Massive Transfusion: Part II
March 23, 2014
Reprints
Massive Transfusion: Part II
This is the second in a two-part series on massive transfusion. Part I focused on damage control resuscitation and use of blood. In part II, the authors examine adjunct therapy, scoring systems, and complications of massive transfusion.
— Sandra M. Schneider, MD, Editor
Executive Summary
- There are several relatively new adjuvant treatments for patients with massive hemorrhage. Of these, TXA is most widely recommended. It is inexpensive and has been shown to reduce mortality when given within three hours of injury.
- A new test, viscoelastic hemostatic assay, identifies the patient's stage of coagulability and fibrinogen status. The result of the test is a curve, which can help determine the need for fibrinogen, clotting factors, and platelets.
- Massive transfusion is an independent predictor of increased mortality, increased infection rate, SIRS, andmulti-organ failure.
- Complications of massive transfusion include acid/base derangement, electrolyte abnormalities (particularly hyperkalemia with rapid infusion), immune system changes, acute lung injury, and fluid overload.
Adjunct Therapy
Tranexamic Acid (TXA). TXA is a synthetic lysine analogue with anti-fibrinolytic properties. It exerts this action by inhibiting the conversion of plasminogen to plasmin. Plasmin, a protease, acts as a fibrinolytic agent by cleaving fibrin and platelet glycoprotein Ib receptors. It has been commercially available around the world for a number of years. In 2011, it was added to the World Health Organization's list of essential medicines, and is now available over-the-counter throughout Europe.1
It was well known that TXA use reduces blood loss in elective surgery; however, in 2010 a large multi-centered, randomized, controlled study, known as the CRASH-2 trial, confirmed this understanding. It showed that all-cause mortality and risk of death due to bleeding was significantly reduced with the administration of TXA.2
A systematic review of the CRASH-2 data, published in 2011, suggested the need for early administration of TXA. TXA administered within one hour of injury reduced the risk of death due to hemorrhage from 7.7% to 5.3%. If administered between one and three hours after injury, it was shown to reduce the risk of death due to hemorrhage from 6.1% to 4.8%. However, if administered after three hours, an increase in mortality from 3.1% to 4.4% was observed.3
Further evidence for the use of TXA in bleeding patients was presented in the 2012 military study known as the MATTERs (Military Application of Tranexamic Acid in Trauma Emergency Resuscitation) study. This retrospective, observational study compared TXA administration to no TXA in patients transfused with at least one unit of packed red blood cells (PRBCs). Those receiving TXA had lower unadjusted mortality than those not receiving TXA, despite being more severely injured. This benefit was greatest in the group of patients who received massive transfusion, in which TXA was also independently associated with survival and less coagulopathy. The study concluded that treatment with TXA should be implemented into clinical practice as part of a resuscitation strategy following severe wartime injury and hemorrhage.4
As a result of these data, military guidelines on the administration of TXA were amended.5 The guidelines currently stress that 1 g TXA should be administered IV over 10 minutes within three hours of injury, prior to the administration of fluids. This should be followed by a second 1 g IV dose over 8 hours. TXA is relatively inexpensive, averaging less than $100 per treatment in most U.S. hospitals. TXA is now included in resuscitation protocols at many civilian trauma centers. This protocol is currently being added to civilian trauma centers throughout the United States.
In Europe, anti-fibrinolytic drugs such as TXA are almost universally used during major surgery, as inhibition of the fibrinolytic pathway after tissue injury has been consistently shown to reduce blood loss. There is evidence for benefit in cardiac, orthopedic, urological, gynecological, and obstetric surgery.6
A 2013 prospective study by Sentürk et al addressing its use in cesarean section found that TXA significantly reduced blood loss during the procedure, and that its use was not associated with an increase in thromboembolic complications.7 Gungorduk et al looked at routine use of TXA for spontaneous vaginal delivery. They also found that administration during the third stage of labor reduced blood loss.8 In a review of the literature for postpartum hemorrhage, although initial therapy revolves around uterotonic drugs and the assessment for retained placenta, TXA may be considered first line for persistent hemorrhage.9
Recombinant Factor VIIa (rFVIIa). Factor VII is one of a series of vitamin K-dependent glycoproteins with an essential role in the activation of the clotting cascade. Factor VII also enhances thrombin generation on activated platelets, independent of factors VIII and IX. Recombinant factor VIIa is currently FDA approved for use in patients with factor VII deficiency and in hemophilia with inhibitors. It is also used off label for treating hemorrhage and coagulopathy.
Boffard et al conducted two parallel randomized, controlled clinical trials to assess the efficacy and safety of rFVIIa as an adjunct therapy in bleeding patients. One study assessed its use in blunt trauma, while the other assessed penetrating trauma. The study concluded that rFVIIa significantly reduced the need for PRBC transfusion in both blunt and penetrating trauma without an increase in thrombotic events. However, the study did not show a a statistically significant decrease in mortality.10
A further retrospective study of civilian and military patients by Perkins et al showed similar results. They found that early administration of rFVIIa (prior to transfusion of 8 units PRBCs) reduced PRBC transfusion by 20% in patients requiring massive transfusion. Again, no mortality benefit was observed, and complications, such as thrombotic events, were not found to be statistically significant.11
However, a retrospective review of military data showed that early use of rFVIIa was associated with decreased 30-day mortality in severely injured combat casualties requiring massive transfusion. Again, it was not shown to be associated with an increased risk of severe thrombotic events.12
More recently, in the largest placebo-controlled data set for the efficacy and safety of rFVIIa to date, the CONTROL trial once again showed a decrease in PRBC transfusion rates; however, no mortality benefit was observed. They did, however, report a possible increase in the incidence of myocardial infarction.13 Clinical trial dosing strategies have varied, but most have reported an initial dose of 200 μg/kg followed by repeat dosing of 100 μg/kg at 1 and 3 hours.14 However, one of the limitations of rVIIa is its cost. For 200 μg/kg dose, a cost of upwards of $12,960 has been reported.15
Given the inconclusive evidence for the efficacy and safety of rFVIIa, coupled with its high cost, it is not currently recommended for routine use in massive transfusion protocol.16,17
Prothrombin Complex Concentrate (PCC). Prothrombin complex concentrate (PCC), otherwise known as factor IX complex, is a combination of vitamin K-dependent glycoprotein clotting factors, as well as proteins C and S. PCC is prepared from the cryoprecipitate of large plasma pools. Various processing techniques enable the production of either three-factor (II, IX, and X) or four-factor (II, VII, IX, and X) concentrates. Overall, clotting factor concentration is approximately 25 times higher than in normal plasma.18 In terms of clotting factor concentration, 2000 mL of fresh frozen plasma (FFP) is comparable to a single dose of PCC. Unlike serum-based products such as FFP, PCC is lyophilized, allowing rapid reconstitution, as opposed to thawing.19 Although not as expensive as rFVIIa, which averaged approximately $7500 per patient in one study, PCC remains somewhat costly, averaging approximately $1000 per patient in the same study.20
PCC is currently FDA approved for congenital coagulation disorders and for reversal of oral anticoagulants, such as warfarin, in the setting of hemorrhage.21 For rapid reversal of warfarin in the setting of hemorrhage, 25 U/kg, irrespective of pre-injury coagulation profile, is recommended.22 However, the most effective dosing regimen for PCC in critically bleeding trauma patients remains unknown. Trauma patients may require a lower dose because they have an increased potential of thrombus formation due to the subsequent loss of anti-thrombin.
PCC use in the setting of acute hemorrhage has been hypothesized to be a more effective, beneficial, and cost-effective alternative to currently available therapy. These benefits may result from a more potent, sustainable, and rapid international normalized ratio (INR) reversal when compared to FFP.19 Data on the use of PCC in trauma are limited; however, a few recent studies are encouraging. Joseph et al described their experience in the use of PCC for coagulopathic trauma patients. They found that PCC rapidly and effectively treats coagulopathy after traumatic injury, irrespective of warfarin use.20 The study also showed a decrease in blood product transfusion. The authors advocated that PCC be considered as an effective tool to treat coagulopathy in trauma. However, they suggested that further prospective studies be conducted to investigate its efficacy and safety.23
A multi-center, retrospective trial assessing FFP compared to PCC coupled with supplementary fibrinogen linked the use of PCC and fibrinogen with a reduced need for blood products and a reduced hospital length of stay, despite a significantly worse hemodynamic status on presentation to the hospital.24
PCC is a potential therapy for critically hemorrhaging patients, but cannot be recommended as first-line therapy at this time due to the limits of its supporting data. Rather, it should be reserved for refractory bleeding until more data are available.
Recombinant Fibrinogen. Recombinant fibrinogen concentrate is a promising alternative to allogeneic blood products such as cryoprecipitate and FFP. It has been suggested that recombinant fibrinogen possesses a few advantages when compared to these traditional sources. Recombinant fibrinogen allows for a standardized dose, it can be more rapidly administered in a smaller volumes, and has a good safety profile.25
Early results demonstrate that fibrinogen concentrate is associated with a reduction in the use of blood products. Rehe-Meyer et al conducted a prospective, placebo-controlled, double-blind study in patients undergoing aortic replacement surgery to determine the efficacy of fibrinogen concentrate. Individuals were randomized to fibrinogen concentrate or placebo, administered intraoperatively. Patients in the fibrinogen concentrate group received fewer blood product components than the patients in the placebo group (median of 2 versus 13 units, respectively). In fact, total avoidance of transfusion was achieved in 13 of 29 patients (45%) in the fibrinogen concentrate group. In comparison, 100% of the placebo group required transfusion.26
Although the data are encouraging, further studies are necessary to confirm the role of fibrinogen concentrate in the management of acute hemorrhage.
Diagnosis of Massive Transfusion Requirement
Determining the need for massive transfusion early in the hospital stay is essential to prevent unnecessary delays in care. Certain injury patterns, such as multiple proximal amputations, evisceration with hypotension, and penetrating abdominal trauma, come with the inherent independent predictor of need for massive transfusion. The need for massive transfusion in other injuries can be less apparent upon hospital presentation.
In an attempt to devise accurate but easy-to-use mechanisms to help identify massive transfusion candidates, many different predictive scoring algorithms have been proposed. These scoring systems are required to be simple enough for the caregiver to calculate in the emergency department. They must only use laboratory values and/or data that can be obtained rapidly upon arrival while maintaining high sensitivities and specificities.
Scoring Systems
Maegele et al conducted a retrospective assessment of the various predictive algorithms and found that the two best performers among the models developed to date were the Trauma Associated Severe Hemorrhage (TASH) score and the Vandromme score.27
The TASH score, developed in 2006 and revalidated in 2011, was derived from 6044 severely injured blunt trauma patients treated in multiple Level 1 trauma centers in Germany. It incorporates seven weighted variables to produce a summative score from 0-28.28,29 (See Table 1.) Systolic blood pressure, gender, initial hemoglobin, focused assessment with sonography for trauma (FAST), heart rate, base excess, and open femur fracture or clinically unstable pelvic fractures are incorporated. The higher the score, the more likely the patient is to require massive transfusion. A score greater than 16 infers a 50% probability of requiring massive transfusion.27
Table 1: TASH Score
| Variable | Value | Score |
| Hemoglobin (g/dL) | < 7 | 8 |
| < 9 | 6 | |
| < 10 | 4 | |
| < 11 | 3 | |
| < 12 | 2 | |
| ≥ 12 | 0 | |
| Base Excess (mmol/L) | < -10 | 4 |
| < -6 | 3 | |
| < -2 | 1 | |
| ≥ -2 | 0 | |
| Systolic Blood Pressure (mmHg) | < 100 | 4 |
| < 120 | 1 | |
| ≥ 120 | 0 | |
| Heart Rate (bpm) | > 120 | 2 |
| ≤ 120 | 0 | |
| FAST | Positive | 3 |
| Negative | 0 | |
| Clinically unstable pelvic fracture | Yes | 6 |
| No | 0 | |
| Open femur fracture | Yes | 3 |
| No | 0 | |
| Gender | Male | 1 |
| Female | 0 | |
|
The seven weighted variables of the Trauma Associated Severe Hemorrhage (TASH) scoring system that are used to evaluate patients, recreated from Maegele et al.27 |
||
The Vandromme score, proposed in 2011 and based on a smaller dataset, incorporates five clinical measures: blood lactate, heart rate, INR, hemoglobin, and systolic blood pressure.30 (See Table 2.) A score greater than 3 was found to have a specificity of 98% in predicting the need for massive transfusion.27
Table 2: The Vandromme Scoring System
| Variable | Value | Score |
| Blood lactate (mmol/L) | < 5 | 1 |
| ≥ 5 | 0 | |
| Heart rate (bpm) | > 105 | 1 |
| ≤ 105 | 0 | |
| INR | > 1.5 | 1 |
| ≤ 1.5 | 0 | |
| Hemoglobin (g/dL) | < 11 | 1 |
| ≥ 11 | 0 | |
| Systolic blood pressure (mmHg) | < 110 | 1 |
| ≥ 110 | 0 |
An evidence-based review by Neal et al suggested that a third scoring system, the Assessment of Blood Consumption (ABC) score, was the more easily used.14 (See Table 3.) The ABC score was developed by Nunez et al using retrospective data from 596 patients presenting to a single Level 1 trauma center.31 It uses four non-laboratory, non-weighted variables to create a score from 0-4. These variables include penetrating mechanism, systolic blood pressure, heart rate, and FAST. The decision to exclude laboratory values makes the ABC score the fastest to calculate in the ED. An ABC score of 2 is reported to be 75% sensitive and 86% specific for predicting the need for massive transfusion.27
Table 3: The Assessment of Blood Consumption (ABC) Scoring System
| Variable | Value | Score |
| Penetrating mechanism | Yes | 1 |
| No | 0 | |
| Systolic blood pressure (mmHg) | < 90 | 1 |
| ≥ 90 | 0 | |
| Heart rate (bpm) | > 120 | 1 |
| ≤ 120 | 0 | |
| FAST exam | Positive | 1 |
| Negative | 0 |
In 2013, Callcut et al used the PROMMTT data to identify triggers associated with the need for massive transfusion, creating a massive transfusion score (MTS). Initial triggers were defined as INR greater than 1.5, systolic blood pressure less than 90, hemoglobin less than 11, base deficit greater than or equal to 6, positive FAST, heart rate greater than or equal to 120, and penetrating mechanism. If all triggers were known, an MTS was created, with one point assigned to each trigger met. They found that all triggers except penetrating mechanism and heart rate were valid independent predictors of massive transfusion, with INR being the most predictive. They also concluded that patients with MTS less than 2 were unlikely to require massive transfusion (negative predictive value of 89%), and for MTS greater than or equal to 2, the sensitivity for predicting massive transfusion requirement was 85%.32
Since there is no prospective, comparative analysis to demonstrate superiority or inferiority of any of these scoring algorithms, no consensus scoring system can currently be determined.14 However, it is clear that the scoring systems can be helpful guides in determining who may ultimately require massive transfusion.
Point-of-Care Viscoelastic Hemostatic Assays (POC VHA)
Point-of-care viscoelastic hemostatic assay (POC VHA), or thrombelastography (TE), has the potential to become the new standard of care for the early detection of coagulopathy. Its use is already incorporated into European guidelines.33 Although elevated plasma assays (PT and aPTT) are independent predictors of mortality in trauma, they are of limited value in the clinical setting of treating acute hemorrhage. The cell-based model of hemostasis stresses the role of platelets in the formation of intact thrombin and the stability of the forming thrombus. For this reason, using whole blood assays can provide the caregiver with a more complete picture of ongoing coagulopathy.34
Two different assays, known as the Thrombelastograph Hemostasis Analyzer system (TEG®) and Rotational Thromboelastometry (ROTEM®), are available. They provide information on clot development, stability, and lysis. Such tests provide real-time data about clot dynamics and, thus, specifics about a patient's coagulopathy.
To perform the TEG assay, whole blood is added to a warmed cuvette. A pin, connected to a detector system, is suspended within the cuvette. The cuvette effectively reproduces slow venous flow by rotating relative to the pin through an angle of 4° to 45°. As the blood clots, fibrin strands form between the cuvette and pin. These fibrin strands transmit the rotational force of the cuvette to the pin and a tracing is generated. Information regarding coagulopathic deficiencies is inferred based on the shape of this tracing.35 In ROTEM, the cuvette is stationary and the pin rotates.
VHAs have been used in cardiac and transplant surgery for more than 25 years to guide transfusion. A study by Ak et al showed use of VHA significantly reduced the use of blood component therapy and overall blood loss.36 A more recent Cochrane review suggested that VHA is superior to conventional assays in predicting blood transfusion requirements.37
Principle VHA measures include reaction time (R) or activated clotting time (ACT), angle (α), maximum amplitude (MA), maximal clot strength (G), clot lysis (ly), and clot lysis time (CLT). (Refer to Figure 1 to better understand these measurements and the following discussion.)
Figure 1: TEG Tracing Breakdown
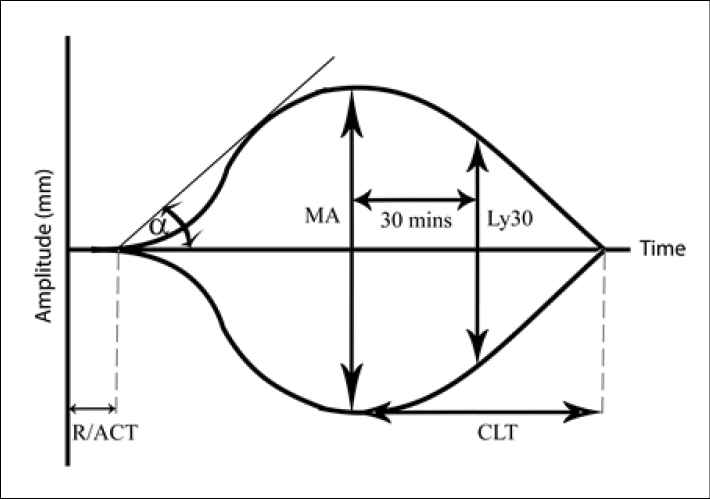
Figure 2: TEG Profiles
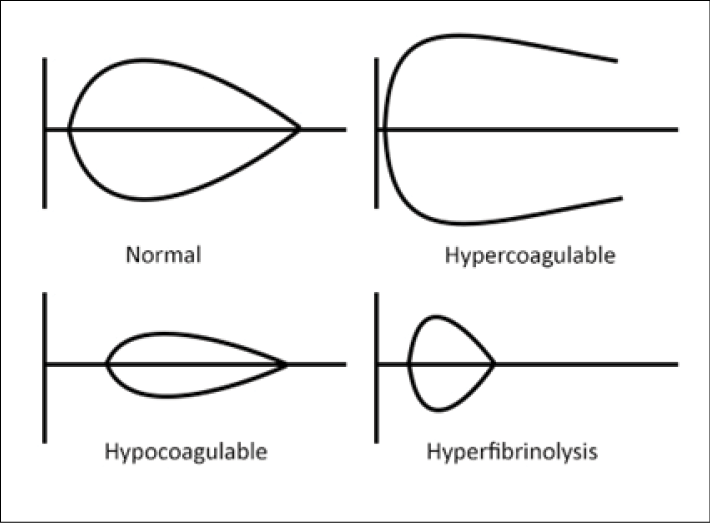
The R or ACT refers to the time it takes for a clot to begin to form. This is reflective of clotting factor concentration. R/ACT will thus be prolonged in consumptive or severe dilutional coagulopathy. The angle (α), in its most simplistic form, is a measure of the rate of clot formation and is reflective of fibrinogen activity. In hypofibrinogenemia, α will be smaller, whereas in hypercoagulable states it can be larger. Maximal amplitude (MA) and its mathematical counterpart, maximal clot strength (G), express the overall clot strength and are measures of platelet and fibrinogen activity. G can be calculated by the equation (5000 × A)/(100 - A) and possesses the units dynes/cm2.38 Thrombocytopenia and hypofibrinogenemia will have smaller MA and G. Clot lysis (ly) and clot lysis time (CLT) are measures of the rate of fibrinolysis. Shorter ly and CLTs are indicative of increased fibrinolysis.
Cotton et al conducted a prospective study to evaluate TEG efficacy and its correlation with traditional coagulation tests. Of 583 trauma activations during the data collection, 272 met the study criteria. Traditional coagulation tests (PT/APTT/INR/Plt) were compared to POC TEG (rTEG) readings. Early rTEG values (ACT) were available within 5 minutes. Later rTEG values (MA and angle) were available within 15 minutes, whereas traditional tests were available in 48 minutes. The results were two-fold. ACT showed correlation with traditional coagulation tests, whereas MA and angle correlated with platelet count. In addition, high ACT (> 128 seconds) predicted massive transfusion, while low ACT (< 105 seconds) predicted that no transfusion was required.39
In similar fashion, Pezold et al found that G provides consistent, independent prediction of the need for massive transfusion and of death early in resuscitation.38 This finding was mirrored in Kashuk et al's experiences with rTEG, in which G was associated with increased survival.40 Although assays such as TEG and ROTEM provide values that are considered "normal" for a given variable, there are limited data to verify such values and, thus, thresholds to begin treatment are not well established. This issue was recently addressed in a study by Chapman et al that examined fibrinolysis thresholds.41
It had previously been accepted that the normal upper limit TEG 30-minute fibrinolysis (LY30) value was 7.5%. However, this was hypothesized to be inappropriate in severe trauma, as the mortality had been noted to rise at much lower levels. Chapman et al showed that within the MTP patients, those with LY30 of 3% or greater were shown to be at a much higher risk of dying from hemorrhage (45.5% vs 4.8%).41 This study outlines the need for further investigation to establish evidence-based thresholds for treatment based on VHA tracings. It is also important to note that these variables differ between different VHA assays. Absolute values obtained from TEG, rTEG, and ROTEM tracings are not comparable. For this reason, specific values have not been published in this paper.
Goal-directed Therapy Based on VHA
Goal-directed treatment algorithms based on TEG tracings have been devised. Like MTPs in which the specifics can vary, the theory remains the same. Through the use of real-time TEG tracings, individual clotting deficiencies can be rapidly identified and treatment can be tailored to those deficiencies.
Given that a prolonged R/ACT depicts a deficiency in clotting factors, FFP (10-20 mL/kg) or cryoprecipitate (3-5 mL/kg) should be administered. When the angle (α) is smaller than expected, hypofibrinogenemia should be considered and a functional fibrinogen (FF) analysis should be performed. A low FF confirms hypofibrinogenemia and fibrinogen should be administered. This can be in the form of FFP (20-30 mL/kg), cryoprecipitate (5 mL/kg), or recombinant fibrinogen (25-50 mg/kg).
A shorter than expected MA could be indicative of hypofibrinogenemia, thrombocytopenia, or both. FF analysis should be performed to determine the cause. If the FF is low, again, fibrinogen should be administered. If the FF is normal, then the patient is more likely thrombocytopenic, and platelets (5-10 mL/kg) should be given.
As previously stated, CLT is shortened by fibrinolysis. In such a setting, the patient would likely benefit from the administration of TXA (1-2 g IV).34
Repeat TEG measurements can to be taken after 30 minutes to assess improvement of the prior coagulopathy and identify the need for further blood products or adjuncts.
Complications of Massive Transfusion
Blood transfusion has been recognized as an independent predictor of multiple organ failure, systemic inflammatory response syndrome (SIRS), increased infection, and increased mortality in multiple studies.42 The need for massive transfusion and its apparent associated mortality notwithstanding, massive transfusion is not without its own complications. These complications include acid/base derangements, electrolyte abnormalities, transfusion-related immunomodulation (TRIM), transfusion-related acute lung injury (TRALI), and transfusion-associated circulatory overload (TACO).42
Systemic Inflammatory Response Syndrome (SIRS). Severe traumatic injury results in a post-traumatic SIRS. This, in turn, leads to a post-traumatic anemia, as traumatic inflammation causes bone marrow suppression.43 This can be compounded by blood transfusion, since it has also been independently associated with an increased incidence of SIRS and death.44
Multiple Organ Failure. Sauaia et al recognized blood transfusion to be an independent risk factor for multiple organ failure in trauma patients.45 They found that age > 55 years, ISS ≥ 25, > 6 units PRBCs in the first 12 hours, base deficit > 8 mEq/L, and lactate > 2.5 mmol/L were independent predictors of multiple organ failure. The mechanism behind multiple organ failure is still poorly understood. It is believed to be a result of SIRS and cytokine mediated.
Acid/Base Derangements. As mentioned previously, a metabolic acidosis often develops in patients with hemorrhagic shock secondary to global hypoperfusion and lactic acidosis. This can be compounded by the transfusion itself as well as crystalloid infusion. Aggressive resuscitation should be continued despite this, as restoration of perfusion is paramount.
Citrate is metabolized by the liver to bicarbonate. This can create an iatrogenic metabolic alkalosis, which, in the presence of shock, can be protective. It should be noted that in patients with liver failure, acidosis is compounded by the liver's inability to catabolize lactate and citrate.46
The reversal of acidosis with alkalinizing agents such as sodium bicarbonate remains controversial and should be reserved for patients with hemodynamic instability due to severe metabolic acidosis.
Electrolyte Abnormalities: Potassium (K). Hyperkalemia and hypokalemia are consistently seen post massive transfusion. Hyperkalemia is usually due to renal insufficiency and crush injuries, the result of rhabdomyolysis and subsequent myonecrosis. However, in a 2008 study by Aboudara et al, 38.5% of transfused trauma patients (excluding crush injury) were noted to have serum potassium greater than 5.5 mEq/L, compared to only 2.9% of non-transfused patients.47 Despite this disparity, no clinical sequelae of hyperkalemia were observed. Potassium concentrations in PRBCs can range tremendously. It appears that with increased PRBC storage time, potassium concentrations increase due to the effect of ATPase pump inactivation and irradiation.42
There is also a transient hyperkalemia seen when transfusion rates exceed 100-150 mL/min. These rates are not uncommon in the acutely exsanguinating patient with a central venous catheter. The Level-1® Rapid infuser, used at many Level 1 trauma centers throughout the United States, infuses PRBCs at 150 mL/min through an 18-gauge peripheral line, at 300 mL/min through a 14-gauge peripheral line, and more than 500 mL/min through a 6-French catheter or central line.48 Rapid transfusion via central venous catheters is associated with hyperkalemic cardiac arrest in vulnerable populations, including critically ill adults.49
Hyperkalemia, however, can be transient, and hypokalemia often follows. The mechanism for this phenomenon is multifactorial. Once transfused, PRBC ATPase is reactivated, causing rapid influx of potassium into the cell.50 This can be compounded by catecholamine surge and the metabolic alkalosis, which results from unavoidable concurrent citrate administration.
Hypocalcemia/Hypomagnesemia — Citrate Toxicity. The liver can only metabolize approximately 3 g of citrate per 5 minutes. In patients with liver failure or who receive blood transfusions that exceed 1 unit in 5 minutes, citrate can accumulate in blood.51 Free citrate, being a negative ion, will bind to calcium and magnesium in the blood, resulting in hypocalcemia and hypomagnesemia known as citrate toxicity. Clinical signs of citrate toxicity include tetany, prolonged QT, decreased myocardial contractility, and resultant hypotension. If left untreated, this can progress to ventricular fibrillation and PEA arrest.
Transfusion-related Immunomodulation (TRIM). Transfusion-related immunomodulation (TRIM) is used to describe the frequent observation that PRBC transfusion is associated with immunosuppression and increased inflammation.52 Although the precise mechanisms of TRIM remain unclear, it is thought that TRIM is contingent on an interchange between transfusion effects, genetic predisposition, and comorbidities.
It has been proposed that TRIM may be mediated by: allogeneic mononuclear cells, white blood cell-derived soluble mediators, and/or soluble HLA peptides circulating in allogeneic plasma.52 However, platelets and vascular endothelial cells likely contribute to TRIM as well.53
TRIM has been linked to cancer recurrence, postoperative infection, viral activation, multiple organ failure, and resultant mortality.52 A 2003 meta-analysis verified the relationship of transfusion to postoperative bacterial infection.54 The researchers found that the odds ratio for the risk of postoperative bacterial infection for transfused versus non-transfused patients was 3.45 (and 5.26 for the subset of trauma patients).54
Transfusion-related Acute Lung Injury (TRALI). Acute lung injury (ALI) is defined as bilateral pulmonary infiltrates on chest X-ray in the absence of left atrial hypertension and SpO2 < 90% on room air or a PaO2/FIO2 (P/F ratio) ≤ 300 mm Hg. ALI becomes acute respiratory distress syndrome (ARDS) when the P/F ratio drops to ≤ 200 mm Hg.55 TRALI is any ALI that ensues within six hours of transfusion that cannot be attributed to any other risk factors associated with ALI or ARDS.56
Clinically, TRALI often presents with the sudden onset of hypoxemic respiratory distress shortly after transfusion. A retrospective study presented the most common signs and symptoms, outside of the aforementioned definition, to be: pink, frothy airway secretions from the endotracheal tube (56%), fever (33%), hypotension (32%), and cyanosis (25%).57
TRALI is the chief cause of transfusion-related mortality in the United States.58 Historical estimates of TRALIassociated mortality have ranged from 5% to 8%.59 The incidence of TRALI varies depending on the blood product transfused: platelets > FFP > PRBC. It has been reported that the risk of TRALI in PRBC transfusion lies within the range of 1 in 5,000 units transfused.60 In comparison, FFP caries the risk of 1 in 2,000 units transfused, and platelets the risk of 1 in 400 units transfused.60 More recently, it has been suggested that the actual risk may be higher.42
The mechanism behind TRALI is still not fully understood; however, it is generally recognized to be a result of a "two-hit" mechanism.61 The primary "hit" refers to neutrophil sequestration in the lung microvasculature followed by their priming. The secondary "hit" involves neutrophil activation by blood product, which results in lung injury. Endothelial cells in the lung microvasculature are believed to account for the sequestration through adhesion molecules and priming through cytokine mediators, prior to transfusion. Once activated by the transfused blood products, the primed neutrophils release cytokines, reactive oxygen species, oxidases, and proteases that damage the pulmonary capillary endothelium.
Since the risk of TRALI increases with increased blood product volume, those receiving massive transfusion are especially at risk. As such, once hemorrhage is controlled, restrictive transfusion practices should be employed.42 Treatment of TRALI is primarily supportive: Oxygen should be administered and, if necessary, endotracheal intubation with the use of lung-protective mechanical ventilation should be initiated. In addition, minimizing fluid administration should be considered.
Transfusion-associated Circulatory Overload (TACO). TACO can be difficult to distinguish clinically from TRALI, as both often present as dyspnea. Although their initial clinical presentation may be similar, the mechanisms behind the two are very different. Whereas TRALI is a phenomenon of increased lung permeability, TACO is a result of hydrostatic pulmonary edema.42 Separating the two can be difficult; however, serum B-type natriuretic peptide levels may be helpful.62 Since TACO results from fluid overload, it often responds to diuretics.
Conclusion
Part I of this series introduced the concept of damage control resuscitation. This focuses on limited use of crystalloids, early transfusion, permissive hypotension, temporizing the lethal triad, and obtaining early surgical control of hemorrhage. Although the optimal ratio for blood product replacement in patients requiring massive transfusion is not known, the most recent data advocate for ratios of PRBC: FFP approaching 1:1, with most MTPs pushing treatment with PRBCs: FFP: platelets of 1:1:1. Tranexamic acid should also be considered, with cryoprecipitate as a source of fibrinogen for persistent bleeding.
Identification of which patients will require massive transfusion is challenging. Many scoring systems, while promising, require complex calculations, may be time-consuming, and require prospective validation. The TASH and Vandromme scoring systems are the highest performing scoring systems. The use of POC VHAs provides insight into patients' underlying coagulopathy. Data are available in real time and can be used to tailor treatment toward individual deficiencies.
Although the data for massive transfusion in non-trauma patients are sparse, there is still a role for utilizing these transfusion and resuscitative concepts for patients with massive blood loss.
References
- World Health Organization. The selection and use of essential medicines. WHO Technical Report Series: 2011:72.
- Shakur H, Roberts I, Bautista R, et al. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): A randomised, placebo-controlled trial. Lancet 2010;376:23-32.
- Roberts I, Shakur H, Afolabi A, et al. The importance of early treatment with tranexamic acid in bleeding trauma patients: An exploratory analysis of the CRASH-2 randomised controlled trial. Lancet 2011;377:1096-101, 101 e1-2.
- Morrison JJ, Dubose JJ, Rasmussen TE, et al. Military Application of Tranexamic Acid in Trauma Emergency Resuscitation (MATTERs) Study. Archives Surg (Chicago, Ill : 1960) 2012;147:113-119.
- Goodloe JM, Howerton DS, McAnallen D, et al. How can tranexamic acid help in EMS? TXA offers new hope for an old challenge in traumatic hemorrhagic shock. EMS World 2013;42:I5-6.
- Ortmann E, Besser MW, Klein AA. Antifibrinolytic agents in current anaesthetic practice. Br J Anaesthesia 2013;111:549-563.
- Senturk MB, Cakmak Y, Yildiz G, et al. Tranexamic acid for cesarean section: A double-blind, placebo-controlled, randomized clinical trial. Arch Gynecology & Obstetrics 2013;287:641-645.
- Gungorduk K, Asicioglu O, Yildirim G, et al. Can intravenous injection of tranexamic acid be used in routine practice with active management of the third stage of labor in vaginal delivery? A randomized controlled study. Am J Perinatology 2013;30:407-413.
- Gogarten W. [Postpartum hemorrhage — an update]. Anasthesiologie, Intensivmedizin, Notfallmedizin, Schmerztherapie: AINS 2011;46:508-514.
- Boffard KD, Riou B, Warren B, et al. Recombinant factor VIIa as adjunctive therapy for bleeding control in severely injured trauma patients: Two parallel randomized, placebo-controlled, double-blind clinical trials. J Trauma 2005;59:8-15; discussion -8.
- Perkins JG, Schreiber MA, Wade CE, et al. Early versus late recombinant factor VIIa in combat trauma patients requiring massive transfusion. J Trauma 2007;62:1095-1099; discussion 1099-1101.
- Spinella PC, Perkins JG, McLaughlin DF, et al. The effect of recombinant activated factor VII on mortality in combat-related casualties with severe trauma and massive transfusion. J Trauma 2008;64:286-293; discussion 293-294.
- Dutton RP, Parr M, Tortella BJ, et al. Recombinant activated factor VII safety in trauma patients: Results from the CONTROL trial. J Trauma 2011;71:12-19.
- Neal MD, Marsh A, Marino R, et al. Massive transfusion: An evidence-based review of recent developments. Arch Surgery (Chicago, Ill : 1960) 2012;147:563-571.
- Stein DM, Dutton RP, Hess JR, et al. Low-dose recombinant factor VIIa for trauma patients with coagulopathy. Injury 2008;39:1054-1061.
- McMorrow RC, Ryan SM, Blunnie WP, et al. Use of recombinant factor VIIa in massive post-partum haemorrhage. Eur J Anaesthesiology 2008;25:293-298.
- Magon N, Babu KM, Kapur K, et al. Recombinant activated factor VII in post partum haemorrhage. Nigerian Medical Journal 2013;54:289-294.
- Schulman S, Bijsterveld NR. Anticoagulants and their reversal. Transfusion Medicine Reviews 2007;21:37-48.
- Ferreira J, DeLosSantos M. The clinical use of prothrombin complex concentrate. J Emerg Med 2013;44:1201-1210.
- Joseph B, Hadjizacharia P, Aziz H, et al. Prothrombin complex concentrate: An effective therapy in reversing the coagulopathy of traumatic brain injury.J Trauma Acute Care Surg 2013;74:248-253.
- Lankiewicz MW, Hays J, Friedman KD, et al. Urgent reversal of warfarin with prothrombin complex concentrate. J Thrombosis and Haemostasis 2006;4:967-970.
- Pabinger I, Brenner B, Kalina U, et al. Prothrombin complex concentrate (Beriplex P/N) for emergency anticoagulation reversal: A prospective multinational clinical trial. J Thrombosis and Haemostasis 2008;6:622-631.
- Joseph B, Amini A, Friese RS, et al. Factor IX complex for the correction of traumatic coagulopathy. J Trauma Acute Care Surg 2012;72:828-834.
- Schochl H, Nienaber U, Maegele M, et al. Transfusion in trauma: Thromboelastometry-guided coagulation factor concentrate-based therapy versus standard fresh frozen plasma-based therapy. Critical Care (London, England) 2011;15:R83.
- Levy JH, Welsby I, Goodnough LT. Fibrinogen as a therapeutic target for bleeding: A review of critical levels and replacement therapy. Transfusion Oct. 9, 2013. [Epub ahead of print]
- Rahe-Meyer N, Solomon C, Hanke A, et al. Effects of fibrinogen concentrate as first-line therapy during major aortic replacement surgery: A randomized, placebo-controlled trial. Anesthesiology 2013;118:40-50.
- Maegele M, Brockamp T, Nienaber U, et al. Predictive models and algorithms for the need of transfusion including massive transfusion in severely injured patients. Transfus Med Hemother 2012;39:85-97.
- Yucel N, Lefering R, Maegele M, et al. Trauma Associated Severe Hemorrhage (TASH)-Score: Probability of mass transfusion as surrogate for life threatening hemorrhage after multiple trauma.J Trauma 2006;60:1228-1236; discussion 1236-1237.
- Maegele M, Lefering R, Yucel N, et al. Early coagulopathy in multiple injury: An analysis from the German Trauma Registry on 8724 patients. Injury 2007;38:298-304.
- Vandromme MJ, Griffin RL, McGwin G, Jr., et al. Prospective identification of patients at risk for massive transfusion: An imprecise endeavor. Am Surgeon 2011;77:155-161.
- Nunez TC, Voskresensky IV, Dossett LA, et al. Early prediction of massive transfusion in trauma: Simple as ABC (assessment of blood consumption)? J Trauma 2009;66:346-352.
- Callcut RA, Cotton BA, Muskat P, et al. Defining when to initiate massive transfusion: a validation study of individual massive transfusion triggers in PROMMTT patients. J Trauma Acute Care Surg 2013;74:59-65, 7-8; discussion 6-7.
- Rossaint R, Bouillon B, Cerny V, et al. Management of bleeding following major trauma: An updated European guideline. Critical Care (London, England) 2010;14:R52.
- Johansson PI, Stensballe J, Ostrowski SR. Current management of massive hemorrhage in trauma. Scand J Trauma, Resuscitation and Emerg Med 2012;20:47.
- Bolliger D, Seeberger MD, Tanaka KA. Principles and practice of thromboelastography in clinical coagulation management and transfusion practice. Transfusion Medicine Reviews 2012;26:1-13.
- Ak K, Isbir CS, Tetik S, et al. Thromboelastography-based transfusion algorithm reduces blood product use after elective CABG: A prospective randomized study. J Cardiac Surgery 2009;24: 404-410.
- Afshari A, Wikkelso A, Brok J, et al. Thrombelastography (TEG) or thromboelastometry (ROTEM) to monitor haemotherapy versus usual care in patients with massive transfusion. Cochrane Database Syst Rev 2011:CD007871.
- Pezold M, Moore EE, Wohlauer M, et al. Viscoelastic clot strength predicts coagulation-related mortality within 15 minutes. Surgery 2012;151:48-54.
- Cotton BA, Faz G, Hatch QM, et al. Rapid thrombelastography delivers real-time results that predict transfusion within 1 hour of admission. J Trauma 2011;71:407-414; discussion 414-417.
- Kashuk JL, Moore EE, Wohlauer M, et al. Initial experiences with point-of-care rapid thrombelastography for management of life-threatening postinjury coagulopathy. Transfusion 2012;52:23-33.
- Chapman MP, Moore EE, Ramos CR, et al. Fibrinolysis greater than 3% is the critical value for initiation of antifibrinolytic therapy. J Trauma Acute Care Surg 2013;75:961-967; discussion 967.
- Sihler KC, Napolitano LM. Complications of massive transfusion. Chest 2010;137:209-220.
- Robinson Y, Hostmann A, Matenov A, et al. Erythropoiesis in multiply injured patients. J Trauma 2006;61:1285-1291.
- Dunne JR, Malone DL, Tracy JK, et al. Allogenic blood transfusion in the first 24 hours after trauma is associated with increased systemic inflammatory response syndrome (SIRS) and death. Surgical Infections 2004;5:395-404.
- Sauaia A, Moore FA, Moore EE, et al. Early predictors of postinjury multiple organ failure. Arch Surg 1994;129:39-45.
- Funk GC, Doberer D, Kneidinger N, et al. Acid-base disturbances in critically ill patients with cirrhosis. Liver International 2007;27:901-909.
- Aboudara MC, Hurst FP, Abbott KC, et al. Hyperkalemia after packed red blood cell transfusion in trauma patients.J Trauma 2008;64:S86-91; discussion S.
- Medical S. Level 1 H-1200 Fast Flow Fluid Warmer Operator's Manual. In: Smiths Medical; 2010.
- Smith HM, Farrow SJ, Ackerman JD, et al. Cardiac arrests associated with hyperkalemia during red blood cell transfusion: A case series. Anesthesia and Analgesia 2008;106:1062-1069.
- Forester D. Hypokalemia, blood transfusions, and body temperature. Crit Care Med 1988;16:360-361.
- Kramer L, Bauer E, Joukhadar C, et al. Citrate pharmacokinetics and metabolism in cirrhotic and noncirrhotic critically ill patients. Crit Care Med 2003;31:2450-2455.
- Vamvakas EC, Blajchman MA. Transfusion-related immunomodulation (TRIM): An update. Blood Reviews 2007;21:327-348.
- Sparrow RL. Red blood cell storage and transfusion-related immunomodulation. Blood Transfusion = Trasfusione del Sangue 2010;8 Suppl 3:s26-30.
- Hill GE, Frawley WH, Griffith KE, et al. Allogeneic blood transfusion increases the risk of postoperative bacterial infection: A meta-analysis. J Trauma 2003;54:908-914.
- Bernard GR, Artigas A, Brigham KL, et al. Report of the American-European Consensus conference on acute respiratory distress syndrome: Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Consensus Committee. J Critical Care 1994;9:72-81.
- Toy P, Popovsky MA, Abraham E, et al. Transfusion-related acute lung injury: Definition and review. Crit Care Med 2005;33:721-726.
- van Stein D, Beckers EA, Sintnicolaas K, et al. Transfusion-related acute lung injury reports in the Netherlands: An observational study. Transfusion 2010;50:213-220.
- Fatalities Reported to FDA Following Blood Collection and Transfusion: Annual Summary for Fiscal Year 2011. http://www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/ReportaProblem/TransfusionDonationFatalities/ucm302847.htm. Accessed Jan. 24, 2014.
- Looney MR, Gropper MA, Matthay MA. Transfusion-related acute lung injury: A review. Chest 2004;126:249-258.
- Wallis JP, Lubenko A, Wells AW, et al. Single hospital experience of TRALI. Transfusion 2003;43:1053-1059.
- Silliman CC. The two-event model of transfusion-related acute lung injury. Crit Care Med 2006;34:S124-S131.
- Gajic O, Gropper MA, Hubmayr RD. Pulmonary edema after transfusion: How to differentiate transfusion-associated circulatory overload from transfusion-related acute lung injury. Crit Care Med 2006;34:S109-S113.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
