Influenza
January 12, 2014
Reprints
Influenza
Authors:
Kurt Weber, MD, Attending Physician/Faculty, Orlando Health EM Residency, Director, Corporate Office of Research Operations, Orlando, FL; Clinical Assistant Professor of Emergency Medicine, Florida State University College of Medicine, Tallahassee.
Kathryn Bondani, MD, Resident Physician, Orlando Health EM Residency, Orlando, FL.
Philip Giordano, MD, Attending Physician/Faculty, Orlando Health EM Residency, Chief of Research Operations, Orlando Health, Orlando, FL; Clinical Assistant Professor of Emergency Medicine, Florida State University College of Medicine, Tallahassee.
Peer Reviewer:
Charles L. Emerman, MD, Chairman, Department of Emergency Medicine, MetroHealth Medical Center, Case Western Reserve University, Cleveland, OH.
Influenza is possibly the most widespread and well known of infectious diseases. Seasonal epidemics can be found on all continents, and no age segment of the population is untouched. Among these groups, both symptoms and severity will vary. Definitive diagnosis in the emergency department (ED) is problematic, as rapid bedside tests vary in sensitivity and specificity and must be interpreted in the context of local disease prevalence. Initiation of antiviral treatment is an issue an emergency physician (EP) should consider. The following review will give the emergency physician information to manage these patients.
Executive Summary
- Patients at high risk for influenza complications include the very young (younger than 2 years), pregnant women, the elderly, and those with significant co-morbid conditions.
- Oseltamivir should be started as soon as possible for any hospitalized patient with suspected influenza.
- Antiviral therapy should be initiated for high-risk outpatients and those with progressive disease who are not being admitted.
- Do not expect quick clinical improvement with antiviral therapy.
- For community-acquired pneumonia during influenza season, consider Staphylococcus aureus as a potential pathogen.
Historical Perspectives
Influenza-like illnesses have been described since the time of Hippocrates, varying from mild upper respiratory symptoms to cases of fulminant hemorrhagic disease.1 The peak of the annual flu season can be classified as an "epidemic" the episodic pandemics are what history remembers.
A pandemic is "an epidemic occurring worldwide or over a very wide area, crossing international boundaries, and usually affecting a large number of people."2 Influenza pandemics occur about three times per century, with the most famous being the Spanish flu of 1918. The loss of human life in the 1918 flu pandemic was phenomenal. The mortality rate was 2-20%, contrasting with mortality rate of 0.1% for the seasonal flu.3 In total, 5% of the world's population died, with estimates of 25 million dead in 25 weeks. For comparison, HIV/AIDS has accounted for 25 million deaths in its first 25 years.3
The first pandemic of this century was the "swine flu" pandemic of 2009. While it ultimately was milder than previous pandemics, with a case fatality rate of 0.03%, the 2009 flu earned its designation by infecting 482,300 people in 199 countries.4
Virology
Influenza viruses are RNA viruses in the Orthomyxoviridae family known to cause disease in a variety of animal species. The family can be split into three genera (influenza A, B, and C), all of which cause infection in humans.5 Of the three, influenza A is both the most prevalent and most severe.6 It is responsible for the annual flu season as well as the pandemics seen throughout history. Influenza B circulates at lower levels and produces less severe disease.7 Influenza C is capable of producing severe disease, but is much less common and not routinely included in the rapid diagnostic testing kits.8
All three genera have a similar structure that includes a viral envelope surrounding a central RNA genome core. (See Figure 1.) The central genomic core is made up of eight single-stranded RNA.9 The envelope contains two large glycoproteins, hemagglutinin (HA) and neuraminidase (NA), which serve as the targets for both antiviral agents and host antibodies. HA is responsible for binding the free-floating virus to target cells and introducing the viral genome. NA mediates the release of viral progeny from the infected cells.10
Figure 1: Structure of Influenza Virus
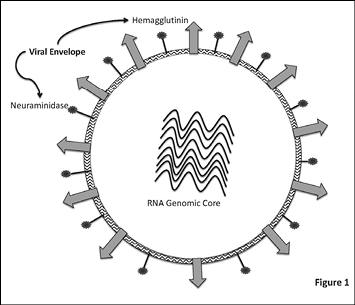
There are 17 different hemagglutinin antigens (H1-H17) and 10 different neuraminidase antigens (N1-N10) that form the basis of nomenclature in the subtypes of influenza A.11,12 For example, an influenza A virus with a type 5 hemagglutinin and type 1 neuraminidase encoded in its genome would be known as H5N1. Influenza A variants can also be grouped according to the host species to which the flu viruses are endemic (e.g., bird flu, human flu, swine flu, equine flu). Currently, N1H1, H1N2, and H3N2 are the only known influenza A subtypes circulating among humans.12 Sporadic cases of different variants may arise but do not result in sustained transmission.13
Antigenic Shift Versus Drift
Influenza evolves through two important mechanisms: antigenic drift and antigenic shift.14,15 (See Figure 2.) Antigenic drift occurs as random mutations create a variety of strains with small antigenic differences. Because of their similarities to the prior generations, it's likely humans will have immunity to most variants.16 However, a small antigenic change in the right place can provide a new strain with just the advantage it needs to infect and transmit itself successfully from human to human. This strain will move steadily through the population, causing a seasonal epidemic.16
Figure 2: Antigenic Drift and Antigenic Shift
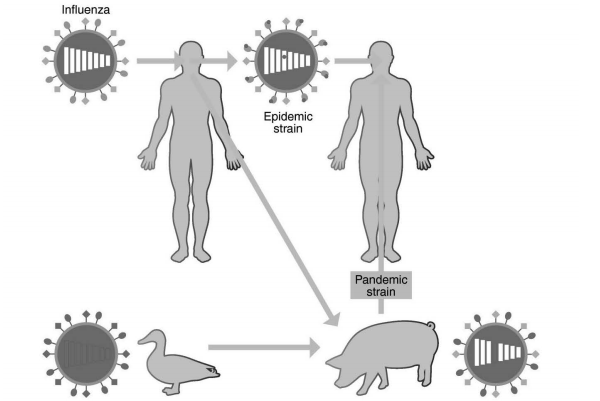
Minor genomic mutations in antigenic drift result in an epidemic strain passed from human to human. In antigenic shift, influenza from 2 separate species (human and bird) mix in a third host species (pig) to create a novel virus capable of producing a human pandemic.14,15 Reprinted with permission of Oxford University Press.
Antigenic shift or reassortment occurs when an influenza virus acquires completely new surface antigens. Reassortment is when two different viruses infect the same host cell and exchange genetic material during replication.17 This most often occurs when strains endemic to different species mix. This can result in a unique strain not seen by humans previously. With lack of immunity, these novel strains can spread widely and quickly, causing a pandemic.18 The virus responsible for the 1918 pandemic, for example, incorporated genetic material from human, swine, and avian origins.19
Transmission
Infected persons become contagious after a 1-3 day incubation period just before clinical symptoms manifest. The virus will shed for another 5-7 days, with the second or third day being the highest infectivity period.20
The influenza virus can spread person to person by direct transmission of respiratory secretions when a person sneezes or coughs, producing aerosolized virions.21 A sneeze will release tens of thousands of virus particles, most of which will quickly settle from the air; however, inhaling just one virus particle can result in active infection.21,24,25 People can also acquire the virus through picking up particles from contaminated surfaces.22 The virus can survive outside the body for variable lengths of time. On hard, non-porous surfaces, the virus may persist one to two days, but only five minutes on skin.25 Protected in mucus, the virus can survive up to 17 days, and indefinitely when frozen.26,27
Clinical Course
Presentation. Consider influenza in anyone who presents with abrupt onset of cough and nasal congestion along with high fever and shaking chills. These symptoms have been reported as the most sensitive findings, yet their low specificities illustrate the overlap between influenza and other respiratory viral illnesses, particularly the common cold.28 (See Table 1.) In periods of local outbreaks when all three symptoms are present, the prevalence of the flu may top 70%.29
Table 1: Common Symptoms of Influenza
| Symptom | Sensitivity | Specificity |
| Fever | 68-86% | 25-73% |
| Cough | 84-98% | 7-29% |
| Nasal congestion | 68-91% | 19-41% |
| Adapted from Call S et al28 | ||
Influenza differentiates itself with extreme fatigue and myalgias throughout the body, particularly the back and legs. The myalgias can be severe and a major cause for the loss of work and school days. Less frequently, conjunctival injection and rashes are noted. While mild vomiting may be seen in adults, it is not a predominant symptom, and diarrhea is unusual in seasonal variants. Children commonly exhibit more gastrointestinal symptoms than adults during the illness.30 Because of the variance of symptoms, particularly among different age groups, and dependency on local disease prevalence, reliable clinical prediction rules for influenza infection have not been developed.30 To help guide the practicing clinician, the Infectious Disease Society of America (IDSA) has published guidance on when to consider influenza.31 (See Table 2.)
Table 2: Who Should Be Considered for Influenza Infection
During Periods of High Prevalence (flu season)
- Anyone with fever and acute onset of respiratory symptoms
- People with fever and exacerbation of lung disease
- Infants and young children with fever and no other signs or symptoms
- Severely ill with fever or hypothermia
Anytime during the year if patient is linked to an influenza outbreak
Adapted from IDSA guidelines31
Complications
While the overwhelming majority of influenza infections will be a mild, self-limiting process, certain cases evolve to a severe respiratory illness that requires hospitalization.32 What starts out as mild upper respiratory tract infection can quickly progress into a severe viral pneumonitis, usually by day 4 or 5 of illness.33 This pneumonitis is associated with severe hypoxemia, ARDS, and fulminant shock, characterized by diffuse infiltrates on chest radiography.34,35 (See Figure 3.)
Figure 3: Diffuse Infiltrates on Chest Radiography
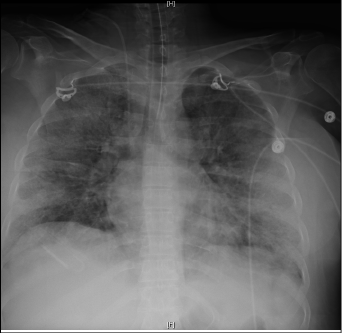
The most lethal complication is bacterial pneumonia.36,37 The classic organism described is Staphylococcus, including methicillin-resistant Staphylococcus aureus (MRSA), but Streptococcus pneumoniae and Streptococcus pyogenes have been documented as well.38 Bacterial superinfection has been implicated in up to 38% of deaths during influenza outbreaks and can be seen early (2-3 days) in the illness.38,39
Preexisting medical comorbidities are also affected in influenza infections, particularly underlying lung disease. Both asthma and chronic obstructive pulmonary disease (COPD) exacerbations are prolonged during co-infection and highly correlated with the need for hospitalization and respiratory failure.36,37
Rarely, complications are seen in other body systems. Neurologic manifestations, including confusion, seizure, and encephalopathy, have been described.40
Diagnostic Testing
The recent introduction of new, more effective antiviral treatments has stimulated the development of diagnostic testing methods for influenza. (See Table 3.) These tests rely on either direct detection of the virus within respiratory samples or indirect identification via the patient's immune response.
Table 3: Diagnostic Testing
| Technology | Time to Result | Pros | Cons |
| Rapid influenza diagnostic test (RIDT) | 15-30 minutes | Can detect both A & B; minimal technical expertise; point of care setting | Cannot detect subtypes (e.g., H1, H3, etc.); variable sensitivity and specificity |
| Immunofluorescence | 2 hours | Fast; can test for multiple respiratory viruses in same assay | Cannot distinguish H subtypes; requires technical expertise and fluorescence microscope |
| Viral culture | 48 hours - 14 days | "Gold standard" allows for testing of antiviral susceptibility, stockpiling of virus | Can be difficult to isolate virus; cannot be used for ED management |
| rt-PCR | 4-6 hours | Most sensitive and specific | Technology not widely available; testing time not ideal for ED setting |
Types of Tests
Rapid Influenza Diagnostic Tests (RIDTs). RIDTs are testing kits for the rapid detection of the influenza virus through recognition of viral antigens and are the most commonly utilized tests in the emergency department setting. There are two broad categories of RIDTs:41 those that can detect both influenza A and B but cannot distinguish between the two types, and those that can detect both influenza A and B and can distinguish between the two types. There is currently no RIDT that can specifically distinguish between influenza A virus subtypes. Respiratory samples are provided via nasopharyngeal (NP) swab, nasopharyngeal aspirate, or, rarely, throat swabs.
Sensitivity varies widely among types of RIDTs and have been reported as anywhere between 10-71%, with specificities greater than 90%.42,43 Sensitivity varies greatly based on collection time in comparison to onset of illness, study population, type of specimen, and circulating subtypes of disease.44 Sensitivity has also been found to be much higher in pediatric populations, likely due in part to higher concentrations of viral shedding by children.45 Since viral shedding peaks within the first 48 hours after symptoms, optimal sensitivity is obtained when collected within this time period.46 Recently, the CDC in conjunction with the Biological Advanced Research and Development Authority and the Medical College of Wisconsin tested 11 commercially available, FDA-approved RIDTs using stock samples of circulating virus strains.47 While all tests were able to detect the virus at its highest concentrations, many struggled with detection at lower levels of viral dilution.
Immunofluorescence Microscopy. For detection of influenza virus by immunofluorescence microscopy, respiratory samples are incubated with fluorescent-stained viral monoclonal antibodies and then examined under a fluorescence microscope for dye uptake.46 Uptake of dye indicates the presence of infection. The sensitivity and specificity of this type of test relies heavily on the number of infected cells present in the respiratory sample and is technique- and technician-dependent.45,46 Benefits of this type of testing include the ability for respiratory samples to be incubated with a variety of different monoclonal antibodies, allowing for simultaneous detection of multiple and alternative respiratory viruses, as well as a relatively rapid result time of about two hours.45,46 Owing to its difficult technique and varying sensitivity, immunofluorescence is not routinely available in hospital and commercial laboratories.
Viral Culture. Viral culture has traditionally served as the "gold standard" for diagnosing influenza.45,46 Isolation of the influenza virus is complex and requires evaluation of samples for at least 7 to 14 days. A newer technique, known as shell viral culture, has the ability to shorten detection time to 48 hours; however, this shortened time is still too long to be useful for individual patient care.46,47 It has been estimated that viral culture may miss 3% to 46% of influenza-positive patients, as viral shedding is minimal later in disease course and extremely difficult to isolate via culture.45,47 Viral cultures are still routinely used in reference laboratories, as they are useful in providing information on antiviral susceptibility of circulating strains, as well as allowing collection of stock virus to be used in research.41,42
Reverse-Transcription Polymerase Chain Reaction Assays. RT-PCR is currently considered to be the most sensitive and specific test for the diagnosis of influenza and is rapidly replacing viral isolation as the gold standard.41,42,46,47 In multiple studies, RT-PCR has been shown to identify more samples as positive than other diagnostic methods, including viral culture.45,47 Currently, platforms for RT-PCR are available in large state public health institutions and reference laboratories but are not widely available in rural or small community hospitals.41 This lack of widely available laboratories, the long time period required for testing (4-6 hours), and the technical skill required makes RT-PCR not feasible for management of individual patients in the emergency department setting, but still useful for confirmatory testing and population surveillance.
Using Diagnostic Tests for Treatment Decisions. When making a decision to use influenza testing in patient care, the clinician has to take into account several variables. First, most initial testing will be completed by RIDTs, which, as previously discussed, can have variable sensitivity rates; false-negative findings may occur because of low quantities of viral shedding, inappropriately collected samples, or emergence of novel viral subtypes that are not recognized by current rapid tests.44
One should also take into account the prevalence of influenza in their population. (See Figure 4.) During times of high prevalence, the positive predictive value of RIDTs is highest, meaning that a positive result is likely a true positive and a negative result more likely to be a false negative.46,48 During times of low prevalence of disease, the positive predictive value of RIDTs is lowered, meaning that a positive test result is more likely to be a false positive and a negative result more likely a true negative.44,48 In high prevalence periods, all patients do not require testing, as clinical judgment can approach sensitivities of 79-87%.48 During periods of low prevalence, both the CDC and the WHO recommend confirmatory testing by viral culture or RT-PCR.44,48
Figure 4: Interpreting the RIDT
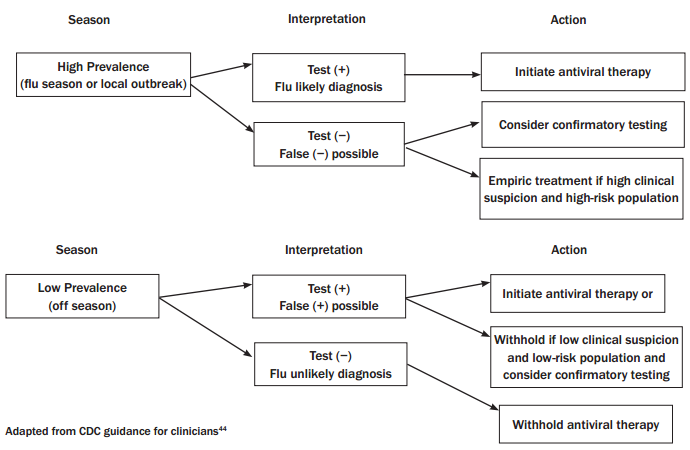
Clinicians should be aware of the cost vs. benefit aspects of influenza testing. The average cost of both a rapid influenza test and a course of antiviral treatment is approximately $50.43 When influenza prevalence and clinical suspicion are both high, it is more cost effective to treat empirically. The opposite is true when influenza prevalence is low.
Treatment
Adamantanes. Adamantanes were initially approved for treatment of influenza A in 1966. These drugs interfere with viral uncoating inside the cell by inhibiting the M2 ion channel protein.49 These drugs have no effect against influenza B, as they lack this protein.49,50 The oral drug amantadine was the first drug in this class. Concerns about adamantadine's central nervous system side effects, including confusion, decreased seizure threshold, and insomnia,45,50 led to the research and development of the newer drug rimantadine, which has a less severe side-effect profile. Multiple studies done in the 1960s to 1970s showed that adamantanes were effective in shortening the duration and severity of influenza A illness.49,50 No studies have shown a decrease in the rate of influenza complications with adamantane treatment.49
Since the 2006 influenza season, resistance to adamantanes has grown to more than 90% in the United States, with more recent resistance rates approaching 100%.51,52,53 Due to this high level of resistance, routine use of adamantanes for influenza treatment or prophylaxis is not recommended.
Neuraminidase Inhibitors. Neuraminidase inhibitors are a newer class of antivirals that inhibit the neuraminidase, thus blocking the virus's ability to reproduce through inhibition of budding.52 It is believed that replication of influenza cells within the respiratory tract reaches its peak within 24 to 72 hours after onset of illness, and initiation of neuraminidase therapy is recommended as soon as possible to target the replication stage of the virus and halt further host cell infection.52 As opposed to the amantadanes, these drugs are active against both influenza A and B. Currently two drugs are available.
Zanamivir. Zanamivir is administered by inhalation through a diskus. The drug becomes highly concentrated in the respiratory tract with low systemic absorption; only 5-15% of the total dose being absorbed and excreted in the urine.54 As the drug is inhaled, there is potential for respiratory side effects. Studies of FEV during treatment of patients with underlying asthma or chronic lung disease have had mixed results,54,55 and there have been some case reports of serious, sometimes fatal, bronchospasm associated with use;54 thus, zanamivir is contraindicated in patients with asthma and other chronic respiratory diseases.54,55
Zanamivir is FDA approved for treatment of influenza in patients older than 7 years of age and the prophylaxis of influenza in patients older than 5 years of age. Treatment dose is 10 mg (or two inhalations) twice a day for five days, and the prophylaxis dose is 10 mg once a day for 10 days.54
Oseltamivir. Oseltamivir is an oral capsule or powder for liquid suspension that is readily absorbed from the gastrointestinal tract and converted into its active carboxylated form.56 It achieves high plasma concentration, thus facilitating its activity outside of the respiratory tract. The most common side effects are nausea and vomiting, which occur in 5-10% of patients and may be reduced when taken with food.51,53,56 Serious hypersensitivity reactions such as Stevens-Johnson syndrome and toxic epidermal necrolysis have been rarely reported and should lead to prompt discontinuation of the drug.56
Oseltamivir is FDA approved for treatment of influenza in patients older than 2 weeks of age who have been symptomatic for no greater than 48 hours and for influenza prophylaxis for those older than 1 year of age. (See Table 4.) The dosage should be adjusted for those with renal impairment.56
Table 4: Oseltamivir Dosing Guidelines
| Population | Treatment Dosage | Prophylaxis Dosage |
| Adults and adolescents | 75 mg twice daily x 5 days | 75 mg once daily x 10 days |
| Ages 1-12 and > 40 kg | 75 mg twice daily x 5 days | 75 mg once daily x 10 days |
| Ages 1-12 and 23-40 kg | 60 mg twice daily x 5 days | 60 mg once daily x 10 days |
| Ages 1-12 and 15-23 kg | 45 mg twice daily x 5 days | 45 mg once daily x 10 days |
| Ages 1-12 and < 15 kg | 30 mg twice daily x 5 days | 30 mg once daily x 10 days |
| Ages 2 weeks to 1 year | 3 mg/kg twice daily x 5 days | Not applicable |
| Adapted from oseltamivir prescribing guidelines distributed by Genentech56 | ||
Efficacy in Treatment. Neuraminidase inhibitors initiated within 48 hours of symptoms onset decrease the severity and duration of influenza A and B illness. Multiple studies done in a wide range of geographical locations and patient populations have confirmed these findings.51,52,55,56
Early treatment seems to be the key to optimal neuraminidase inhibitor efficacy. The IMPACT trial done in 1999-2000 looked at the time between initiation of oseltamivir therapy and the duration and severity of influenza illness in more than 1400 patients 12 to 70 years of age.55 Results showed that initiation of therapy within 12 hours had the largest impact, and initiation at 48 hours the smallest impact on duration and severity of disease. Initiation of therapy at time intervals between 12 and 48 hours affected the disease duration and severity proportionally.
Data are limited on whether neuraminidase treatment can lower complication rates; however, several small-scale studies have shown that treatment with oseltamivir can lower incidence of lower respiratory tract complications, lower antibiotic usage, and decrease hospitalization rates.51,52,57,58
Resistance. In contrast to adamantanes, development of resistance to neuraminidase inhibitors is uncommon. Prior to 2007, annual surveillance identified a less than 1% resistance rate among circulating viral strains.59 During the 2007-2008 influenza season, the incidence of resistance increased to approximately 10% in the H1N1 viral strain in the United States.59 Despite this seasonal anomaly, current viral surveillance data indicate that approximately 99% of currently circulating influenza strains remain sensitive to neuraminidase inhibitors.52
Future Treatments
A parenteral form of zanamivir is being investigated,60,61 and recent trials show that it may reduce viral shedding and prevent illness. Peramivir is another new neuraminidase inhibitor that is undergoing trials for IV and IM administration60,61,62 and also appears to be effective in reducing duration and severity of illness.62,63 Laninamivir, a long-acting, single-dose inhalational neuraminidase inhibitor, is also under investigation, and early studies show it to be non-inferior to oral oseltamivir.60,61,63
Whom To Treat
According to the CDC in their 2011 recommendations on the use of antivirals,53 clinicians should initiate prompt treatment of confirmed or suspected influenza in persons at high risk for influenza complications or those who require hospitalization. The CDC identifies the following groups as high risk:
- children younger than 5 years old (especially those younger than 2 years old);
- Adults older than 65 years;
- people with chronic pulmonary (including asthma), cardiovascular, renal, hepatic, hematologic (including sickle cell), metabolic disorders (including diabetes), or neurologic disorders (including spinal cord disorders, mental retardation, cerebral palsy, and seizures);
- people with immunosuppression via medications or HIV;
- people who are pregnant or within 2 weeks post-partum;
- people younger than 18 years of age on chronic aspirin therapy (due to increased risk of Reyes syndrome);
- American Indians/Alaskan Natives;
- people who are morbidly obese;
- residents of nursing homes and other chronic care facilities.
In addition, treatment should be considered in those with confirmed or suspected illness who are not at high risk as long as treatment can be initiated within 48 hours of symptom onset.53 These patients with low risk of complications do not require treatment, but may see some benefit of shortened duration and a decrease in lost work days.52
Patients with suspected or confirmed illness requiring hospitalization should be started on antiviral treatment even if outside of 48 hours from symptom onset,41,42 as recent observational studies show that treatment up to 96 hours from symptom onset can reduce risk of severe outcomes.
Prophylaxis
The secondary illness attack rates among close contacts of those with confirmed influenza (especially family members residing in the same household) have been reported as 10-40%.66,67 Several large, randomized, controlled trials have shown that chemoprophylaxis with oseltamivir or zanamivir is 70-80% effective in protecting close contacts when given within 48 hours of exposure.67-69 Persons who should be considered for antiviral chemoprophylaxis include family or other close contacts of a person with suspected or confirmed influenza who are at higher risk for influenza complications but are not protected by vaccination.53,70 Patients who receive chemoprophylaxis may still acquire the influenza virus and potentially be able to transmit the virus even if clinical illness is prevented.53,66,71 Chemoprophylaxis should only be considered if antivirals can be started within 48 hours of the most recent exposure.64,68,69 As previously discussed, zanamivir is approved for chemoprophylaxis in patients older than 5 years of age, and oseltamivir is approved for patients older than 1 year of age.
Some thought has been given to pre-exposure prophylaxis, especially in very high-risk populations such as nursing home residents and severely immunosuppressed patients.56 A six-week study of oseltamivir chemoprophylaxis among nursing home residents demonstrated a 92% reduction in influenza illness.56,77 Pre-exposure prophylaxis must be administered for the length of time that exposure may occur.56 This prolonged use of antivirals may lead to a higher incidence of viral resistance and should only be used in those who are very high risk who cannot be otherwise protected.53,71 Duration of pre-exposure prophylaxis is varied to anywhere from 28-42 days.53,71 There are no data on therapy for more than six weeks. Dosing remains identical to post-exposure prophylaxis.
Isolation
The spread of influenza from person to person is primarily through large particle respiratory droplet transmission, i.e., when an infected person coughs or sneezes near an uninfected person.53 Emergency department waiting rooms and triage areas should remain vigilant about appropriate respiratory hygiene and cough etiquette during periods of high prevalence of influenza infection. Health care team members should actively ask all patients about respiratory symptoms and should attempt to isolate those with suspected respiratory illness.72 Posting visual alerts to provide patients as well as health care personnel with instructions on facemask use, cough etiquette, and hand hygiene is a reasonable step to aid in infection control.72
Once in the emergency department, droplet precautions should be implemented for patients with suspected influenza.72 These precautions require patients to be placed in single treatment areas whenever possible. Health care workers should wear facemasks when entering rooms, and these masks should be discarded after leaving the patient's room. While the CDC currently recommends N95 respirators, a recent randomized, controlled trial found that surgical masks appeared to be no worse than N95 respirators, and transmission rates leading to confirmed diagnosis were similar in the two populations.73 If the patient on droplet precautions must be moved for treatment or testing purposes, the patient should wear a facemask. Communication between departments about suspicions of influenza is important in helping decrease spread of the disease.72
For emergency department procedures that may cause increased infectious aerosols, such as intubation or cardiopulmonary resuscitation, the number of health care workers should be limited to only essential personnel and personal protective equipment, including a surgical mask, gown, and gloves, should be worn.72 Unprotected health care personnel and visitors should not be allowed in rooms for these procedures.
Special Considerations for Treatment and Prevention
Pregnancy. Pregnant women are especially susceptible to developing severe illness from influenza virus and are at an increased risk of hospitalization due to complications.74,75,76 During the recent H1N1 pandemic, pregnant women were four times more likely to be hospitalized, had higher rates of ICU admission, and had higher rates of death than the general population.74,77 Those women who have underlying conditions, including asthma, obesity, pregestational or gestational diabetes, and hypertension, appear to be at the greatest risk of hospitalization and death.74 Risk of influenza complications, including preterm labor and fetal death, appears to be highest in the second and third trimesters.74,76
Multiple studies have shown no increased risk in stillbirth or other birth complications with administration of vaccination or antiviral treatment.74-76 Current public health recommendations from the Advisory Committee on Immunization Practices and ACOG recommend influenza vaccination during the second and third trimesters as well as early treatment with antiviral medications without reliance on diagnostic testing.64,68 All currently available antivirals are pregnancy category C medications and dosing is identical to that for non-pregnant adults.64
Pediatrics. Children younger than the age of 2 years have high rates of hospitalization due to influenza, with the highest rates of both hospitalization and death occurring in those younger than 6 months.77,78
Infants younger than 6 months of age get some influenza protection from natural maternal influenza antibodies.77 Breastfeeding has also been shown to have some protective effects.77 Since 2003, influenza immunization has been recommended for infants ages 6 to 23 months and this was expanded to all children in 2010.
Efficacy of neuraminidase inhibitors in children has been shown in multiple large studies.51,54 In one large trial, oseltamivir treatment within 48 hours shortened the length of illness by 36 hours and decreased the incidence of otitis media by 44%.51
Elderly. Of the current estimated 36,000 influenza-related deaths in the United States each year, 90% occur in older adults and are most related to cardiovascular and pneumonia complications.79 In addition, almost two-thirds of all influenza-related hospitalizations for influenza are persons older than 65 years of age.72 Not surprisingly, severe influenza infection risk escalates with an increase in comorbidities, with the most vulnerable older adults experiencing 60 times the risk of hospitalization and death compared to that of healthy 65- to 75-year-olds.79 The highest risk factors include very advanced age, prior admission for influenza or pneumonia, chronic conditions including heart, lung, and renal disease, malignancy, previous stroke, and dementia.79
Prompt antiviral treatment of those with confirmed or suspected influenza68 is imperative, and post-exposure prophylaxis is indicated in any patient with known exposure. There is no change in treatment or prophylaxis dosing based on age alone.56,68
Immunocompromised. Despite the underlying cause of immunosuppression, all groups are higher risk for severe influenza infection resulting in hospitalization or death.82
In any patient who develops suspected or confirmed influenza, prompt treatment with antivirals should be initiated.56,91 Evidence is very limited, but oseltamivir post-exposure chemoprophylaxis has been shown to be effective for some immunocompromised patients and is strongly recommended for this patient group.91
Summary
Influenza is and will continue to be one of the most important infectious disease entities in emergency medicine. Influenza has classic mechanisms of evolution that make seasonal epidemics a certainty and historic pandemics a possibility.
Available medication for influenza includes an inhaled medication (zanamivir) and oral medication (oseltamivir). For most patients, these antivirals are not indicated unless they can be started within 48 hours. However, some patients at high risk for severe disease may benefit from treatment initiated after this timeframe.
References
- Martin P, Martin-Granel E. 2,500-year evolution of the term epidemic. Emerg Infect Dis 2006;6:976980.
- Miquel Porta, ed. Dictionary of Epidemiology. Oxford University Press. 2008:179.
- Taubenberger JK, Morens DM. 1918 Influenza: The mother of all pandemics. Emerg Infect Dis 2006;12(1):15-20.
- Pandemic (H1N1) 2009 World Health Organization (WHO) update 70, http://www.who.int/csr/don/2009_10_16/en/index.html Accessed November 11, 2013.
- Taubenberger JK, Morens DM. The pathology of influenza virus infections. Annu Rev Pathol 2008;3:499522.
- Korteweg C, Gu J. Pathology, molecular biology, and pathogenesis of avian influenza A (H5N1) infection in humans. Am J Path 2008;172(5):11551170.
- Centers for Disease Control and Prevention (CDC). Estimates of deaths associated with seasonal influenza — United States, 1976-2007. MMWR Morb MortalWkly Rep 2010;59(33):10571062.
- Matsuzaki Y, Katsushima N, Nagai Y, et al. Clinical features of influenza C virus infection in children. J Infect Dis 2006;193 (9):12291235.
- Bouvier NM, Palese P. The biology of influenza viruses. Vaccine 2008;26(4):D4953.
- Webster RG, Bean WJ, Gorman OT, et al. Evolution and ecology of influenza A viruses. Microbiol Rev 1992;56(1):152179.
- Wilks S, de Graaf M, Smith DJ, et al. A review of influenza haemagglutinin receptor binding as it relates to pandemic properties. Vaccine 2012;30(29):4369-4376.
- CDC. Influenza Type A Viruses and Subtypes.http://www.cdc.gov/flu/avianflu/influenza-a-virus-subtypes.htm. Retrieved 13 June 2013.
- Imai M, Kawaoka Y. The role of receptor binding specificity in interspecies transmission of influenza viruses. Curr Opin Virol 2012;2(2):160-167.
- Ito T, Couceiro JN, Kelm S, et al. Molecular basis for the generation in pigs of influenza A viruses with pandemic potential. J Virol 1998 Sep;72(9):7367-7373.
- DeFranco A. (2007). Immunity Primers in Biology. Corby, Northants: Oxford University Press.
- Wolf YI, Viboud C, et al. Long intervals of stasis punctuated by bursts of positive selection in the seasonal evolution of influenza A virus. Biol Direct 2008;1(1): 34.
- Labella AM, Merel SE. Influenza. Med Clin North Am 2013;97(4):621-645.
- Parrish C, Kawaoka Y. The origins of new pandemic viruses: The acquisition of new host ranges by canine parvovirus and influenza A viruses. Annual Rev Microbiol 2007;59: 553586.
- Garten RJ, Davis CT, Russell CA, et al. Antigenic and genetic characteristics of swine-origin 2009 A(H1N1) influenza viruses circulating in humans. Science 2009;325(5937):197201.
- Cao B, Li X-W, Mao Y, et al. Clinical features of the initial cases of 2009 pandemic influenza A (H1N1) virus infection in China. N Engl J Med 2009;361:2507-2517.
- Korteweg C, Gu L. Pathology, molecular biology, and pathogenesis of avian influenza A (H5N1) infection in humans. Am J Pathol 2008;172(5):11551170.
- Tellier R. Review of aerosol transmission of influenza A virus. Emerg Infect Dis 2008;12(11):16571662.
- Cole E, Cook C. Characterization of infectious aerosols in health care facilities: An aid to effective engineering controls and preventive strategies. Am J Infect Control 1998;26(4):453-464.
- Weber TP, Stilianakis NI. Inactivation of influenza A viruses in the environment and modes of transmission: A critical review. J Infect 2008;57(5):361373.
- Bean B, Moore BM, Sterner B, et al. Survival of influenza viruses on environmental surfaces. J Infect Dis 1989;146(1):4751.
- Thomas Y, Vogel G, Wunderli W, et al. Survival of influenza virus on banknotes. Appl Environ Microbiol 2008;74(10):30023007.
- De Benedictis P, Beato MS, Capua I. Inactivation of avian influenza viruses by chemical agents and physical conditions: a review. Zoo Pub Health 2007;54:51-68.
- Call S, Vollenweider M, Hornung C, et al. Does this patient have influenza? JAMA 2005;293(8):987997.
- Monto AS, Gravenstein S, Elliott M, et al. Clinical signs and symptoms predicting influenza infection. Arch Intern Med 2000;160(21):32433247.
- Woolpert T, Brodine S, Lemus H, et al. Determination of clinical and demographic predictors of laboratory-confirmed influenza with subtype analysis. BMC Infect Dis 2012;12(1):129.
- Harper SA, Bradley JS, Englund JA, et al. Seasonal influenza in adults and children — diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: Clinical practice guidelines of the Infectious Diseases Society ofAmerica. Clin Infect Dis 2009;48(8):10031032.
- Kumar A, Zarychanski R, Pinto R, et al. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA 2009;302:1872-1879.
- Writing Committee of the WHO. Clinical Aspects of Pandemic (H1N1) 2009 Influenza. N Engl J Med 2010;362:1708-1719.
- Kumar A, Zarychanski R, Pinto R, et al. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA 2009;302:1872-1879.
- Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients with 2009 H1N1 influenza in the United States, April-June 2009. N Engl J Med 2009;361:1935-1944.
- Louie JK, Acosta M, Winter K, et al. Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA 2009;302:1896-1902.
- Chertow DS, Memoli MJ. Bacterial coinfection in influenza: A grand rounds review. JAMA 2013;309(3):275-282.
- Mauad T, Hajjar LA, Callegari GD, et al. Lung pathology in fatal novel human influenza A (H1N1) infection. Am J Respir Crit Care Med 2010;181:72-79.
- The ANZIC Influenza Investigators. Critical care services and 2009 H1N1 influenza in Australia and New Zealand. N Engl J Med 2009;361:1925-1934.
- Noriega LM, Verdugo RJ, Araos R, et al. Pandemic influenza A (H1N1) 2009 with neurological manifestations, a case series. Influenza Other Respir Viruses 2010 4(3), 117120.
- Peterson S, Dugas AF, Rothman R. Evaluation of 11 commercially available rapid influenza diagnostic tests. Ann Emerg Med 2013;61(5):573-575.
- Barry MA. A 29-year-old woman with flu-like symptoms: Review of influenza diagnosis and treatment. JAMA 2010;304(6);671-678.
- Stein J, Louie J, Flanders S, et al. Performance characteristics of clinical diagnosis, a clinical decision rule and a rapid influenza test in the detection of influenza infection in a community sample of adults. Ann Emerg Med 2005;46(5):412-419.
- Centers for Disease Control and Prevention. Guidance for clinicians on the use of rapid influenza diagnostic tests. Available at:http://www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm. Accessed October 10, 2013.
- Ruest A, Michaud S, Deslandes S, et al. Comparison of the Directigen Flu A+B test, the QuickVue influenza test and clinical case definition to viral culture and reverse transcription-PCR for rapid diagnosis of influenza virus infection. J Clin Micro 2003;41(8):3487-3493.
- Retric M, Comanor L, Petti CA. Role of the laboratory in diagnosis of influenza during seasonal epidemics and potential pandemics. J Infect Dis 2009;194:S98-110.
- Roa PL, Catalan P, Giannella M, et al. Comparison of real-time RT-PCR, shell vial culture and conventional cell culture for the detection of the pandemic influenza A (H1N1) in hospitalized patients. Diag Micro and Inf Dis 2011;69:428-431.
- World Health Organization. Recommendations on the use of rapid testing for influenza diagnosis. Available at:http://www.who.int/influenza/resources/documents/RapidTestInfluenza_WebVersion.pdf. Accessed October 10, 2013.
- Hayden FG. Antivirals for influenza: Historical perspectives and lessons learned. Antiviral Research 2006;71:372-378.
- Couch RB. Prevention and treatment of influenza. N Engl J Med 2000;343:1778-1787.
- Moscana A. Neuraminidase inhibitors for influenza. N Engl J Med 2005;353:1363-1373.
- Dugas A, Rothman R. Antiviral agents for the treatment and chemoprophylaxis of influenza. Ann Emerg Med 2011;58(3):299-303.
- Centers for Disease Control and Prevention. Antiviral agents for the treatment and chemoprophylaxis of influenza: recommendations of the Advisory Committee on Immunization Practices. Morb Mortal Wkly Rep 2011;60(1):1-25.
- Relenza (zanamivir) prescribing information. Available at http://us.gsk.com/products/assets/us_relenza.pdf. Accessed October 22, 2013.
- Aoki FY, Macleod MD, Carewicz O, et al. Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicro Chemo 2003;51:123-129.
- Tamiflu (oseltamivir phosphate) prescribing information. Available at http://www.gene.com/download/pdf/tamiflu_prescribing.pdf. Accessed October 22, 2013
- Treanor J, Hayden FG, Vrooman P, et al. Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza. JAMA 2000;283(8):1016-1024.
- Jefferson T, Jones M, Doshi P, et al. Neuraminidase inhibitors for preventing and treating influenza in healthy adults. Cochrane Database Syst Rev 2010;2:CD001265.
- Sheu T, Deyde VM, Okomo-Adhiambo M, et al. Surveillance for neuraminidase inhibitor resistance among human influenza A and B viruses circulating worldwide from 2004 to 2008. Antimicro Agents Chemo 2008;52(9):3284-3293.
- Hayden FG. Developing new antiviral agents for influenza treatment: What does the future hold?. Clin Infect Dis 2009;48:S3-S13.
- Fry A, Perez A, Finelli L. Use of intravenous neuraminidase inhibitors during the 2009 pandemic: Results from population-based surveillance. JAMA 2011;306(2):160-162.
- Kohno S, Kida H, Mizuguchi M, et al. Efficacy and safety of intravenous peramivir for treatment of seasonal influenza virus infection. Antimicro Agents Chemo 2010;54(11):4568-4574.
- Watanabe A, Chang SC, Kim MJ, et al. Long-acting neuraminidase inhibitor laninamivir octanoate versus oseltamivir for treatment of influenza. Clin Infect Dis 2010;51(10):1167-1175.
- Center for Disease Control and Prevention. Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices — United States, 2013-2014. Morb Mort Wkly Rep 2013;62(7):1-48.
- Birnkrant D, Cox E. The emergency use authorization of peramivir for treatment of 2009 H1N1 influenza. N Engl J Med 2009;361:2204-2207.
- Welliver R, Monto A, Carewicz O, et al. Effectiveness of oseltamivir in preventing influenza in household contacts. JAMA 2001;285(6):748-754.
- Hayden FG, Belshe R, Villanueva C, et al. Management of influenza in households: A prospective, randomized comparison of oseltamivir treatment with or without postexposure prophylaxis. J Infect Dis 2004;189:440-449.
- Glezen WP. Prevention and treatment of seasonal influenza. N Engl J Med 2008;359:2579-2585.
- Ng S, Cowling B, Fang V, et al. Effects of oseltamivir treatment on duration of clinical illness and viral shedding and household transmission of influenza virus. Clin Infect Dis 2010;50(5):707-714.
- Harper S, Bradley J, Englund J, et al. Seasonal influenza in adults and children — diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: Clinical practice guidelines of the Infectious Disease Society of America. Clin Infect Dis 2009;48(8):1003-1032.
- Khazeni N, Bravata D, Holty JE, et al. Systemic Review: Safety and efficacy of extended duration antiviral chemoprophylaxis against pandemic and seasonal influenza. Ann Intern Med 2009;151:464-473.
- Centers for Disease Control and Prevention. Prevention strategies for seasonal influenza in healthcare settings: guidelines and recommendations. Available at http://www.cdc.gov/flu/professionals/infectioncontrol/healthcaresettings.htm. Accessed October 22, 2013.
- Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs. N95 respirator for preventing influenza among health care workers. JAMA 2009;(302(17):1865-1871.
- Siston AM, Rasmussen SA, Honein M, et al. Pandemic 2009 influenza A (H1N1) virus illness among pregnant women in the United States. JAMA 2010;303 (15):1517-1525.
- Haberg SE, Trogstad L, Gunnes N, et al. Risk of fetal death after pandemic influenza virus infection or vaccination. N Engl J Med 2013;368(4):333-340.
- Pasternak B, Svanstrom H, Molgaard-Nielson D, et al. Risk of adverse fetal outcomes following administration of a pandemic influenza A (H1N1) vaccine during pregnancy. JAMA 2012;308(2):165-174.
- Zaman K, Roy E, Arifeen SE, et al. Effectiveness of maternal influenza immunization in mothers and infants. N Engl J Med 2008;359:1555-1564.
- Poehling K, Edwards K, Weinberg G, et al. The underrecognized burden of influenza in young children. N Engl J Med 2006;355:31-40.
- McElhaney J, Zhou X, Talbot HK, et al. The unmet need in the elderly: How immunosenescence, CMV infection, co-morbidities and frailty are a challenge for the development of more effective influenza vaccines. Vaccine 2012;30:2060-2067.
- Barker W, Borisute H, Cox C. A study in the impact of influenza on the functional status of frail older people. Arch Intern Med 1998;158:645-650.
- Targonski P, Jacobson R, Poland G. Immunosenescence: Role and measurement in influenza vaccine response among the elderly. Vaccine 2007;25(16):3066-3069.
- Kunisaki K, Janoff E. Influenza in immunosuppressed populations: A review of infection frequency, morbidity, mortality and vaccine response. Lancet Infect Dis 2009;9:493-504.
- Beck C, McKenzie B, Hashim A, et al. Influenza vaccination for immunocompromised patients: Systematic review and meta-analysis by etiology. J Infect Dis 2012;206:1250-1259.
- Vu D, Peck A, Nichols WG, et al. Safety and tolerability of oseltamivir prophylaxis in hematopoietic stem cell transplant recipients. Clin Infect Dis 2007;45:187-193.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
