Diagnosis and Management of Respiratory Syncytial Virus in the Emergency Department
March 1, 2014
Reprints
Diagnosis and Management of Respiratory Syncytial Virus in the Emergency Department
Authors:
Katarzyna Gore, MD, Department of Emergency Medicine, Advocate Christ Medical Center, Oak Lawn, IL.
Samuel Lam, MD, RDMS, Department of Emergency Medicine, Advocate Christ Medical Center, Oak Lawn, IL.
Peer Reviewer:
Steven M. Winograd, MD, St. Barnabas Hospital, Core Faculty, Emergency Medicine Residency Program, Albert Einstein Medical School, Bronx, NY.
This article is adapted from one that originally appeared in the December 15, 2013 issue of Emergency Medicine Reports.
Respiratory syncytial virus is common during winter months, causing a spectrum of disease from upper respiratory infections to respiratory failure. Acute care physicians are challenged to appropriately evaluate and manage these children. The authors review common concerns and diagnostic and therapeutic controversies to optimize care of these children.
— Ann M. Dietrich, MD, Editor
Executive Summary
- In young infants, RSV has been found to be associated with spells of apnea, with a reported incidence in excess of 20% in some cases.
- Wilwerth et al identified the following risk factors for apnea:
- Preterm infants (born less than 37 weeks) younger than 48 weeks post conception on presentation;
- Term infants age 1 month or younger on admission;
- Those with a previously witnessed apnea episode by a doctor or parent.
- The current recommendation from the American Academy of Pediatrics (AAP) is not to routinely image children clinically diagnosed with bronchiolitis.
- In well-appearing febrile infants who test positive for RSV, blood and urine testing alone may be adequate to rule out SBI, particularly in those who are older than 29 days old.
Introduction
For many emergency physicians, the notion of respiratory syncytial virus (RSV) often brings up the mental image of young patients with bronchiolitis, so it is easy to forget that the virus can affect all age groups and cause a wide spectrum of illnesses, from mild upper respiratory illness to respiratory failure. The purpose of this article is to broaden the understanding of RSV, its epidemiology, pathophysiology, diagnosis, and treatment, and to familiarize the reader with evidence-based clinical management guidelines.
History and Epidemiology
RSV was first discovered in chimpanzees and initially named chimpanzee coryza agent.1 Researchers subsequently realized that the virus originated in humans, so Chanock et al later proposed a name change to respiratory syncytial virus.2
RSV is seen worldwide and affects people of all ages. The very young, very old, and immunocompromised are at highest risk for life-threatening infections. Its incidence varies by time of year, occurring mostly in the winter and early spring months in temperate climates. In most of the United States, the peak months are November through March.3 (See Figures 1 and 2.)
|
Figure 1. CDC RSV Seasonal Duration by Region 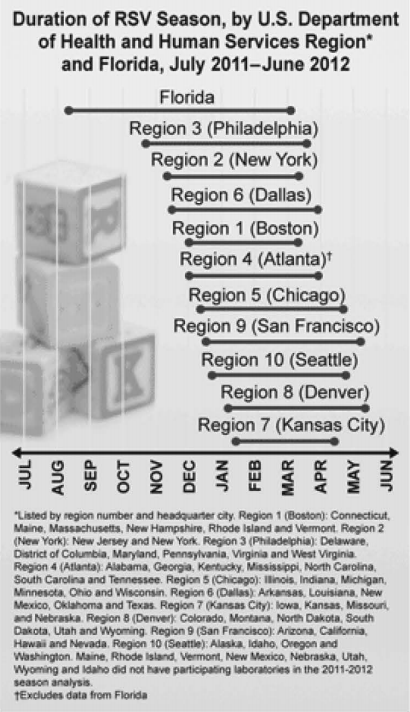
Source: Centers for Disease Control and Prevention web site. |
Figure 2. CDC Seasonal Variation of Antigen and Viral Positivity 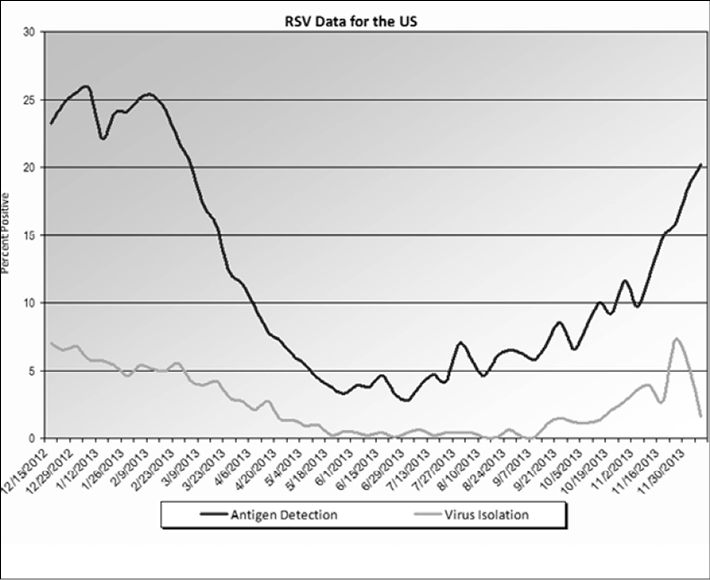
Source: Centers for Disease Control and Prevention |
RSV is the most common cause of lower respiratory infection in children younger than 1 year of age. Almost all children become exposed to the virus by age 3. Approximately 2-3% of those infected require hospitalization, of which 2-5% develop respiratory failure and require intubation. In the United States, RSV is associated with between 132,000 and 172,000 hospitalizations annually in children younger than 5 years of age.4 Globally, RSV is responsible for as many as 34 million episodes of acute lower respiratory tract infections, 3.4 million hospitalizations, and up to 199,000 deaths per year in children younger than 5 years of age.5 Groups that are at highest risk are premature infants, those with congenital heart and pulmonary diseases, as well as immunodeficient infants. Boys (more so than girls), children exposed to secondhand smoke, and those living in poverty are also more prone to RSV infection and illness.6
Pathophysiology
There are two strains of RSV (A and B), and it is part of the Paramyxoviridae family (genus Pneumovirus). (See Figure 3.) It is an enveloped RNA virus that is nonsegmented, which means it cannot undergo reassortment and change its virulence as easily as viruses such as influenza or rotavirus.7 The virus creates large sheets of multinucleated cells that are formed by the merging of smaller, neighboring cells through the help of its surface proteins. These "syncytia" have been widely studied in vitro, yet exactly how these syncytia contribute to pathogenesis (or whether they do at all) is uncertain.
Figure 3. Respiratory Syncytial Virus
Electron microscope image of respiratory syncytial virus.
Source: Centers for Disease Control and Prevention
Transmission of the virus is mostly thought to be through contact with large droplets that can survive on surfaces for hours.8 The virus appears to become unstable when aerosolized, so small-particle transmission is not a major mode of infection.9 The duration of viral shedding appears to be variable and dependent on whether the infection is primary or reinfection, the severity of the infection, and the age of patient. Younger patients with primary infections tend to shed for longer periods of time. In one study, the average shedding duration in infants was 6.7 days, with a range of 1-21 days.10 In another study, the average shedding time was 3.9 days, with a range of 1-17 days in healthy working adults.11 It has been shown that the spread of nosocomial RSV infection can be significantly decreased with contact isolation and use of gloves and gowns by providers.12
It is thought that the RSV infection pathway starts with a nasopharyngeal inoculation via direct contact with the virus. The virus then makes its way down into the epithelium of the respiratory tract by either cell-to-cell transfer or aspiration. In the lower airways, lymphocytic infiltration and mucus hypersecretion begin, followed by necrosis of the bronchial epithelium. The sloughing of these necrotic cells and its associated edema lead to airway resistance and air trapping.13 Young infants tend to be more adversely affected by this process, as airway resistance is inversely proportional to the airway radius to the fourth power. In infants with pneumonia, there is also marked inflammation of the lung interstitial tissue and alveoli.
It appears that both humoral and cellular pathways are activated during the infection process. The inflammatory reactions are partly responsible for the development of immunity; however, they are also responsible for the production of byproducts that cause symptoms of bronchiolitis. For instance, it appears that the types of cytokines and chemokines that become activated play a role in the severity of disease.14,15 Reinfections are possible due to generally incomplete immunity development, although subsequent infections tend to be milder in severity.
RSV Association with Reactive Airway Disease
RSV infection and its relationship to asthma has long been a topic of controversy. Many studies have shown a correlation between the two.16 In a recent article, 206 children diagnosed with severe RSV bronchiolitis before 1 year of age were prospectively enrolled. These children were followed up to their 7th birthday, and the parents were asked whether asthma was diagnosed by a physician. The study found that 48% of children with severe RSV bronchiolitis were subsequently diagnosed with asthma.17
Another study evaluated 95,310 children in a large cohort study looking at the relationship between birth date in relation to RSV season and the development of asthma. They found that infants born 122 days prior to the winter RSV virus peak had the highest risk of developing clinically significant bronchiolitis. In addition, infants who were 121 days old during peak season were at highest risk of developing asthma. Finally, having bronchiolitis at any age was associated with an increased risk of developing asthma. Based on these findings, these researchers made the bold statement that "evidence from our analyses provide compelling support for a causal role of winter viruses in the development of asthma."18 To support their theory, the authors pointed out that infants have lost most of their maternal antibodies by 4 months of age, and immunoglobulin activity is at its nadir. Furthermore, acute viral infections at this age may lead to chronic airway sequelae, as well as effects on immune regulation and development. Although these are highly plausible explanations from a pathophysiology standpoint, the controversy is far from settled at this point.
Clinical Presentation
RSV infection appears to cause different symptoms in premature neonates. In a study by Forster et al in the neonatal intensive care unit (NICU) setting, the presenting symptom was bradycardia in 75% of the cases who tested positive for RSV. Other significant symptoms include cyanosis, body temperature instability, and hypercapnia.19
In young infants, RSV has been found to be associated with spells of apnea, with a reported incidence in excess of 20% in some cases.20 The mechanism of how RSV causes apnea is unclear, although it is generally thought to be centrally mediated. In a retrospective cohort study of 691 hospitalized infants 6 months of age or younger with bronchiolitis, Wilwerth et al identified the following risk factors for apnea:
- Preterm infants (born less than 37 weeks) younger than 48 weeks post conception on presentation;
- Term infants age 1 month or younger on admission;
- Those with a previously witnessed apnea episode by a doctor or parent.21
Another case control study by Arms et al found that most infants with apnea were younger than 2 months and were symptomatic for less than five days.22 Finally, a study by Kneyber et al found that age younger than 2 months was the strongest predictor for apnea on admission, and was the strongest predictor for recurrent episodes of apnea in their patient population.23
In infants and young children, upper respiratory symptoms are the most common presentation of children with RSV infection. Twenty percent to 30% develop lower respiratory infections such as bronchiolitis and pneumonia, and a smaller percentage develop upper airway diseases such as croup and tracheobronchitis.24 Severity of disease can vary from mild wheezing to severe respiratory distress and even respiratory failure. Prematurity, in particular, is a significant risk factor for more severe disease.25 In a cohort study by Swingler et al in children with bronchiolitis in the ambulatory setting, the median duration of illness was 12 days; however, 9% were still symptomatic after 28 days.26 In another study by Petruzella et al, the median duration of bronchiolitis symptomatology was 15 days, with 25% of patients still symptomatic after 21 days.27 In children with reinfections, the presenting symptoms may be similar to primary infections but are often less intense, of shorter duration, and predominantly involving the upper respiratory tract.
Differential Diagnosis
RSV should be included in the differential diagnosis for any patient presenting with an upper or lower respiratory illness. Patients presenting in peak fall and winter months should be considered at high risk. It should also be considered in patients who are suspected of influenza but who do not test positive for influenza. Other viruses such as influenza, rhinovirus, adenovirus, human metapneumovirus, and parainfluenza can cause similar clinical symptoms. Table 1 reviews clinical differentiation of common clinical pneumonia syndromes of childhood.
Table 1. Common Clinical Pneumonia Syndromes of Childhood
| Syndrome | Typical Cause | Age Group | Clinical Features | Radiographic Findings |
|---|---|---|---|---|
|
Bacterial |
Streptococcus pneumoniae |
All ages, MC* 1 mo to 6 yrs |
Abrupt onset, high fever, focal findings, chest and/or abdominal pain |
Focal infiltrate |
|
Atypical infancy |
Chlamydia trachomatis |
3 wks to 3 mos |
Tachypnea, no fever, staccato cough, crackles |
Interstitial infiltrate |
|
Atypical |
Mycoplasma pneumoniae |
> 5 yrs |
Insidious onset, low-grade fever, systemic symptoms (headache, malaise, etc.) |
Diffuse, bilateral infiltrates, often "patchy" |
|
Viral |
Multiple viruses |
All ages, MC* 3 mos to 5 yrs |
URI symptoms, wheezing, +/- low-grade fever, diffuse exam findings |
Variable, diffuse interstitial infiltrates common |
|
* MC = Most common
Adapted from: Pediatrics in Review 2008;29:152. |
||||
Diagnostic Approach
The gold standard of RSV diagnosis is the detection of live virus in cell culture. Other means of diagnosis include detection of viral antigen by immunoassays and detection of viral nucleic acids by polymerase chain reaction (PCR). The specimen required is generally a nasopharyngeal aspirate or swab.13 The antigen tests have the advantages of being readily available, having a rapid turnaround time, and having sensitivities greater than 90%. The PCR test has a longer turnaround time, but is more sensitive than the antigen tests. Serology is not helpful in children or adults because repeated infections can result in a stable and sustained level of elevated RSV-specific antibody throughout a person's life.
In children who present with the classic bronchiolitis picture during peak RSV season, a clinical diagnosis may be made without further testing. In children who present out of the usual seasonal distribution and who do not present with classic symptoms, RSV testing may be useful. Other routine laboratory tests are generally of minimal value.
One question that is often raised by emergency physicians is when to image patients with suspected RSV infection. To address this issue, Kneyber et al derived a model for predicting a normal chest X-ray based on age, birth weight, presence of rhinitis, presence of retractions, and arterial oxygen saturation.28 The current recommendation from the American Academy of Pediatrics (AAP) is not to routinely image children clinically diagnosed with bronchiolitis unless they fall into high-risk groups (age younger than 12 weeks, history of prematurity, underlying cardiopulmonary disease, or immunodeficiency). (See Figures 4 and 5.) This is partly based on the fact that patients who underwent routine imaging had higher rates of antibiotic use without change in recovery time. However, radiography may be useful when the hospitalized child does not improve at the expected rate, if the severity of disease requires further evaluation, or if another diagnosis is suspected.29 (See Figure 6.)
|
Figure 4. Chest Radiograph of a Child with Bronchiolitis 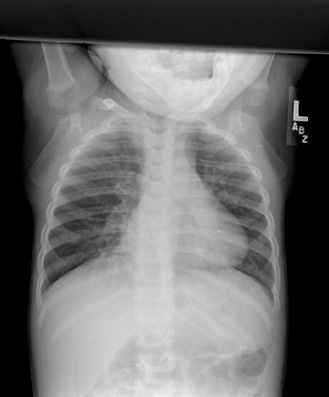
|
Figure 5. Chest Radiograph of a Child with Bronchiolitis 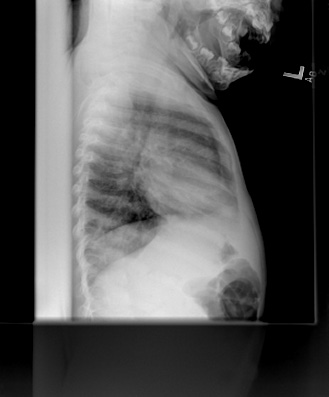
|
Figure 6. Cardiomyopathy 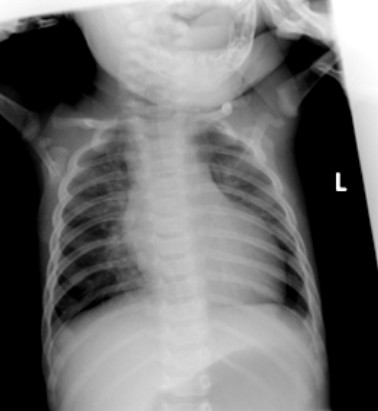
|
Other studies have focused on the predictive value of chest radiography. In a Portuguese multicenter study, neonates with consolidation on chest X-ray and confirmed RSV had worse outcomes in terms of length of NICU stay and mechanical ventilation days.30 However, in another study, chest X-rays had little predictive value on the clinical severity of disease in hospitalized children on general pediatric floors. The researchers suggested, therefore, that the use of radiography be limited to the intensive care setting.31
Another question often asked by emergency physicians is whether febrile young infants diagnosed with RSV should undergo a full septic workup to exclude other sources of serious bacterial infection (SBI). Levine et al conducted a multicenter, prospective study that looked at 1248 infants aged 60 days or younger presenting to the emergency department with a febrile illness. Almost all of these children underwent testing of blood, urine, stool, and cerebrospinal fluid. Two hundred sixty-nine children (22%) in the group were documented to have RSV infection. The rate of SBI in RSV-positive children (7%) was significantly lower compared with RSV-negative children (12.5%). Most cases of SBI in those who were RSV-positive were urinary tract infections, and none had concomitant bacterial meningitis. The rate of bacteremia was 1.1% in the RSV-positive group (all in infants younger than 29 days of age) and 2.3% in the RSV-negative group. On the other hand, the rate of SBI was not significantly different in neonates younger than 29 days who tested positive or negative for RSV.32 These findings were largely echoed by other similar studies.33-36 Hence, in well-appearing febrile infants who test positive for RSV, blood and urine testing alone may be adequate to rule out SBI, particularly in those who are older than 29 days old.
Management and Prevention
The treatment of RSV infection is highly dependent on the presentation and severity of disease. In general, treatment is mainly supportive; however, many adjunctive therapies have been evaluated over the years.
In young children without serious respiratory distress, many times supplemental oxygen and frequent suctioning of secretion can be sufficiently therapeutic. Mechanical ventilation may be necessary at times for those presenting in respiratory failure.13 It is also important for clinicians to assess hydration. Very young infants who are breathing at rates greater than 60 breaths per minute may not feed adequately and are very prone to dehydration and aspiration. These infants, therefore, should be admitted for hydration and observation until their respiratory rates allow for safe feeding.29
Bronchodilators
Bronchodilators such as albuterol have long been used and studied in RSV bronchiolitis. Unfortunately, there is no clear consensus as to their effectiveness. It may be easier to understand why wheezing in RSV bronchiolitis may not respond to bronchodilators by returning to the pathophysiology of the disease. In reactive airway disease such as asthma, the wheezing is a symptom related to bronchoconstriction and air trapping. As beta and alpha agonists work to dilate the airway smooth muscles, there is usually significant improvement in symptomatology. In viral bronchiolitis, the sloughing of cells and their collection in small airways, along with the excess secretions, are the reasons for air trapping, so bronchodilators have limited effect. In addition, it has been shown that these aerosolized medications have poor penetration into the bronchioles of infected infants due to the thick secretions.37
In a large Cochrane review, 28 randomized, controlled trials were analyzed to compare bronchodilators (excluding epinephrine) to placebo in infants with bronchiolitis. In this review, it was concluded that bronchodilators did not improve oxygen saturation, reduce hospital admission rate, reduce hospital length of stay, or reduce time to resolution of illness.38 Similar meta-analyses specific to RSV infections have concluded that bronchodilators are not efficacious in the treatment of RSV bronchiolitis. Nevertheless, several studies on intubated patients showed improvement of pulmonary function studies performed using ventilators after bronchodilator treatment.37
In a study by Skjerven et al, nebulized racemic adrenaline was compared with inhaled saline in hospitalized infants younger than 12 months of age diagnosed with moderate to severe bronchiolitis. In this multicenter, randomized, double-blind trial, 404 patients were evaluated for length of stay, use of oxygen, need for tube feeding, need for ventilatory support, and relative improvement in the clinical score from baseline. There did not appear to be a statistically significant difference between those who had received nebulized adrenaline and saline in any of these measures.39 These findings echo an earlier randomized, controlled study by Wainwright et al, in which there did not appear to be a statistically significant difference in length of hospital stay, need for supplemental oxygen or intravenous fluids, or respiratory effort scores between young infants who received nebulized racemic epinephrine and saline in the inpatient setting.40
Although the AAP guideline states that bronchodilators should not be used routinely in the management of bronchiolitis, many clinicians are still using these medications in their practice. This is likely based on the notion that viral bronchiolitis cannot be fully distinguished from reactive airway disease in young children, so a trial of the (relatively harmless) bronchodilators is appropriate. This practice is also acknowledged in the AAP guidelines, which state that "a carefully monitored trial of alpha-adrenergic or beta-adrenergic medication is an option. Inhaled bronchodilators should be continued only if there is a documented positive clinical response to the trial using an objective means of evaluation."29
Hypertonic Saline
Nebulized 3% saline solution has also been studied as a treatment for RSV bronchiolitis. In a meta-analysis by Zhang et al, 11 trials enrolling more than 1000 patients who had mild to moderate bronchiolitis were included. Nebulized 3% saline was compared with nebulized 0.9% saline. They found that hospitalized children in the hypertonic saline group had a shorter average length of hospital stay than those in the 0.9% saline group. However, treatment with nebulized 3% saline did not result in any significant outcome benefit in emergency department (ED) patients. The authors concluded that although there may be a role for nebulized hypertonic saline in the inpatient setting, its acute use in the ED appeared to be limited from the current studies.41
Steroids
In a Cochrane review, there was no statistically significant benefit on hospital stay or clinical outcomes with the use of corticosteroids when compared with placebo.42 This is also reflected in the AAP guidelines, which advise against the use of both enteral and inhaled corticosteroids in acute viral bronchiolitis.29
Antiviral
Aerosolized ribavirin has been studied as a treatment for RSV. The majority of the research has been done on young children in hospitalized settings. Although some studies have shown that ribavirin shortens hospitalization time, reduces duration of mechanical ventilation, and improves clinical scores, all were limited by small sample size, low power, and wide confidence intervals.43-46 Furthermore, these benefits have not been consistently demonstrated in other studies. For example, in a prospective, randomized study by Everard et al, there was no significant difference between the ribavirin and placebo groups in the length of hospital stay or clinical improvement at 24 hours.47 Another small prospective, randomized study by Guerguerian et al attempted to look at the benefits of ribavirin on mechanically ventilated children without other significant comorbidities, and found no significant difference between the aerosolized ribavirin and aerosolized saline groups.48
Other studies have attempted to determine the effects of ribavirin on the recurrence of disease. One study focused on the incidence of recurrent episodes of reactive airway disease and hospitalizations after early treatment with ribavirin during initial RSV infection. This study found that ribavirin-treated children had fewer and less severe reactive airway disease and recurrent respiratory infections than those in the control group. Although it was a prospective study, there were only 49 subjects.49 In another small prospective study, children given ribavirin tended to have fewer wheezing episodes and reactive airway complaints on long-term follow-up.50
Due to the lack of convincing evidence for the use of ribavirin, the AAP advises against its routine use in the general population. They do acknowledge, however, that its use may be considered in individuals with severe disease, or those who are at risk for severe disease (e.g, immunocompromised and/or hemodynamically significant cardiopulmonary disease).29
Vaccination. Due to the lack of effective therapy for RSV infections, many have focused on possible preventative efforts. In the 1960s, a vaccine was developed and tested in children, but the trials were stopped due to its adverse outcomes. The findings from these early trials showed that not only was the vaccine not protective against severe RSV infection, but many vaccinated children had an augmented reaction to initial RSV exposure and presented with more severe disease. Nearly 80% of the children vaccinated were hospitalized for respiratory infections, compared to 5% of the control subjects. Two infants who received the vaccine also died from subsequent RSV infection. Although the exact mechanisms of the paradoxical response are not fully understood, studies have suggested that the activation of cellular and humoral immunity was altered, and that the proliferation of lymphocytes was exaggerated in those who were vaccinated.51
In recent years, there have been renewed efforts at RSV vaccine development. Several vaccines utilizing various mechanisms have been created, but so far none has been proven effective in clinical human trials.52
Antibody. Palivizumab is a humanized mouse monoclonal antibody that can be given at the beginning of the RSV season for a total of five doses approximately one month apart. The antibody is very costly and is not recommended for the general population. It is recommended for use in children with chronic lung disease, premature infants, and children with hemodynamically significant congenital heart disease, and is likely beneficial in children with immune deficiencies.29,53 In two randomized, controlled trials, it was found that patients at high risk who were pretreated with palivizumab had significantly lower hospitalization rates than the control groups.54 The cost is approximately $288/kg body weight, with a recommended dose of 15 mg/kg. Due to its significant cost, it is uncertain whether this is cost effective in all patients for whom it may be recommended.55 A summary of treatment options for RSV is presented in Table 2.
Table 2. Summary of Treatment Options for RSV
| Infants/Children |
|
Bronchodilators (albuterol, racemic epinephrine)
|
|
Hypertonic (3%) saline
|
|
Corticosteroids
|
|
Antiviral (ribavirin)
|
|
Palivizumab
|
Disposition
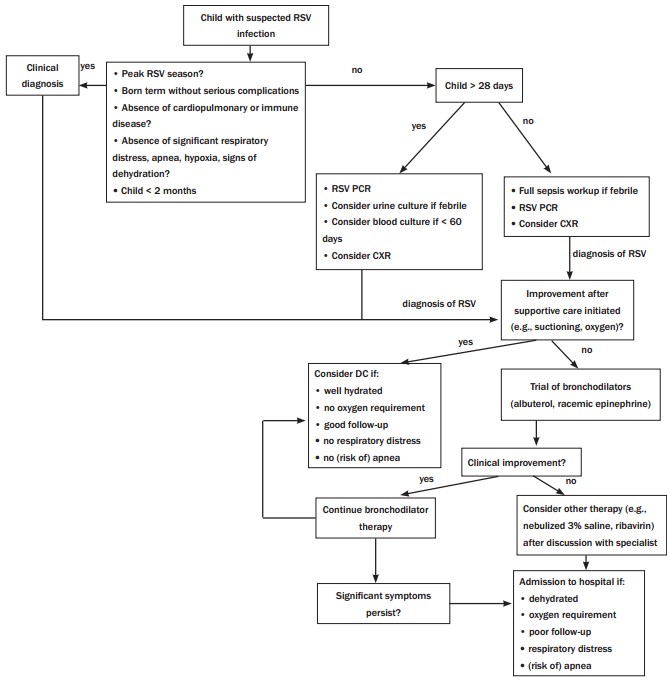
Disposition of patients diagnosed with RSV should be made based on their clinical status, degree of respiratory distress, hydration status, and presence of risk factors or comorbidities. Admission for supportive care should be considered for those at risk for decompensation and respiratory failure. The enclosed algorithm provides a guideline for the workup and disposition of such patients. (See Figure 7.)
Figure 7. RSV Evaluation:Children
Conclusion
RSV is a common cause of lower respiratory infection in children. Groups that are high risk include premature infants and children with congenital heart and pulmonary issues. Chest radiographs are not routinely indicated. Recognition of high-risk patients and optimizing therapy may decrease morbidity.
References
- Morris JA, Blount RE, Savage RE. Recovery of cytopathogenic agent from chimpanzee with coryza. Proc Sc Exp Biol Med 1965;92:544-549.
- Chanock R, Roizman B, Myers R. Recovery from infants with respiratory illness of virus related to chimpanzee coryza agent (CCA). Isolation properties and characterization. Am J Hyg 1957;66:281-290.
- US Center for Disease Control and Prevention. The National Respiratory and Enteric Virus Surveillance System (NREVSS) 2013. Available at: http://www.cdc.gov/surveillance/nrevss/rsv/natl-trend.html. Accessed on November 13, 2013.
- Stockman LJ, Curns, AT, Anderson LJ et al. Respiratory syncytial virus-associated hospitalizations among infants and young children in the United States, 1997-2006. Pediatr Infect Dis J 2012;31(1):5-9.
- Nair H, Nokes DJ, Gessner BD, et al. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: A systematic review and meta-analysis. Lancet 2010;375:1545-1555.
- Hall CB, Weinberg GA, Iwane MK, et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med 2009;360(6): 588-598.
- Welliver RC. Respiratory syncytial virus and other respiratory viruses. Pediatr Infect Dis J 2003;22(2 Supp):S6-S12.
- Hall CB, Douglas RG. Modes of transmission of respiratory syncytial virus. J Pediatr 1981;99(1):100-103.
- Rechsteiner J, Winkler KC. Inactivation of respiratory syncytial virus in aerosol. Journal of General Virology 1969;5(3):405-410.
- Hall CB, Douglas RG Jr., Geiman JM. Respiratory syncytial virus infections in infants: Quantitation and duration of shedding. J Pediatr 1976;89(1):11-15.
- Hall CB, Long CE, Schnabel KC. Respiratory syncytial virus infections in previously healthy working adults. Clin Infect Dis 2001;33(6):792-796.
- Leclair JM, Freeman J, Sullivan BF, et al. Prevention of nosocomial respiratory syncytial virus infections through compliance with glove and gown isolation precautions. N Engl J Med 1987;317(6): 329-334.
- Long SS, Pickering LK, Prober CG, et al. Principles and Practice of Pediatric Infectious Diseases: Expert Consult — Online, Elsevier Health Sciences 2012: 1130-1133.
- Garofalo RP, Patti J, Hintz KA, et al. Macrophage inflammatory protein-1α (not T helper type 2 cytokines) is associated with severe forms of respiratory syncytial virus bronchiolitis. J Infect Dis 2001;184(4):393-399.
- Legg JP, Hussain IR, Warner JA, et al. Type 1 and type 2 cytokine imbalance in acute respiratory syncytial virus bronchiolitis. Am J Respir Crit Care Med 2003;168(6):633-639.
- Régnier SA, Huels J. Association between respiratory syncytial virus hospitalizations in infants and respiratory sequelae: Systematic review and meta-analysis. Pediatr Infect Dis J 2013;32:820.
- Bacharier LB, Cohen R, Schweiger T, et al. Determinants of asthma after severe respiratory syncytial virus bronchiolitis. J Allergy Clin Immunol 2012;130(1): 91-100.e103.
- Wu P, Dupont WD, Griffin MR, et al. Evidence of a causal role of winter virus infection during infancy in early childhood asthma. Am J Respir Crit Care Med 2008;178(11):1123-1129.
- Forster J, Schumacher RF. The clinical picture presented by premature neonates infected with the respiratory syncytial virus. Eur J Pediatr 1995;154(11): 901-905.
- Ralston S, Hill V. Incidence of apnea in infants hospitalized with respiratory syncytial virus bronchiolitis: A systematic review. J Pediatr 2009;155(5):728-733.
- Willwerth BM, Harper MB, Greenes DS. Identifying hospitalized infants who have bronchiolitis and are at high risk for apnea. Ann Emerg Med 2006;48(4):441-447.
- Arms JL, Ortega H, Reid S. Chronological and clinical characteristics of apnea associated with respiratory syncytial virus infection: A retrospective case series. Clin Pediatr (Phila) 2008;47(9):953-958.
- Kneyber MC, Brandenburg AH, de Groot R, et al. Risk factors for respiratory syncytial virus associated apnea. Eur J Pediatr 1998;157(4):331-335.
- American Academy of Pediatrics. Respiratory syncytial virus. In: Pickering LK, ed. Red Book: 2012 Report of the Committee on Infectious Diseases, 29th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2012: 609-618.
- Henderson FW, Collier AM, Clyde WA Jr, et al. Respiratory-syncytial-virus infections, reinfections and immunity. A prospective, longitudinal study in young children. N Engl J Med 1979;300(10):530-534.
- Swingler GH, Hussey GD, Zwarenstein M, et al. Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med 2000;154(10):997-1000.
- Petruzella FD, Gorelick MH. Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics 2010;126(2):285-290.
- Kneyber MC, Moons KG, de Groot R, et al. Predictors of a normal chest X-ray in respiratory syncytial virus infection. Pediatr Pulmonol 2001;31(4):277-283.
- Lieberthal AS, Bauchner H, Hall CB, et al. Diagnosis and management of bronchiolitis. Pediatrics 2006;118(4): 1774-1793.
- Goncalves A, Rocha G, Guimaraes H, et al. Value of chest radiographic pattern in RSV disease of the newborn: A multicenter retrospective cohort study. Crit Care Res Pract 2012;2012:861867.
- Dawson KP, Long A, Kennedy J, et al. The chest radiograph in acute bronchiolitis. J Paediatr Child Health 1990;26(4):209-211.
- Levine DA, Platt SL, Dayan PS, et al. Risk of serious bacterial infection in young febrile infants with respiratory syncytial virus infections. Pediatrics 2004;113(6):1728-1734.
- Bordley W, Viswanathan M, King VJ, et al. Diagnosis and testing in bronchiolitis: A systematic review. Arch Pediatr Adolesc Med 2004;158(2):119-126.
- Titus MO, Wright SW. Prevalence of serious bacterial infections in febrile infants with respiratory syncytial virus infection. Pediatrics 2003;112(2):282-284.
- Byington CL, Enriquez FR, Hoff C, et al. Serious bacterial infections in febrile infants 1 to 90 days old with and without viral infections. Pediatrics 2004;113(6):1662-6-34.
- Ralston S, Hill V, Waters A. Occult serious bacterial infection in infants younger than 60 to 90 days with bronchiolitis: A systematic review. Arch Pediatr Adolesc Med 2011;165:951-956
- Black CP. Systematic review of the biology and medical management of respiratory syncytial virus infection. Respir Care 2003;48(3):209-233.
- Gadomski AM, Bhasale AL. Bronchodilators for bronchiolitis. Cochrane Database Syst Rev 2010; (12):CD001266.
- Skjerven HO, Hunderi JO, Brügmann-Pieper SK, et al. Racemic adrenaline and inhalation strategies in acute bronchiolitis. N Engl J Med 2013;368(24):2286-2293.
- Wainwright C, Altamirano L, Cheney M, et al. A multicenter, randomized, double-blind, controlled trial of nebulized epinephrine in infants with acute bronchiolitis. N Engl J Med 2003;349(1):27-35.
- Zhang L, Mendoza-Sassi RA, Wainwright C, et al. Nebulized hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst Rev 2013;7:Cd006458.
- Fernandes RM, Bialy LM, Vandemeer B, et al. Glucocorticoids for acute viral bronchiolitis in infants and young children. Cochrane Database Syst Rev 2013;6:CD004878
- Hall CB, McBride JT, Walsh EE, et al. Aerosolized ribavirin treatment of infants with respiratory syncytial viral infection. A randomized double-blind study. N Engl J Med 1983;308:1443-1447.
- Hall CB, McBride JT, Gala CL, et al. Ribavirin treatment of respiratory syncytial viral infection in infants with underlying cardiopulmonary disease. JAMA 1985;254(21):3047-3051.
- Rodriguez WJ, Kim HW, Brandt CD, et al. Aerosolized ribavirin in the treatment of patients with respiratory syncytial virus disease. Pediatr Infect Dis J 1987;6:159-163.
- Smith DW, Frankel LR, Mathers LH, et al. A controlled trial of aerosolized ribavirin in infants receiving mechanical ventilation for severe respiratory syncytial virus infection. N Engl J Med 1991;325:24-29.
- Everard ML, Swarbrick A, Rigby AS, et al. The effect of ribavirin to treat previously healthy infants admitted with acute bronchiolitis on acute and chronic respiratory morbidity. Respir Med 2001;95(4):275-280.
- Guerguerian AM, Gauthier M, Lebel MH, et al. Ribavirin in ventilated respiratory syncytial virus bronchiolitis. Am J Respir Crit Care Med 1999;160(3):829-834.
- Edell D, Khoshoo V, Ross G, et al. Early ribavirin treatment of bronchiolitis: Effect on long-term respiratory morbidity. Chest 2002;122(3):935-939.
- Rodriguez WJ, Arrobio J, Fink R, et al. Prospective follow-up and pulmonary functions from a placebo-controlled randomized trial of ribavirin therapy in respiratory syncytial virus bronchiolitis. Ribavirin Study Group. Arch Pediatr Adolesc Med 1999;153(5):469-474.
- Dudas RA, Karron RA. Respiratory syncytial virus vaccines. Clin Microbiol Rev 1998;11(3):430-439.
- Rudraraju R, Jones BG, Sealy R, et al. Respiratory syncytial virus: Current progress in vaccine development. Viruses 2013;5(2):577-594.
- Shadman KA, Wald ER. A review of palivizumab and emerging therapies for respiratory syncytial virus. Expert Opin Biol Ther 2011;11(11):1455-1467.
- Hu J, Robinson JL. Treatment of respiratory syncytial virus with palivizumab: A systematic review. World J Pediatr 2010;6(4):296-300.
- Wang D, Cummins C, Bayliss S, et al. Immunoprophylaxis against respiratory syncytial virus (RSV) with palivizumab in children: A systematic review and economic evaluation. Health Technol Assess 2008;12(36):iii, ix-x, 1-86.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
