Emergencies in the obstetric patient can be double trouble
By Christopher O. Wood, MD, Resident, Department of Emergency Medicine, Indiana University School of Medicine, Indianapolis. Gregory P. Moore, MD, JD, Associate Clinical Professor, Department of Emergency Medicine, Indiana University School of Medicine, Indianapolis; Reid Hospital, Richmond, IN; Kaiser Permanente Sacramento/Roseville, CA. Rebecca D. Wood, JD, Attorney, Pentera Group Inc., Indianapolis.
Editor’s note: This is the third part in the ED Legal Letter series on abdominal pain. The first two parts detailed common presentations of adult abdominal pain. Part four will discuss pediatric abdominal pain. This third part will discuss ectopic pregnancy and trauma in pregnancy. The cases presented will describe negligence of unsuspecting emergency physicians who did not follow acceptable risk management strategies in dealing with obstetric patients and encountered subsequent litigation. These common chief complaints of patients presenting to the emergency department (ED) can lead to high-risk complications for the patient and unborn child if not adequately evaluated and promptly treated. The risk management strategies in this article, if followed, will improve patient outcomes and reduce the potential for litigation.
Ectopic Pregnancy
Case #1. Nicholas vs. Brooklyn Hospital Center.1 On July 6, 1999, Ella Nicholas presented to Woodhull Medical Center in Brooklyn, NY, with complaints of abdominal and pelvic pain. Mrs. Nicholas was seen by the ED physician, who told her that she was in good health. There were no further instructions or tests. After this visit, she took a two-week vacation and developed a ruptured ectopic pregnancy that resulted in death on Aug. 1, 1999. Her husband sued the medical center, claiming that the ED physician failed to perform an appropriate examination, including failure to perform an ultrasound. The defendants argued that the complaints did not necessitate an ultrasound, and that her care was acceptable. This action eventually settled for $2 million.
Discussion
The incidence of ectopic pregnancy in the United States has increased significantly during the past few decades, with more than 100,000 cases annually,2 accounting for approximately 2% of reported pregnancies. Complications from ectopic pregnancy are the leading cause of first-trimester mortality, and related deaths account for 9% of all pregnancy-related deaths in the first trimester.3 The estimated yearly cost of hospitalization, disability, and lost wages due to ectopic pregnancy exceeds $1 billion.4
Bilateral ectopic pregnancies have been reported, but are rare. Heterotopic pregnancies occur in 1/10,000 to 1/30,000 and are increasing with use of infertility drugs. Risk factors for ectopic pregnancy include tubal surgery,5 previous ectopic pregnancy,6 IUD use,7 pelvic inflammatory disease,8 endometriosis, infertility treatment, and in-utero exposure to diethylstilbestrol.
Any woman of childbearing age who presents with irregular menses, vaginal bleeding, or abdominal pain may have an ectopic pregnancy. There is significant risk of missing an ectopic pregnancy. Ectopic pregnancy is missed up to 50% of the time at the first office visit, and 36% of the time at the first ED visit.9 Vital signs may or may not be helpful in distinguishing ectopic pregnancy from alternative diagnoses. Hypotension is a variable finding and may be seen in 10-50% of patients.10 Tachycardia or bradycardia may be seen. Tachycardia often is seen in association with hypovolemia and is an early sign of shock due to hemorrhage. Bradycardia also may be seen in ectopic pregnancy and may be due to increased vagal tone associated with peritoneal irritation due to intra-abdominal bleeding.
The classic triad for ectopic pregnancy is lower abdominal pain, amenorrhea, and vaginal bleeding. However, this triad is poorly sensitive or specific. These three symptoms occur together in only 40-70% of cases of proven ectopics.11 Abdominal pain is the most reliable symptom and is present in 95-100% of cases. Pain due to unruptured ectopic is classically steady and localized to one or both lower quadrants, but pain may be intermittent and colicky. One author found that women with ectopic pregnancy were more likely to have moderate-to-severe sharp pain, lateral or bilateral tenderness on abdominal or pelvic exam, and a uterus less than 8-week size with cervical motion tenderness on pelvic exam.12 Midline pain decreased the risk of ectopic. Pain with urgency to defecate in acutely ill patients suggests the possibility of intra-abdominal bleeding and may be associated with rupture.13 Ruptured ectopic presents with acute pain of fewer than 24 hours duration, whereas those with unruptured ectopic commonly complain of more persistent pain for more than 24 hours.14 Pain may be exaggerated by urination or defecation due to peritoneal irritation. Movement or bending over also may worsen pain.
Back pain and shoulder pain may be seen due to hemorrhage with peritoneal irritation. About 50% of patients with hemoperitoneum have peritoneal signs on exam. Adnexal tenderness occurs in most patients and usually is ipsilateral but may be bilateral. Adnexal mass is a variable finding, present in up to 40-50% of patients and usually is ipsilateral.11 One group found that in 40 patients with proven ectopic, adnexal mass could not be palpated even under anesthesia.15 Cervical motion tenderness can be seen due to hemoperitoneum, and fullness in the cul-de-sac can signify rupture.16 Vaginal bleeding is reported in 60-80% of patients. More than 80% of patients will describe a missed period. Most patients will present 4-5 weeks after their missed period, during weeks 4-12 of pregnancy.
Advances in diagnostic testing have helped reduce morbidity and mortality by allowing for earlier and more accurate diagnosis. Use of beta-hCG testing and ultrasonography reduced the case fatality rate from 35.5 per 10,000 ectopic pregnancies in 1970 to 3.4 per 10,000 pregnancies in 1987.17 Any woman of childbearing age with abdominal pain should have a urine pregnancy test (UPT) performed on arrival to the ED. UPT is both sensitive and specific for pregnancy,18 and false-negative results are rare. Serum beta-hCG testing should be done in those women with a positive UPT at risk for ectopic. Serum testing is used in combination with ultrasound to differentiate intrauterine pregnancy (IUP) from threatened abortion or ectopic. Mean levels will be lower for ectopic vs. IUP. Levels should double every 48 hours in IUP up to about 6-7 weeks gestational age.19 Longer doubling times (about seven days) suggest abnormal IUP or ectopic.20 Serum testing is of major value in establishing the discriminatory zone above which a normal IUP should be visualized on ultrasound. Absence of intrauterine gestational sac on transvaginal ultrasound (TVUS) when hCG levels exceed the discriminatory zone suggests ectopic.19 For transabdominal ultrasound, absence of gestational sac with a serum beta-hCG level greater than 6500 mIU/mL is suggestive for ectopic.21 For transvaginal ultrasound, a gestational sac should be seen with levels greater than 1500 mIU/mL. Patients with levels greater than 2000 and no definite IUP on TVUS are at high risk of ectopic pregnancy.22
Pelvic ultrasound has become a key diagnostic aid in identifying and ruling out ectopic pregnancy. Ultrasound (US) is indicated in all women with pelvic pain or vaginal bleeding and a positive UPT23, 24 that have not had previous ultrasound to identify an IUP. Emergency physicians commonly begin evaluation with bedside transabdominal ultrasonography (TAUS). Bedside US in the ED is fast, reliable, accurate, and improves outcomes.22, 25, 26 One study argues for a cost-effective strategy where emergency physicians screen all patients at risk for ectopic with US at the first visit and obtain formal US if ED US is indeterminate or shows no IUP.27 Evidence of a normal IUP includes appearance of a normal-appearing, double-ringed gestational sac and yolk sac, fetal pole, or heartbeat.25 This usually excludes ectopic pregnancy, as heterotopic pregnancy is rare.
TVUS provides greater resolution of pelvic structures vs. TAUS and is superior in diagnosing ectopics.24 TVUS is used in women with nondiagnostic results on TAUS. An adnexal mass plus the presence of free pelvic fluid correlates best with ectopic.28 Endometrial stripe thickness is reduced and is predictive of risk of ectopic pregnancy when hCG less than 1000.29 Free fluid or blood in the cul-de-sac or intraperitoneal gutters suggests rupture but no finding on TVUS is a reliable indicator of rupture. Even though TVUS is superior in diagnosis of ectopic pregnancy, about 15% of TVUS are indeterminate,30 and 25% of these patients eventually will be diagnosed with ectopic.31 The rate of change of serial hCG is predictive of ectopic pregnancies in symptomatic patients with indeterminate TVUS.32 Patients with increasing hCG and an empty endometrial cavity on US are at highest risk, and two-thirds will go on to have a confirmed ectopic.
All women suspected of ectopic pregnancy and who have unknown Rh status should have Rh testing performed. Anti-D immune globulin should be administered to Rh-negative patients. Management of patients with suspected ectopic pregnancy hinges on a combination of ultrasonography in conjunction with beta-hCG testing and a consideration of the clinical picture. (See Figure 1.)33
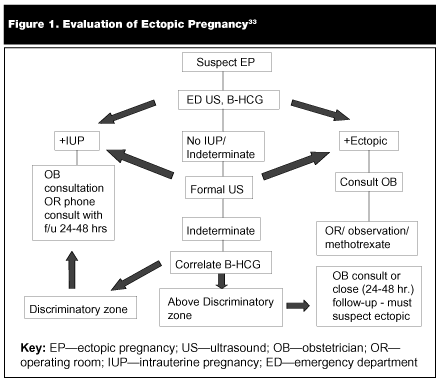
Care should be taken when discharging patients with low beta-hCG levels and indeterminate examinations, as a significant number of patients in this subgroup will develop ectopic pregnancy. To minimize the likelihood of ruptured ectopic pregnancy outside of the hospital, patients and family members require detailed instructions (including risks and symptoms that warrant immediate return for re-evaluation), and close follow-up with serial beta-hCG examinations should be arranged. Patients should be involved in decision-making, and discussions should be documented.
Pitfalls in Management
1. Failure to maintain a high index of suspicion for ectopic pregnancy. Ectopic should be considered in all women of childbearing age with abdominal pain or vaginal bleeding, and urine pregnancy testing is mandatory.
2. Failure to consider ectopic in women with risk factors. Women with a history of tubal surgery, previous ectopic, IUD use, or PID have an increased risk of ectopic pregnancy, and the diagnosis must be considered.
3. Over-reliance on vital signs and physical exam findings. A high index of suspicion is required for early and accurate diagnosis.
4. Over-reliance on laboratory testing and sonographic findings. Serum beta-hCG testing and ultrasonography are aids in the diagnosis and management of women with suspected ectopic; however, they do not replace clinical judgment. The quality of ultrasonography is operator-dependent, and the absence of an adnexal mass or free fluid on ultrasound does not exclude ectopic pregnancy.
5. Failure to arrange for follow-up or provide detailed discharge instructions. Obstetric consultation is critical in women with findings suggestive of ectopic pregnancy. Careful planning with detailed instructions and close obstetric follow-up is crucial for women who are to be discharged home.
Preeclampsia and Eclampsia
Case #1. Doolittle v. Phipps.34 Lynn Doolittle was approximately 30 weeks pregnant in October 1992 when she developed severe abdominal pain, headaches, nausea, and vomiting. She presented to Physicians Immediate Care and was seen by Dr. Phipps. Dr. Phipps found Mrs. Doolittle to have elevated blood pressure and a significant amount of proteinuria. Despite this, she was discharged with a diagnosis of upper respiratory tract infection and was treated with bed rest and over-the-counter medication. Mrs. Doolittle visited relatives the next day, where she developed worsening headaches and hypertension. Later that evening, she was taken to a nearby hospital and was diagnosed with preeclampsia. She was transferred to another hospital and, after arrival, developed seizures and placental abruption. Emergency cesarean section was performed, but her son suffered brain damage due to fetal hypoxia and intracerebral hemorrhage. He developed cerebral palsy and seizure disorder as a result of his injuries. A $1.5-million settlement was reached.
Discussion
Severe preeclampsia is an obstetric emergency with potential fatal outcome for both mother and fetus. The major goal for ED physicians is the early diagnosis of preeclampsia and initiation of therapy to prevent maternal complications without compromising fetal well-being. Hypertension in pregnancy is the most common medical disorder of pregnancy, affecting 5-15% of pregnancies.35 Of these women, 70-80% have preeclampsia.36
Preeclampsia is defined as a maternal blood pressure of greater than 140 mmHg systolic or greater than 90 mmHg diastolic, usually beginning after 20 weeks gestation.37 It usually is associated with proteinuria and/or edema. It is a pregnancy-specific syndrome of reduced organ perfusion related to vasospasm, activation of the coagulation cascade, and perturbations in blood volume and pressure control. It most commonly affects women at extremes of reproductive age, younger than 19 years or older than 35 years old.38 It is the second-leading cause of maternal mortality in the United States and is a major cause of neonatal morbidity and mortality.
Risk factors for preeclampsia include nulliparity, diabetes mellitus, underlying renal disease, history of maternal hypertension, previous history of preeclampsia, obesity, lack of prenatal care, multiple gestation, and hydatidiform mole. Eclampsia is defined as the presence of seizures in a woman with preeclampsia that cannot be attributed to other causes. Traditionally, it has been thought of as the end-stage of preeclampsia, but some argue that it is a subset rather than the end of a continuum.39
Preeclampsia may occur at any point during gestation or during the postpartum period. Most women present near term, and it is seldom seen at fewer than 20 weeks gestation. Preeclampsia at fewer than 20 gestation weeks may signify the presence of a hydatidiform mole, multiple gestation, or collagen vascular disease. Eclampsia is the most dangerous complication of preeclampsia. Most eclampsia occurs antepartum; however, postpartum eclampsia does occur and can present without classic pre-eclamptic signs.40 Other complications include HELLP syndrome (Hypertension, Elevated Liver enzymes, Low Platelets), disseminated intravascular coagulation, pulmonary edema, acute renal failure, hepatic hematoma, and placental abruption.
The classic presentation of preeclampsia occurs in the woman in her third trimester of pregnancy who presents with hypertension, proteinuria, and generalized edema, but the presentation often is variable. Women with mild disease often are asymptomatic and have only hypertension and mild proteinuria. Signs and symptoms indicating severe eclampsia include blood pressure greater than 160 mmHg systolic or 90 mmHg diastolic, headache, visual disturbance, shortness of breath, abdominal pain, nausea and vomiting, and rapid weight gain or onset of generalized edema. (See Table 1.)

The diagnosis of preeclampsia is clinical, and a high index of suspicion is required to make the diagnosis. Hypertension is the sine qua non of preeclampsia,41 and a search for the illness should be made in any pregnant woman with elevated blood pressure. History and physical examination should be directed to look for signs and symptoms of severe disease such as petechiae, dyspnea and rales, epigastric or right upper quadrant abdominal tenderness, oliguria, altered mental status, and hyperreflexia. These findings should prompt the emergency physician to rapid action to prevent eclampsia and the onset of seizures. In all women with suspected preeclampsia, intravenous access should be obtained and fetal monitoring should be initiated. Foley catheterization should be performed in all women with proven preeclampsia to monitor urine output for signs of oliguria. Laboratory studies should include urinalysis, complete blood count, platelet count, electrolytes (including magnesium), BUN and creatinine, liver function tests, LDH, and coagulation profile. Radiologic imaging is of limited benefit. Ultrasonography is useful to monitor fetal well-being. Computed tomography of the head is unnecessary unless the mother develops focal neurologic symptoms or altered mental status.42
The goals of the ED management of women with preeclampsia are fourfold: 1) to prevent maternal complications without compromising fetal well-being; 2) treat hypertension to prevent maternal cardiovascular and neurologic complications; 3) prevent seizures; and 4) provide monitoring of maternal and fetal condition. Hospitalization is required to prevent maternal seizures and to monitor the fetus. Early obstetric consultation is critical. For cases of mild preeclampsia, management primarily consists of bed rest and observation of maternal and fetal well-being. For severe preeclampsia, the initiation of seizure prophylaxis and antihypertensive therapy is indicated. The definitive treatment is delivery of the fetus.
Magnesium sulfate is the drug of choice to prevent seizures in preeclampsia. It has been shown to be superior to diazepam and phenytoin in the prevention of seizures in women with preeclampsia.43,44 An initial bolus of 4 g of a 20% solution is infused over 10-15 minutes, followed by a continuous infusion of 2 g/h. Mental status, respiratory rate, urine output, and patellar reflexes are monitored frequently while the infusion is continued. Toxicity occurs with levels that exceed 7 mEq/L. Loss of patellar reflexes is an early sign of magnesium toxicity and occurs at levels of 8-10 mEq/L. Respiratory depression occurs with levels exceeding 11 mEq/L, and respiratory arrest occurs with levels greater than 13 mEq/L. If signs of toxicity develop, the infusion should be discontinued. Overdose also may be reversed with intravenous calcium gluconate, 1 g of a 10% solution over 3-5 minutes.
Antihypertensive medications are indicated in women with severe preeclampsia (blood pressure greater than or equal to 160/110). The goal of therapy is to lower the maternal blood pressure to a diastolic pressure of 90-100 mmHg. Further reduction in maternal blood pressure may impair placental perfusion and produce fetal distress. It should be remembered that blood pressure control does not cure preeclampsia or prevent progression to eclampsia. The main goal of blood pressure control is to prevent cardiovascular and neurologic complications.
Hydralazine is the first-line agent for blood pressure control. It is given intravenously with an initial dose of 5 mg, followed by repeated doses of 5-10 mg IV every 20 minutes if there is no response. A second-line agent should be considered if there is no response after a total dose of 20 mg. Labetolol is a second-line agent when hydralazine fails. Sodium nitroprusside is a third-line option in rare cases when both hydralazine and labetalol fail to lower maternal blood pressure. If nitroprusside is used, invasive blood pressure monitoring is recommended, as this medication can precipitate severe hypotension that can lead to uteroplacental insufficiency with fetal distress.
Management of full-blown eclampsia requires prompt action on the part of the emergency physician to prevent further complications in both the mother and fetus. Airway and breathing must be addressed and airway management initiated if necessary. A search for other causes of seizure should be made. Immediate administration of magnesium sulfate is critical to terminate ongoing seizures and prevent further seizures in women with eclampsia.45 Dosing of magnesium sulfate is similar for that in women in preeclampsia. Up to 10-15% of women will have a second seizure,41 and a second loading dose of 2-4 g IV may be administered. In cases of recurrent seizures, treatment with intravenous diazepam and/or phenytoin may be attempted.
Preeclampsia: Pitfalls in Management
1. Failure to recognize preeclampsia. A search for preeclampsia should be made in all pregnant women with hypertension.
2. Failure to admit patients with preeclampsia. Women with preeclampsia, regardless of severity, require urgent OB consultation and hospital admission for fetal and maternal monitoring.
3. Delay in administration of magnesium sulfate. Therapy with magnesium sulfate should be initiated immediately in all women with severe preeclampsia to prevent seizures.
4. Delay in treatment of seizures. Treatment for eclampsia should be started immediately in any pregnant woman who presents with hypertension and seizures.
5. Delay in treatment of hypertension. Although lowering blood pressure does not cure preeclampsia or prevent seizures, careful blood pressure control is necessary to prevent maternal cardiovascular and neurologic complications.
Spontaneous Abortion
Case #1. Pressley v. Oakwood Hospital.46 Angelia Pressley, a 19-year-old woman at 18 weeks gestation, presented to Oakwood Hospital’s ED with complaints of lower abdominal cramps and vaginal spotting. Dr. Kossak, the OB attending, placed Ms. Pressley on a fetal monitor for one hour. Ms. Pressley was discharged home 2½ hours later. After arriving home, Ms. Pressley continued to have cramping and later miscarried. It was later discovered that Ms. Pressley had a urinary tract infection (UTI). Ms. Pressley sued the hospital and Dr. Kossak, claiming that the plaintiff failed to recognize premature labor, failed to prescribe intravenous antibiotics for the UTI and IV fluids to stop the labor, and that she was discharged home before the urinalysis was completed. The defendants contended that Ms. Pressley’s evaluation and treatment were appropriate, that cramping had ceased at the time of discharge, and that even with antibiotics and IV fluids, Ms. Pressley still would have miscarried. The jury returned a verdict of $100,000 in favor of the plaintiff.
Discussion
Spontaneous abortion is defined as the expulsion of the products of conception without external interference prior to 20th week of gestation. The majority occur before 16 weeks, with a most of these occurring before eight weeks’ gestation. There are five types of spontaneous abortion. A threatened abortion occurs when there is vaginal bleeding in first half of pregnancy with closed cervical os. An inevitable abortion occurs with vaginal bleeding in the first half of pregnancy with progressive cervical dilation but no passage of the products of conception. An incomplete abortion occurs with the partial passage of the products of conception with an open cervical os. A "missed" abortion refers to a nonviable embryo or fetus that is retained in utero. The term is less accurate since ultrasonography and serial beta-hCG testing can provide accurate diagnosis of fetal death. First-trimester or second-trimester fetal death are more accurate terms. In a completed abortion, all of the products of conception have been passed, and the cervical os is closed. Threatened abortion occurs in 10-20% of recognized pregnancies, and 50-75% of threatened abortions end in miscarriage.47-49 Up to 30% of all pregnancies may result in spontaneous abortion with early pregnancy loss.50
Abdominal pain and vaginal bleeding in a pregnant woman are classic signs of spontaneous abortion. In threatened abortion, vaginal bleeding is mild. Lower abdominal pain or cramping may be absent, but when present, the risk of progressing to complete miscarriage is increased.51 Moderate vaginal bleeding is seen with an inevitable abortion, and pain due to uterine contractions is more persistent. Vaginal bleeding is heavier and sometimes excessive in cases of incomplete abortion. This occurs because the partially separated placenta prevents myometrial contraction; life-threatening hemorrhage may result. Bleeding is minimal or absent with missed abortion; in most cases, women will present with an intermittent brown discharge. In completed abortions, sudden cramping and vaginal bleeding occur, followed by slight vaginal spotting and diminished cramping after the products of conception are fully expelled.
Serious complications from miscarriage are rare. Mortality from spontaneous abortion is low, but life-threatening hemorrhage can occur with incomplete abortion. Chorioamnionitis and disseminated intravascular coagulation are potential complications in women with missed abortion. Other complications include recurrent spontaneous abortion and maternal depression due to the psychological trauma due to miscarriage. Fortunately, women who miscarry have a high likelihood of delivering a live infant with the next pregnancy. Apgar found that in women with one or two previous miscarriages, there is an 80-85% chance of the next pregnancy resulting in a live-born child.48 Wilcox also found that most women with early loss had normal fertility and delivered a live infant; one-third of women conceived in next cycle and 95% eventually conceived.50 This information may be conveyed to comfort an ED patient who has lost a fetus.
In the ED, a careful history and physical examination is necessary. Physical examination should include a careful abdominal examination searching for evidence of tenderness, peritonitis, and uterine size. During the pelvic examination, one should assess: 1) whether the cervix is open or closed; 2) if clots or tissue are present in the vaginal vault; 3) the degree of vaginal bleeding; and 4) uterine size, tenderness, and the presence of any adnexal masses or tenderness. When in doubt, the cervix may be probed with a pair of ring forceps to determine whether the internal os is open or closed. Cotton swabs should be avoided, and probing should be avoided completely in the second trimester. Any clots or tissues found in the vault should be examined for chorionic villi.
Laboratory testing is limited in the diagnosis and management of spontaneous abortion. A complete blood count may be helpful in cases of persistent bleeding to assess the degree of blood loss. All women with suspected spontaneous abortion require Rh typing and administration of anti-D immunoglobulin if Rh-negative. A search for urogenital infection is indicated, as treatment of asymptomatic bacteriuria and bacterial vaginosis may prevent preterm labor and later miscarriage.52
As with ectopic pregnancy, ultrasonography is helpful in the diagnosis and management of spontaneous abortion. An ultrasound aids in determining the location of the pregnancy, assessing fetal viability, and identifying the presence of retained products of conception. TAUS should detect the presence of fetal heart tones (FHT) in most pregnancies by 7-8 weeks and in virtually all pregnancies by 9-10 weeks. If FHT are identified, the risk of miscarriage, even in the face of vaginal bleeding, is only 5.5%.53 In several studies, the absence of FHT at 9-10 weeks gestational age accurately predicted miscarriage in nearly all cases.54,55 However, failure to detect FHT at fewer than 9-10 weeks cannot reliably predict status or outcome, especially in patients with a closed cervix. A gestational sac should be visible on TVUS by 5 weeks, a yolk sac by 6 weeks, and fetal heart motion by 8 weeks gestational age.
The role of serum beta-hCG testing in the management of spontaneous abortion is to help assess the viability of the fetus when ultrasonography is indeterminate or when gestational age is fewer than 6-7 weeks.56 Stable or declining hCG levels, or evidence of fetal demise on ultrasound, are strong evidence of nonviability and miscarriage.
Management of spontaneous abortion in the ED first focuses on ensuring the hemodynamic stability of the mother. Once stability is ensured, management is based on the type of spontaneous abortion. All Rh-negative women should receive anti-D immunoglobulin.
In the first trimester, a 50-mcg dose is given and a full 300-mcg dose is given in the second trimester. For threatened abortion, patient education and support are key goals in management. Up to 50% of women with threatened abortion will go on to miscarry, and there is no treatment to prevent miscarriage.
The woman with a threatened abortion should be advised that routine activities will not harm the pregnancy. She should avoid intercourse, tampons, and douching as long as she is having bleeding. If tissue is passed, it should be saved and brought to the obstetrician or ED for examination. She should receive explicit instructions to return immediately for fever, a significant increase in bleeding, worsening pain, or other symptoms that might indicate ectopic pregnancy. For inevitable or incomplete miscarriage, dilation and curettage is standard treatment to remove remaining intrauterine contents. It also is recommended for cases of first- and second-trimester fetal death due to the theoretical risk of disseminated intravascular coagulation and the psychological effects of carrying a dead fetus.51 For completed abortion, management is complicated because it is rarely clear whether the miscarriage truly is completed. Women who miscarry and have no or minimal evidence of endometrial tissue on ultrasound, mild bleeding, and gestational age fewer than eight weeks may be managed expectantly.57 On the other hand, dilation and curettage is indicated in women with significant residual tissue. If a woman is to be managed expectantly, she should be instructed to return for fever, cramping or pain, increased bleeding, or the passage of tissue. Gynecologic follow-up should be arranged to ensure completion of the miscarriage.
Pitfalls in Management
1. Failure to recognize spontaneous abortion. Spontaneous abortion should be considered in any pregnant woman with vaginal bleeding or abdominal pain. Fetal viability should be performed.
2. Failure to consider ectopic pregnancy. Ectopic pregnancy should be considered in all women who are being evaluated for possible spontaneous abortion.
3. Failure to identify and treat urogenital infection. Asymptomatic bacteriuria and bacterial vaginosis have been identified as causes of preterm labor and premature rupture of membranes. Failure to identify and treat these conditions may result in later complications.
4. Failure to check Rh status and provide anti-D immunization. All Rh-negative pregnant women with spontaneous abortion should receive anti-D immuno-globulin in the ED before admission or discharge.
5. Failure to consult an OB. Consultation with an OB should be performed in cases where the gestational age of the fetus is at the borderline of viability.
Trauma in Pregnancy
Case #1. Sutton v. Winter Haven Hospital.58 Stephanie Sutton was eight months pregnant when she was involved in a motor vehicle accident on Jan. 18, 1995.
Dr. Stamper, the ED physician, assumed her care. Mrs. Sutton claimed that she asked Dr. Stamper to examine her baby in utero, but this was not done. During her evaluation, the ED nursing staff had difficulty obtaining a consistent fetal heartbeat. The nursing staff contacted Mrs. Sutton’s OB, Dr. Fearing, to inform her of the accident and concerns for the baby’s well-being.
According to the plaintiffs, Dr. Fearing did not immediately come to the hospital, nor did she order continuous fetal monitoring. In addition, they claimed that Dr. Fearing did not consider emergent cesarean delivery after being notified of the nursing staff’s inability to obtain a consistent fetal heartbeat. The Suttons argued that negligence on part of Dr. Stamper, Dr. Fearing, and the hospital nursing staff resulted in the stillbirth of their son a few days later.
The defendants denied negligence, claiming that the baby had died prior to arrival to the ED due to complete placental abruption and umbilical cord infarction at the time of the accident. The Suttons, however, maintained that a gradual abruption occurred at the time of the accident, which progressed to a complete abruption that led to her son’s death. Drs. Fearing and Stamper claimed that the ED nursing staff were negligent in failing to report the inability to obtain heart tones, and if this had been reported in a timely fashion, actions would have been taken to save the child’s life. The jury returned a $501,141 verdict in favor of the Suttons.
Discussion
Traumatic wounds and injuries occur in 6-7% of all pregnancies. Trauma is the most common cause of maternal death, accounting for 46.3% of fatalities in pregnant women.59 The management of the pregnant trauma patient poses two unique challenges to the emergency physician. First, the pregnant woman undergoes several anatomic and physiologic alterations during gestation that alter evaluation and management. Second, the ED physician has two lives to consider, each with a different response to trauma.60 Optimal care is critical to ensure the best possible outcome for both mother and child.
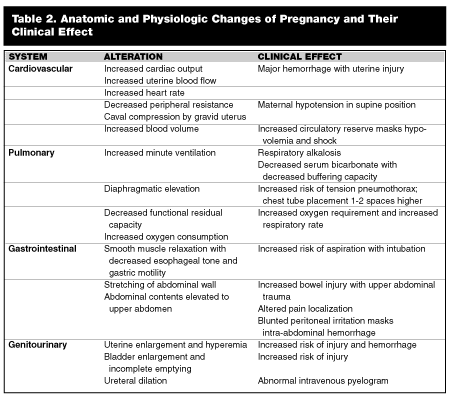
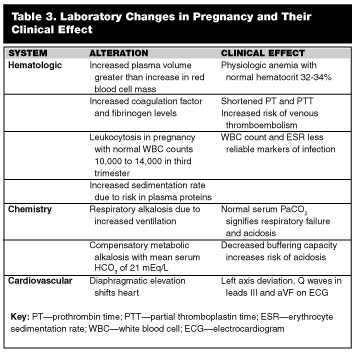
There are multiple anatomic and physiologic changes that occur during pregnancy. (See Table 2.) Laboratory values also are altered in pregnancy, and results of laboratory tests must be interpreted in light of these changes. (See Table 3.) These alterations must be accounted for when managing the pregnant trauma patient.
Most cases of trauma in pregnancy are due to blunt trauma. Although interpersonal violence and direct falls are potential causes of blunt abdominal trauma, motor vehicle collisions account for the majority of traumatic injuries in pregnancy. A major problem in many of these cases is the lack of or improper use of seat belts.61 Unbelted women have a higher risk of fetal death, and improperly placed lap belts can cause a significant increase in force transmitted through the uterus.62 This transmitted force may result in either placental abruption or uterine rupture. Placental abruption occurs when the stiff, inelastic placenta shears away from the elastic uterine wall. This results in disruption in placental blood flow, resulting in fetal hypoxia and distress. Signs of abruption include vaginal bleeding, uterine tenderness, contractions, or irritability. Up to 70% of fetal losses occur due to abruptio placentae,63 with fetal mortality reaching more than 50%.64 In addition, women with placental abruption are at a higher risk of disseminated intravascular coagulation. Uterine rupture is a relatively rare occurrence and most often is seen with high-speed motor vehicle collisions, which distribute large forces to the uterus. Maternal shock, abdominal pain, and palpable fetal parts are typical findings. Fetal demise is common, as placental blood flow is disrupted completely by uterine rupture. Direct fetal injury may arise due to maternal pelvic fractures, and fatal in utero injuries often are due to intracranial hemorrhage or skull fracture.
Penetrating trauma in pregnancy is a less common event, but it comprises a disproportionately large percentage of trauma cases in urban areas.65 The gravid uterus alters injury patterns in cases of penetrating trauma. The uterus protects the abdominal viscera from injury but places the fetus at high risk of injury and death. Fetal injury rates of up to 89% and mortality of up to 71% have been reported.66
Also, there is an increased risk of injury to the bowel or solid organs with upper abdominal wounds due to elevation by the gravid uterus.
Management of the pregnant trauma patient follows the ABCs of basic and advanced trauma life support. The primary survey focuses on the mother, with a limited examination of the fetus to assess gestational age and fetal heart tones. The mother, due to a limited oxygen reserve and increased oxygen demand during pregnancy, quickly can become hypoxic, causing fetal distress. Therefore, oxygen therapy should be started early and continued throughout the primary and secondary surveys until maternal hypoxia, shock, and fetal distress have been ruled out. Early intravenous access is critical for volume resuscitation. Care must be taken when assessing for maternal hypovolemia, as signs of shock can and will be masked by an increase in maternal blood volume during pregnancy. Signs of hypotension and shock may not appear until significant blood loss has occurred, and fetal compromise may be advanced by this time. Early fluid resuscitation with crystalloid and blood are thus indicated. Lactated Ringer’s solution is the resuscitation fluid of choice. Unless spinal cord injury is suspected, all women at more than 20 weeks gestation should be tilted to the left to avoid hypotension due to compression of the inferior vena cava by the gravid uterus. At the end of the primary survey, a focused examination of the uterus is performed to assess gestational age. Fundal height provides a quick estimation of gestational age and potential viability. Fetal heart tones can be assessed by Doppler ultrasound as early as 10 weeks of age. If the uterus is fewer than 24 weeks in size, or if fetal heart tones are absent, treatment should focus solely on the mother.
A sterile speculum examination should be performed to visualize for genital tract injury, cervical dilation, and any vaginal bleeding or leakage. Evidence of vaginal bleeding suggests the presence of placental abruption, while clear fluid may represent rupture of membranes. Fetal evaluation during the secondary survey centers on assessing for fetal heart rate and movement. All women greater than 20 weeks gestation should be placed on continuous tococardiographic monitor. Fetal monitoring is essential, as the fetus is the "canary in the coal mine" that serves to warn of occult maternal hypovolemia.67 Signs of fetal hypoxia and distress include tachycardia or bradycardia, decreased beat-to-beat variability, and late decelerations.
In the pregnant trauma patient, routine trauma blood work should be sent. In addition, all mothers should have blood type with Rh factor screening performed. Coagulation studies should be sent in cases of multiple trauma or if abruption is suspected. A Kleihauer-Betke (KB) test also should be performed to assess for fetomaternal hemorrhage (FMH) due to trauma. FMH can result in Rh sensitization of Rh-negative mothers. Most labs that use the KB test screen for FMH of 5 mL or more. Unfortunately, the amount of FMH sufficient to sensitize an Rh-negative woman is less than this 5-mL level. Therefore, all Rh-negative women should receive anti-D immune globulin. A single 300-mcg dose RhIG protects against 30 mL of FMH; additional doses are given when the amount of FMH exceeds this amount.
Radiographic studies play a role in the diagnosis and management of maternal injuries, and necessary diagnostic studies should not be withheld out of concern for fetal radiation. However, these studies should be performed with regard for the fetus’ protection, and radiation exposure should be minimized. Ultrasonography is the first-line imaging modality to evaluate for maternal blunt abdominal trauma. Ultrasound is fast, noninvasive, and allows for simultaneous assessment of fetal viability and maternal injuries. Ultrasonography is sensitive, specific, and accurate for detecting intra-abdominal injuries in blunt trauma.68 It also allows assessment of fetal heart rate, movement, gestational age, and amniotic fluid volume. It is not, however, reliable for the diagnosis of placental abruption, as up to 50% of injuries may be missed.69 Diagnostic peritoneal lavage (DPL) also can be safely performed to evaluate for intra-abdominal injury, and computed tomography may be used in the stable patient with indeterminate ultrasound or DPL findings.
Ultimate management depends on the stability of the mother and fetus. Maternal stabilization is key to preventing fetal distress. When the mother has been stabilized, care then can focus on the child. Fetal loss can occur even in cases of minor trauma or when abdominal injury is absent; therefore, all pregnant trauma patients greater than or equal to 20 weeks gestation should be monitored. Monitoring is essential to help ensure fetal survival. One group found that up to 60% of fetal deaths after emergent cesarean section occur with the underuse of monitoring and delay in recognition of fetal distress.70 Another study recommends a minimum of four hours of cardiotocographic monitoring to assess for signs of fetal distress or contractions that may signify placental abruption.69 If the mother and fetus are unstable, maternal resuscitation is critical to ensure fetal well-being. Any life-saving procedures should be performed prior to cesarean section. Perimortem cesarean section may be performed emergently in cases of maternal cardiopulmonary arrest with gestational age greater than 20 weeks and fetal heart tones present.
Pitfalls in Management
1. Failure to secure the airway and provide oxygenation. The pregnant woman has a limited oxygen reserve and can become hypoxic quickly. In addition, the fetus cannot tolerate prolonged periods of hypoxia.
2. Reliance on maternal heart rate and blood pressure to diagnose blood loss. Pregnant women have increased vascular reserve and may not show signs of hypovolemia until significant blood losses have occurred. Fetal distress is the most sensitive sign of maternal hypovolemia. Early IV access and fluid resuscitation is key to maintaining intravascular volume and ensuring uterine perfusion.
3. Reliance on ultrasonography to diagnose placental abruption. Ultrasound is not sensitive for abruption. Fetal distress is the most sensitive indicator of placental abruption, and fetal monitoring is critical.
4. Failure to monitor for signs of intrauterine pathology. Fetal loss may occur even in cases of minor trauma. All women greater than 20 weeks gestation require a minimum of four hours of cardiotocographic monitoring to assess for fetal distress and other signs of placental abruption.
5. Failure to recognize fetal distress. Normal fetal heart rate is 120-160 beats per minute. Signs of fetal distress include bradycardia, tachycardia, decreased variability, and decelerations.
References
1. Sonny Nicholas, indiv. and as Adm. of the Est. of Ella Nicholas v. The Brooklyn Hospital Center and "J.Y." intended to be the physician who signed the emergency room record of July 6, 1999, Kings County (NY) Supreme Court, Index No. 9098/01.
2. Tilden FF, Powers RD. Ectopic pregnancy: Avoiding missed diagnosis and reducing morbidity. Emerg Med Rep 1996;17:199-206.
3. Centers for Disease Control. Ectopic pregnancy — United States 1990-1992. MMWR 1995;44:46-49.
4. Ander DS, Ward KR. Medical management of ectopic pregnancy: The role of methotrexate. J Emerg Med 1997;15:177-182.
5. Peterson HB, Xia Z, Hughes JM, et al. The risk of ectopic pregnancy after tubal sterilization. N Engl J Med 1997;336-762-767.
6. Ankum WM, Mol BWJ, Van der Veen F, et al. Risk factors for ectopic pregnancy: A meta-analysis. Fertil Steril 1996;65:1093-1099.
7. Franks AL, Beral V, Cates W, et al. Contraception and ectopic pregnancy risk. Am J Obstet Gynecol 1990;163:1120-1123.
8. Marchbanks PA, Annegers JF, Coulam CB, et al. Risk factors for ectopic pregnancy. JAMA 1988;259:1823-1827.
9. Tintinalli JE, Ruiz E, Krome RL, eds. Emergency Medicine: A Comprehensive Study Guide. 4th ed. New York City: McGraw-Hill;1996:556.
10. Gonzalez FA, Waxman M. Ectopic pregnancy, a retrospective study of 501 consecutive patients. Diag Gynecol Obstet 1981;3:181-186.
11. Bryson CP. Beta-subunit of human chorionic gonadotropin, ultrasound, and ectopic pregnancy: A prospective study. Am J Obstet Gynecol 1983;146:163-165.
12. Dart RG, Kaplan B, Varaklis K. Predictive value of history and physical examination in patients with suspected ectopic pregnancy. Ann Emerg Med 1999;33:283-290.
13. Hadjis NS, McAuley GJ, Ruo L, et al. Acute abdominal pain and urgency to defecate in the young and old: A useful symptoms complex? J Emerg Med 1999;17:239-242.
14. Singh KB, Poole CA, Otterson WN, et al. Characteristics of indigent women with ruptured and unruptured tubal pregnancies. J Reprod Med 1992;37:745-748.
15. Pagano R. Ectopic pregnancy: A seven-year survey. Med J Aust 1981;2:586-588.
16. Gonzalez FA, Waxman M. Ectopic pregnancy, a prospective study on differential diagnosis. Diag Gynecol Obstet 1981; 3:101-109.
17. Ory ST. New options for diagnosis and treatment of ectopic pregnancy. JAMA 1990;267:534-537.
18. O’Connor RE, Bibro CM, Pegg PJ, et al. The comparative sensitivity and specificity of serum and random urine hCG determinations in the ED. Am J Emerg Med 1993;11:434-436.
19. American College of Obstetrics and Gynecology. Medical management of tubal pregnancy: Clinical management guidelines for obstetrician-gynecologists. ACOG Practice Bulletin No. 3, 1998 Dec.
20. Bateman BG, Nunley WC, Klop LA, et al. Vaginal sonography findings and hCG dynamics of early intrauterine and tubal pregnancies. Obstet Gynecol 1990;75:421-427.
21. Kadar N, DeVore G, Romero R. Discriminatory hCG zone: Its use in the sonographic evaluation for ectopic pregnancy. Obstet Gynecol 1981;58:156-161.
22. Mateer JR, Valley VT, Aiman EJ, et al. Outcome analysis of a protocol including bedside endovaginal sonography in patients at risk for ectopic pregnancy. Ann Emerg Med 1996;27:283-289.
23. Trompler VA, Peacock JE, Sullivan DJ. Ectopic pregnancy: Early diagnosis is essential. ED Legal Letter 1996;7:79-90.
24. Dart RG. Role of pelvic ultrasonography in evaluation of symptomatic first-trimester pregnancy. Ann Emerg Med 1999;33:310-320.
25. Kaplan BC, Dart RG, Moskos M, et al. Ectopic pregnancy: Prospective study with improved diagnostic accuracy. Ann Emerg Med 1996; 28:10-17.
26. Mateer JR, Aiman EJ, Brown MH, et al. Ultrasonographic examination by emergency physicians of patients at risk for ectopic pregnancy. Acad Emerg Med 1995;2:867-873.
27. Durston WE, Carl ML, Guerra W, et al. Ultrasound availability in the evaluation of ectopic pregnancy in the ED: Comparison of quality and cost-effectiveness with different approaches. Am J Emerg Med 2000;18:408-417.
28. Tongsong T, Pongsatha S. Transvaginal sonographic features in diagnosis of ectopic pregnancy. Int J Gynecol Obstet 1993;43:277-283.
29. Dart RG, Dart LM, Mitchell P, et al. The predictive value of endometrial stripe thickness in patients with suspected ectopic pregnancy who have an empty uterus at ultrasonography. Acad Emerg Med 1999;9:602-609.
30. Dart RG, Dart LM, Mitchell P, et al. The utility of a dilation and evacuation procedure in patients with symptoms suggestive of ectopic pregnancy and indeterminate transvaginal ultrasonography. Acad Emerg Med 1999;6:1024-1029.
31. Braffman BH, Coleman RG, Ramchandani P, et al. Emergency department screening for ectopic pregnancy: A prospective U.S. study. Radiology 1994;190:797-802.
32. Dart RG, Mitterando J, Dart LM. Rate of change of serial beta-human chorionic gonadotropin values as a predictor of ectopic pregnancy in patients with indeterminate transvaginal ultrasound findings. Ann Emerg Med 1999;34:703-710.
33. Stephanee Evers, MD, personal communication, 2003.
34. Jordan Doolittle by Lynn and Scott Doolittle v. Jerald Phipps, MD, and Physicians Immediate Care, Winnebago County (IL) Circuit Court. Medical Malpractice: Verdicts, Settlements, and Experts 2002;16(12):25.
35. Brown MA. Pregnancy-induced hypertension: Pathogenesis and management. Aus N Z J Med 1991;21:257-273.
36. Brown MA, Whitworth JA. The kidney in hypertensive emergencies — victim and villain. Am J Kidney Dis 1992;20:427-442.
37. Working Group on High Blood Pressure in Pregnancy. Report of the national high blood pressure education program working group on high blood pressure in pregnancy. Am J Obstet Gynecol 2000;183:S1-S22.
38. Doan-Wiggins L. Hypertensive disorders of pregnancy. Emerg Clin North Am 1987;5:495-508.
39. Katz VL, Farmer R, Kuller JA. Preeclampsia into eclampsia: Toward a new paradigm. Am J Obstet Gynecol 2000;182:1389-1396.
40. Veltkamp R, Kupsch A, Polasek J, et al. Late onset postpartum eclampsia without preeclamptic prodrome: Clinical and neuroradiological presentation in two patients. J Neurol Neurosurg Psychiatry 2000;69:824-827.
41. Benrubi GI, et al. Contemporary Issues in Emergency Medicine. New York City: Churchill Livingstone;1990:89-100.
42. Brown CEL, Purdy P, Cunningham FG. Head computed tomographic scans in women with eclampsia. Obstet Gynecol 1988;159:915-920.
43. Eclampsia Collaborative Trial Group. Which anticonvulsant for women with eclampsia? Evidence from the Collaborative Eclampsia Trial. Lancet 1995;345:1455-1463.
44. Lucas MJ, Leveno KJ, Cunningham FG. A comparison of magnesium sulfate with phenytoin for the prevention of eclampsia. N Engl J Med 1995;333:201-205.
45. Rosen P, et al. Emergency Medicine: Concepts and Clinical Practice. Fifth ed. Chicago:Mosby;1998:2421-2424.
46. Angelia Pressley v. Oakwood Hospital and Medical Center and Marie Kossak, MD, Wayne County (MI) Circuit Court, Case No. 97741050NM.
47. Abbott J, Zaccardi M, Lowenstein SR. Threatened abortion: A prospective study of predictors of outcome in an emergency department population (abstract). Ann Emerg Med 1989;18:439.
48. Apgar BS, Churgay CA. Spontaneous abortion. Prim Care 1993; 20:621-627.
49. Everett CB. Incidence and outcome of bleeding before the 20th week of pregnancy: Prospective study from general practice. BMJ 1997;315:32-34.
50. Wilcox AJ, Weinberg CR, O’Connor JF, et al. Incidence of early loss of pregnancy. N Engl J Med 1988;319:189-194.
51. Sinkinson CA, Baker DP. The complaint of vaginal bleeding during pregnancy: A double dose of anxiety. Emerg Med Rep 1989; 10:1-8.
52. Vause S, Johnson T. Management of preterm labor. Arch Dis Child 2000; 83:F79-85.
53. Tongsong T, Srisomboon J, Wanapirak C, et al. Pregnancy outcome of threatened abortion with demonstrable fetal cardiac activity: A cohort study. J Obstet Gynaecol 1995;21:331-335.
54. Eriksen PS, Philipsen T. Prognosis in threatened abortion evaluated by hormone assays and ultrasound scanning. Obstet Gynecol 1980; 55:435.
55. Hertz JB, Mantoni M, Svenstrup B. Threatened abortion studied by estradiol-17 beta in serum and ultrasound. Obstet Gynecol 1980; 55:324.
56. Rosen P, et al. Emergency Medicine: Concepts and Clinical Practice. Fifth ed. Chicago: Mosby; 2002:2413-2416.
57. Schiff E, Ben-Baruch G, Moran O, et al. Prediction of residual trophoblastic tissue in first-trimester abortions and low levels of human chorionic gonadotropin beta-subunit. Am J Obstet Gynecol 1990;162:797-801.
58. Stephanie Sutton and James Sutton, her husband v. Winter Haven Hospital Inc.; Gessler Clinic, PA; Patricia Fearing, MD; and William J. Stamper, MD, Polk County (FL) Circuit Court, Case No. GC-G-96-2256.
59. Fildes J, Reed L, Jones N, et al. Trauma: The leading cause of maternal death. J Trauma 1992;32:643-645.
60. Pimentel, L. Mother and child: Trauma in pregnancy. Emerg Clin North Am 1991;3:549-563.
61. Shah KH, Simons RK, Holbrook T, et al. Trauma in pregnancy: maternal and fetal outcomes. J Trauma 1998;45:83-86.
62. Rosen P, et al. Emergency Medicine: Concepts and Clinical Practice. Fifth ed. Chicago: Mosby; 2002:256-266.
63. Corsi PR, Rasslan S, de Oliveira LB, et al. Trauma in pregnant women: Analysis of maternal and fetal mortality. Injury 1999;30:239-243.
64. Rodgers FB, Rozycki GS, Osler TM, et al. A multi-institutional study of factors associated with fetal death in injured pregnant patients. Arch Surg 1999:134:1274-1277.
65. Fildes J, Reed L, Jones N, et al. Trauma: The leading cause of maternal death. J Trauma 1992;32:643-645.
66. Buschbaum HJ, et al. Trauma in Pregnancy. Philadelphia: WB Saunders; 1979:82-142.
67. Schmidt J, Moore GP. Management of multiple trauma. Emerg Med Clin N Am 1993;11:29-51.
68. McKenney MG, Martin L, Lentz K, et al. 1,000 consecutive ultrasounds for blunt abdominal trauma. J Trauma 1996;40:607-610.
69. Pearlman MD, Tintinalli JE, Lorenz RP. A prospective controlled study of outcome after trauma during pregnancy. Am J Obstet Gynecol 1990;162:1502-1507.
70. Morris JA Jr., Rosenbower TJ, Jurkovich GJ, et al. Infant survival after cesarean section for trauma. Ann Surg 1996;223:481-488.
This is the third part in the ED Legal Letter series on abdominal pain. The first two parts detailed common presentations of adult abdominal pain. Part four will discuss pediatric abdominal pain. This part discusses ectopic pregnancy and trauma in pregnancy.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
