Drug Criteria & Outcomes: Frovatriptan formulary evaluation
Drug Criteria & Outcomes: Frovatriptan formulary evaluation
By Melinda Spray,
PharmD
Written while on clinical clerkship with
Auburn University at Huntsville (AL) Hospital
Triptans
Sumatriptan (Imitrex) —
GlaxoSmithKline
Naratriptan (Amerge) — Glaxo-Wellcome
Rizatriptan (Maxalt, Maxalt MLT) — Merck
Zolmitriptan (Zomig) — AstraZeneca
Almotriptan (Axert) — Pharmacia
Frovatriptan (Frova) — Elan Corp.
Description
Of the triptans listed above, frovatriptan succinate is the triptan most recently approved by the Food and Drug Administration. The triptans are 5-hydroxytryptamine (5-HT) 1B/1D receptor agonists. The major use for this class of drugs is for the acute treatment of migraines with or without an aura in adults (age > 18 years). The current oral triptan on Huntsville Hospital System’s Formulary Interchange Program is zolmitriptan.
Mechanism of action
Frovatriptan binds to and stimulates the 5-HT 1B/1D receptors on extracerebral, intracranial arteries. This inhibits excessive vasodilation of these arteries, which occurs during a migraine.
Pharmacokinetics
The triptans are all very similar in many aspects; however, kinetic profiles differ from drug to drug. Frovatriptan’s beneficial pharmacokinetic difference from the other triptans is its long half-life. Frovatriptan’s disadvantage is a slower onset of action. The pharmacokinetics of the triptans are shown in Table 1, below.
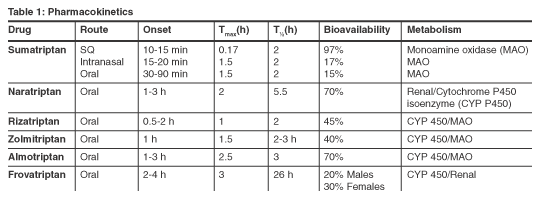
Indications and dosing
All triptans are indicated for the acute treatment of migraines with or without an aura in adults. Subcutaneous sumatriptan injection also is indicated for the acute treatment of cluster headaches. Dosing information is listed in Table 2 below.

Adverse events
Adverse events are similar with all drugs included in this class. These reactions are generally mild. Rare adverse events that could occur with the use of any triptan include coronary vasospasm, myocardial infarction, ventricular tachycardia, and ventricular fibrillation. Adverse reactions are listed by drug in Table 3, below.
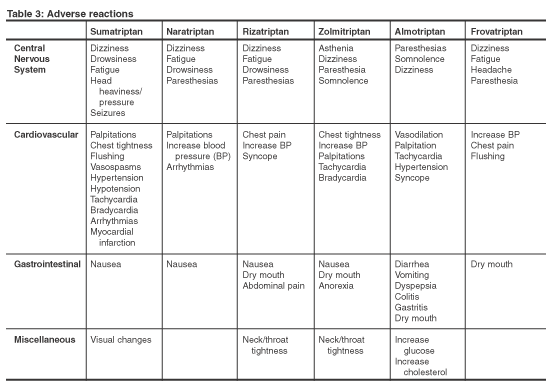
Contraindications
The contraindications for frovatriptan are similar to those of other triptans (see Table 4 below). Frovatriptan is not contraindicated in patients with severe renal or hepatic dysfunction.
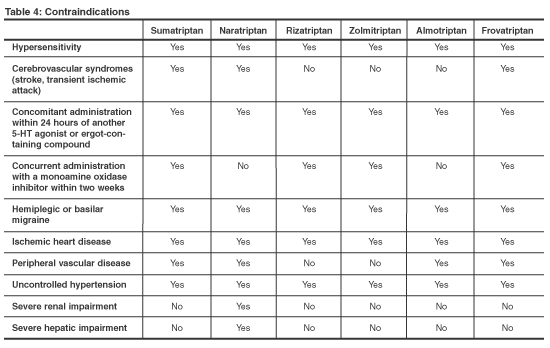
Drug interactions
Drug interactions generally are similar among the drugs of the triptan class (see Table 5 below). However, the triptan metabolic pathways differ, which could lead to different drug interactions. Some of the reactions are not documented with all the triptans; however, they still could occur. Many of the drug interactions that cause an increase in serum frovatriptan do not require dosage adjustment of the drug. Frovatriptan dosages as high as 40 mg have been observed to be safe.
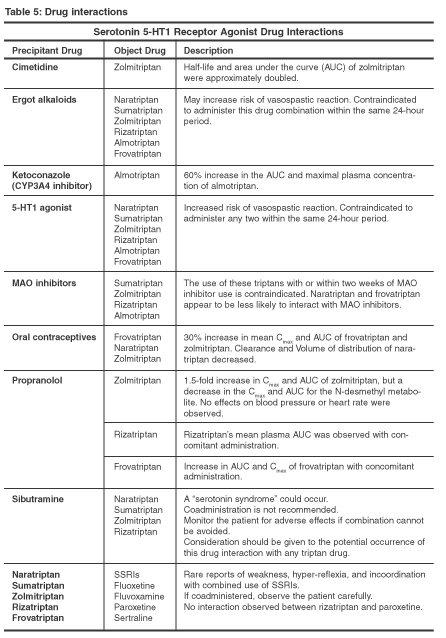
Cost
The hospital cost of frovatriptan is similar to that of most other triptan drugs at equivalent doses (in the $10-$12 range for a single dose).
Clinical studies
Two large-scale dose-finding studies were used to establish the therapeutic dose of frovatriptan. These studies established frovatriptan 2.5 mg as the dose that balanced efficacy and safety.
There also are trials evaluating pharmacokinetics and drug interactions. One published article compared tolerability and safety of frovatriptan and sumatriptan; however, this article did not compare efficacy or headache recurrence rate. The data demonstrated that up to three doses of frovatriptan 2.5 mg over a 24-hour period were well-tolerated by patients with migraine with short-term and long-term use.
There are five placebo-controlled trials establishing the efficacy of frovatriptan. Two of these studies are unpublished and unavailable from the drug company at this time. The published efficacy studies are VML 251/96/06 (study 1), VML 251/96/07 (study 2), and VML 251/96/09 (study 3). The three published studies were multi-center, randomized, double-blind, placebo-controlled, parallel-group clinical trials. Study 3 also used an active comparator. The demographic characteristics of the trial participants were similar among the treatment and placebo groups of all three studies, as well as among the three study groups. The participants of all three trials were mostly women (85-90%), and the mean age of the trial participants was 40.2-42.3 years. The primary efficacy endpoint of study 1 was two-hour headache response. Study 2 primary efficacy endpoints were two-hour and four-hour headache response and 24-hour headache recurrence. Study 3 primary efficacy endpoints were two-hour headache response and 24-hour headache recurrence.
Headache response at hour 4 is shown in Table 6, below. Two-hour headache response ranged from 37% to 46% with frovatriptan and 21% to 27% with placebo. The two-hour pain-free rates for frovatriptan were 9-14% compared with 1-5% with placebo. Four-hour headache response ranged from 56% to 65% with frovatriptan and 31% to 38% with placebo. Both of these efficacy endpoints established a statistically significant difference between the efficacy of frovatriptan and the efficacy of placebo.
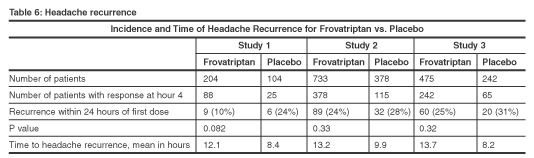
Frovatriptan has a long half-life and therefore is expected to have a lower headache recurrence rate. Studies 2 and 3 investigated headache recurrence as a primary endpoint, and study 1 also investigated incidence of headache recurrence. Table 6, above, shows the results for all three studies. These results do not show a statistically significant difference between frovatriptan and placebo with regard to headache recurrence.
Summary and recommendation
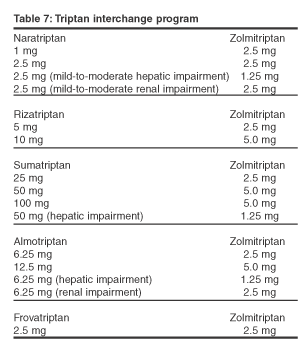
The placebo-controlled trials establish that frovatriptan’s efficacy is superior to placebo. However, these trials are published together, and all the information about the trials is not published in this article. There currently are no published studies comparing frovatriptan with another triptan. For this reason, it is difficult to evaluate frovatriptan’s place in therapy at the Huntsville Hospital System. Frovatriptan’s long half-life is a possible strength; however, there was not a statistically significant difference between the headache recurrence rates of frovatriptan and placebo. A weakness of frovatriptan is its slow onset of action when compared to other drugs of the triptan class. For these reasons, it is recommended that frovatriptan not be added to the Huntsville Hospital formulary at this time. Frovatriptan should be interchanged to the equivalent dose of the formulary drug, zolmitriptan, if no substitution is written with the drug order. (See Table 7, below.)
References
- Elan. Frovatriptan product information. San Diego; April 2002.
- Elan. Frovatriptan (Frova) package insert. San Diego; December 2001.
- Agents for Migraine: Serotonin 5-HT1 Receptor Agonists. Drug Facts and Comparisons. St. Louis: Facts and Comparisons; 2002:849-856.
- Ryan R, Geraud G, Goldstein J, et al. Clinical efficacy of frovatriptan: Placebo controlled studies. Headache 2002;42(suppl2):S84-S92.
- Personal communication. Cindy Hall, Buyer, Pharmacy Department. Huntsville (AL) Hospital System, September 2002.
- Jellin JM, editor. Migraines. Pharmacist’s Letter January 2002.
- Swidan S. Review of treatment strategies for successful migraine management: Focus on efficacy and safety of triptans. P & T 2002;27:402-409.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
