing the number of CFU. This technique relies on quantitative culture of paired blood samples, one of which is obtained through the central catheter hub and the other from a peripheral venipuncture site. In most studies, when blood obtained from the CVC yielded a colony count at least 5- to 10-fold greater than that for blood obtained from a peripheral vein, this was predictive of catheter-related BSI.16 Among tunneled catheters, for which the method is most accurate, a quantitative culture of blood from the CVC that yields at least 100 CFU/mL is considered diagnostic without a companion culture of a peripheral blood sample.17 Most clinical microbiological laboratories are not capable of performing this type of analysis.
A new method, which correlates well with quantitative blood cultures, makes use of continuous blood culture monitoring for positivity (eg, radiometric methods) and compares the differential time to positivity for qualitative cultures of blood samples drawn from the CVC and a peripheral vein. When studied with tunneled catheters, this method has offered accuracy comparable to that of quantitative cultures of blood samples and has had greater cost-effectiveness.18,19 In a study of differential time to positivity, a definite diagnosis of catheter-related bacteremia could be made in 16 of the 17 patients who had a positive result of culture of a blood sample from the CVC at least 2 h earlier than they had a positive result of a peripheral blood culture; the overall sensitivity was 91% and specificity was 94%.16 Most hospitals do not have quantitative blood culture methodologies, but many will be able to use differential time to positivity for diagnosis.
Putting the Picture Together
Bacteremia is common in critically ill patients but is infrequently the only sign of infection. The sepsis syndrome may be initiated by a specific infection, but this is not invariably true. Treatment of the septic patient involves hemodynamic and specific organ support and antibiotic coverage. Specific organ and fluid cultures should be used to confirm infections in patients with symptoms suggesting pneumonia, urinary tract infection, etc. Because bacteremia is common and not pathological in "healthy" as well as ICU patients, blood cultures should be reserved for those patients in whom bacteremia would be considered pathologic (eg, suspected CVC infection, suspected endocarditis, etc). Negative cultures may be reassuring, but positive blood cultures may be misleading. If repeated cultures are positive for the same organism, a primary focus of infection should be sought and antibiotic coverage should include, but not be limited to, the identified organism.
In the absence of other findings of specific risk factors, fever is not an indication to obtain blood cultures in the ICU patient. If blood cultures are obtained, persistent fever or recurrent fever alone is not an indication for repeated blood cultures. To avoid problems in interpretation, strict protocols must be used for skin preparation, sampling size, and handling of specimens for blood cultures. CVCs are a particular risk of nosocomial infection in the ICU. Strict adherence to sterile placement and handling must be used. Identifying CVC colonization and infection is difficult but a low threshold for line removal is important. The rate of CVC colonization and related BSI are quality markers of an ICU system of care and may become a mandatory required measure for all ICUs in the future.
References
1. Angus DC, et al. Crit Care Med. 2001;29:1303-1307.
2. Bates DW, et al. Ann Intern Med. 1990;113(7):495-500.
3. Bates DW, Lee TH. JAMA. 1992;267(14):1962-1966.
4. Hotchkiss RS, Karl IE. N Engl J Med. 2003;348: 138-150.
5. Berry FA Jr, et al. Anesth Analg. 1973;52(6):873-876.
6. Berry FA Jr, et al. Pediatrics. 1973;51:476-479.
7. Ziegler EJ, et al. N Engl J Med. 1991;324(2):429-436.
8. McCloskey RV, et al. Ann Intern Med. 1994;121:1-5.
9. Anonymous. Crit Care Med. 1998;26(2):392-408. http://www.sccm.org/profresources/G_toc.html
10. Andris DA, et al. Nutrition. 1998;14:427-432.
11. Maki DG, et al. N Engl J Med. 1977;296:1305-1309.
12. Brun-Buisson C, et al. Arch Intern Med. 1987;147: 873-877.
13. Cleri DJ, et al. J Infect Dis. 1980;141:781-786.
14. Sherertz RJ, et al. J Clin Microbiol. 1990;28:76-82.
15. Sherertz RJ, et al. J Clin Microbiol. 1997;35:641-646.
16. Fan ST, et al. Eur J Clin Microbiol Infect Dis. 1989;8: 142-144.
17. Capdevilla JA, et al. Eur J Clin Microbiol Infect Dis. 1992;11:403-407.
18. Blot F, et al. J Clin Microbiol. 1998;36:105-109.
19. Blot F, et al. Lancet. 1999;354:1071-1077.
Sepsis and its consequences are common causes of death in the United States. Detection of infection and its proper treatment are essential for survival in all patients, but especially those in the ICU.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
