Obesity
April 1, 2014
Reprints
Executive Summary
Obesity in the United States is an epidemic affecting approximately 72 million Americans. Individuals with body mass index (BMI) > 30 kg/m2 have nearly a 100% increased risk of premature death as well as increased risk of developing more than 40 obesity-related diseases. With the recent recognition by the American Medical Association that obesity is a disease, it is incumbent upon primary care physicians to address our obese patients in a disease-model.
- The World Health Organization characterizes obesity as a BMI greater than 30 kg/m2.
- A useful tool for assessing patient readiness for obesity treatment is the 5As: Ask, Assess, Advise, Agree and Arrange/Assist.
- The American Society of Bariatric Physicians recently published new guidelines regarding the evaluation and treatment approaches for obese patients.
- Treatment approaches include dietary caloric restriction and nutritional counseling, exercise, elimination of common medications that contribute to weight gain, and consideration of pharmacologic intervention.
- Bariatric surgery remains the most effective modality in patients who are surgical candidates and have failed reasonable attempts of weight loss.
In June 2013, the American Medical Association officially recognized obesity as a disease.1 For many years, obesity was viewed as simply a behavioral disorder where the patient was not able to engage in enough physical activity to burn the calories consumed. This new recognition has enormous consequences since most insurance companies previously have denied coverage for this disease.
The recent epidemic of diabetes is the most obvious manifestation of the associated rise in obesity in the United States. Approximately 72 million Americans are obese and about 18 million have morbid obesity.2 Individuals with a body mass index (BMI) > 30 kg/m2 have a 50-100% increased risk of premature death as well as increased risk of developing more than 40 obesity-related diseases including diabetes, heart disease, and cancer.2 This article will define obesity, discuss pathophysiology, and provide a practical approach to effective treatment.
Clinical Vignette
Patient M is a 64-year-old male who came to a clinic because he was not able to lose weight, despite multiple attempts on his own. He works as a school bus driver, usually eats two meals per day, has breakfast occasionally, and is not very active. He also has hypertension, type 2 diabetes, and hypertriglyceridemia. His current weight is 282 pounds, with a BMI of 39.1 kg/m2. He currently takes metformin 500 mg po bid, aspirin 81 mg po daily, glypizide 5 mg po bid, hydrochlorothiazide 25 mg po daily, lisinopril 20 mg po daily, and amlodipine 5 mg po daily for his blood pressure. The patient is not able to tolerate statins because of severe myalgias. His current HbA1c is 11.3% and his percentage of body fat is 38% as measured by bioelectrical impedance.
This patient has multiple medical problems and is not able to lose weight despite trying "diets" on his own. He usually eats two meals a day, so overeating does not appear to be a major issue. His work schedule prevents him from physical activity. The above clinical findings indicate that his obesity is significantly affecting various organs. He has worsening or uncontrolled diabetes and elevated triglycerides.
This scenario is quite common in the primary care setting. Clinicians understand that if obesity is not effectively treated and reversed, patients' clinical condition will undoubtedly worsen over time. Primary care physicians need to know how to approach obese patients and what treatment options are available based on current evidence.
Definitions
- Disease is defined as a condition of the living animal or plant body or of one of its parts that impairs normal functioning and is typically manifested by distinguishing signs and symptoms.3
- Obesity is an increase in the number and/or size in the adipocytes leading to increased adipose tissue.4 This has serious consequences on how the human body functions.
- Calorie is the amount of energy needed to raise the temperature of one gram of water by 1° C.
Epidemiology and Classification
According to the third National Health and Nutrition Examination Survey (NHANES III) and Centers for Disease Control and Prevention (CDC) data, in 2010, 78 million adults and 12.5 million children were classified as obese based on BMI;5 the estimated cost per year to the health care system is about $147 billion.6 Obesity is more prevalent in the Hispanic Latino and African American populations. For children, different guidelines apply, and growth charts are used to measure a child's risk of becoming obese.7
Obesity can be classified by multiple parameters. The most widely used is BMI, which identifies the relationship between height and weight and estimates the amount of fat, although this is not a measurement of adiposity. BMI is not the most reliable or accurate method to measure fat.8 For example, many athletes might have BMIs that would be in the obese range, but their actual percentage of fat is normal or low.
Since obesity is defined as an increase in adipose mass, it makes more sense to pay attention to the degree of adiposity.
The World Health Organization classifies obesity based on the following categories:9
- Underweight, BMI < 18 kg/m2
- Normal, BMI 18.5-24.9 kg/m2
- Overweight, BMI 25-29.9 kg/m2
- Obesity Class I, BMI 30-34.9 kg/m2
- Obesity Class II, BMI 35-39.9 kg/m2
- Obesity Class III, BMI > 40 kg/m2 (this term replaces morbid obesity)
See Table 1 for classification of obesity and treatment options based on BMI.
Table 1: Classification of Obesity and Treatment Options Based on Body Mass Index
| BMI | Kg/m2 |
Lifestyle Modification |
Behavioral Counseling |
Pharmacotherapy | Surgery |
| Normal | 18.5-24.9 | Yes | |||
| Overweight | 25-29.9 | Yes | Yes | Yes* | |
| Obese I | 30-34.9 | Yes | Yes | Yes | |
| Obese II | 35-39.9 | Yes | Yes | Yes | Yes* |
| Obese III | 40 or above | Yes | Yes | Yes | Yes |
*Requires comorbidity like hypertension, type 2 diabetes, or dyslipidemia
Adapted from NHLBI Obesity Guidelines 2010.10
Waist Circumference
Visceral fat is one of the most important predictors of adiposity and possible complications, which include risk of developing cardiovascular disease, type 2 diabetes, certain types of cancer, and all-cause mortality.10-13 Waist circumference can be measured with a tape placed just above both iliac crests and going all the way around through the umbilicus. For the U.S. population, the upper limit for men is 40 inches and for women 35 inches, other ethnic populations may have different references. (See Table 2.)
Table 2: Waist Circumference as a Measure of Adiposity
| Male | Female | |
| Waist Circumference | > 40 inches (102 cm) | > 35 inches (88 cm) |
Percentage of Fat
The American Council on Exercise provides a useful table about the percentage of fat.14 (See Table 3.) Normally, women have more fat than men. In the clinical setting, it is useful to measure adiposity, since many patients who are not obese based on BMI or waist circumference may have a higher percentage of fat and may already express multiple features of the metabolic syndrome.
Table 3: Percentage of Fat
| Male | Female | |
| Essential Fat | 2-5% | 10-13% |
| Athletes | 6-13% | 14-20% |
| Fitness | 14-17% | 21-24% |
| Acceptable | 18-24% | 25-31% |
| Obesity | > 25% | >32% |
Source: American Council on Exercise. ACE Lifestyle & Weight Management Consultant Manual: The Ultimate Resource for Fitness Professionals. American Council on Exercise; 2009.
Measuring Adiposity
Both direct and indirect methods are available to help physicians estimate the amount of adipose tissue. Indirect methods include anthropometric measurements that include weight and height, with their relationship being used to calculate BMI (Formula: weight (kg)/[height (m)]2), waist circumference, hip-to-waist ratio, and skin fold measurements. Bioelectric impedance uses an electric current that when passed through the body is able to measure the resistance. As fat causes more resistance than muscle, this information can be used to calculate the amount of fat.15 These simple methods require minimal equipment and are a good alternative for office use as handheld devices are inexpensive.
Direct methods to measure adiposity include underwater measurement of weight, dual-energy X-ray absorptiometry (DXA), computed tomography (CT), and magnetic resonance imaging (MRI).16
Obesity clinics typically use anthropometric measurements with bioelectric impedance and or additional imaging studies such as DXA. These data points are helpful to monitor the patient's progress, as many are involved in exercise programs that make them gain muscular mass. Weight alone is not a reliable tool, since muscle weighs more than fat and since patients can lose a large amount of fat mass with little change in weight. This can cause frustration for patients who rely on body weight alone as a way to monitor their progress.
Physiology of Adipocytes
One of the largest organs in the body, the adipocytes have an important neuroendocrine function. They regulate how energy is stored, provide feedback to the hunger center through leptin, and play a role in blood pressure regulation, angiogenesis, and immune system17 through the multiple hormones, or adipokines, produced.
Leptin is a hormone that regulates energy balance. Once a person has stored enough fat, the adipocytes release leptin, which promotes satiety at the hunger center on the hypothalamus.18 In the setting of insulin and leptin resistance, this feedback does not work properly, and hormones like resistin, TNFa, and IL-6 may promote further inflammation, making patients appear more edematous and causing fluid retention. It also produces angiotensin II,19 which increases blood pressure and could be why many patients exhibit the metabolic syndrome (insulin resistance, high triglycerides, and high blood pressure). The adipocytes also produce other adipokines, so it is an active organ and not just a simple storage unit.
Pathogenesis
The pathogenesis of obesity is controversial and multifactorial, and several theories have been postulated. The energy imbalance theory, which is the most common theory, states that obesity occurs when a person eats more than he or she needs or fails to perform adequate physical activity. Energy in has to equal energy out, based on the first law of thermodynamics, energy conservation.20
The genetic component emphasizes hereditary cause. Although the vast majority of the obese population has no genetic defect, there is clearly a genetic or familial predisposition; for example, obesity is more common in children if both parents are obese.21 In addition, it is complicated by the common presence of shared behaviors and the environment within families. People basically grow up eating what is served at home, so if obesogenic food is prepared, family members likely will be more exposed to it. The most common genetic abnormalities related to obesity are the melanocortin receptor 4 (MC4R) deficiency,22 Prader-Willi syndrome, and Bardet-Biedel syndrome.
Epigenomics is a critical concept that investigates how food and the environment can affect gene expression and energy regulation. These environmental toxins are called endocrine disruptors23 and can occupy or mimic the function of multiple hormones. Natural foods like leafy greens, beans, and garlic can mimic the action of estrogen, showing some beneficial effect on bone mineral density, insulin resistance, and cardiovascular risk factors in women after menopause. Chemical compounds that are most commonly related to obesity are biphosphenol A found in plastics and known to cause hyperinsulinemia, and increased fat storage,24,25 and brominated flame retardants commonly found in colored soft drinks such as Mountain Dew, Gatorade, etc. These substances can alter the metabolism even in the perinatal period. If patients are exposed to multiple disruptors, including highly processed food (junk food) and soft drinks, they are more likely to develop obesity.
Hormonal Imbalance
Insulin plays an important role in how bodies store energy, especially fat. The two most powerful stimuli to the release of insulin are carbohydrates, especially glucose, and amino acids.26 Insulin has many important roles. It facilitates glucose entry into the cells, specifically muscle and adipose tissue for metabolic use.27 Glycolysis is the metabolic breakdown of glucose to produce energy in the form of ATP. Insulin suppresses hepatic glucose and triglyceride production, impairs adipose tissue lipolysis and muscle proteolysis, and stimulates glucose uptake in muscle.28 Insulin also acts like a growth hormone.29 The theory is that consuming high amounts of carbohydrates in the diet saturates the insulin receptors. Over time, a person will need more and more insulin to achieve a similar effect and to lower the blood sugar, a phenomenon known as insulin resistance.30 Since insulin makes a person store fat, the adipose tissue will grow, the signals for satiety via leptin also become saturated, and leptin resistance develops. The majority of obese patients have high levels of leptin.29
Sleep deprivation and poor quality sleep can also lead to obesity. Lack of sleep can impact appetite regulation, impair glucose metabolism, and increase blood pressure. Getting less than 6 hours of sleep has been associated with elevated BMI in observational studies.31,32 Inadequate sleep impacts the secretion of ghrelin, a signal hormone that increases appetite. As nearly 20% of the U.S. population get less than 6 hours of sleep, this equates to about 53 million people at risk of associated obesity.31,32
Endocrine causes of obesity include hypothalamic obesity, growth hormone deficiency, Cushing syndrome, and hypothyroidism.33 Although they are a small percentage (1%),34 they should be included in the differential diagnosis, especially in overweight children. Infectious agents such as human adenovirus-36 also have been associated with higher BMIs and possible obesity.35 Even electromagnetic field (EMF) radiation has been found to have some correlation with increased risk, especially in the perinatal period.36
Much of the current research is focusing on the gut microbiome. The human body is an ecosystem that shares life with billions of microbial organisms. Foods can change the balance of this ecosystem, i.e., certain types of bacteria promote the storage of fat while others do not, depending on what they are fed.37-40
Clinical Manifestations
The usual symptoms of obesity include increased weight gain, hunger, insomnia, and fatigue. Once a person has more fat cells, an increased number of hormones and neurotransmitters will further cause different disease manifestations, including increased blood pressure, increased levels of angiotensin produced by fat cells, and direct stimulation of insulin in the kidneys to increase renin with sodium and water retention. Persistent insulin resistance will lead to other manifestations such as type 2 diabetes, polycystic ovarian syndrome, obesity hypoventilation, obstructive sleep apnea, the overload of fructose to our liver causing fatty infiltration, and non-alcoholic steatohepatitis (NASH).35,41
Evaluation of the Patient with Obesity
Clinical evaluation should include a complete history and physical examination, with special attention devoted to prior weight loss attempts, learning dietary habits, monitoring weight oscillation, and discussing environmental support. Laboratory tests should include complete blood count, complete metabolic panel, cholesterol panel, hemoglobin A1c, basal insulin, leptin,42 adiponectin,43 and thyroid-stimulating hormone.
The laboratory tests above usually confirm a clinical suspicion that a patient is likely to be insulin and leptin resistant; the hemoglobin A1c provides an idea of how much carbohydrates the patient consumes. The lipid panel also provides important information, and physicians should pay special attention to triglycerides, since higher levels correlate with insulin resistance and carbohydrate consumption, low HDL levels, and small particles of LDL-p.44 It may be preferable to order a lipid particle analysis (nuclear magnetic resonance spectroscopy) because it provides more information about a patient's insulin resistance and provides a baseline to monitor therapy. Leptin, when combined with BMI, provides a better estimate of adiposity,42 and adiponectin levels can detect insulin resistance earlier.43
Body composition analysis is also important. As mentioned earlier, the most used methods are bioelectric impedance and DXA scanning.
Treatment Recommendations
In October 2013, the American Society of Bariatric Physicians published new guidelines, the Obesity Algorithm, to summarize evaluation and treatment approaches for overweight and obese patients.45 When evaluating patients with obesity, a helpful tool to assess patient readiness to be treated is the 5As.46,47
- Ask. Ask for permission to discuss body weight and readiness for change.
- Assess adiposity, BMI, waist circumference, and percent body fat. Explore drivers of weight gain and complications with excess weight.
- Advise the patient about the health risk of obesity, the benefits of modest weight loss, the need for a long-term plan, and treatment options.
- Agree on realistic weight-loss expectations and targets, behavioral changes, and specific details of treatment.
- Arrange/Assist in identifying and addressing barriers, providing resources, consulting with appropriate providers, and following up.
Nutritional Therapy
One of the most important parts of obesity treatment is nutrient deficiency. This is a key concept, since many people assume that since they overeat it is not possible to be nutrient deficient at the same time. With high insulin levels and fat storage, they live in a constant state of inflammation48 that depletes the patient of both macro- and micronutrients. There is no consensus on the best dietary approach for treating obesity or maintaining optimal health. Hundreds of diets are available in the literature and lay press. If possible, it is important to spend 30-45 minutes with each patient discussing what they should eat and what foods they should avoid.
Principles of Caloric Restriction
Based on the law of thermodynamics, a patient should be able to lose weight by creating a caloric deficit, either by increasing physical activity or restricting energy intake. The American Heart Association, the American College of Cardiology, and the Obesity Society 2013 guidelines give the following dietary recommendations:
- Prescribe 1200-1500 kcal/day for women and 1500-1800 kcal/day for men (kcal levels are usually adjusted for the individual's body weight).
- Prescribe a 500 kcal/day or 750 kcal/day energy deficit; or
- Prescribe an evidence-based diet that restricts certain food types (such as high-carbohydrate foods, low-fiber foods, or high-fat foods) to create an energy deficit by reduced food intake.
- Prescribe a very low calorie diet where a person consumes ≤ 800 calories per day; this should be attempted only under medical supervision.2
Caloric restriction has good short-term results, but compliance is an issue because of increased hunger and the body's ability to adjust the metabolic rate. In a state of deprivation, the body will try to hold on to every single calorie, making weight loss more difficult.
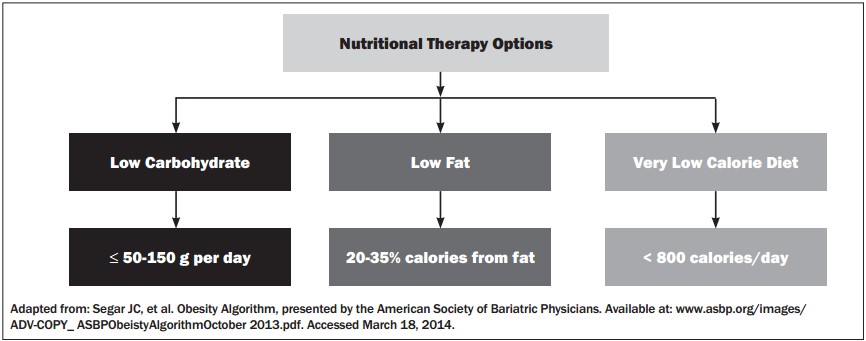
The 2013 Obesity Algorithm, developed by the American Society of Bariatric Physicians, includes algorithms for carbohydrate restriction (< 150-50 g of carbohydrates per day), fat restriction (< 20-35% of calories from fat), and very low calorie diets (< 800 calories per day). (See Figures 1, 2, and 3.)
Figure 1: American Society of Bariatric Physicians Algorithm: Carbohydrate Restriction
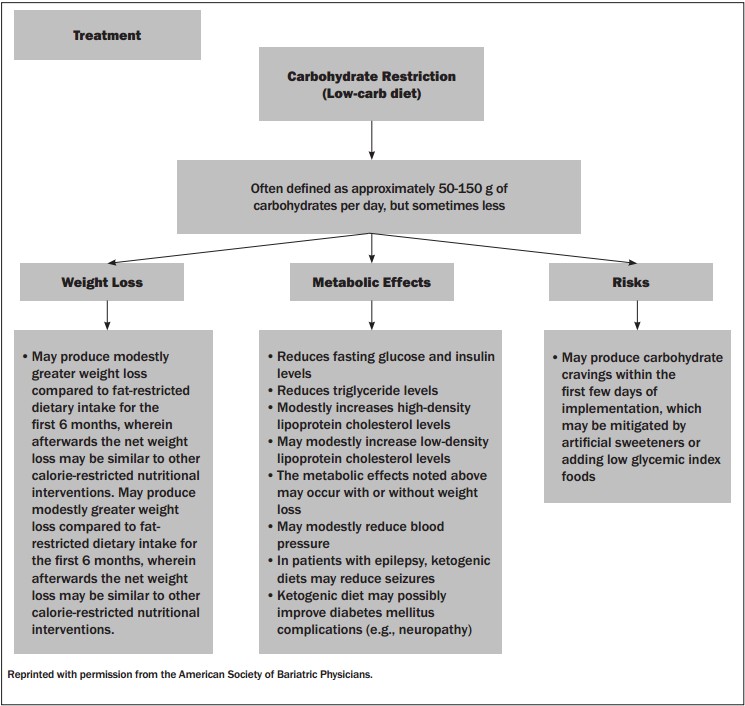
Figure 2: American Society of Bariatric Physicians Algorithm: Fat Restriction
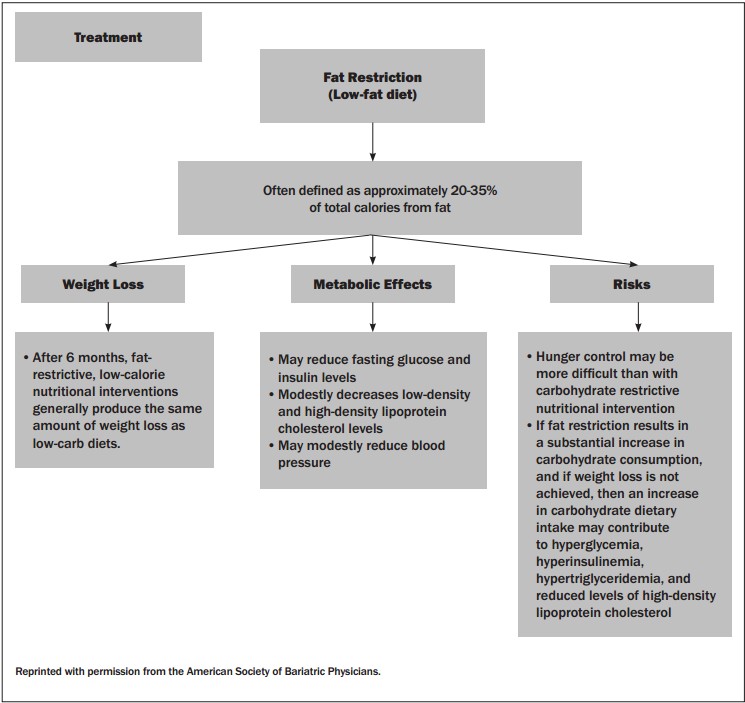
Figure 3: Characteristics of Nutritional Therapy Options for Obesity
Primary care physicians should be familiar with both low-carbohydrate and low-fat diets. Very low calorie diets have a high risk of complications, so they should be recommended under careful supervision.
Diets Based on Macro Composition
Diets are classified based on the percentage of macronutrients, and depend on the amount of protein, fat, and carbohydrates allowed. The A-Z trial compared the Atkins (low carbohydrate), Zone (40; 30; 30), LEARN (Lifestyle, Exercise, Attitudes, Relationships, and Nutrition, < 10% fat), and Ornish (strict low fat < 7%) diets. Patients with low carbohydrate intake lost more weight and experienced more favorable overall metabolic effects at 12 months when compared to the other three.49,50 (See Table 4.)
Table 4: Classification of Diets Based on Macro Composition
| Diet | Carbohydrates | Fat | Protein |
| Standard American | 45-65% | 20-35% | 10-35% |
| Low-carbohydrate | 20-40% | 40-60% | 10-35% |
| Very low carbohydrate | >20% | 60-80% | 10-35% |
| Low-fat | 50-70% | <15% | 10-35% |
| Zone diet | 40% | 30% | 30% |
| Ornish diet | 74% | 7% | 18% |
Low-carbohydrate diets may be effective alternatives to low-fat diets. Better lipid profiles are found on low-carbohydrate diets and better glycemic control on the Mediterranean diet.51 The Mediterranean diet, which follows the traditional cuisine of countries including Greece, Southern Italy, and Spain, usually is based on high consumption of olive oil, whole grains, legumes, fresh fruits, nuts, fish, and dairy in the form of cheese and yogurt; on moderate consumption of alcohol, especially wine; and on less consumption of red meats.52 In Northern Italy and other areas of the Mediterranean, the main fats used for cooking are lard and butter, with olive oil mainly used in salads and cold plates.53
The Paleolithic or ancestral diet is based on the idea that people should consume foods that are not processed and thus are closer to the natural state. This diet assumes that ancestors were hunters and gatherers who ate mainly organs of the animals that they hunted, nuts, vegetables, and roots, which are normally lower in carbohydrates and have a low glycemic index. They also consumed fruits seasonally, but did not consume cereals, grains, dairy, or any other processed food.54
Physical Activity
Approximately 60% of energy is metabolized at rest with no physical activity, which is the basal metabolic rate. Ten percent is the thermic effect of food (TEF), which is the energy required to metabolize food, so when people fast or avoid being fed they lose this important 10%. Twenty percent of the energy people use is the non-exercise, active thermogenesis (NEAT), which includes any single movement made during the day, such as walking to work, typing, performing yard work, undertaking agricultural tasks, and cooking. Only 10% of energy expenditure is related to exercise or programmed physical activity to promote fitness and endurance.55 (See Figure 4.)
Figure 4: Energy and Physical Activity

To increase energy expenditure, patients can increase muscular mass by doing resistance training or weight lifting, eating frequent meals to have the benefit of the TEF, staying more active, walking more, using the stairs, trying not to sit for long periods of time, and exercising.45
The benefits from exercise include increased release of growth hormone, upregulation of mitochondria, improved insulin sensitivity and glucose homeostasis, improved weight maintenance, and overall optimization of all systems.45,56,57
There is actually potential for weight gain with exercise, especially if the patient gains muscle mass and loses fat. Therefore, weight may not reflect the overall benefit of exercise, so physicians must measure fat percentages if concerned. With increased physical activity, physiologically induced hunger could potentially increase, making the patient more vulnerable to selecting the wrong kind of nutrients after exercise. Therefore, it is recommended that patients track their food at all times.
The 2013 American Society of Bariatric Physicians obesity algorithm suggests physicians assess patient motility first to establish the best exercise recommendations. Some patients with class III obesity (BMI = 40-49.9 kg/m2) often are essentially bed bound and unable to walk, so they may benefit from aquatic exercises, physical therapy, or arm exercises. Patients with limited mobility but who are able to walk may benefit from walking, swimming, and aquatic exercises. Patients with no limitations may choose from exercises listed in Table 5 based on their fitness and preference.45,58
Table 5: Types of Exercise58
| Types of Exercises | Examples |
| Aerobic | Running, zumba |
| Anaerobic | Weight lifting |
| Flexibility | Yoga, stretching |
|
High-intensity Interval Resistance Training |
Running at maximum effort for 100 feet, sets of 4 |
Behavior Therapy
As part of integrative care, each patient should be assessed individually for the need for behavioral interventions. Physicians should provide basic behavioral support, since patients often have associated depression and eating disorders and could be victims of discrimination and bullying. Techniques such as motivational interviewing are often used, and some clinics have a mental health specialist or a therapist as part of the team.
Pharmacologic Treatment of Obesity
One of the most critical parts of treating an obese patient is reviewing the medications that may be contributing to the problem. It is important that physicians become familiar with medications that alter how fat is stored, thermogenesis, and appetite regulation. (See Table 6.) These medications should be discontinued if possible or replaced by alternatives that do not promote weight gain.
Table 6: Medications that Promote Weight Gain or Fat Storage45
|
|
|
|
|
|
|
|
|
The most common medications that contribute to weight gain are sulfonylureas, beta-blockers, insulin, and corticosteroids. Some physicians may not be comfortable in discontinuing a patient's medications, but it is difficult to lose weight when one is constantly stimulating the storage of fat with external insulin or medications that promote the release of insulin, as all of these therapies are associated with hypoglycemia and increase in hunger.
Medications That Affect Appetite Regulation
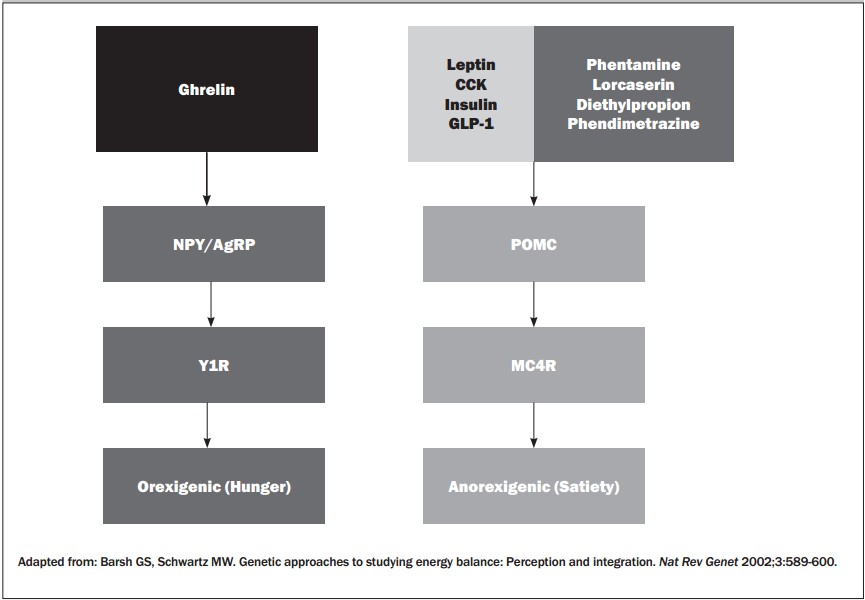
Basic appetite physiology. The hunger center is located in the arcuate nucleus of the hypothalamus. Two pathways interact with multiple systems to sense nutrition status, and these pathways receive orexigenic (hunger) and anorexigenic stimuli. When the stomach is empty it starts to release the hunger hormone ghrelin, which stimulates the arcuate nucleus to produce neuropeptide Y and agouti-related protein, producing the sensation of hunger.59,60 The only stimulus known to decrease ghrelin levels is food. It is not clear which macronutrient in food has a greater impact or if the release is simply caused by the distention of the gastric walls. The other pathway is stimulated by leptin, which is produced in the adipocytes. Leptin activates the proopiomelanocortin (POMC) neurons and the release of alpha-melanocyte-stimulating hormone that binds to the MC4 receptors, promoting satiety. It is in this last pathway where the appetite suppressants, sympathetic amines, and lorcaserin have their effects.59,60 (See Figure 5.)
Figure 5: Genetic Approaches to Studying Energy Balance
Sympathomimetic amines. Phentermine, diethylpropion, phendimetrazine, and benzphetamine are considered anorexigenic because they act by promoting satiety in the hypothalamus. They are considered schedule IV controlled substances by the Drug Enforcement Agency because of the theoretical possibility of addiction.61 The most common side effects are insomnia, increased anxiety, diarrhea, and palpitations. These medications can achieve a 3-5% weight loss. The typical dose is once daily in the morning to decrease the stimulant effect and prevent insomnia. It is recommended to start with the lowest dose and titrate upward depending on the results.62-64 Only the combined therapy of topiramate/phentermine (Qsymia) has been approved for long-term use with sustained weight loss of up to 10% at 1 year compared to placebo.
This drug combination appears to have a synergistic effect in promoting satiety and on food ingestion. It has been reasonably well tolerated, with adverse reactions (including paresthesia, dizziness, dysgeusia, insomnia, constipation, and dry mouth) occurring in 5% or more of study subjects. Long-term safety has not yet been established and it is contraindicated in pregnancy.65,66
Lorcaserin (Belviq) is a 5-HT2C receptor agonist that has serotonergic activity and acts like an anorectic by promoting satiety via POMC neurons at the hypothalamus. Most common adverse effects are upper respiratory infections, headache, and diarrhea. It is also a schedule IV drug and contraindicated in pregnancy. Patients on lorcaserin have been able to lose 5-10% of body weight with two major clinical studies lasting 1 and 2 years.67,68 Both lorcaserin and phentermine/topiramate are approved for prolonged therapy.
These medications were used in conjunction with lifestyle change recommendations.
Agents that interfere with absorption. Tetrahydrolipstatin or orlistat (Xenical) is a lipase inhibitor that decreases the absorption of fat. It is available in two strengths: 60 mg, which can be found as an over-the-counter medication, and 120 mg, which is the prescribed strength. Side effects include oily spotting per rectum, flatulence, diarrhea, and decreased absorption of other nutrients, especially the liposoluble vitamins A, D, E, and K. Patients on Orlistat can lose ≥ 3% of body weight at 1 year.69
Surgical Treatment for Obesity
Although surgical treatment for obesity is currently the most effective therapy, it is reserved for patients with class II obesity with comorbidities, such as hypertension, type 2 diabetes, and sleep apnea, and patients with class III obesity. The types of obesity surgeries are listed in Table 7.
Table 7: Surgical Treatment of Obesity
| Types of Surgeries | |
| Purely Restrictive |
Laparoscopic gastric banding Sleeve gastrectomy |
| Mostly Restrictive | Roux-en-Y gastric bypass |
| Mostly Malabsortive |
Duodenal switch Biliopancreatic diversion Long loop gastric bypass |
The 2013 American Society of Bariatric Physicians Obesity Algorithm suggests careful selection of surgical candidates. To be considered a surgical candidate, patients must have tried reasonable attempts to improve their health, received expert opinion from an obesity medicine specialist, and demonstrated the ability to maintain a good lifestyle after the surgery. Careful preoperative evaluation with a specialized team is also recommended.45
The most popular surgical options are laparoscopic adjustable gastric banding, sleeve gastrectomy, and Roux-en-Y gastric bypass. Each option carries its own risks and consequences of disrupting the anatomy and physiology, and patients should be aware of short- and long-term complications of each procedure.45,70 (See Table 8.) There is little doubt as to the effectiveness and increasing safety of bariatric surgery. Approximately 150,000 bariatric surgeries are performed annually in the United States with mortality rates of 0.1% and overall risk of major complications of 4%.
Table 8: Complications of the Surgical Treatment of Obesity
| Acute Complications | Chronic Complications | |
|
Laparoscopic adjustable gastric banding |
Band too tight, dysphagia, hemorrhage, infection, pneumonia, deep vein thrombosis, and death |
Weight regain or no loss, band slippage, erosion, infection, esophageal dilatation, depression, nutrient deficiency |
| Sleeve gastrectomy |
GI obstruction, bleeding, anastomotic leaks, infection, pulmonary embolism,deep vein thrombosis, dehydration,and death |
Weight regain, marginal ulcer, reactive hypoglycemia (dumping syndrome), fistula, anastomotic leak, gallstones, vitamin deficiency, bacterial overgrowth, depression |
| Gastric bypass | GI obstruction, hemorrhage, anastomotic leaks, infection, arrhythmias, pneumonia,venous thromboembolism,rhabdomyolisis, and death |
Weight regain, marginal ulcers, dumping syndrome, gallstones, calcium deficiency, protein caloric malnutrition and multiple vitamin deficiencies, depression |
Adapted from: Segar JC, et al. Obesity Algorithm, presented by the American Society of Bariatric Physicians. Available at: www.asbp.org/images/ ADV-COPY_ ASBPObeistyAlgorithmOctober 2013.pdf. Accessed March 18, 2014.
Intense Behavioral Therapy for Obesity
Since November 2011, the Centers for Medicare and Medicaid Services has recognized the treatment for obesity as an important part of preventive care and covers 22 intense behavioral therapy (IBT) sessions in a 12-month period with no copay. This service, provided by primary care offices, includes:71
- One face-to-face visit every week for the first month;
- One face-to-face visit every other week for months 2-6; and
- One face-to-face visit every month for months 7-12, if the beneficiary meets the 3 kg (6.6 pounds) weight loss requirement during the first 6 months.
The HCPCS Code for IBT for obesity is G0447.
Treatment Options for Patient M
Taking a complete history, with special emphasis on dietary and sleep habits, usually takes about 30 minutes. During the interview, the team discovered that the patient drinks 4-5 regular soft drinks a day and his preferred snacks are cookies and chips. He does not cook and eats most meals outside his home. If he eats breakfast, it usually consists of a bowl of cereal (24 g of sugar), two slices of bread (24 g of sugar), and a glass of orange juice (28 g of sugar). He believes this a healthy option. Between breakfast and dinner, he has one portion of snack and for lunch he usually has some kind of sandwich or burger (bread 24 g of sugar) and the same thing at dinner. He drinks 4-5 cans of soda (a 32-ounce soft drink contains about 39 g of sugar). It is estimated he consumes 319 g of carbohydrates per day. During a second 30-minute session of motivational interviewing exploring the patient's readiness to change, when asked why he eats so much sugar, he responded that he was not aware he consumed that much. He stated that nobody has told him that high sugar consumption can lead to serious health consequences.
He was provided with several treatment options. First, since he is diabetic, he was asked to choose between a low-fat diet or a low-carbohydrate diet. The benefits and concerns with both types of treatment were explained. He also was given the option to take appetite suppressants as part of the treatment, which included phentermine alone, phentermine/topiramate combination, or lorcaserin. This patient would be a good candidate for surgical treatment if he is not able to improve with lifestyle changes.
Physicians must realize that medications also can be a big part of the problem. It was decided to optimize his diabetes treatment to decrease insulin resistance and not promote it. To do this, glipizide was discontinued and his metformin was increased to 1000 mg po twice a day. The patient could benefit from incretin mimetic therapy and he was started on liraglutide 0.6 mg subcutaneous daily.
The patient declined appetite suppressants because he was concerned about the potential side effects, including insomnia and diarrhea.
The patient decided to try a low-carbohydrate diet because he did not like the idea of counting calories and he was a meat eater. He agreed to be followed every 2 weeks for the first month and then monthly thereafter. The expectation was for this patient to be able to lose 1 pound per week. The results of the treatment are shown in Table 9.
Table 9: Results of Treatment for Patient "M"
| Sept | Oct | Nov | Dec | Jan | Feb | Mar | April | |
| Weight | 282 | 272 | 264 | 254 | 258 | 252 | 251 | 247 |
| HgbA1C | 11.3 | 8.6 | 6.5 | |||||
| Body Fat% | 38% | 32% |
In summary, this patient had a 12% weight loss of 35 pounds over 7 months and experienced improvement in his diabetic control and overall health. His HgbAIC decreased from 11.3% to 6.5%, and blood pressure also improved over time.
Conclusions and Recommendation
The management of obesity is multidisciplinary, but it is expected that primary care providers provide the core responsibility for orchestrating care. Obesity medicine as a specialty is growing, and more and more providers are able to provide this type of care. There still are many unanswered questions, but recognition of obesity as a disease will open new frontiers.
If consultative help is needed, just like for any condition, primary care physicians should consider referring patients to physicians who have specialized in obesity medicine.72
In my personal experience, obesity is a nutrient deficiency state.73,74 Just restricting food and asking patients to exercise more is not rational, unless the nutrition portion is addressed first. There is no evidence that removing junk food from one's diet is healthy, but it makes perfect sense. It is sad to admit as a physician that guidelines are being built by the pharmaceutical industry and the manufacturers of foods that promote obesity. The only way to overcome this epidemic is with proper nutrition, real food, and effective physician engagement.
References
- American Medical Association. Reference Committee on Constitution and Bylaws. Resolution 420, June 2013. Available at: www.ama-assn.org/resources/doc/sps/a13-highlights.pdf. Accessed March 18, 2014.
- Mechanick JI, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient — 2013 update: Cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and American Society for Metabolic & Bariatric Surgery. Surg Obes Relat Dis 2013;9:159-191.
- Merriam Webster Online Dictionary. Obesity. Available at: www.merriam-webster.com/dictionary/disease. Accessed March 18, 2014.
- Bray GA, Bouchard C, eds. Handbook of Obesity. 3rd Edition. London: Informa Healthcare; 2008.
- Flegal KM, et al. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. JAMA 2012;307:491-497.
- Zamosky L. The obesity epidemic. Medical Economics Feb. 25, 2013.
- Barlow SE, Expert Committee. Expert committee recommendations regarding the prevention, assessment and treatment of child and adolescent overweight and obesity: Summary report. Pediatrics 2007;120(Suppl 4):S164-S192.
- Shah NR, Braverman ER. Measuring adiposity in patients: The utility of body mass index (BMI), percent body fat, and leptin. PLoS One 2012;7:e33308.
- Obesity: Preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 2000;894:i-xii, 1-253.
- National Institutes of Health. National Heart, Lung, and Blood Institute. NHLBI Obesity Education Initiative. The Practical Guide. Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. October 2000.
- Janssen I, et al. Waist circumference and not body mass index explains obesity-related health risk. Am J Clin Nutr 2004;79:379-384.
- Simpson JA, et al. A comparison of adiposity measures as predictors of all-cause mortality: The Melbourne Collaborative Cohort Study. Obesity 2007;15:994-1003.
- Koster A, et al. Waist circumference and mortality. Am J Epidemiol. 2008;167:1465-1475.
- American Council on Exercise. ACE Lifestyle & Weight Management Consultant Manual: The Ultimate Resource for Fitness Professionals. American Council on Exercise; 2009.
- Bioelectrical impedance analysis in body composition measurement: National Institutes of Health Technology Assessment Conference Statement. Am J Clin Nutr 1996;64(3 Suppl):524S-532S.
- Heymsfield SB, et al. Evaluation of total and regional adiposity. In: Handbook of Obesity. Bray GA, Bouchard C, eds. New York: Marcel Dekker; 2004:33.
- Procaccini C, et al. Leptin as an immunomodulator. Mol Aspects Med 2012;33:35-45.
- Lavens D, et al. Review: Negative regulation of leptin receptor signaling. Eur Cytokine Netw 2006;17:211-219.
- Underwood PC, Adler GK. The renin angiotensin aldosterone system and insulin resistance in humans. Curr Hypertens Rep 2013;15:59-70.
- Feinman RD, Fine EJ. "A calorie is a calorie" violates the second law of thermodynamics. Nutr J 2004,3:9.
- Kral TVE, Faith MS. Influences on child eating and weight development from a behavioral genetics perspective. J Pediatr Psychol 2009;34:596-605.
- Mergen M, et al. A novel melanocortin 4 receptor (MC4R) gene mutation associated with morbid obesity. J Clin Endocrinol Metab 2001;86:3448.
- Lubrano C, et al. Obesity and metabolic comorbidities: Environmental diseases? Oxid Med Cell Longev 2013;2013:640673.
- Alonso-Magdalena P, et al. Pancreatic insulin content regulation by the estrogen receptor ER alpha. PLoS ONE 2008;3:e2069.
- Miyawaki J, et al. Perinatal and postnatal exposure to bisphenol a increase adipose tissue mass and serum cholesterol level in mice. J Atheroscler Thromb 2007;14:245-252.
- Nuttall FQ, et al. Effect of protein ingestion on the glucose and insulin response to a standardized oral glucose load. Diabetes Care 1984;7:465-470.
- Kahn BB. Lilly lecture 1995. Glucose transport: pivotal step in insulin action. Diabetes 1996;45:1644-1654.
- Magkos F, et al. Metabolic actions of insulin in men and women. Nutrition 2010;26:686-693.
- Gautron L, Elmquist JK. Sixteen years and counting: An update on leptin in energy balance. J Clin Invest 2011;121:2087-2093.
- Moran A, et al. Differentiation of glucose toxicity from beta cell exhaustion during the evolution of defective insulin gene expression in the pancreatic islet cell line, HIT-T15. J Clin Invest 1997;99:534-539.
- Lack of sleep is linked to obesity, new evidence shows. ScienceDaily Available at: http://www.wiley.com/WileyCDA/PressRelease/pressReleaseId-103225.html. Accessed March 18, 2014.
- Knutson KL. Does inadequate sleep play a role in vulnerability to obesity? Am J Hum Biol 2012;24:361-371.
- Speiser PW, et al. Childhood obesity. J Clin Endocrinol Metab 2005;90:1871-1887.
- Reinehr T, et al. Definable somatic disorders in overweight children and adolescents. J Pediatr 2007;150:618-622.
- Cohen DE, Fisher EA. Lipoprotein metabolism, dyslipidemia, and nonalcoholic fatty liver disease. Semin Liver Dis 2013;33:380-388.
- EMF Exposures in the Womb Can Lead to Childhood Obesity. Microwave News July 27, 2012. Available at: http://microwavenews.com/news-center/emf-exposures-womb-can-lead-childhood-obesity. Accessed March 18, 2014.
- Joyce SA, Gahan CG. The gut microbiota and the metabolic health of the host. Curr Opin Gastroenterol 2014;30:120-127.
- Martinez FD. The human microbiome. Early life determinant of health outcomes. Ann Am Thorac Soc 2014;11(Suppl 1):S7-S12.
- Rajpal DK, Brown JR. Modulating the human gut microbiome as an emerging therapeutic paradigm. Sci Prog 2013;96(Pt 3):224-236.
- Cotillard A, et al. Dietary intervention impact on gut microbial gene richness. Nature 2013;500:585-588. Erratum in: Nature 2013;502:580.
- Sirmans SM, Pate KA. Epidemiology, diagnosis, and management of polycystic ovary syndrome. Clin Epidemiol 2013;6:1-13.
- Ruhl CE, et al. Body mass index and serum leptin concentration independently estimate percentage body fat in older adults. Am J Clin Nutr 2007;85:1121-1126.
- Semple RK, et al. Plasma adiponectin as a marker of insulin receptor dysfunction: Clinical utility in severe insulin resistance. Diabetes Care 2008;31:977-979.
- Miccoli R, et al. Insulin resistance and lipid disorders. Future Lipidology 2008;3:651-664.
- Segar JC, et al. Obesity Algorithm, presented by the American Society of Bariatric Physicians. Available at: www.asbp.org/images/ ADV-COPY_ ASBPObeistyAlgorithmOctober 2013.pdf. Accessed March 18, 2014.
- Vallis M, et al. Clinical review: Modified 5 As: Minimal intervention for obesity counseling in primary care. Can Fam Physician 2013;59:27-31.
- Alexander SC, et al. Do the five A's work when physicians counsel about weight loss? Fam Med 2011;43:179-184.
- Cottam DR, et al. The chronic inflammatory hypothesis for the morbidity associated with morbid obesity: Implications and effects of weight loss. Obes Surg 2004;14:589-600.
- Gardner CD, et al. Micronutrient quality of weight-loss diets that focus on macronutrients: Results from the A TO Z study. Am J Clin Nutr 2010;92:304-312.
- Gardner CD, et al. Comparison of the Atkins, Zone, Ornish, and LEARN diets for change in weight and related risk factors among overweight premenopausal women: The A TO Z Weight Loss Study: A randomized trial. JAMA 2007;297:969-977. Erratum in: JAMA 2007;298:178.
- Shai I, et al. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N Engl J Med 2008;359:229-241.
- Willett WC. The Mediterranean diet: Science and practice. Public Health Nutr 2006;9:105-110.
- Albertini M. Guida all'Italia Gastronomica. Touring Club Italiano; 1984.
- Lindeberg S. Palaeolithic diet ("stone age" diet). Scand J Nutr 2005;49:75-77.
- Levine JA. Nonexercise activity thermogenesis (NEAT): Environment and biology. Am J Physiol Endocrinol Metab 2004;286:E675-E685.
- Godfrey RJ, et al. The exercise-induced growth hormone response in athletes. Sports Med 2003;33:599-613.
- Warburton DE, et al. Health benefits of physical activity: The evidence. CMAJ 2006;174:801-809.
- Bupa Health Information Team. Walking and health. October 2012. Available at: http://www.bupa.co.uk/individuals/health-information/directory/w/walking-health. Accessed March 18, 2014.
- Vetter ML, et al. Behavioral and pharmacologic therapies for obesity. Nat Rev Endocrinol 2010;6:578-588.
- Barsh GS, Schwartz MW. Genetic approaches to studying energy balance: Perception and integration. Nat Rev Genet 2002;3:589-600.
- International Narcotics Control Board. List of Psychotropic Substances under International Control. Green List 24th ed, May 2010. Annex to the annual statistical report on psychotropic substances. Available at: http://www.upu.int/uploads/tx_sbdownloader/listCustomsPsychotropicSubstancesUnderInternationalControlEn.pdf. Accessed March 18, 2014.
- Nelson DL, Gehlert DR. Central nervous system biogenic amine targets for control of appetite and energy expenditure. Endocrine 2006;29:49-60.
- Cabrerizo García L, et al. Update on pharmacology of obesity: Benefts and risks. Nutr Hosp 2013;28(Supl 5):121-127.
- Ioannides-Demos LL, et al. Pharmacotherapies for obesity: Past, current, and future therapies. J Obes 2011;2011:179674.
- Allison DB, et al. Controlled-release phentermine/topiramate in severely obese adults: A randomized controlled trial (EQUIP). Obesity (Silver Spring) 2012;20:330-342.
- Smith SM, et al. Phentermine/topiramate for the treatment of obesity. Ann Pharmacother 2013;47:340-349.
- Smith SR, et al. Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med 2010;363:245-256.
- Fidler MC, et al. A one-year randomized trial of lorcaserin for weight loss in obese and overweight adults: The BLOSSOM trial. J Clin Endocrinol Metab 2011;96:3067-3077.
- Torgerson JS, et al. XENical in the prevention of diabetes in obese subjects (XENDOS) study: A randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care 2004;27:155-161.
- DeMaria EJ. Bariatric surgery for morbid obesity. N Engl J Med 2007;356:2176-2183.
- Department of Health and Human Services. Centers for Medicare and Medicaid Services. Intensive Behavioral Therapy (IBT) for Obesity. ICN 907800 August 2012. Available at: http://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/downloads/ICN907800.pdf. Accessed March 18, 2014.
- American Society of Bariatric Physicians. Available at: www.aspb.org. Accessed March 18, 2014.
- Markovic TP, Natoli SJ. Paradoxical nutritional deficiency in overweight and obesity: The importance of nutrient density. Med J Aust 2009;190:149-151.
- Seneff S. The Obesity Epidemic: is the Metabolic Syndrome a Nutritional Deficiency Disease? October 9, 2009. Available at: http://people.csail.mit.edu/seneff/obesity_epidemic.html. Accessed March 18, 2014.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
