Pediatric Viral Exanthems: Distinguishing the Benign from the Serious
Authors: Catherine A. Marco, MD, FACEP, Associate Professor, Surgery, Medical College of Ohio; Attending Physician, St. Vincent Mercy Medical Center, Toledo, OH; Valerie R. Lint, DO, MS, St. Vincent Mercy Medical Center, Toledo, OH; Jennifer A. Schuster, MD, St. Vincent Mercy Medical Center, Toledo, OH .
Reviewer: Alfred Sacchetti, MD, FACEP, Chairman, Emergency Medicine, Our Lady of Lourdes Medical Center, Camden, NJ.
Rashes are common in children, and therefore, a frequent emergency department (ED) concern. The ED physician must be familiar with common rash patterns in children—the majority of which can be diagnosed by a complete history and physical examination.
The authors review the classic course of common pediatric diseases associated with rashes, including varicella-zoster virus, herpes simplex virus (HSV), roseola, and rubella. Understanding the classic patterns, disease progression, high-risk populations, and potential complications allow the ED physician to avoid unnecessary testing in low-risk patients with a classic presentation, and aggressively approach potentially significant rashes in high-risk populations (e.g., neonatal HSV).
This article also is designed to increase ED physicians’ awareness of treatment strategies associated with common viral exanthems.
Note: This article contains information about an off-label use of oral antivirals. — The Editor
Introduction
Dermatologic findings are common among pediatric emergency patients. More than 50 infectious agents have been identified that may cause exanthems in the pediatric population.1 Results of one study demonstrated that 72% of cases of fever and rash in the pediatric population were caused by viruses; 20% by bacteria.2 Many exanthems have specific patterns of lesions, distributions, and clinical history; some of the more common exanthems are detailed in this article. However, many viral exanthems are nonspecific, and establishing a definitive etiology may not be feasible.
Important historical elements include duration of symptoms; associated symptoms (e.g., fever, headache, gastrointestinal symptoms); time course of lesions; distribution; associated symptoms (e.g., itching, pain); and exacerbating and relieving factors. Medical history, medication usage, and allergies may be useful information. The physical examination should be conducted in a focused manner. General appearance and vital signs should be taken and stabilized. Details regarding the lesions should be documented, including size, color, secondary findings (e.g., scale, excoriations), and distribution. A differential diagnosis should be established, appropriate treatment initiated, and timely follow-up ensured. In stable cases where the diagnosis is uncertain, supportive care and timely follow-up are appropriate.
Varicella-Zoster Virus
Varicella-zoster virus is a double-stranded DNA virus and a member of the Herpes viridae family. It is the unique etiologic agent that causes both varicella (chickenpox) and herpes zoster (shingles). The history of these two distinct, yet closely linked, viral exanthems date to 1888 when children developed primary varicella after exposure to individuals with acute zoster infections.3 The origin of the word chickenpox is hypothesized to be from the word "chiche-pois," French for chickpea, used to describe the vesicle size. The term zoster is a Greek word that describes the binding of one’s armor, similar to the distribution of the characteristic zoster rash.4 Without immunization, the lifetime risk of varicella-zoster virus infection is about 95%, with most reported cases (90%) involving chickenpox in children 14 years and younger.5 The lifetime risk of zoster is approximately 10-30%, with marked increases among the elderly (older than 85 years).6 Varicella has a global distribution, but it is more prevalent in temperate climates, occurring most often during the months of March to May. However, zoster does not share in these same epidemiologic facts of season, geographic, or climate trends. Varicella is primarily a childhood illness. Although children rarely acquire zoster, children who are immunosuppressed (who have HIV infection or cancer or who take immunosuppressive drugs) are susceptible. It is considered one of the most infectious viral diseases in humans with an estimated 80-90% transmission rate among household contacts.7 It is contagious by direct contact with infected vesicular fluid and inoculation by airborne transmission.
Acute varicella among young, healthy, nonpregnant individuals often follows a benign, self-limiting disease course. Prior to the appearance of the rash, a 1-2 day prodromal period may occur, which includes headache, malaise, and fever. However, children may present with the rash and fever simultaneously. The typical pruritic rash of varicella begins on the head, spreads centripetally, finally reaching the extremities. Proximal extremities are more affected than distal. The rash occurs in various stages of evolution from macules to papules, and then to vesicles on an erythematous base, commonly described as "dewdrop-on-a-rose-petal." After the vesicles have formed, they develop to pustules that eventually form crust and scab, while new crops of vesicles form during a 2-5 day period. Therefore, the chicken pox lesions classically will be found at various stages of this transformation. Adults, older children, and secondary cases of varicella acquired from household contacts typically will have a more severe course, with more lesions, more systemic complaints and a higher risk of serious complications, such as pneumonia, encephalitis, and death. Other severe sequelae that may occur include skin bacterial superinfection (the most common complication among healthy children), neurologic complications, (e.g., acute cerebellar ataxia and meningoencepahalitis), varicella pneumonia (an infrequent complication among children), asymptomatic transient hepatitis, immune-mediated thrombocytopenia, and rare ophthalmologic complications (e.g., keratitis and anterior uveitis). Varicella-zoster virus infection during pregnancy carries risks both to the mother and the infant.
After the resolution of primary varicella, the virus enters the latency phase, where it lies dormant in ganglia, most commonly the thoracic and trigeminal ganglia. With time, the cell-mediated immunity to varicella will decline. In conjunction with immunosuppressive factors, the latent virus may become active. The reactivation of varicella-zoster virus appears in one or more ganglia in the appearance of herpes zoster, also known as shingles. (See Figure 1.)
Figure 1. Herpes Zoster

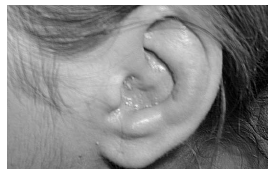
Prior to the appearance of the lesions, a prodrome of burning, pain, itching, or tingling occurs and may last for several days. The vesicles are distributed along a dermatomal pattern, and may persist for up to four weeks if not treated with antiviral agents. Significant morbidity occurs among the elderly with the persistence of pain and dyesthesia, known as postherpetic neuralgia. Additionally, the disease course among immunocompromised patients tends to be more severe, involving multiple nerve roots, and in some cases leading to disseminated visceral disease. It is important to also examine for the presence of the ocular manifestations of varicella-zoster virus, since the virus can affect many ocular structures. Specifically, herpes zoster ophthalmicus affects the areas distributed by the ophthalmic division of the trigeminal nerve.8 It can lead to chronic ocular inflammation, visual loss, tissue scarring, and debilitating pain. Ocular manifestations should be considered in any individual with involvement of the tip of the nose, known as Hutchinson’s sign. In 1864, Hutchinson discovered that tissue involvement along the distribution of the nasociliary nerve is associated with ocular complications. Herpes zoster oticus may present with devastating otalgia coinciding with vesicular involvement of the external ear canal and pinna. (See Figure 2.) Ramsey-Hunt syndrome is the disease entity in which herpes zoster oticus produces a facial paralysis. The onset of pain in and around the ear, mouth, and the face may precede the rash for hours to days. Additional findings may include tongue lesions, alteration in taste, and the inability to fully close the ipsilateral eye.
Figure 2.
Ramsey-Hunt Syndrome (Herpes Zoster Oticus)
The varicella vaccine was first developed in Japan in 1974. This live attenuated vaccine (Oka strain) was approved in 1995 for the use in the United States. The administration of this vaccine has affected the incidence of varicella in addition to the 80% decline in varicella-related hospitalizations.9 The vaccine’s effectiveness is estimated to be around 70-90%, with only mild, breakthrough cases. Children can receive the single-dose vaccine between 12-15 months of age; it is recommended that two doses be given 4-8 weeks apart for older children and adults. However, there remains controversy on the vaccine’s lifetime immunity and the need for a booster.
In healthy children, treatment of varicella is supportive and consists of antipruritics, skin hygiene, antipyretics, and adequate hydration. The use of antiviral agents such as acyclovir has been shown to be of clinical and economical benefit, however they have not been proven to decrease the complication rate among healthy children. Therefore, their use typically is reserved for newborns, preterm infants, children older than 13 years, and adults. Because replication occurs during the first few days, it is best to start the antiviral therapy within 24 to 48 hours from the onset of the rash. Intravenous acyclovir is recommended for the immunocompromised patient to help prevent dissemination and shorten the course. Moreover, antiviral agents should be administered for all patients who present with the zoster eruption within three days of the appearance of the skin lesions. Acyclovir, valacyclovir, and famciclovir are the primary antivirals available. Disseminated zoster should be treated with intravenous acyclovir. The addition of a steroid may reduce the incidence of postherpetic neuralgia, if administered within 72 hours of the ap-pearance of lesions.
Herpes Simplex Virus
HSVs are DNA viruses that occur worldwide in pandemic proportions and commonly result in ED visits due to their frequency and intense pain-causing syndromes.
The viruses can exist as two separate types: herpes simplex virus type 1 (HSV-1) and herpes simplex virus type 2 (HSV-2); each has characteristic antigenic differences, and separate clinical and epidemiologic manifestations.10 HSV infections can range from benign herpes labialis, to painful genital herpes and even life-threatening neonatal herpes. The emergency clinician will face HSV infections commonly in the ED and should be aware of the diagnostic challenges and latest treatments—particularly for neonatal herpes and disseminated HSV—for which missed diagnosis and delayed treatment can be devastating.
Herpes viruses are common etiologies for acute skin infections that may be acquired at an early age and reach 80% incidence in the general adult population.11 Transmission of the virus is by direct contact with mucous membranes or non-intact skin. The infection rate of HSV-1 is more frequent among children (younger than 5 years) of lower socioeconomic class, and increases to 70-80% by adolescent and early adulthood. Additionally, genital herpes is one of the most common sexually transmitted diseases worldwide.
Neonatal herpes simplex virus, a devastating disease of the newborn, is estimated at one per 3,000 to 20,000 live births.12 HSV-2 is responsible for the majority of neonatal HSV cases, but 15-30% of the cases are due to HSV-1. Primary infection of genital HSV occurring during the third trimester of pregnancy, in a patient who has not had complete seroconversion by the onset of labor, leads to a 33% incidence of transmission to the infant. However, recurrent maternal infection has only a 3% incidence of viral transmission, due to protective maternal immunity against HSV-2. Occult, asymptomatic cervical shedding of the virus nevertheless leads to many, unpredicted neonatal infections.
Other HSV infections that warrant mention, but are beyond the scope of this article, include Kaposi varicelliform eruption (eczema herpeticum), herpes whitlow (vesicular outbreaks on the hands and fingers), herpes gladiatorum (eruption on the torso of athletes and wrestlers), herpetic sycosis (follicular infection of the bearded area) and disseminated HSV infection (usually among immunocompromised patients.)
The primary infection of both HSV-1 and HSV-2 are the most severe states, characterized by clusters of painful vesicles on an erythematous base. Systemic symptoms including fever, myalgias, and headache often are present. The characteristic lesions may persist for several weeks; however, resolution of the primary infection does not correlate with eradication of the virus.13 The virus remains latent in the neural ganglion cells where reactivation may occur, resulting in less severe, episodic outbreaks in the same patient. During the sub-clinical phase, asymptomatic shedding may occur and sexual partners should be cautioned regarding possible transmission.
HSV-1 is more likely to recur orally leading to herpes labialis, also known as cold sores. Primary infection is characterized by painful vesicles on the lips, gingiva, palate, or the tongue, often preceded by a prodromal period of fever, sore throat, and tender lymphadenopathy. Also known as herpetic gingivostomatitis, children age 1-5 years are most commonly afflicted with symptoms lasting 10-14 days, although any age may suffer from the disease.14 Recurrences usually present as lesions on the vermillion border preceded by pain, burning, itching, and paresthesias. Recurrences may be prompted by factors such as stress, fatigue, illness, dental trauma, and ultraviolet light.
HSV-2 infections most commonly affect the genital area. Outbreaks can occur within two days to two weeks post viral exposure and lead to a painfully debilitating state. Symptoms associated with this outbreak include fever, malaise, edema, inguinal lymphadenopathy, dysuria, and vaginal/penile discharge. Up to 25% of women with primary HSV-2 infections may develop the complication of aseptic meningitis.
The diagnosis of neonatal HSV can be challenging. The presentation includes nonspecific signs and symptoms such as irritability, lethargy, fever, and failure to thrive at one week of life. Most of the neonates do not develop skin lesions that suggest the diagnosis; thus, HSV should be considered in the differential of nonspecific illness in the neonatal period, especially among premature infants and those who have a low birth weight. Infants with congenital HSV infection—4% of all neonatal HSV infections—may be born with microcephaly, hydrocephalus, chorioretinitis, and vesicular skin lesions.
Oral and labial HSV-1 infections primarily are treated supportively with topical anesthetic agents for symptomatic relief. Oral antivirals (e.g., acyclovir) also may be used, but have not been approved for pediatric use.
Treatment regimens for HSV-2 differ for primary and recurrent infections. All primary HSV genital infections should be treated with antiviral medications, which include acyclovir, famciclovir, or valacyclovir.15 All three regimens work to inhibit viral replication, thereby reducing the duration of the painful lesions and the viral shedding. All three agents may be used for suppressive therapy for patients with recurrent outbreaks during a period of six years or longer.
Neonatal HSV must be treated aggressively and the ED physician should treat all infants 8 weeks and younger with vesicular skin lesions with empiric acyclovir.16 Prophylactic parenteral acyclovir should be started for infants born to mothers suspected of having HSV, particularly for infants born by vaginal delivery. All neonates suspected of having neonatal HSV should undergo a full septic workup and admission.
Erythema Infectiosum and Other Parvovirus B19 Infections
Erythema infectiosum was first recognized in 1889 as fifth disease because of its position among the list of other common childhood exanthems and the chronologic order in which they were first reported.17 These included measles, scarlet fever, rubella, and a once-distinct disease called Dukes disease. In 1974, it was discovered to be caused by the virus parvovirus B19 and subsequently renamed erythema infectiosum for the corresponding blood bank code label.18 The virus belongs to the large Parvoviridae family and is a commonly transmitted human pathogen. Erythema infectiosum occurs worldwide, but seems to be more prevalent in temperate, tropical climates. It is common in childhood, with peak rates between the ages of 5 and 14 years. Transmission of the virus occurs via respiratory droplets, making household transmission likely.
More than half of the cases are asymptomatic, with a rare prodromal phase that includes low-grade fever, headache, pharyngitis, myalgias, nausea, diarrhea, and joint pain.19,20 The distinct facial erythema provides the basis for its other common name— "slapped cheek" disease, a result of red papules coalescing to form the red, edematous symmetrical cheek rash, which spares the periorbital areas and nasal bridge. The patient is no longer infectious when the cheek rash appears. Approximately two days after the onset of the facial erythema, the rash extends to the extremities in a unique, fishnet-like pattern that usually fades in 6-14 days. (See Figure 3a and 3b.) The slapped cheek appearance typically fades in four days. Certain triggers such as sunlight, stress, and temperature change may stimulate a recurrence during the two to three weeks following the extremity eruption.
Figure 3. Fifth Disease
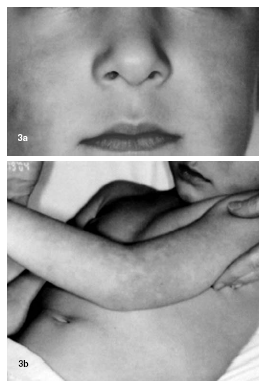
Figure 3a: Note erythematous cheeks.
Figure 3b: Reticular rash on limbs.
Fifth disease is only one of several dermatologic, hematologic, and rheumatologic conditions caused by Parvovirus B19; other disease states include transient aplastic crisis, arthropathy, persistent anemia, and hydrops fetalis. It also has been suggested that Parvovirus B19 may play a role in Kawasaki’s disease, Henoch-Schonlein purpura, juvenile rheumatoid arthritis (Still’s disease), and giant cell arteritis.21 Severe outcomes may occur in immunocompromised patients, the fetus, and patients with hemoglobinopathies. Patients with immunocompromised conditions, such as sickle cell, also may develop a serious, prolonged chronic anemia caused by an abrupt cessation of red-cell production resulting from a B19 infection. Other severe sequelae include the transplacental transmission to the fetus, which may lead to miscarriage or hydrops fetalis.
Management of patients with erythema infectiosum is symptomatic. Most patients do well with simple treatment measures including antipyretics, oral hydration, and antiprurituc agents, if needed. Other treatments may be indicated for significant complications, including blood transfusions for patients with chronic hemolytic anemias who develop transient aplastic crisis and serial fetal ultrasounds for pregnant patients with positive IgM and IgG levels supporting parvovirus infection. There are no published recommendations for children returning to school once the diagnosis is made; however, children may return to school with the appearance of the facial rash. However, patients with aplastic crisis may be contagious for extended periods.
Pityriasis Rosea
Pityriasis rosea is a self-limited papulosquamous eruption frequently seen among adolescents and young adults during the fall and winter months. It is characterized by a classic rash that assumes a storiform or Christmas tree pattern and often is preceded by a small, circular lesion termed a "herald patch."
The exact etiology is still unclear, despite clinical and epidemiological evidence that suggest an infectious cause. Clinical support for a viral origin includes the prodromal upper respiratory symptoms, the characteristic rash pattern, and the spontaneous resolution of the lesions.22 Epidemiological features that support a viral cause include community outbreaks and the peak incidence during the cold months.23 Results from one study provided evidence of a viral etiology for pityriasis rosea by detecting human herpes virus-7 DNA in peripheral blood mononuclear cells from patients with acute pityriasis rosea.24 However, these results have not been confirmed with subsequent studies.25-27 The incidence of pityriasis rosea peaks at the ages of 20 to 29 years without a significant gender bias.28-30
The diagnosis of pityriasis rosea is made clinically, based upon history, physical examination, and the unique timing and appearance of the rash. Prior to the appearance of the generalized rash, a herald patch may be seen in 50 to 90% of cases. (See Figure 4a.) This single, circular lesion with defined borders precedes the rash by several days to a couple of weeks and often is confused with tinea corporis or nummular eczema. The lesion can be found most commonly on the neck or lower trunk area, but does occasionally appear on the extremities. The pathognomonic finding of the disease is the subsequent eruption of multiple, round to oval-shaped, salmon-colored plaques that follow the lines of skin cleavage creating a Christmas tree pattern. (See Figure 4b). These plaques are covered with a fine, white scale, which may be described as a "cigarette-paper" scale forming a collarette. The individual lesions may appear hyperpigmented in African-American patients, but are generally pink in nature. The rash appears rapidly over the trunk, neck, and proximal extremities. Unlike secondary syphilis, the palms and soles generally are spared. Pruritis is a common complaint and often persists 1 to 2 months before resolving spontaneously. A significant number of cases are preceded by symptoms of a mild, upper respiratory infection, but pityriasis itself is not considered contagious. Pityriasis rosea may have an atypical presentation including face and oral mucosal lesions in addition to involving the palms and soles.
Figure 4. Pityriasis Rosea in a 13-year-old Male
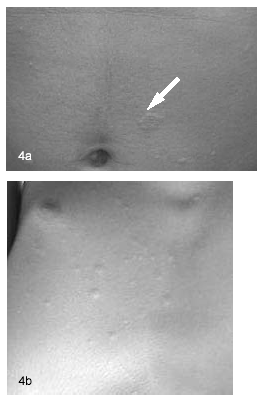
Figure 4a. Note herald patch on abdomen.
Figure 4b. Rash of pityriasis rosea.
Other diseases to be considered in the differential diagnosis include tinea corporis, tinea versicolor, secondary syphilis, guttate psoriasis, nummular eczema, lichen planus, and drug eruptions.
Educating patients about the extended course of the illness is essential. Unlike other viral exanthems, pityriasis may last up to three months. While no workup for pityriasis is necessary, atypical presentations or symptoms that persist beyond the typical time course may indicate that further diagnostic tests are indicated.
Treatment is supportive and may include topical corticosteroids and antihistamines to provide relief of the prominent itching. Ultraviolet B light (UVB) may decrease the severity of the disease if used within the first week of eruption but should be done in conjunction with a dermatologist. Recurrence is rarely seen, and occurs twice as commonly in blacks (6%) as compared with whites (2% to 3%).31
Pityriasis is a self-limited disease frequently seen in the ED due to the prominent pruritis. For the emergency clinician, classic presentations require supportive treatment. For atypical or prolonged presentations, other more serious differential diagnosis, such as syphilis, should be ruled out.
Rubella
The rubella virus is a single-stranded RNA togavirus. It is spread by the respiratory route via nasopharyngeal droplets. The incubation period of rubella is estimated to be 2 to 3 weeks. Exposed persons are infectious prior to the outbreak of the exanthem; the infectious period continues for about five days after rash develops.32,33
Clinically, rubella typically begins as a mild respiratory illness.34 Fever, headache, malaise, and anorexia are common symptoms. However, young children often have no prodromal symptoms prior to the appearance of the rash. Tender suboccipital, posterior cervical, and postauricular adenopathy are common. Typically, rubella is a mild, viral illness and results in minimal morbidity and mortality. Thrombocytopenia and encephalitis occur infrequently in children. Complications are more prevalent in adolescents and adults.
The rubella exanthem begins as macules and papules on the face, scalp, and neck.35 The rosy pink lesions quickly spread to the rest of the body, including the hands and feet. Forchheimer’s sign, seen in 20% of rubella cases, is a soft palate eruption of petechial spots. The exanthem appears and resolves over a three-day course, thus the term "three-day measles." Pruritus is common with the rash. Mild desquamation often occurs.
Rubella has become rare since the advent of the rubella vaccine in 1969. Incidence of rubella is less than one in 100,000. Although rare, congenital rubella remains a devastating condition. A fetus has the greatest chance of contracting rubella in the first 12 weeks of gestation. Risk declines as gestation progresses. Deafness, heart disease, and cataracts compose the classic triad of congenital rubella syndrome. Hearing loss results from sensorineural damage. Patent ductus arteriosus is the most common cardiac manifestation. Cataracts are usually bilateral, and retinopathy also may be present. Neurological conditions, such as encephalitis and mental retardation, are also common. Chronic rubella infection results in growth retardation, hepatospleno-megaly, thrombocytopenia, jaundice, and purple skin lesions. An infant with congenital rubella is infectious for the first year of life.
The diagnosis of rubella can be made clinically. Suboccipital lymphadenopathy, rosy pink lesions, and an exanthem lasting three days are the clinical hallmarks of rubella. Serology may be performed for definitive diagnosis. Therapy is supportive only.
Rubeola
The measles virus is a single-stranded RNA paramyxovirus. Rubeola is endemic, highly contagious, and is a cause of significant morbidity and mortality. Prior to the advent of the measles vaccination, 95% of the population contracted measles by age 15 years. The virus is spread by both aerosolized respiratory droplets and direct contact. The virus remains virulent for at least two hours as droplets; the incubation period is typically 10-12 days. The carrier is infectious from the prodromal period until four days after the exanthem outbreak.36
The number of reported cases of measles has dropped dramatically in the United States since the advent of the measles vaccine. In 1998 and 1999, fewer than 70 cases were reported in the United States compared with the 4-5 million cases per year prior to immunization. Measles is most likely to infect pre-schoolers in low-income homes or in heavily populated areas. Most cases result from either noncompliance with recommended immunizations or primary vaccination failure. The endemic and virulent nature of measles obviates the need for its eradication. Worldwide, it is the No. 1 cause of vaccine-preventable death. The United States has decreased cases by 99% through routine series of two measles-mumps-rubella (MMR) vaccinations. The World Health Organization still is trying to decrease measles incidence in other parts of the world where it remains prevalent.37 Rubeola is seen most often in the spring and winter.
Clinically, rubeola first appears as an influenza-like illness. The prodrome includes a high fever of 104-105ºF, malaise, and the classic C’s of measles—cough, coryza, and conjunctivitis. The symptoms are present for 2-4 days prior to exanthem outbreak. The measles exanthem begins as erythematous macules and papules along the hairline and behind the ears. It spreads from the head in a downward and outward pattern. As time passes the initial lesions coalesce. The rash changes from a blanching, bright red rash to a brownish, non-blanching hue within 2-4 days of initial onset. Mild desquamation often occurs.
Koplik spots are pathognomonic for measles. The enanthem is on the buccal mucosa at the second molar. Lesions are bluish-white on an erythematous base. The Koplik spots are present during the prodromal phase and usually nonexistent at rash onset.
Laboratory results often show leukopenia with lymphocytopenia. Generalized lymphadenopathy and splenomegaly may be seen as extracutaneous manifestations of measles.38 Complications may include pneumonia, otitis media, and diarrhea. Children younger than 5 years most often incur complications. Pneumonia is the most common cause of death in children with measles. Hepatitis, thrombocytopenia, and encephalitis also may occur.
Three types of encephalitis may occur as complications from rubeola. Acute post-infectious measles encephalitis (APME), occurs about two weeks after rash onset. Clinical features include fever, altered mental status, seizures, and headache. Periventricular demyelination has been found in these patients. APME mortality is 10-20%. Measles inclusion body encephalitis (MIBE), manifests 1-6 months after primary measles infection. It affects immunocompromised individuals as a result of continued viral replications in neurons. Typically, no fever occurs, but altered mental status and seizures are present. One in 1 million cases of measles in the United States leads to the development of subacute sclerosing panencephalitis (SSPE), which lies dormant for 7-10 years after the initial measles infection. Inflammation and demyelination of cortical and subcortical gray and white matter occurs, and clinical symptoms arise, including slowly progressive mental retardation, motor dysfunction, seizures, coma, and death.39
Atypical measles has declined since the advent of the live attenuated vaccine. Exposure to wild type measles following killed measles vaccination may result in atypical measles. Although rare, this also has occurred following live attenuated vaccination. Koplik spots, conjunctivitis, and runny nose are absent. The illness comprises fever, myalgias, headache, cough, and rash. However, symptoms are more severe, and the exanthem differs in location and appearance. Rash begins at the wrists and ankles as well as the palms and soles. The rash spreads inward and upward. This presentation may be confused with Rocky Mountain spotted fever. The exanthem varies from maculopapular to vesicular and may be erythematous to purpuric. Pneumonia almost always is found in these patients, and pleural effusions are common.
Vaccination is available in a combination with mumps and rubella vaccine—MMR—or as a single agent vaccine. Both are live attenuated vaccines. Any patient with anaphylaxis to eggs or neomycin may have a severe, allergic reaction to the measles vaccine. Vaccination is contraindicated in pregnancy, immunocompromised patients, and cancer patients. Previously raised questions of the measles vaccination as the cause of inflammatory bowel disease and autism have been studied. No evidence has been found to support such claims. In approximately 10% of patients, mild fever and rash may develop after immunization. Transient thrombocytopenia is a recognized side effect of vaccination.
If a measles case is suspected, the individual should be isolated. Anyone exposed to measles who has not been vaccinated, who is pregnant, or younger than 1 year should receive prophylaxis with immunoglobulin within six days of contact. The measles vaccine should be given five months later.
Diagnosis of measles can be made clinically, but it is difficult prior to onset of the exanthem. For definitive diagnosis, acute and convalescent titers can be measured or enzyme-linked immunosorbent assay (ELISA) can be performed. During epidemics, collection of specimens should be done for genotyping.
Treatment of measles is primarily supportive, and should include oral hydration, antipyretics, and analgesics. Pneumonia, otitis media, persistent fever, or other significant illness may necessitate hospital admission and/or antibiotic treatment. Ribavirin may be helpful in complicated measles complications. If the patient is in an area deficient in vitamin A, 400,000 units should be given as it has been shown to decrease morbidity and mortality.
Roseola Infantum
Roseola is thought to be of viral origin, yet no specific entity has been isolated. The rotavirus and human herpes virus-6 have been reported as sources of roseola. Several other viruses also have been reported to be associated with roseola. The incubation period is 1-2 weeks. Roseola infects males and females equally. Most cases occur in spring and early fall. Children between 6 months and 3 years of age account for nearly all cases of roseola.40
Clinically, fever is the first symptom, and may be 104-105ºF. Fever may be persistent or intermittent. The febrile child is playful and nontoxic in appearance. Occasionally, febrile seizures occur. On physical exam, cervical lymphadenopathy, pharyngeal erythema with or without exudates, and otitis media may be found.
The exanthem appears suddenly after fever resolution, which is where the name "roseola subitum" originated. The rash begins on the trunk and spreads upward to the neck and proximal extremities. It is pink in color and can be macular, papular, or maculopapular. The exanthem may be as short in duration as a few hours, or it may last up to 2 days. No pruritus or flaking occurs.41
Diagnosis is made clinically. High fever in a well-appearing child, rapid decline of fever, and sudden onset of rash are the keys to clinical diagnosis. Leukopenia with lymphocytosis may be seen on laboratory examination. Therapy is supportive and may include antipyretics and oral hydration.
Infectious Mononucleosis
Infectious mononucleosis may affect any pediatric age group and is caused by the Epstein-Barr virus (EBV), a DNA herpes virus.42 Because infection is often mild and undiagnosed in early childhood, most clinically recognized cases occur in adolescents and young adults.43 Most adults have been exposed and have titers against the Epstein-Barr virus. There is no typical seasonal variation. The virus is transmitted via salivary secretions. The incubation period is typically 30-50 days. Clinical symptoms typically include fever, headache, malaise, fever, pharyngitis, and lymphadenopathy. Splenomegaly and hepatomegaly also may occur. Symptoms may last from a few days to several months. Significant complications of hemolytic anemia, aplastic anemia, pneumonitis, and neurologic problems occur rarely.
The exanthem of infectious mononucleosis has been estimated to occur in approximately 5-50% of cases. The typical appearance is a nonspecific maculopapular eruption, although petechial, scarletiniform, erythema multiforme, and urticarial lesions also may be seen.44 The lesions typically appear about day four of the illness. The trunk and arms often are involved early, and lesions may spread to the face and remainder of the body. Patients who receive ampicillin or amoxicillin are at increased risk of developing the rash, and up to 80% of patients with mononucleosis who are treated with ampicillin may develop the rash. The drug-related rash is thought to be due to production of drug-specific antibodies with immune complex deposition in the skin. The drug-related eruption typically occurs 7-10 days after initiation of antibiotic therapy. The rash is not associated with subsequent penicillin allergies.
Infectious mononucleosis also may be associated with other dermatologic findings. Erythema nodosum, exacerbation of pre-existing acne vulgaris, urticaria, and anaphylaxis have been reported.
The potential for splenic rupture should be considered in cases of infectious mononucleosis. Splenomegaly occurs in approximately 8-50% of patients45-47 and often is undetected clinically. Splenic rupture occurs in 0.1-0.5% of cases,48 and usually occurs in the first three weeks of illness, with most (80%) cases occurring in patients age 25 years and younger.49
Laboratory studies are considered optional, as the diagnosis is primarily clinical. The heterophile antibody test is available, and the sensitivity varies from 63-84%; specificity is between 84-100%. The Monospot heterophile antibody test commonly is used, and becomes positive 5-7 days after onset of symptoms, and may be falsely negative if performed early in the disease course. EBV serology may be performed to confirm acute infection. Liver transaminases may be elevated modestly, and peak at 2-3 weeks.
Treatment consists of supportive care, including adequate rest, hydration, nutrition, antipyretics, and analgesics. Steroids may be prescribed for patients with significant pharyngitis and dysphagia. Patient education is essential, particularly regarding prognosis and supportive care. Contact sports should be avoided, although the precise time until return to full activity is debatable. Published recommendations include abstinence from contact sports for 3 weeks to 6 months.
Hand, Foot, and Mouth Disease
Hand, Foot and Mouth Disease (HFMD) refers to a viral exanthem first described in 1958.20 It usually is caused by enteroviruses, including coxsackievirus A16 and enterovirus 71. Cases have occurred less commonly from infection with coxsackie A4-7, A9, A10, B1-3, and B5. HFMD is highly contagious and is spread by fecal-oral contamination. Pediatric patients younger than 10 years—typically younger than 4 years—are affected most commonly. Epidemics often occur in the late summer and early fall, although sporadic cases often are seen. In the United States, epidemics may be seen approximately every three years.
Clinical manifestations are quite predictable. Following the incubation period of 3-6 days, a prodrome may occur with fever, malaise, cough, anorexia, and abdominal pain. Within the next few days, clinical manifestations typically include oral lesions, followed by vesicular lesions on the hands and/or feet. Lesions may begin as erythematous papules and macules, and/or vesicles, which typically progress to erosions or ulcers, and disappear during the ensuing 5-10 days. The lateral and dorsal aspects of fingers commonly are involved. Lesions often occur in a pattern along skin lines. Oral lesions occur most commonly on the hard palate, tongue, and buccal mucosa. Oral intake may be compromised by pain.
Complications are rare.50 Scarring is rare. Reported complications include myocarditis, meningoencephalitis, pulmonary edema, and death. Neurologic complications including myoclonus, paralysis, meningitis, and encephalitis have been reported.51-53
Treatment consists of supportive measures to decrease discomfort and improve oral intake. Malaise and pain may be treated with antipyretic agents, such as acetaminophen or ibuprofen. Oral lesions may be treated symptomatically with diphenhydramine elixir, or magnesium hydroxide. Although acyclovir has shown improvement of symptoms in one small study,54 the lack of large-scale studies demonstrating efficacy currently precludes routine use.
Other Enteroviral Exanthems
A variety of enteroviruses may cause symptoms including fever, malaise, gastrointestinal complaints, meningitis, and rash. Infections tend to peak in summer and autumn months. The enterovirus exanthem typically exhibits a nonspecific maculopapular eruption; although petechiae, mimicking a meningococcal infection, may be seen.55 Petechiae have been reported with coxsackievirusA9, A4, B2-5, echovirus 9, and echovirus 3,4, and 7 infections.56
Conclusions
Pediatric viral exanthems occur commonly among the emergency pediatric population. Accurate historical information and physical examination can be crucial in establishing the correct diagnosis. Many viral exanthems are benign and resolve spontaneously with supportive care. Certain exanthems require specific interventions and have been addressed in this manuscript.
References
1. Cherry JD. Viral exanthems. Curr Probl Pediatrics 1983;13:1.
2. Goodyear HM, Laidler PW, Price EH et al. Acute infectious erythemas in children: A clinico-microbiological study. Br J Dermatol 1991;124:433-438.
3. Chen, Minsue T. Clinical manifestations of varicella-zoster virus infection. Dermat Clin 2002;20:267-282.
4. Starr C, Pavan-Langston D. Varicella-zoster virus: Mechanisms of pathogenicity and corneal disease. Ophthalmol Clin North Am 2002;7-15.
5. Wallington T, Weir E. Varicella control and vaccine coverage: Issues and challenges. CMAJ 2002;166:631-632.
6. Thomas SL, Hall AJ. What does epidemiology tell us about risk factors for herpes zoster? Lancet Infect Dis 2004;4:26-33.
7. Brentjens M, Yeung-Yue KA, Lee PC, et al. Vaccines for viral diseases with dermatologic manifestations. Dermatol Clin 2003;349-369.
8. Roque MR, Foster, CS. Herpes Zoster. Emedicine. Available at: www.emedicine.com/oph/topic257.htm.
9. Hoey J. Varicella vaccine update: Need for a booster? CMAJ 2003; 168:589.
10. Yeung-Yue KA, Brentjens MH, Lee PC, et al. Herpes simplex viruses 1 and 2. Dermatol Clin 2002; 20:315-331.
11. Torres G. Herpes Simplex. Emedicine. Available at: www.emedicine.com/derm/topic179.htm.
12. Rudnick CM, Hoekzema, GS. Neonatal herpes simplex virus infections. Am Fam Physician 2002;65:1138-1142.
13. Braverman PK. Sexually transmitted diseases in adolescents. Clin Ped Em Med 2003;869-889.
14. Hairston BR, Bruce AJ, Rogers RS 3rd. Viral diseases of the oral mucosa. Dermatol Clin 2003;21:17-32.
15. Colgan R, Michocki R, Greisman L, et al. Antiviral drugs in the immunocompetent host: Part I. treatment of hepatitis, cytomegalo-virus, and herpes infections. Am Fam Physician 2003; 67: 757-762, 675.
16. Bale JF Jr. Congenital infections. Neurol Clin 2002;20:1039-1060.
17. Mancinci A. Exanthems in childhood: An update. Pediatr Ann 1998; 27:163-170.
18. Cherry JD. Parvovirus infections in children and adults. Adv Pediatr 1999;46:245-269.
19. Metry D, Katta R. New and emerging pediatric infections. Dermatol Clin 2003;21:269-276
20. Scott L, Stone, MS. Viral Exanthems. Dermatology Online Journal 1993; 9:4. Available at: http://dermatology.cdlib.org/93/reviews/viral/scott.html. Accessed Feb. 11, 2004.
21. Young N, Brown K. Mechanisms of disease: Parvovirus B19. N Engl J Med 2004;350:586-597.
22. Sharma PK, Yadav TP, Gautam RK, et al. Erythromycin in pityriasis rosea: A double-blinded, placebo-controlled clinical trial. J Am Acad Dermatol 2000:42:241-244.
23. Messenger AG, Knox EG, Summerly R, et al. Case clustering in pityriasis rosea: Support for role of an infective agent. Br Med J 1982;284:371-373.
24. Drago F, Ranieri E, Malaguti F, et al. Human herpes virus 7 in patients with electron microscopy investigations and polymerase chain reaction in mononuclear cells, plasma and skin. Dermatology 1997:195:374-378.
25. Kempf W, Adams V, Kleinhans M, et al. Pityriasis rosea is not associated with human herpesvirus 7. Arch Dermatol 1999;135:1070-1072.
26. Chuh AA, Chiu SS, Peiris JS. Human herpesvirus 6 and 7 DNA in peripheral blood leukocytes and plasma in patients with pityriasis rosea by polymerase chain reaction: A prospective case control study. Acta Derm Venereol 2001;81:289-290.
27. Wong WR, Tsai CY, Shih SR, et al. Association of pityrisasis rosea with human herpesvirus-6 and human herpesvirus-7 in Taipei. J Formos Med Assoc 2001;100:478-483.
28. Cheong WK, Wong KS. An epidemiological study of pityriasis rosea in Middle Road Hospital. Singapore Med J 1989;30:60-62.
29. Harman M, Aytekin S, Akdentiz S, et al. An epidemiological study of Pityriasis Rosea in the Eastern Anatolia. Eur J Epidemiol 1998;14: 495-497.
30. Tay YK, Goh CL. One-year review of pityriasis rosea at the National Skin Centre, Singapore. Ann Acad Med Singapore 1999;28:829-831.
31. McLaurin C. Unusual patterns of common dermatoses in blacks. Cutis 1983; 32:352-360.
32. Bialecki C, Feder HM Jr, Grant-Kels JM. The six classic childhood exanthems: A review and update. J Am Acad Dermatol 1989; 891-903.
33. Cherry JD. Viral exanthems. Dis Mon 1982;28:1-56.
34. Gable EK, Liu, G, Morrell DS. Pediatric Exanthems. Prim Care 2000;27:353-369.
35. Weintraub R. Viral infections. Maj Probl Clin Pediatr 1978;19: 184-208.
36. Stalkup JR. A review of measles virus. Dermatol Clin 2002;20: 209-215.
37. Muscat M, Glismann S, Bang H. Vaccine preventable diseases. Euro Surveill 2003;6:123-129.
38. Zenel JA. Visual Diagnosis: An infant who has fever and rash. Diagnosis: Measles. Pediatr Rev 2000;3:105-107.
39. Duke T, Mgone CS. Measles: Not just another viral exanthem. Lancet 2003;36:763-773.
40. Bligard CA, Millikan LE. Acute exanthems in children. Clues to differential diagnosis of viral disease. Postgrad Med 1986;5:150-154, 159-167.
41. Behbehani AM. A concise review: Common viral exanthems and enanthems. J Kans Med Soc 1981;10:442-446.
42. Papesch M, Watkins R. Epstein-Barr virus infectious mononucleosis. Clin Otolaryngol 2001;26:3-8.
43. Morris MC, Edmunds WJ. The changing epidemiology of infectious mononucleosis. J Infection 2002;45:107-132.
44. Ikediobi NI, Tyring SK. Cutaneous manifestations of Epstein-Barr virus infection. Dermatol Clin 2002;20:284-289.
45. Rea TD, Russo JE, Kafon W, et al. Prospective study of the natural history of infectious mononucleosis caused by Epstein-Barr virus. J Am Board Fam Pract 2001;14:234-242.
46. Crawford DH. Epstein-Barr virus. In: Principles and Practises of Clinical Virology. 3rd ed. Chichester: John Wiley and Sons Ltd;1994:109-124.
47. Halstead ME, Bernhardt DT. Common infections in the young athlete. Pediatr Annals 2002;31:42-48.
48. Maki DG, Reich RM. Infectious mononucleosis in the athlete: Diagnosis, complications, and management. Am J Sports Med 1982; 10:162-173.
49. Kinderknecht JJ. Infectious mononucleosis and the spleen. Curr Sports Med Rep 2002;1:116-120.
50. Ho ML. Enterovirus 71: The virus, ints infections and outbreaks. J Microbiol Immunol Infect 2000;33:205-216.
51. Huang C, Liu C, Chang Y et al. Neurologic complications in children with enterovirus 71 infection. N Engl J Med 1999; 341:936-942.
52. Liu C, Tseng HW, Wang SM et al. An outbreak of enterovirus 71 infection in Taiwan, 1998. Epidemiologic and clinical manifestations. J Clin Virol 2000;17:23-30.
53. McMinn PC. An overview of the evolution of enterovirus 71 and its clinical and public health significance. FEMS Microbiol Rev 2002; 26:91-107.
54. Shelley W, Hasim M, Shelley E. Acyclovir in the treatment of hand-foot-and-mouth disease. Cutis 1996; 57:232-234.
55. Sukhai RN, Munneke R. Enteroviral meningitis with a petechial rash in three children. Eur J Pediatr 2002;161:226-227.
56. Feigin RD, Cherry JD. Textbook of Pediatric Infectious Diseases. 4th ed. Philadelphia: WB Saunders;1998:731.
The authors review the classic course of common pediatric diseases associated with rashes, including varicella-zoster virus, herpes simplex virus (HSV), roseola, and rubella. Understanding the classic patterns, disease progression, high-risk populations, and potential complications allow the ED physician to avoid unnecessary testing in low-risk patients with a classic presentation, and aggressively approach potentially significant rashes in high-risk populations (e.g., neonatal HSV). This article also is designed to increase ED physicians awareness of treatment strategies associated with common viral exanthems.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
