Special Feature: Pharmacology Update - Atypical Antipsychotics
Pharmacology Update: Atypical Antipsychotics
By Richard Harrigan, MD
Antipsychotic agents are a diverse class of drugs used to treat both psychiatric and nonpsychiatric conditions. The latter include control of nausea and emesis; pain and nausea reduction in various headache syndromes; chemical restraint of violent or agitated patients; hiccup suppression; and treatment of various movement disorders (e.g., Tourette’s syndrome, Huntington’s chorea). Once termed neuroleptics due to a propensity of the older agents to affect neurologic function, this term largely has been discarded. Antipsychotics are currently stratified into two categories: the older, typical agents, and the newer, atypical antipsychotics, which offer improved efficacy and a somewhat different side effect profile. Atypical antipsychotics were developed to address not only the positive symptoms of psychosis (e.g., hallucinations, delusions, and disordered thought) but also the negative symptomatology (e.g., withdrawal, flat affect, and loss of drive). This review will focus on the basic pharmacology, adverse effects, and toxicologic manifestations of the atypical antipsychotics. (See Table 1.)
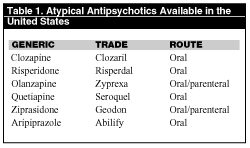
Pharmacology
Typical, or conventional, antipsychotics (which include a variety of drugs and drug classes, such as: phenothiazines [e.g., chlorpromazine]; butyrophenones [e.g., haloperidol]; and thioxanthenes [e.g., thiothixene]) are well documented as controlling the positive manifestations of psychosis as well as agitation due to other disorders (e.g., substance abuse, dementia). To differing degrees they block dopaminergic, alpha-adrenergic (principally a1), muscarinic cholinergic, and histaminic receptors, yielding a varied side effect profile linked to their site of antagonism: extrapyramidal symptoms and tardive dyskinesia, orthostatic hypotension, anticholinergic effects, and sedation, respectively.1
The atypical antipsychotics are all from differing structural classes, and display different neurochemical profiles. Generally speaking, they preferentially antagonize dopamine-2 (D2) receptors in the mesolimbic region of the brain over D2 receptors in the mesocortical and nigrostriatal pathways; therefore, they are less likely to cause cognitive blunting and extrapyramidal symptoms, respectively, than the conventional antipsychotics. They also demonstrate minimal effects at tubuloinfundibular dopaminergic sites, lessening the effect on serum prolactin levels and avoiding the galactorrhea, amenorrhea, and gynecomastia associated with some typical agents.2 In addition to mesolimbic D2 selectivity, these atypical agents feature high binding affinity for certain serotonergic sites (e.g., 5-HT2A); antagonism at these receptors in the nigrostriatal and mesocortical regions serves to disinhibit dopaminergic transmission at these sites, decreasing extrapyramidal manifestations and cognitive blunting effects, respectively. The antiserotonergic effects in the limbic neurons also seem to have a direct antipsychotic effect.2 However, the atypical antipsychotics are similar to their typical predecessors in that they competitively block a number of other neurochemical sites; affinity for these other sites varies from drug-to-drug, and is dose-dependent. (See "Adverse Effects," below.)
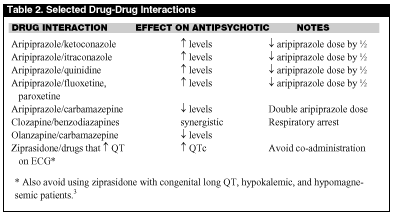
All atypical antipsychotics are rapidly absorbed, with peak concentrations occurring in 1-6 hours — an important feature when confronted with the overdose patient. These drugs are highly protein bound, lipophilic, and feature a large volume of distribution, thus rendering dialysis an ineffective means of toxicity management. All undergo hepatic metabolism. Clinically important drug-drug interactions do exist. (See Table 2 for several key interactions.) A general rule is to use these drugs with caution when other drugs with similar side effect profiles (e.g., central nervous system or respiratory depression, orthostasis, etc.) are co-administered. Cytochrome P450 interactions occur to varying degrees with these agents and should be considered. All atypical antipsychotics are pregnancy category C, with the exception of clozapine, which is category B.
Adverse Effects
Most adverse effects can be anticipated by reviewing the pharmacologic properties of these drugs. Generally speaking, extrapyramidal disorders, tardive dyskinesia, and neuroleptic malignant syndrome appear less frequently with the atypical antipsychotics than with the typical agents; among the atypicals, risperidone (especially at increased dosages) seems to carry the highest risk for these dopamine receptor-related effects, as it does for prolactin elevation.2 All can cause a reversible elevation in hepatic transaminases.2 Blockade at a1-adrenergic sites (especially clozapine, risperidone, olanzapine, ziprasidone, quetiapine, and aripiprazole) may cause orthostatic hypotension, miosis, and reflex tachycardia. Antihistaminic effects (especially clozapine, olanzapine, and quetiapine) principally include sedation. Muscarinic antagonism, causing both peripheral and central anticholinergic effects, is seen more with clozapine and olanzapine.2 There has been a link to weight gain, hyperlipidemia, and the development or exacerbation of diabetes mellitus.2-4 Agranulocytosis is seen in approximately 1% of clozapine-treated patients; even rarer with this agent is an idiosyncratic eosinophilic myocarditis; both are more likely to occur during the first weeks of therapy.2 Ziprasidone may cause QT prolongation on the electrocardiogram — although apparently not to the extent that thioridazine does — and its use should be avoided with drugs or syndromes that prolong the QT interval. Ziprasidone has not been linked to torsade de pointes thus far.3,5 Priapism has been associated with clozapine, risperidone, olanzapine, and quetiapine, and probably is mediated by a1-adrenergic antagonism.6-8 Priapism traditionally has been linked with the antidepressant trazadone, and several phenothiazine (as well as other typical) antipsychotics, in addition to several antihypertensives; mechanistically speaking, it could be encountered with any of the atypical antipsychotic agents.8
Acute Overdose
The high therapeutic index of atypical antipsychotics renders lethal ingestion a rare occurrence. However, toddlers ingesting a single tablet of clozapine, olanzapine, and risperidone have been significantly poisoned;2 pediatric ingestion of any of these agents, even in small amounts, should be taken seriously. Symptomatic overdose generally features an exaggerated spectrum of the adverse effects described above, particularly CNS depressant and cardiovascular effects. Clozapine has been reported to cause CNS depression and seizures.9 Risperidone poisoning is usually well-tolerated; however, it has been associated with neurologic (sedation, dystonia, and spasms), cardiovascular (sinus tachycardia, prolongation of the QTc, and wide-complex tachycardia), and electrolyte disturbances.10,11 Toxicity from olanzapine manifests principally with CNS sedation; miotic pupils have also been described, thus imitating opioid and clonidine overdose.12 Quetiapine poisoning may cause depressed mental status — as with any of these agents, at times profound — hypotension, and tachycardia; it has been shown to cause significant QT prolongation (710 msec) after massive ingestion.13,14 Ziprasidone has been reported to cause mild sedation and some QTc prolongation after overdose.15,16 Experience with aripiprazole is limited, and product information thus far reports only somnolence and vomiting after overdose.17
Treatment is largely supportive, and routine toxicologic standards should be employed. There are no specific antidotes known to be effective for these agents when taken in overdose. The emergency physician should be prepared to protect the airway if CNS sedation is profound. Telemetric monitoring should be used, and an electrocardiogram obtained to assess the QTc interval. Hypotension should be treated with intravenous fluids initially; norepinephrine or phenylephrine appear to be good choices for refractory hypotension considering the propensity of atypical antipsychotics to cause a1-adrenergic blockade. Seizures should be treated with benzodiazepines, and barbiturates if necessary.2
Dr. Harrigan, Associate Professor of Emergency Medicine, Temple University Hospital and School of Medicine, Philadelphia, PA, is Editor of Emergency Medicine Alert.References
1. Andersson C, et al. Emerging roles for novel antipsychotic medications in the treatment of schizophrenia. Psychiatr Clin North Am 1998;21:151.
2. Burns MJ. The pharmacology and toxicology of atypical antipsychotic agents. J Toxicol Clin Toxicol 2001;39:1.
3. Caley C, et al. Ziprasidone: The fifth atypical antipsychotic. Ann Pharmacother 2002;36:839.
4. Sernyak MJ, et al. Association of diabetes mellitus with use of atypical neuroleptics in the treatment of schizophrenia. Am J Psychiatry 2002;159:561.
5. Glassman AH, et al. Antipsychotic drugs: Prolonged QTc interval, torsade de pointes, and sudden death. Am J Psychiatry 2001;158:1774.
6. Rosen SI, et al. Clozapine-induced priapism. J Urol 1992;148;876.
7. Pais V, et al. Priapism from quetiapine overdose: First report and proposal of mechanism. Urology 2001;58:462vii.
8. Compton MT, et al. Priapism associated with conventional and atypical antipsychotics medications: A review. J Clin Psychiatry 2001;62:362.
9. Welber MR, et al. Clozapine overdose: A case report. J Emerg Med 1995;13:199.
10. Acri AA, et al. Effects of risperidone in overdose. Am J Emerg Med 1998;16:498.
11. Duenas-Laita A, et al. New clinical manifestations of acute risperidone poisoning. J Toxicol Clin Toxicol 1999;37:893.
12. O’Malley GF, et al. Olanzapine overdose mimicking opioid intoxication. Ann Emerg Med 1999;34:279.
13. Hustey FM. Acute quetiapine poisoning. J Emerg Med 1999;17:995.
14. Harmon TJ, et al. Loss of consciousness from acute quetiapine overdosage. J Toxicol Clin Toxicol 1998;36:599.
15. Burton S, et al. Ziprasidone overdose. Am J Psychiatry 2000;157:835.
16. House M. Overdose of ziprasidone. Am J Psychiatry 2002;159:1061.
17. Abilify (aripiprazole) product information. Bristol-Myers Squibb/Otsuka Pharmaceutical;2002.
Antipsychotic agents are a diverse class of drugs used to treat both psychiatric and nonpsychiatric conditions. This review will focus on the basic pharmacology, adverse effects, and toxicologic manifestations of the atypical antipsychotics.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
