New research confirms role of heart disease as a treatment by-product
Studies shed light on this and other med problems
While the exact cause remains to be found, it is no longer a mystery whether certain antiretroviral drugs can lead to metabolic changes associated with heart disease. Recent studies have made it clear that there is a direct connection between some protease inhibitors (PIs) and other anti-HIV medications and health problems that could affect HIV patients over the long term.
"In the past couple of years, there’s been increasing concern about atherosclerosis and coronary heart disease in patients with antiretroviral therapy," says Marshall Glesby, MD, PhD, assistant professor of medicine at Weill Medical College at Cornell University in New York City.
"The most carefully studied patients have a number of abnormalities associated with heart disease, including abnormal lipids, changes in body shape, diabetes, increased truncal fat, and others." More-over, there’s good evidence of a direct cause and effect with regard to certain PIs, Glesby says. "From data on certain PIs given for short periods of time to people without HIV infection, you can see increased elevation in cholesterol, triglycerides, and insulin resistance," he explains.
For example, one recent study found that PI use is associated with coronary artery calcification, atherogenic lipid changes, and increased erythrocyte volume.1
Some additional metabolic complications of antiretroviral therapy include hyperlipidemia, elevated lactate and lactic acidosis, and osteopenia/osteoporosis.
Research presented at the 4th International Workshop on Adverse Drug Reactions and Lipo-dystrophy in HIV, held in September in San Diego add to the body of evidence that antiretroviral therapy can lead to hypertriglyceridemia. Research also has found that patients receiving antiretroviral regimens containing the nucleoside reverse transcriptase inhibitors (NRTIs) stavudine were at an increased risk of hypertriglyceridemia.2,3
Other long-term problems associated with HIV and antiretrovirals are morphologic changes, including the loss of subcutaneous fat and fat accumulation, such as gynecomastia and a dorsocervical fat pad, says Paul Sax, MD, clinical director of the HIV Program at Brigham & Women’s Hospital and a medical professor at Harvard Medical School, both in Boston.
NRTIs are associated with fat wasting or fat atrophy, and some of the NRTIs are associated with peripheral neuropathy, while PIs are closely associated with increased insulin resistance, he says.
Recent research has concluded that both stavudine- and zidovudine-based antiretroviral therapy are associated with significant gluteal subcutaneous adipocyte depletion, and that lamivudine is associated with more adipocyte depletion than the drugs didanosine or zalcitabine.4
In analyzing data from fat redistribution and metabolic changes (FRAM) research, Sax says, investigators have drawn these conclusions about antiretroviral treatment and metabolic changes:
- Some protease inhibitors cause fat accumulation and insulin resistance.
- Mitochondrial toxicity associated with NRTIs leads to fat wasting.
- When PIs and NRTIs are combined in therapy, there is an acceleration of fat wasting due to fat cell apoptosis.
Other antiretroviral complications resulting in long-term side effects include the association between some NRTIs, including DDI and D4T, and peripheral neuropathy, Sax says.
"Non-nucleoside reverse transcriptase inhibitors (NNRTIs) have a different side-effects profile that doesn’t appear relevant to morphologic changes," Sax notes. "Although they do sometimes cause alterations in lipids, it’s been observed that these alterations are favorable."
Current research has examined the incidence of accelerated atherosclerosis and PI medications, and there seems to be solid evidence that PIs contribute to symptoms associated with heart disease. However, the jury still is out on whether this means there is an increased risk of death from heart disease among HIV patients treated with PI therapy.
"The bigger issue and question is to determine whether people with HIV have an increased risk of cardiovascular disease and to what extent that is due to metabolic and lipid changes with therapy," says Judith Currier, MD, an associate professor of medicine at the David Geffen School of Medicine at the University of California, Los Angeles. Currier also is director of clinical trials unit of the UCLA Care Center.
She spoke about coronary heart disease and PIs at the 40th Annual Meeting of the Infectious Diseases Society of America (IDSA), held in October in Chicago.
"Several studies say there’s an increased risk with PI use, and others don’t," Currier says. "What we need to focus on now is trying to determine to which extent it’s due to HIV in itself, the metabolic changes associated with treatment or the prevalence of other risk factors for cardiovascular disease that exist in this population."
There is no question that some HIV drugs cause patients to experience lipid and metabolic changes, but the contribution of these changes to heart disease risk in this population is not known, she adds. "We’re still a ways off deciphering these factors."
A study presented at the IDSA conference concluded that the highest mortality rate when HIV-infected and noninfected individuals with and without coronary heart disease (CHD) were compared was among the HIV-infected people with CHD.5
Investigators found a significant increase in the risk of CHD among HIV patients in the 18 to 34 age group, but not among older HIV patients, says Currier, who was a co-author of the IDSA study about CHD and HIV.
The study found that the youngest group (ages 18-24) of HIV patients who also had CHD diagnoses were at an 8.67 times greater risk of death from all causes than HIV-infected individuals of that age range who did not have a CHD diagnosis, says Beth Burtcel, PharmD, manager of Virology and Scientific Operations for Bristol-Myers Squibb Co. in Plainsboro, NJ. Burtcel also co-authored the IDSA study on CHD and HIV.
"These patients could have died of something besides a CHD event," Burtcel notes. "But the take-home message is that for younger individuals with CHD and HIV, the risk of death is significantly higher."
There was not such a large difference in the rate of death among the 18-24 age group of people who had CHD but did not have HIV infection, Burtcel adds.
The study used California’s MediCal data in the retrospective study, examining claims from more than 28,000 HIV-infected men and women and more than three million non-HIV infected people.
Both the HIV-positive and negative cohorts had to be free of all CHD diagnoses and claims for at least one year to be included in the study, Burtcel says.
Antiretroviral medication and its possible role in CHD were not investigated as part of the IDSA study, Burtcel adds.
"This study just gives us a general idea of what role coronary heart disease is playing in HIV-infected people," Burtcel says. "These data suggest there is an association between CHD and all-cause mortality rates in HIV-infected individuals."
Clinicians should put these findings in perspective and not rush to switch antiretroviral therapy that otherwise is saving a patient’s life, Sax advises.
Recent data on AIDS deaths from the Centers for Disease Control and Prevention (CDC) in Atlanta may offer some broader perspective on the issue about antiretroviral medications and increased risk of coronary heart disease. CDC data offer continued testimony to the increased longevity of HIV patients since the advent of PIs and other potent antiretroviral drugs. (See charts below)
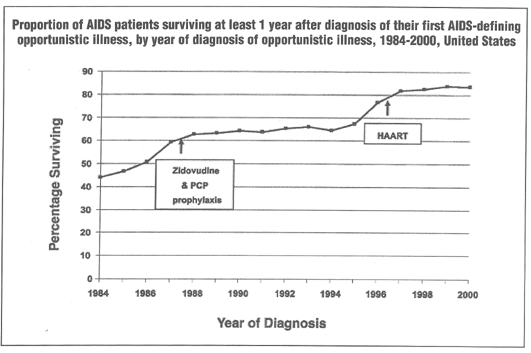 |
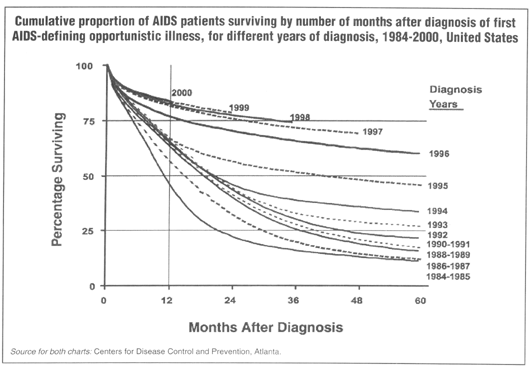 |
"We HIV specialists are unanimous in agreeing that people with AIDS need therapy," Sax says. "But the important thing is to be aware of the side effects so that you can treat them and manage them if necessary."
Also, clinicians should factor these long-term side effects into their decisions about when to start a healthy, HIV-positive patient on antiretroviral treatment, Sax adds. "We may be delaying treatment a little bit longer than before for patients who are asymptomatic."
Some guidelines have been introduced in the past year to assist HIV clinicians in making decisions about managing lipid disorders and metabolic complications. For instance, the Inter-national AIDS Society USA recently published its guidelines on managing metabolic complications, and in August, the Adult AIDS Clinical Trials Group (AACTG) issued its metabolic complications guide.
Meantime, HIV clinical practice already has changed because of the metabolic complications, says David Haas, MD, director of Vanderbilt AIDS Clinical Trials Center and associate professor of medicine at the Vanderbilt Medical School in Nashville, TN.
"Right now, management of HIV has become as much about management of medication side effects such as lipid abnormalities as it is about management of the virus itself," Haas says.
"I think treating HIV infection has been like being on a roller coaster where enthusiasm for treating sometimes has been offset by concerns of drug toxicity," he adds. "For some patients, their cardiovascular disease poses greater risk in the immediate future than their HIV disease."
One new PI, called atazanavir, that has been available in expanded access programs but still awaits approval by the Food and Drug Admini-stration, is a possible alternative to antiretrovirals that cause metabolic problems, Haas says.
The once-a-day drug has been shown to have favorable lipid profiles in HIV patients, according to new research.
References
1. Meng Q, Lima JAC, Lai H, et al. Coronary artery calcification, atherogenic lipid changes, and increased erythrocyte volume in black injection drug users infected with human immunodeficiency virus-1 treated with protease inhibitors. Am Heart J 2002; 144:642-648.
2. Galli M, Gervasoni C, Adorni F, et al. Factors associated with the development of high serum triglyceride levels in patients receiving two NRTI combinations. Presented at the 4th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV. San Diego; September 2002. Abstract 40.
3. Morlese J, Qazi N, Orkin C, et al. Both stavudine and zidovudine-based therapy is associated with gluteal subcutaneous adipocyte depletion. Presented at the 4th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV. San Diego; September 2002. Abstract 30.
4. Walmsley S, Montaner J, Saag M, et al. Nucleoside reverse transcriptase inhibitor treatment as a risk factor for hyperlipidemia: Results from the FOCUS trial. Presented at the 4th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV. San Diego; September 2002. Abstract 52.
5. Burtcel B, Kawabata H, Dezil C, et al. Increased mortality in HIV infected Individuals with Coronary Heart Disease. Presented at the 40th Annual Meeting of the Infectious Diseases Society of America. Chicago; October 2002. Poster 466.
While the exact cause remains to be found, it is no longer a mystery whether certain antiretroviral drugs can lead to metabolic changes associated with heart disease.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
