The Recognition and Management of Diabetic Ketoacidosis in Children
Authors: Richard J. Scarfone, MD, Assistant Professor of Pediatrics, University of Pennsylvania School of Medicine, Philadelphia, PA; Andrea Kelly, MD, Assistant Professor of Pediatrics, University of Pennsylvania School of Medicine; Philadelphia, PA.
Peer Reviewer: Gary R. Strange, MD, MA, FACEP, Professor and Department Head, Department of Emergency Medicine, University of Illinois, Chicago.
Although diabetes mellitus (DM) is second only to asthma as the most prevalent chronic disease of childhood, the literature has very few comprehensive reviews of diabetic ketoacidosis (DKA), the most frequent cause of death in children with diabetes. The importance of an early diagnosis and appropriate management should not be underestimated.
Emergency department (ED) physicians must have a clear understanding of the pathophysiology that is present in a patient with DKA, especially in a young child with new-onset DM. An understanding of the unique needs of the child in DKA is critical for appropriate decision-making to ensure an optimal outcome for the patient. Children with DKA have the potential for different complications than adults and are at higher risk for cerebral edema. Management strategies should focus on correction of fluid deficits, but not overhydration, and correction of acidosis, but not equate acidosis with dehydration. The authors provide a focused review for the ED physician for recognition and management of a child with DKA, with special attention to potentially serious complications. — The Editor
Introduction
Approximately two of every 1,000 children in the United States have Type 1 DM;1 approximately 13,000 new cases are diagnosed each year.2,3 DM is second only to asthma as the most prevalent chronic disease in childhood.4,5 The prevalence among boys and girls is equal, but white children commonly are more afflicted compared with African-American children.1
DKA, defined here as hyperglycemia (serum glucose > 200 mg/dL), ketonemia, and acidosis (pH < 7.30 or HCO3 < 15 mEq/L), is the most common cause of death for children with DM.
A recent report from the United Kingdom found that, during a seven-year span, 83% of all deaths in diabetic patients younger than 20 years were due to DKA.6 The mortality associated with DKA is predominantly related to the development of cerebral edema. Symptomatic cerebral edema with signs of increased intracranial pressure (ICP) occurs in approximately 0.31% of all episodes of DKA.
This article reviews the diagnosis and emergency management of children with DKA and current strategies to minimize potential complications.
Etiology of Diabetes Mellitus
The pathogenesis of DM involves heterogeneous defects in either insulin secretion and/or action that lead to hyperglycemia. To better reflect the various etiologies of DM, the American Diabetes Association recently reclassified most cases as either Type 1 or Type 2.7 The hallmark of Type 1 DM, previously referred to as juvenile or insulin-dependent diabetes, is an absolute deficiency in insulin secretion. With few exceptions, Type 1 DM arises from autoimmune destruction of pancreatic beta cells; hyperglycemia occurs when less than 20% of insulin secretory capacity remains.7 Type 2 DM is a polygenic disorder arising from insulin resistance and insulin deficiency.
Evidence for autoimmunity in Type 1 DM is convincing. Type 1 DM occurs with increased frequency in individuals with other autoimmune diseases such as Hashimoto’s thyroiditis and Grave’s disease.8 Type 1 DM also may co-exist with other autoimmune-mediated diseases such as autoimmune thyroiditis, Addison’s disease, or both in Type II polyglandular autoimmune disease. Inheritance of certain histocompatability antigens (HLA), such as HLA DR3 or DR4 confers a 2-3 fold increase in DM risk; inheritance of both increases the risk 7-10 fold.8 At diagnosis, most individuals are found to have one or more antibodies directed against the beta cell. Rosenbloom reported that 19 of 31 first-degree relatives with three antibodies directed at the beta cell developed DM during a five-year period.9 Also, lymphocytic infiltrates of islet cells are found in Type 1 DM; as inflammation progresses, islets are replaced with hyaline and scar tissue.8
Among patients with DM who have an identical twin, only 30-50% of twins also are affected. Therefore, in addition to inheritance, there must be other etiological factors. Evidence supports an environmental component: disease incidence in-creases as the distance from the equator increases and after viral epidemics. One theory encompassing many of these factors is that environmental triggers in genetically predisposed individuals lead to a production of auto-antibodies, T-cells, and cytokines, causing progressive islet inflammation and insulin deficiency.8
Once considered to be a disease of adulthood, Type 2 DM is emerging as an important entity in children and adolescents, accounting for 8-45% of newly diagnosed cases.10-13 This rate of increase may reflect shifts in population; Type 2 DM affects primarily Americans of African, Hispanic, Asian, and American Indian descent.11 Increases in Type 2 DM in children and adults parallel the obesity epidemic. In fact, at diagnosis, most children with Type 2 DM are obese and undergoing puberty—a period of enhanced insulin resistance.10-13 Initially, children with Type 2 DM may be indistinguishable from those with Type 1; at presentation, as many as one-third have ketonuria, and 5-25% have DKA.10
Pathophysiology of DKA
The biochemical and clinical disturbances found in children with DKA are manifestations of loss of normal hormonal control of carbohydrate, protein, and fat metabolism. Deficient insulin secretion coupled with unrestrained counter-regulatory hormone activity leads to the metabolic abnormalities seen in DKA.
Insulin stimulates glucose uptake by muscle, liver, and fat (See Figure 1 ). It promotes anabolism (i.e., glycogen, protein, and lipid synthesis), while suppressing catabolism (i.e., glycogenolysis, proteolysis, gluconeogenesis, lipolysis, fatty acid oxidation, and ketogenesis). In insulin sufficient states, the increase in blood glucose that occurs following caloric intake stimulates insulin secretion by the beta cell, thereby maintaining blood glucose in a normal range. In the fasting state, insulin is secreted at low levels, allowing regulated hepatic glucose production via glycogenolysis and gluconeogenesis. Further relaxation of insulin’s suppressive effects upon lipolysis, fatty acid oxidation, and ketogenesis allows ketones to serve as an alternative fuel to glucose, a normal fasting adaptation.8,14
Figure 1. Pathophysiology of DKA
Maintenance of fuel homeostasis during fed and fasting states demands that a delicate balance be preserved between the blood glucose-lowering effects of insulin and the blood glucose-enhancing effects of the counter-regulatory hormones: glucagon, cortisol, growth hormone, and catecholamines. (See Figure 1.) In DKA, these normal adaptations to fasting are unregulated. With mild insulin deficiency, post-prandial or fasting hyperglycemia occurs. When blood glucose concentrations exceed the renal threshold (180 mg/dL), glucosuria ensues leading to polyuria, a compensatory polydipsia, and nocturia. Excess urinary losses lead to dehydration as do compensatory hyperpnea/tachypnea, ketoacid-induced emesis, and decreases in oral intake. With significant dehydration, the glomerular filtration rate (GFR) decreases, causing impaired glucose and ketone disposal, aggravating hyperglycemia and ketoacidosis.8,14
Continuation of the insulin deficient state worsens hyperglycemia and permits unrestrained lipolysis, fatty acid oxidation, and ketogenesis. Acidemia initially is prevented by activation of the respiratory and renal buffering systems. Unremitting ketogenesis leads to an increased anion gap. When dehydration occurs, regeneration of bicarbonate and excretion of ketoacids by the kidney are compromised, exacerbating the anion gap acidosis. This acidosis primarily arises from elevated concentrations of the ketones beta hydroxybutyrate and acetoacetic acid. To a much lesser degree, elevated free fatty acids, arising from enhanced lipolysis, and lactate, a consequence of poor tissue perfusion, also contribute to the acidosis.8,14
Potential Complications
Among the complications associated with DKA (See Table 1), symptomatic cerebral edema with brain herniation is the most feared. (See Table 2.) Many children with DKA will have asymptomatic subclinical brain swelling before treatment, but few progress to significantly altered mental status or brain herniation.15 Increasing illness severity is clearly a risk factor for brain herniation. In one series encompassing 12 years, of nine children suffering brain herniation as a DKA complication, eight had an initial serum pH of 7.1 or less.15 Glaser, et al reported that among 7,000 children hospitalized with DKA during a 15-year span, approximately 1% developed clinically significant cerebral edema.16 Among those with cerebral edema, 21% died and 21% had permanent neurological sequelae.16 Further, among those patients with cerebral edema, greater neurological depression, a high initial serum urea nitrogen concentration, and intubation with hyperventilation were associated with poorer outcomes.17
Table 1.
Causes of Morbidity and Mortality
with Diabetes Mellitus

Table 2. Complications from DKA and Its Therapy

Cerebral edema accounts for the majority of deaths due to DKA.6,18 Classically, symptomatic cerebral edema develops within 4 to 12 hours after therapy has been started, often in a child who had been showing signs of improvement, although subclinical cerebral edema has been described in children with DKA before the initiation of therapy.6,16,19,20 The demographic factors that have been reported to be associated with the development of cerebral edema include new-onset DM, younger age, and longer duration of symptoms. Other potential risk factors for the development of cerebral edema include attenuated rise in measured serum sodium concentration during therapy,27 severity of acidosis, bicarbonate treatment, greater hypocapnia, and elevated serum urea nitrogen level at presentation. (See Table 3.)
Table 3. Risk Factors for Cerebral Edema

Clinical signs that reflect the development of increased ICP may include headache, vomiting, depressed mental status, bradycardia, and irregular respirations. (See Table 4.)
Table 4.
Clinical Signs that Reflect
the
Development of Increased ICP

Dysrhythmias also may occur in children with DKA, usually secondary to metabolic derangements including hyponatremia, hypernatremia, hypokalemia, hyperkalemia, hypoglycemia, and hypocalcemia.
Prevention
Approximately 20-40% of patients with DM present with DKA at initial diagnosis.22,23 The key to the prevention of DKA is early recognition and education of patients, caretakers, and physicians.8 A recent study highlighted this fact by showing that education of physicians and school personnel on the signs and symptoms of DM reduced the number of new cases that presented in DKA.24
In the setting of established DM, an intercurrent illness can precipitate DKA. DM management during periods of illness can be challenging for children and families. During those times, increased secretion of counter-regulatory hormones leads to an insulin-resistant state. However, poor oral intake, emesis, and malabsorption can lead to hypoglycemia. A common mistake in managing a child with DM is to assume an absence of urinary ketones in the setting of a normal or low blood glucose level. An even more common error is to withhold insulin because of decreased oral intake or emesis; this may lead to significant ketosis.
Providing specific written guidelines for DM management during intercurrent illnesses (i.e, sick-day rules) as well as telephone access to medical personnel have been shown to decrease the number of DKA admissions due to intercurrent illnesses.25
Recurrent DKA typically occurs in the setting of deliberate insulin omission in children with significant psychosocial stressors. The fact that 20% of pediatric DM patients account for 80% of all DKA admissions26 underscores the magnitude of the problem of recurrent DKA. ED physicians should be familiar with the risk factors for recurrent DKA, which include adolescence, female gender, longstanding diabetes, lower socioeconomic background, and pre-existing psychopathology.26-28
Insulin pumps are becoming more prevalent in long-term DM management, and ED physicians should be familiar with them. Because continuous insulin delivery systems use only short-acting insulin, their failure can lead quickly to DKA. If the blood glucose level is elevated and does not normalize after a therapeutic insulin bolus, the pump site may need to be changed. If the hyperglycemia persists and urinary ketones develop, pump failure should be assumed. However, when a pump user has an intercurrent illness necessitating additional insulin, the basal rate can be increased and boluses delivered every 2-4 hours. Such interventions will depend upon prior planning with medical personnel who will help guide appropriate bolus doses.29
Prevention of DKA remains an elusive goal. The Diabetes Control and Complications Trial followed 1,441 adolescents and young adults for more than six years.30 Patients were randomly assigned to intensive therapy (use of a pump or three or more daily insulin injections and blood glucose monitoring four times daily) or conventional therapy (one or two daily insulin injections and urinary or blood glucose monitoring just once daily). Although determining rates of DKA was not the primary aim of the trial, in the conventional therapy group DKA occurrences among adolescents were significantly greater than that of adults.31 A recent reappraisal of those data suggests that poor adherence with insulin therapy is the primary factor leading to a greater incidence of DKA in this population.32
Clinical Presentation
DKA is defined as ketoacidosis that almost always is associated with varying degrees of dehydration, depending on the duration and severity of the symptoms and the length and degree of diuresis. For previously undiagnosed children, there may be a history of polydipsia and polyuria for several weeks before they come to medical attention. The presence of polyuria helps to distinguish them from children with other causes of dehydration such as gastroenteritis. However, children known to have DM typically have briefer prodromes before becoming seriously ill with DKA. For such children, the history should focus on recent insulin administration, dietary intake, and illnesses marked by fever, vomiting, or diarrhea. Astute ED physicians will assess DKA patients for underlying serious illnesses such as appendicitis or pneumonia, which may precipitate the metabolic derangements. Additionally, pregnancy testing of adolescent females should be considered.
The physical assessment should begin with the patient’s vital signs and a brief neurologic examination. Vital signs will reflect the patients’ attempts to compensate for the dehydration and acidosis, and usually include tachycardia and the classic deep hyperpneic (Kussmaul) respirations. This increased work of breathing in children with clear breath sounds is an indication that DKA may be the underlying problem. Another clinical clue for the diagnosis would be the smell of ketones on the breath. Carefully assess the child clinically for the degree of dehydration with attention to the child’s pulse rate, peripheral pulses, capillary refill, and mucous membranes. The blood pressure is typically normal for the child’s age; uncompensated shock with hypotension is an unusual occurrence in children with DKA. Clinicians should remember that because DKA results in a hyperosmolar dehydration, intravascular fluids are spared at the expense of the extravascular space and the degree of dehydration may be underestimated. The mental status may be normal, but frequently patients are somnolent or even obtunded. Warning signs and symptoms of symptomatic cerebral edema include irregular respirations, inappropriate slowing of heart rate and/or rising blood pressure, decreased oxygen saturation, worsening mental status, such as increased agitation or obtundation, abnormal pupillary responses, cranial nerve palsies, headache, or recurrent vomiting. In addition, a recent article describes incontinence as one of the "major criteria" in the detection of cerebral edema.33
Differential Diagnosis
Children known to have DM who present with ill appearance and dehydration should not pose a diagnostic dilemma. Laboratory tests revealing hyperglycemia, ketonemia, and metabolic acidosis will support the clinical suspicion of DKA. More challenging are children previously not known to have DM who present with significant dehydration or an altered mental status. The differential diagnosis will include other common causes of volume loss, such as hypovolemia, sepsis and cardiac etiologies, as well as intoxications, acute adrenal insufficiency, and hyperosmolar non-ketotic coma. A simple bedside test to determine the serum glucose level should point the clinician immediately toward the correct diagnosis.
Patient Management
Protocols for the management of DKA vary among institutions reflecting some of the controversies regarding optimal therapy and a lack of conclusive evidence to guide certain treatment practices. One study identified substantial differences in the management of pediatric DKA among physicians of different specialties.34 The treatment strategy presented is one based on available evidence and developed by ED physicians at the Children’s Hospital of Philadelphia and frequently is re-assessed and revised based on scientific data; others may manage children slightly differently than what is outlined here. Additionally, this strategy is very similar to a recently published consensus statement on DKA in children and adolescents.21
Initial Stabilization and Laboratory Testing
Children with DKA are among the most seriously ill, non-trauma patients that ED physicians treat. As with management of other critically ill children, ED physicians must first focus attention on airway patency, ventilation, and circulatory status. It is uncommon for children with DKA to require mechanical ventilation; an exception might be deeply obtunded patients without intact gag reflexes. Kussmaul respirations, typically present when the PaCO2 level is below 20 torr, allow for exhalation of carbon dioxide as a means of compensating for the metabolic acidosis. Regarding dehydration, in one study of 219 episodes of DKA among children and young adults, the mean fluid deficit was 7.4%, as assessed by average post-treatment weight gain;35 fluid resuscitation strategies will be outlined in the next section. Close attention must be made to both renal and cerebral function with frequent neurological assessments being performed. The recommended initial laboratory testing is outlined in Table 5.
Table 5. Initial Laboratory and Diagnostic Testing

Fluid Volume
Much controversy has surrounded the issue of initial fluid resuscitation in the treatment of DKA. Some studies have suggested that patients given intravenous fluids rapidly or at high volume were more likely to develop cerebral edema and brain herniation.15,35,36 One of these studies, a retrospective series of 42 children with cerebral edema, determined that the rate of fluid administration was inversely correlated with time to herniation and that children who developed cerebral edema had higher rates of intravenous fluids given within the first 4 hours of therapy compared with those without this complication.15 However, small studies such as this one simply may have identified initial fluid resuscitation as a marker for disease severity. That is, it is probable that patients who are clinically more worrisome at initial presentation are more likely to develop cerebral edema; these are the same patients likely to be treated aggressively with fluids.
Results from a recent multicenter study seemed to decrease the concern about initial fluid rate.16 Here, 61 children hospitalized with DKA and cerebral edema were matched with 174 children who had not developed this complication. An attempt was made to control for degree of illness by matching initial serum glucose concentration and venous pH. In this retrospective study, it was not possible to control for other factors reflecting illness severity such as mental status or capillary refill time. Among the risk factors not found to be associated with cerebral edema were rates of infusion of intravenous fluids, sodium, or insulin, or rate of decrease in serum glucose concentration. Because cerebral edema is a relatively uncommon occurrence, a randomized trial assessing different fluid strategies is impractical. Thus, it is likely that this multicenter case-control study may provide the best data from which clinicians can make management decisions.
The goals of fluid and sodium therapy in a child with DKA are restoration of circulating volume, replacement of sodium and intracellular and extracellular deficits of water, restoration of the GFR with enhanced clearance of glucose and ketones from the blood and avoidance of cerebral edema. Based on available data, it seems prudent for ED physicians to assess each individual patient’s hemodynamic status, appropriately fluid resuscitate children who are severely dehydrated, and avoid overhydration. Although rare, DKA complications such as thrombosis of the femoral vein or cerebral venous sinus may occur secondary to lack of adequate hydration.37
Children should be stratified by initial degree of illness. Documented weight loss, if available, will provide the most accurate assessment of degree of dehydration, but that data must be used with caution. Weights obtained 48 hours or more before admission may reflect weight loss secondary to fat/protein catabolism rather than just water loss. Also, the weight estimate used for fluid calculations should be an ideal body weight; this is particularly important in obese children. Physical examination findings are helpful in estimating fluid needs. Patients with mild to moderate DKA typically have normal or near-normal mental status, moderately elevated heart rate, normal blood pressure and 2-second capillary refill time. These children should receive a 10-20 cc/kg bolus of normal saline (154 mEq/L).
Those with more severe DKA include children who are somnolent or obtunded, with markedly elevated heart rate, Kussmaul respirations, decreased or normal blood pressure, and prolonged capillary refill time. These children will need an initial 20 cc/kg bolus of normal saline. Note that for children who are 10% dehydrated, this bolus replaces just one-fifth the total deficit. A re-examination of each patient’s hydration status should be performed and additional boluses in 10 cc/kg aliquots should be repeated until the patient’s cardiovascular status is more stable. Clinicians should disassociate the degree of acidosis from the deficit volume; these children have a ketoacidosis, not a lactic acidosis.
Following correction of deficit volume, fluid management should be directed at gradual correction of fluid volume loss 35,38 with several sources suggesting an intravenous fluid rate that allows gradual correction during a 48-hour period.21,35,39 The practice at our institution is to administer half the deficit volume plus maintenance fluids during the next 16 hours and that same volume during the subsequent 24-32 hours. In many cases, once the ketosis has resolved, patients can successfully hydrate themselves orally and intravenous fluids may be discontinued. The total fluid volume administered should be reassessed frequently, and the fluid total rarely should exceed 1.5 to 2.0 times the usual daily requirement based on age, weight, or body surface area. Urinary losses should not be added to the calculation of replacement fluids although, urinary output should be noted, and some institutions advocate replacement of losses exceeding 3 cc/kg.21 The recently published consensus statement on DKA management stated that there is no evidence to either support or oppose a more rapid initial fluid replacement or replacement of urinary losses.21
Oral fluid intake should be avoided in the initial hours of care; the majority of oral fluids are hypotonic, and many children will vomit, erasing gains made with intravenous hydration. This overall rate of rehydration, of course, is considerably slower than that used to correct hypovolemic shock due to other causes, but is similar to the approach advocated for other hypertonic states of dehydration. The initial management goals focus on improving, rather than normalizing, vital signs.
Fluid Composition
Hypertonic extracellular fluid leads to the production of intracellular organic osmolytes by brain cells in an attempt to prevent cellular dehydration.40 Studies have shown that rehydration with hypotonic fluids leads to a shift of free water into brain cells and cerebral edema.38,41,42 Investigators have found that a failure of the serum sodium concentration to correct during DKA therapy is associated with cerebral edema,16,35,36,43,44 and the management strategy for a child with DKA should avoid rehydration with large volumes of hypotonic solution.
Serum sodium concentrations are frequently low in DKA. Although free water intake and sodium losses via renal wasting and emesis contribute to this state, serum sodium values are artificially lowered by the hyperosmolar state. Serum glucose shifts intracellular water into the intravascular space, thereby creating a dilutional hyponatremia. To obtain a corrected serum sodium level, for each 100 mg/dL of serum glucose greater than 100 mg/dL, add 1.6 mEq/L to the measured value.
Fluid management for a hypothetical patient is provided in Table 6. As discussed earlier, normal saline should be used for initial fluid boluses. Following this, normal saline may be given until the child is no longer requiring a continuous infusion of insulin. For the typical patient, this point is reached about 12-16 hours after initial ED therapy. For more prolonged episodes of DKA, switching to three-quarters or one-half normal saline after 12-16 hours generally will be adequate to provide for sodium deficits as well as maintenance sodium needs, while avoiding natriuresis. Glucose-containing solutions should be administered when the serum glucose approaches 300 mg/dL and the serum glucose level frequently corrects before the serum pH level does.
Table 6. Fluid Management for a Hypothetical Patient

Hypokalemia and hypophosphatemia are common and preventable complications of DKA. Acidosis exacerbates the renal wasting of electrolytes including potassium; excretion of ketones at the renal tubule obligates further caution and water loss. Also, dehydration activates the renin-angiotensin-aldosterone system promoting urinary potassium loss in exchange for sodium and water. It is easy for ED physicians to be misled; despite total body depletion, serum potassium levels may be normal or even elevated. Correction of acidosis shifts potassium intracellularly, causing an abrupt decline in serum potassium concentration. For these reasons, potassium should be administered, in equal amounts of potassium chloride and potassium phosphate, soon after resuscitation fluids have been given. The concentration of potassium that is administered should be based on the initial serum potassium value. (See Table 7.) One word of caution: A severely dehydrated child may have acute renal failure; it’s best not to begin potassium administration until after the child has urinated. Also, an electrocardiogram should be obtained to monitor for dysrhythmias that might arise due to acidosis or metabolic derangements.
Table 7. Potassium Replacement Therapy

Bicarbonate Therapy
Traditionally, bicarbonate therapy has been recommended to hasten correction of the metabolic acidosis for children with an initial arterial pH of less than 7.1.1,45-47 However, the results of the multicenter study discussed previously question this approach.16 In that study, patients treated with bicarbonate were more than four times more likely to develop cerebral edema compared with patients in a matched control group.16 The numbers of children treated with intravenous boluses of bicarbonate vs. continuous infusions were too small to compare these strategies with sufficient power; these patients were grouped together in the analysis. The authors concluded, "treatment with bicarbonate is associated with an increased risk of cerebral edema and should be avoided in most circumstances."16
Administration of bicarbonate increases the risk of hypokalemia by reversing acidosis and driving potassium intracellularly. In one study, use of bicarbonate did not improve clinical outcomes in children hospitalized with severe DKA.48 This was true even when the initial degree of acidosis was controlled in a multivariate analysis. Similarly, controlled trials in adults have failed to demonstrate a benefit to the use of bicarbonate.45,48,49 Finally, the use of bicarbonate may be associated with worsening central nervous system acidosis; CO2 is formed and crosses the blood-brain barrier.1,45,46
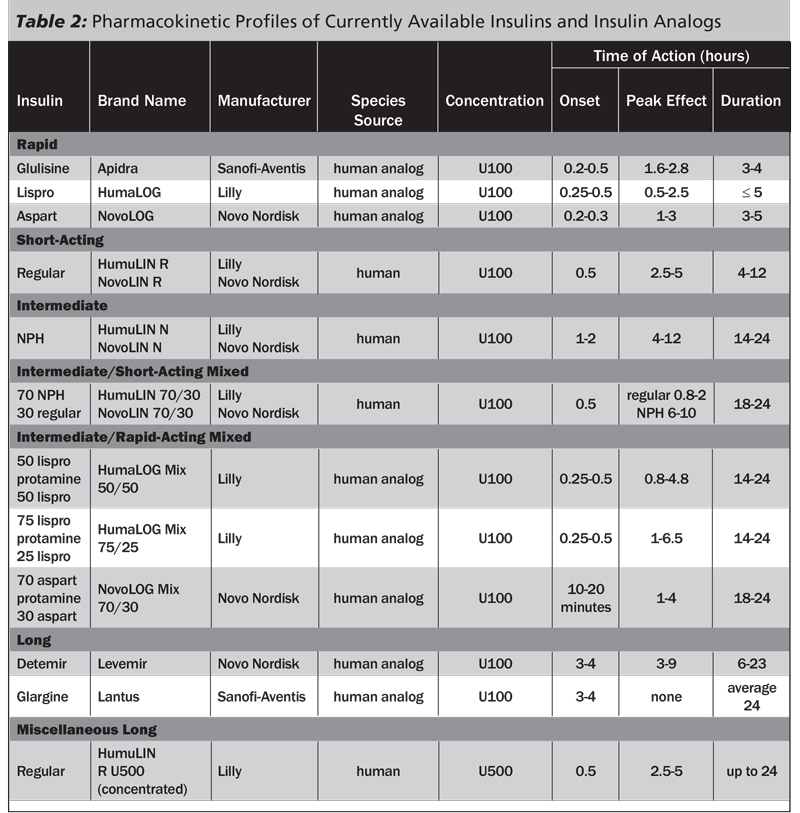
Insulin
Currently, there are few data comparing different dosing regimens of insulin. A priming bolus of intravenous insulin at the initiation of insulin therapy is metabolized within minutes and not proven to be of any benefit. A continuous infusion of Regular insulin at 0.1 units/kg/hour should be started as soon as possible, based on actual body weight. If intravenous access cannot be obtained, then insulin may be given as an intramuscular bolus. The insulin therapy should be titrated with the treatment goals to lower the serum glucose by 100 mg/dL/hour and to correct the base deficit. A common mistake is for clinicians to slow or discontinue the insulin infusion as the serum glucose approaches 300 mg/dL. The serum glucose level frequently corrects before the serum pH does. If the patient remains acidotic, then insulin must be continued. To avoid hypoglycemia, glucose should be administered as the serum glucose approaches 300 mg/dL.
Two-Bag System for Fluid and Electrolyte Administration
Previously, Grimberg and colleagues reported on the use of a two-bag system to administer fluids and electrolytes.50 This method consists of two bags of identical electrolyte content but different dextrose concentrations (0% and 10%). Each bag is hung by the patient’s bedside, and the rate of administration of each bag determines the amount of glucose administered, thus, allowing a fine control of serum glucose levels while insulin is given continuously. Prior to implementing this system, it was necessary for our pharmacist to prepare new bags with ever-higher glucose concentrations to prevent insulin-induced hypoglycemia. The new method allows for a quicker response to declining serum glucose levels.50
Monitoring
Children with DKA potentially are unstable and frequent clinical monitoring of vital signs and mental status should be performed. (See Table 8.) The child’s fluid intake and output should be obtained on an hourly basis, and consideration should be given to placement of a Foley catheter. For moderately to severely ill children, serum glucose levels should be obtained hourly, and serum electrolytes, arterial or venous pH, blood urea nitrogen and creatinine levels every two to four hours. Calcium and phosphorus levels may be monitored every six hours in the initial stages of patient management. An arterial blood gas measurement more accurately reflects the patient’s acid base status and may be considered in children with moderate to severe DKA. Two intravenous lines may be beneficial with one line used to administer fluids and insulin and the other used for obtaining samples for laboratory tests. Documentation of clinical and laboratory data on a flow sheet is very helpful for identifying trends and should include the amounts of fluids given and insulin administered, urine output, serial laboratory test results, and response to therapy.
Table 8. Monitoring of the Child with DKA

Management of Cerebral Edema
The treatment of symptomatic cerebral edema should be started as soon as the condition is suspected. (See Table 9.)
Table 9.
Treatment of DKA-associated
Cerebral Edema

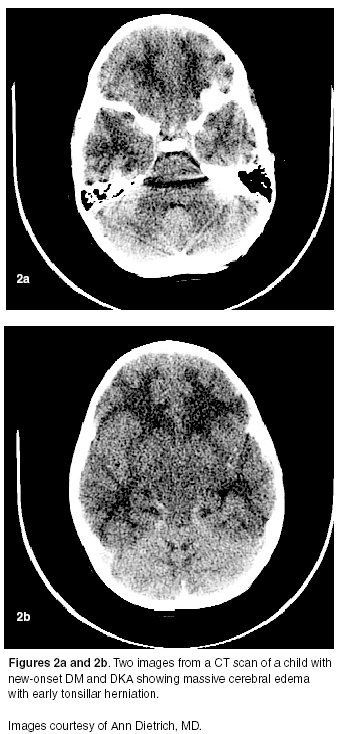
Following stabilization of airway, breathing, and circulation, intravenous fluid rates should be decreased, and patients should be treated promptly with mannitol.21 Hypertonic 3% saline may be considered as an alternative to mannitol.21 Children with symptomatic cerebral edema may require tracheal intubation and mechanical ventilation although aggressive hyperventilation was associated with detrimental outcomes in one retrospective series of children with DKA-related cerebral edema.21 Once the child is more stable, a head CT scan (See Figure 2) should be obtained to assess for other causes of any abrupt change in mental status such as venous sinus thrombosis.
Figure 2. Patient with DKA and Cerebral Edema
Disposition
A child with DKA may need to be hospitalized in an intensive care unit where he can be monitored closely if he has any of the following risk factors: age younger than 5 years, severe acidosis, compromised circulation, or depressed level of consciousness. Other high-risk patients include those with a serum glucose greater than 600 mg/dL, hypernatremia, or hypokalemia. The management of a child with DKA is labor-intensive and is most easily accomplished in a setting with low patient-to-nurse ratios. In addition to an intensive care unit, a unit on the general ward with nurses having expertise in the care of children with DKA also would be acceptable.
Summary
DKA is a common and serious complication of DM. Symptomatic cerebral edema with brain herniation remains the most feared complication of DKA and is associated with a high mortality. ED physicians need to be aware of current management strategies and potential complications. Patient management should be designed to correct dehydration, acidosis, and metabolic derangements while minimizing adverse outcomes. Although a number of controversies in the management of children with DKA remain, newer information has helped to guide therapeutic approaches.16,21 However, more research is needed to delineate the optimal fluid therapy to ensure the best possible outcomes for children with DKA.
References
1. Sperling MA. Diabetes Mellitus. In: Behrman RE, Kliegman RM, Jenson HB, eds. Nelson Textbook of Pediatrics, 16th ed. Philadelphia: WB Saunders Company; 2000: 1767-1791.
2. LaPorte R, Matsushima M, Chang Y-F. Prevalence and incidence of insulin-dependent diabetes. In: Diabetes in America. Bethesda, MD: National Institutes of Health, Department of Health and Human Services;1995:37-45.
3. Melton LJ, Palumbo PJ, Chu CP. Incidence of diabetes mellitus by clinical type. Diabetes Care 1983;6:75-86.
4. Kaufman FR. Diabetes in children and adolescents. Areas of Controversy. Med Clin of North Am 1998;82:721-738.
5. American Diabetes Association. American Diabetes Association: Diabetes 1996 Vital Statistics. Alexandria VA, American Diabetes Association;1996:13-20.
6. Edge JA, Ford-Adams ME, Dunger DB. Causes of death in children with insulin dependent diabetes 1990-96. Arch Dis Child 1999;81: 318-323.
7. The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 1999;22:S5-S19.
8. Sperling M. Diabetes Mellitus. In: Sperling M, ed. Pediatric Endocrinology. 2nd ed. Philadelphia: WB Saunders Company; 2002:323-366.
9. Rosenbloom AL, Schatz DA, Krischer JP, et al. Therapeutic controversy: Prevention and treatment of diabetes in children. J Clin Endocrinol Metab 2000;85:494-522.
10. Association of American Diabetes Association. Type 2 diabetes in children and adolescents. Pediatrics 2000;105:671-680.
11. Dabelea D, Pettitt DJ, Jones KL, et al. Type 2 diabetes mellitus in minority children and adolescents. An emerging problem. Endocrinol Metab Clin North Am 1999;28:709-729.
12. Fagot-Campagna A. Emergence of type 2 diabetes mellitus in children: Epidemiologic evidence. J Pediatr Endocrinol Metab 2000;13: 1395-1402.
13. Fagot-Campagna A, Pettitt DJ, Engelgau MM, et al. Type 2 diabetes among North American children and adolescents: An epidemiologic review and a public health perspective. J Pediatr 2000;136:664-672.
14. Travis L, Brouhard B, Schreiner B. Diabetes Mellitus in Children and Adolescents. Philadelphia: WB Saunders Company;1987:111-168.
15. Mahoney CP, Vicek BW, DelAguila M. Risk factors for developing brain herniation during diabetic ketoacidosis. Pediatr Neurol 1999; 21:721-727.
16. Glaser N, Barnett P, McCaslin I, et al. Risk factors for cerebral edema in children with diabetic ketoacidosis. N Engl J Med 2001;344: 264-269.
17. Marcin JP, Glaser N, Barnett P, et al. Factors associated with adverse outcomes in children with diabetic ketoacidosis-related cerebral edema. J Pediatr 2002;141:793-797.
18. Scibilia J, Finegold D, Dorman J, et al. Why do children with diabetes die? Acta Endocrinol Suppl (Copenh) 1986;279:326-333.
19. Glasgow AM. Devastating cerebral edema in diabetic ketoacidosis before therapy. Diabetes Care 1991;14:77-78.
20. Couch RM, Acott PD, Wong GW. Early onset of fatal cerebral edema in diabetic ketoacidosis. Diabetes Care 1991;14:78-79.
21. Dunger DB, Sperling MA, Acerini CL, et al. European Society for Paediatric Endocrinology/Lawson Wilkins Pediatric Endocrine Society consensus statement on diabetic ketoacidosis in children and adolescents. Pediatrics 2004;113:e113-e140.
22. Faich GA, Fishbein HA, Ellis SE. The epidemiology of diabetic acidosis: A population-based study. Am J Epidemiol 1983;117:551.
23. Pinkey JH, Bingley PJ, Sawtell PA, et al. Presentation and progress of childhood diabetes mellitus: A prospective population-based study. Diabetologia 1994;37:70-74.
24. Vanelli M, Chiari C, Ghizzoni I, et al. Effectiveness of a prevention program for diabetic ketoacidosis in children. An 8-year study in schools and private practices. Diabetes Care 1999;22:7-9.
25. Daneman D. Diabetes-related mortality. A pediatrician’s view. Diabetes Care 2001;24:801-802.
26. Skinner T. Recurrent diabetic ketoacidosis: Causes, prevention, and management. Horm Res 2002;57:78-80.
27. Dumont R, Jacobson A, Cole C, et al. Psychosocial predictors of acute complications of diabetes in youth. Diabet Med 1995;12: 612-618.
28. Kovacs M, Charron-Prochownik D, Obrosky DS. A longitudinal study of biomedical and psychosocial predictors of multiple hospitalizations among young people with insulin-dependent diabetes mellitus. Diabet Med 1995;12:142-148.
29. Krall LP, Beaser RS, eds. Joslin Diabetes Center. Joslin Diabetes Manual 12 ed. Lea & Febiger;1989:14-188;246-268.
30. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977-986.
31. The Diabetes Control and Complications Trial Research Group. Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: Diabetes Control and Complications Trial. J Pediatr 1994;125:177-188.
32. Morris AD, Boyle DI, McMahon AD, et al. Adherence to insulin treatment, glycemic control and ketoacidosis in insulin-dependent diabetes mellitus. Lancet 1997;350:1505-1510.
33. Muir AB, Quisling RG, Yang MC, et al. Cerebral edema in childhood diabetic ketoacidosis: Natural history, radiographic findings, and early identification. Diabetes Care 2004;21:1541-1546.
34. Glaser NS, Kuppermann N, Yee CKJ, et al. Variation in the management of pediatric diabetic ketoacidosis by specialty training. Arch Pediatr Adoles Med 1997;151:1125-1132.
35. Harris GD, Fiordalisi I, Harris WL, et al. Minimizing the risk of brain herniation during treatment of diabetic ketoacidemia: A retrospective and prospective study. J Pediatr 1990;117:22-31.
36. Duck SC, Wyatt DT. Factors associated with brain herniation in the treatment of diabetic ketoacidosis. J Pediatr 1988;113:10-14.
37. Keane S, Gallagher A, Ackroyd S, et al. Cerebral venous thrombosis during diabetic ketoacidosis. Arch Dis Child 2002;86:204-205.
38. Trachtman H. Cell volume regulation: A review of cerebral adaptive mechanisms and implications for clinical treatment of osmolal disturbances: II. Pediatr Nephrol 1992;6:104-112.
39. Harris GD, Fiordalisi I, Yu C. Maintaining normal intracranial pressure in a rabbit model during treatment of severe diabetic ketoacidemia. Life Sci 1996;59:1695-1702.
40. Muir A. Cerebral edema in diabetic ketoacidosis: A look beyond dehydration. J Clin Endocrinol Metab 2000;85:509-513.
41. McManus ML, Churchwell KB, Strange K. Regulation of cell volume in health and disease. N Engl J Med 1995;333:1260-1266.
42. Prockop LD. Hyperglycemia, polyol accumulation, and increased intracranial pressure. Arch Neurol 1971;25:126-140.
43. Bello FA, Sotos JF. Cerebral oedema in diabetic ketoacidosis in children. Lancet 1990;336:64.
44. Harris GD, Fiordalisi I, Finberg L. Safe management of diabetic ketoacidemia. J Pediatr 1988;113:65-68.
45. Hale PJ, Crase J, Nattrass M. Metabolic effects of bicarbonate in the treatment of diabetic ketoacidosis. Br Med J 1984;289:1035-1038.
46. Kaufman FR, Halvorson M. New trends in managing type I diabetes. Contempor Pediatr 1999;16:112-123.
47. Plotnick L. Insulin-dependent diabetes mellitus. Pediatr Rev 1994;15: 137-148.
48. Green SM, Rothrock SG, Ho JD, et al. Failure of adjunctive bicarbonate to improve outcome in severe pediatric diabetic ketoacidosis. Ann Emerg Med 1998;31:41-48.
49. Morris LR, Murphy MB, Kitabchi AE. Bicarbonate therapy in severe diabetic ketoacidosis. Ann Intern Med 1986;105:836-840.
50. Grimberg A, Cerri RW, Satin-Smith M, et al. The "two bag system" for variable intravenous dextrose and fluid administration: Benefits in diabetic ketoacidosis management. J Pediatr 1999;134:376-378.
Although diabetes mellitus is second only to asthma as the most prevalent chronic disease of childhood, the literature has very few comprehensive reviews of diabetic ketoacidosis (DKA), the most frequent cause of death in children with diabetes. The importance of an early diagnosis and appropriate management should not be underestimated. The authors provide a focused review for the ED physician for recognition and management of a child with DKA, with special attention to potentially serious complications.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
