Chromium Supplementation for Weight Loss
Chromium Supplementation for Weight Loss
By Dónal P. O’Mathúna, PhD
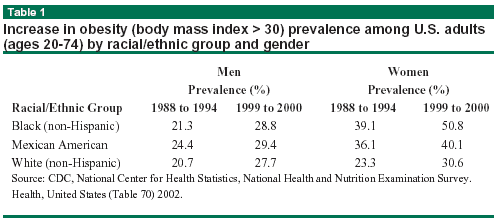
Yet another indication of the serious obesity problem in the United States was the selection of obesity for the April 9, 2003, thematic issue of the Journal of the American Medical Association (JAMA). Women in particular struggle with this issue, as Table 1 (below) demonstrates. The proportion of women categorized as overweight or severely obese has increased similarly over the same years.
Pharmaceutical responses to the problem received setbacks with the 1997 withdrawal of fenfluramine and dexfenfluramine (both associated with valvular regurgitation and primary pulmonary hypertension) and the 2000 withdrawal of phenylpropanolamine, present in popular over-the-counter weight-loss medications and associated with stroke.1 Diets remain perennially popular; however, the JAMA obesity theme issue included a review of popular low-carbohydrate diets that concluded there was insufficient evidence to recommend for or against their use.2
Frustrated by diets and concerned about the adverse effects of pharmaceuticals, many are turning to nonprescription weight-loss products, including dietary supplements. Such products are especially popular with young women.3 One of these is chromium, which generates more than $100 million in sales annually.4 The most popular formulation is chromium picolinate, available in pills, chewing gum, sports drinks, and nutrition bars. Products are marketed as "fat burners" both for those who want to lose weight and athletes who want to build muscle. Other claims made for these supplements are that they increase energy, curb addictions, cure acne, prevent insomnia, relieve depression, and increase life span.
Biochemistry
Chromium is an essential trace element and part of the insulin metabolic pathway. The U.S. Department of Agriculture estimated safe and adequate daily dietary intake of chromium is 50-200 micrograms (mcg) for adults (1 mcg = 0.001 mg).5 The Institute of Medicine (IOM) 2001 report on Dietary Reference Intakes set the Adequate Intake (AI) level for chromium at 35 mcg/d for young men and 25 mcg/d for young women. The AI is the amount expected to meet or exceed the daily requirements in essentially all healthy people.6 Establishing these levels, and measuring them clinically, is hampered by the extremely low levels of chromium in body fluids. There currently is no simple, reliable test for chromium deficiency.
Pharmacology
Chromium deficiency first was reported in 1977 when patients on long-term total parenteral nutrition developed classic diabetic symptoms that were reversed with the addition of chromium to their diets.7 The potentially beneficial role of chromium in treating certain patients with Type 2 diabetes (those who are chromium deficient) currently is receiving much attention.8 Because of the role of insulin in managing body weight, and given that chromium’s mechanism of action involves glucose and insulin metabolism, interest has arisen on the potential role of chromium in weight-loss strategies.
Mechanism of Action
Chromium deficiency leaves cells less sensitive to insulin (or "insulin resistant").9 As a result, glucose is less able to enter cells for energy production and instead is stored as fat. Insulin resistance also hinders the passage of amino acids into muscle cells, reducing protein synthesis.10 Chromium supplementation allegedly reverses these effects, leading to the "burning" of excess fat, weight loss, and increased muscle mass.
Clinical Studies
A meta-analysis of controlled clinical trials of chromium picolinate for weight loss was published in April 2003.11 Seventeen trials met the inclusion criteria of being randomized, placebo-controlled, and double-blind. However, seven could not be used in the meta-analysis because of incomplete data. None of these trials found significantly greater body weight loss in those taking chromium compared to placebo.
Data from the remaining 10 trials were pooled with the meta-analysis finding an overall significant reduction in body weight for those taking chromium picolinate compared to placebo. However, the mean difference in body weight loss was only 1.1 kg over 10-13 weeks (95% confidence interval [CI] of -1.8 to -0.4 kg). The reviewers questioned whether this was clinically significant, especially given that a 1,200 kcal/d diet achieves a mean weight loss of 0.5-0.6 kg/week.11 A similarly small but significant reduction (-1.2%) in body fat was found and a nonsignificant effect on lean body mass.
A general concern with meta-analyses is whether significant overall findings are real or an artifact of the statistical pooling. This is of particular concern with this meta-analysis because, individually, only two trials had statistically significant results favoring chromium. One method of checking the validity of the robustness is to remove from the analysis the data from the most positive trial. When these data were removed from this chromium meta-analysis, the significant reduction in body weight and percent body fat was eliminated. Such a finding requires that the overall result of the meta-analysis be interpreted with caution.11
Further examination of the two trials with positive effects from chromium supplementation reveals important methodological weaknesses. In the first, 154 adults were divided into three groups and received placebo, 200 mcg, or 400 mcg chromium picolinate daily.12 Subjects were instructed to consume "at least two servings" of a protein drink containing their assigned chromium dose. After 72 days, underwater displacement testing showed significantly reduced body fat in the chromium groups compared to placebo, but no significant difference between the two chromium groups. Mean weight loss difference was 1.0 kg on 200 mcg (95% CI of -2.4 to 0.4 kg) or 1.3 kg on 400 mcg (95% CI of -2.3 to -0.3 kg). Amount of drink consumed, overall diet, and exercise frequency were not controlled (whereas diet and exercise were monitored in all other studies included in the meta-analysis).
In the second positive study, the same researchers randomly assigned 130 new subjects to take capsules containing either 400 mcg chromium picolinate or placebo.10 After 90 days, body composition was measured using dual energy X-ray absorptiometry. Changes in actual weight, percent body fat, and fat-free mass did not differ between the two groups, but the chromium group showed significantly reduced fat mass (P = 0.023). After statistical adjustments to control for dietary and exercise differences, significant differences were calculated for actual weight (P < 0.001), percent body fat (P < 0.001), and fat mass (P < 0.001). However, the significant differences were found only in calculated estimates of weight and fat loss based on energy expenditure, not the measured values. The measured mean weight loss difference was 1.1 kg (95% CI of -2.3 to 0.1 kg).
Adverse Effects
Chromium supplements are believed to be safe, with no clinical studies reporting adverse reactions. The IOM report found insufficient evidence to set an Upper Limit.6 Rats given several thousand times the equivalent of 200 mcg/d chromium in humans showed no adverse effects.13 However, a small number of cases of liver toxicity and other adverse effects have recently been reported.8,14 The FDA has received several hundred adverse event reports involving chromium supplements, though most involve dietary supplements containing numerous herbs and other agents.9
In vitro studies have demonstrated that chromium picolinate can produce chromosomal damage in hamster ovary cells15 and can cleave DNA in solution.16 Although the former study used doses vastly in excess of normal physiological levels, the latter occurred within physiological ranges. Chromium picolinate’s unique stability gives it good absorption, but also allows accumulation in body tissues, leading to concerns about long-term side effects.
Drug Interactions
No adverse drug interactions have been reported. However, ascorbic acid, aspirin, and indomethacin markedly increase chromium absorption, while antacids lower absorption.5 Diets high in complex carbohydrates, not simple sugars, increase chromium absorption.5 Potential interactions may occur with drugs affecting glucose or cholesterol levels, or with corticosteroids.17
Formulation
Trivalent Cr3+ is the natural form of chromium found almost exclusively in foods, especially brewer’s yeast, liver, American cheese, cereals, and wheat germ. However, chromium content in foods is highly variable, and processing can either increase or decrease the level.6 The search for the biologically active form of chromium led to the extraction of glucose tolerance factor (GTF) from yeast.18 This complex contains chromium, nicotinate, and amino acids and has led to several new chromium complexes being tested for efficacy and safety. Chromium picolinate is the form most commonly used in supplements, usually 200 mcg capsules. Many products contain smaller amounts of chromium picolinate along with numerous herbs and minerals.
Conclusion
Chromium’s essential role in insulin metabolism is well-established. Supplementation in chromium-deficient diabetics may offer some benefits, including improved weight management.8 However, the majority of controlled trials of chromium picolinate as a weight-loss supplement found no significant benefit for weight loss or percent body fat reduction. A meta-analysis of these trials found a small, but statistically significant benefit. Two trials contributed heavily to this benefit and both had serious methodological weaknesses. However, both were also larger than any of the other trials. Research is lacking on the long-term effects of consuming chromium picolinate.
Recommendation
Chromium supplementation offers very little, if any, benefit to women attempting to lose weight. Although some controversy exists over the possibility of serious harmful effects, these have not been observed in clinical trials. Diabetic patients should not take chromium without first consulting their physicians and monitoring their blood glucose closely. For those attempting to lose weight, lifestyle changes continue to offer most benefit. For example, reducing the amount of prolonged sedentary activities, especially hours watching TV, could potentially reduce the number of women with obesity by 30%.19 Other lifestyle changes more likely to produce weight loss than dietary supplements include reducing caloric intake, increasing exercise, modifying eating behaviors, and enlisting others’ support. These remain the foundations of successful weight-loss programs.
Dr. O’Mathúna
is Professor of Bioethics and Chemistry, Mount Carmel College of Nursing, Columbus,
OH.
References
1. Food and Drug Administration. Letter to manufacturers of drug products containing phenylpropanolamine (PPA). Nov. 3, 2000. Available at: www.fda.gov/cder/drug/infopage/ppa/ppaltr.pdf. Accessed Dec. 1, 2000.
2. Bravata DM, et al. Efficacy and safety of low-carbohydrate diets. JAMA 2003;289:1837-1850.
3. Blanck HM, et al. Use of nonprescription weight loss products. Results from a multistate survey. JAMA 2001;286:930-935.
4. Vincent JB. Biological role for chromium at a molecular level. Accounts Chem Res 2000;33:503-510.
5. Stoecker BJ. Chromium. In: Shils ME, et al, eds. Modern Nutrition in Health and Disease. 9th ed. Baltimore, MD: Lippincott, Williams & Wilkins; 1999: 277-282.
6. Institute of Medicine. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Washington, DC: National Academy Press; 2001.
7. Jeejeebhoy KN. Chromium and parenteral nutrition. J Trace Elem Exp Med 1999;12:85-89.
8. O’Mathúna DP. Chromium supplementation in the treatment of Type 2 diabetes mellitus. Altern Med Alert 2000;3:40-44.
9. Porter DJ, et al. Chromium: Friend or foe? Arch Fam Med 1999;8:386-390.
10. Kaats GR, et al. A randomized, double-masked, placebo-controlled study of the effects of chromium picolinate supplementation of body composition: A replication and extension of a previous study. Curr Ther Res 1998;59:379-388.
11. Pittler M, et al. Chromium picolinate for reducing body weight: Meta-analysis of randomized trials. Int J Obes Relat Metab Disord 2003;27:522-529.
12. Kaats GR, et al. Effects of chromium picolinate supplementation of body composition: A randomized, double-masked, placebo-controlled study. Curr Ther Res 1996;57:747-756.
13. Ravina A, et al. Reversal of corticosteroid-induced diabetes mellitus with supplemental chromium. Diabetic Med 1999;16:164-167.
14. Lana S, et al. Chromium-induced toxic hepatitis. Eur J Intern Med 2002;13:518-20.
15. Stearns DM, et al. Chromium(III) picolinate produces chromosome damage in Chinese hamster ovary cells. FASEB J 1995;9:1643-1648.
16. Speetjens JK, et al. The nutritional supplement chromium(III) tris(picolinate) cleaves DNA. Chem Res Toxicol 1999;12:483-487.
17. Jellin JM, et al. Pharmacist’s Letter/Prescriber’s Letter: Natural Medicines Comprehensive Database. Stockton, CA: Therapeutic Research Facility; 2003.
18. Mertz W. Chromium in human nutrition: A review. J Nutr 1993;123:626-633.
19. Hu FB, et al. Television watching and other sedentary behaviors in relation to risk of obesity and type 2 diabetes mellitus in women. JAMA 2003;289:1785-1791.
O'Mathuna DP. Chromium supplementation for weight loss. Altern Ther Women's Health 2003;5(5):33-36.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
