Influenza in Children: What Emergency Physicians Need to Know This Season
Author: James A. Wilde, MD, FAAP, Associate Professor of Emergency Medicine and Pediatrics, Medical College of Georgia, Augusta.
Peer Reviewer: John P. Santamaria, MD, FAAP, FACEP, Medical Director, After-Hours Pediatrics, Affiliate Professor of Pediatrics, University of Florida School of Medicine, Tampa.
The 2003-2004 influenza season was memorable for most emergency departments (EDs): large volumes of febrile children and concerned parents. Although the majority of otherwise healthy children who present with typical flu symptoms during an influenza outbreak may be diagnosed clinically, there are certain situations where confirmation of the diagnosis may be beneficial and also may limit unnecessary diagnostic testing and reduce unnecessary use of antibiotics. Central also to the argument to not use routine rapid flu testing during influenza outbreaks is the recommendation from the Committee on Infectious Diseases from the American Academy of Pediatrics (AAP) to not use antiviral therapy for uncomplicated influenza in an otherwise healthy child. The author presents a thorough discussion of the scientific evidence and controversies surrounding the use and value of influenza testing, antiviral therapy, and the influenza vaccine. With the media currently focused on vaccine shortages, the emergency physician must be prepared to rationally and scientifically explain diagnostic and therapeutic approaches in children with influenza. The author prepares the ED physician to confidently face the 2004-2005 influenza season. — The Editor
Introduction
Influenza season will soon be upon us, and many patients who experience acute fever, cough, and sore throat symptoms typical of the flu will seek care in the nation’s EDs. The 2004-2005 season may be particularly difficult for medical practitioners due to several topics relating to influenza that have been front-page news during the last 12 months. Chief among them are concerns about the large number of deaths due to influenza that were reported in children last winter, news from Southeast Asia of new influenza variants that may represent the beginning of a long overdue pandemic, and news releases beginning in October 2004 regarding severe disruptions in the influenza vaccine supply. Pediatric EPs can expect a high level of anxiety in patients and their parents due to this widespread publicity.
This article discusses all of these topics and reviews current recommendations regarding the presentation, diagnosis, and management of influenza in children.
Background
Influenza is an infectious disease with a long history. Scholars believe that a description of an epidemic in ancient Athens attributed to Hippocrates represents the first report of influenza in Western literature. Many other reports of influenza outbreaks are recorded beginning about 1,000 years ago.1 The annual epidemics that characterize influenza in the present era probably began in the last 500-1000 years, perhaps as a result of increased travel between world population centers. The most deadly single-season epidemic in recorded history occurred in 1918 when Spanish Flu ravaged the world, killing 550,000 people in the United States and at least 20 million people more around the world.2 Subsequent pandemics occurred in 1957 and 1968, but neither proved to be as deadly. A fascinating study reported in the journal Nature in October 2004 indicates that the unparalleled severity of the 1918 epidemic appears to be due to a mutation in the hemagglutinin gene that triggered a massive cytokine release in the victims. The unusual degree of cytokine release led to severe inflammation and lung damage that caused the victim’s rapid demise.3
The yearly influenza epidemic period generally begins in the United States between November and January (See Table 1).4 The winter of 2003-2004 was unusual; reports of widespread influenza activity began in October and peaked by the first week of December, much earlier than the average onset of the flu season recorded during the last 28 years. Influenza epidemics are characterized by intense periods of activity with an abrupt onset and an abrupt end. The entire epidemic period generally lasts for 8-12 weeks. The explosive nature of these epidemics probably is due to a combination of the very short 1-4 day incubation period5 and the high rate of communicability. A consistent finding in surveillance studies is that influenza epidemics generally begin most intensely in children, then shift to the adult population as the epidemic progresses. More than one-half of infections in the early stages of the epidemic are in children from 5 to 19 years of age.6 Therefore, increased school absence and visits to physicians among children in this age group may be used to alert the community to the onset of the annual flu epidemic.
Table 1.
Month of Peak Influenza Activity
in the United States, 1976-2002*

Influenza has been responsible for an estimated 38,000 excess deaths per year due to pneumonia and influenza (P&I) in the United States during the past 20 years. Ninety percent of these deaths occur in patients older than 65 years.7 These data are not gathered by direct reports of influenza infection because it is not a reportable disease. Instead, the Centers for Disease Control and Prevention (CDC) in Atlanta monitors P&I activity throughout the year in 122 cities across the country. Variations from the baseline level of P&I mortality allow researchers to estimate the relative impact of influenza. Figure 1 displays P&I mortality for the past four years. The absence of peaks above the baseline in the years 2001 and 2003 indicate that the epidemics during those years were particularly mild.
The attack rate for influenza varies widely from year to year,8 from a low of 2-5% in a mild season to a high of 20% or more in a severe season. The attack rate in children tends to be significantly higher, with rates of 45% or more reported.9 These infections are responsible for an estimated 110,000 hospitalizations per year in the United States.10 (See Table 2.)
Table 2.
Age-Specific Annual Rates of Influenza-Associated
Hospitalizations (per 100,000)
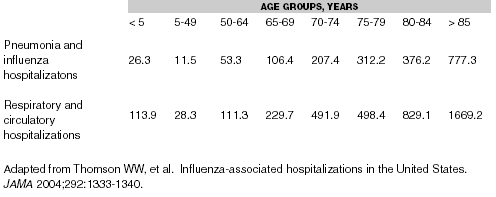
The age distribution of hospitalizations is bimodal, with a peak in the younger-than-5-years age group, a relatively low rate between age 5 years and 49 years, and a second peak beginning in the 50-64 year age group that rises sharply with each subsequent decade of life. The highest rates for influenza-associated hospitalization are in children younger than 5 years and adults older than 65 years.
Influenza activity is monitored worldwide by the World Health Organization (WHO) and in the United States through both WHO USA and the CDC. Data for the United States are collected by the CDC in part through the Sentinel Provider Network, a group of approximately 900 primary care and EPs who report weekly to the CDC. The principal information relayed by members of the Sentinel Provider Network is the proportion of patients managed for influenza-like illness (ILI) during the preceding week. ILI is defined for this purpose as fever (temperature equal to or more than 100°F) and cough and/or sore throat in the absence of a diagnostic test for a specific diagnosis. A rise in the proportion of patient visits for ILI that represents more than two standard deviations above the baseline is closely associated with increased influenza activity. Likewise, a sharp drop in ILI generally heralds the end of the influenza epidemic period. The curve for P&I mortality lags slightly behind the ILI curves and also is useful for establishing both the duration of the influenza epidemic period and the severity of the epidemic itself. (See Figure 1.)
There have been five significant pandemics in the past 120 years.2 (See Table 3.) The last major pandemic was in 1968 when A(H3N2) subtype, known as "Hong Kong Flu," initially appear-ed on the world stage. A mild pandemic due to A(H1N1) occurred in 1977. Since 1977, all yearly epidemics due to influenza A have been due to a combination of either H3N2 or H1N1 subtypes. The 36-year interval from 1968 to the present represents an unusually long period between severe pandemics, fueling speculation that the world is overdue for another in the near future.
Table 3. Major Influenza Epidemics Since 1889

The Virus
Influenza is a respiratory pathogen classified as an orthomyxovirus. Human influenza is divided into Type A and Type B. Influenza Type C is of unclear significance in humans. Influenza viruses are typed based on proteins that are found within the interior of the virion, proteins that undergo no significant mutation and appear to be well conserved between types. Influenza viruses are divided further into subtypes based on two proteins that appear on the surface of the virion: hemagglutinin (H) and neuraminidase (N). Hemagglutinin is required for the virus to attach itself to host cell membranes. Neuraminidase is an enzyme that aids in viral penetration into the interior of the infected cell and further release of replicated virions from the cell. All human influenza epidemics and pandemics in the past century have been caused by strains with one of three hemagglutinins (H1, H2, or H3) and one of two neuraminidases (N1 or N2).
A feature of influenza that distinguishes it from most other respiratory viruses is its high rate of mutation to new strains. These mutations occur in the hemagglutinin and neuraminidase genes. It is because of this constant tendency to mutate that influenza is an annual concern, even among people who have had past influenza infections. Mutation of influenza virus comes in two forms: antigenic drift and antigenic shift.
Antigenic drift refers to subtle changes in the surface H and N proteins. Drifted strains have the same H and N subtypes but are different enough from the precursor subtype that immunity to the precursor does not confer full immunity to the drifted strain. Due to antigenic drift, influenza vaccine components must be updated yearly.
Antigenic shift refers to a major mutation resulting in a different H or N subtype. As a result of a shift, completely novel viruses may appear. Each of the major pandemics of the last 100 years were due to a major shift in the viral surface proteins, leaving the population with little to no existing immunity to the new virus. For example, the Hong Kong flu epidemic of 1968 was due to a shift from the H2N2 subtype that had been prevalent since 1957 to the H3N2 subtype. The H3N2 subtype is still the predominant circulating subtype today.
Both Influenza A and Influenza B undergo antigenic drift, but only Influenza A undergoes antigenic shift. For this reason, only Influenza A is subtyped. The nomenclature for influenza viruses has been standardized to allow for ease of communication throughout the world. The letters and numbers on either side of the backslashes provide a wealth of information about the virus, including (in order) the type, the world location where the virus was first isolated, the strain number, the year of isolation, and the subtype. Therefore, the strain A/USSR/90/77 (H1N1) was an influenza virus of Type A, subtype H1N1, isolated in the USSR in 1977.
Signs and Symptoms
Influenza is one of the few respiratory viruses that commonly causes fever in both children and adults. Most of the other respiratory viruses that circulate throughout the year cause fever in children but generally not in adults. Non-influenza infections are manifested in adults primarily by non-specific upper respiratory tract symptoms, such as cough and nasal congestion.2 The sensitivity of fever plus respiratory symptoms such as cough, rhinorrhea, or sore throat for the diagnosis of influenza during an epidemic period is estimated to be 60-80% in adults.8,11,12 Positive predictive values have been reported from 40% to 86% and can be expected to vary with the stage and severity of the epidemic. The sensitivity of clinical symptoms for the specific diagnosis of influenza in children is not well established but is probably lower than for adults due to co-circulation of other respiratory viruses during the influenza season.
Illness due to influenza typically has an abrupt onset, with both fever and respiratory symptoms evident early. The incubation period is generally 1-4 days. Adults are infectious from one day before to five days after the onset of overt symptoms, but young children may be infectious from six days before to 10 days after the onset of illness.5
Influenza infection in adults is characterized by high fever, myalgias, headache, malaise, cough that tends to be non-productive, sore throat, and rhinorrhea. These symptoms are also common in children, but in addition a significant number of children also develop nausea and vomiting.5 Fever in uncomplicated influenza may last up to 5-6 days, but fever persisting beyond that point should prompt a search for another explanation. At least 20% of children experience acute otitis media as a complication of influenza.13
One of the primary major complications of influenza is secondary bacterial pneumonia. Other major complications include encephalopathy, transverse myelitis, myositis, and myocarditis. In addition, febrile seizures have been reported in up to 20% of children.14
Diagnosis
In the majority of otherwise healthy patients who present with typical flu symptoms during the influenza season, the diagnosis can be established on clinical grounds alone. However, there are situations in which confirmation of the diagnosis may be helpful. There are a number of methods available to confirm the diagnosis of influenza, including serology, viral culture, polymerase chain reaction (PCR), direct fluorescent antibody (DFA) test, and rapid tests based on enzyme-linked immunosorbent assays (ELISA).
Serology is the closest we have to a gold standard for the diagnosis of flu. Serologic methods to diagnose any infectious disease depend on an intact immune system that mounts a significant response to the invading organism. The response is measured by a rise in the level of antibody to the infectious agent. A four-fold rise above the baseline IgG antibody level is accepted as presumptive evidence for an infection. This requires a comparison between paired acute and convalescent titers and provides confirmation of the infection retrospectively, generally 2-4 weeks after the onset of symptoms. This also renders the assay of limited value in the acute diagnosis and management of influenza. Another problem with the use of serology for the diagnosis of influenza is that the assay must be changed each year to account for drifted strains because the assay measures hemagglutination-inhibition (HI) antibody titers. Therefore, serology is offered primarily in reference or research laboratories; it is not available in most community hospitals or clinics. A single serum sample is not sufficient to establish the diagnosis by serology.
Assays based on the PCR have proliferated during the last 10 years and are generally quite sensitive for the detection of influenza and other viruses. However, the technique is extremely labor intensive and requires a high level of expertise. For both of these reasons, PCR also is offered primarily in reference labs, making it of little value in the acute care setting.
Influenza grows quite well in viral culture media. Swabs from the nasopharynx or throat during the acute stage of the infection may detect the virus, but the yield decreases after the third day of illness. The culture result generally is not available for at least 2-3 days, rendering it of limited value in the diagnosis and management of influenza in the outpatient setting. It may be of greater value in the inpatient setting, particularly to establish the presence of a nosocomial infection. Cultures also are helpful to monitor influenza activity in the community, both to detect the beginning of an epidemic period and to identify the circulating type, subtype, and strain. This information can be used to make better decisions regarding specific antiviral therapy.
Assays based on direct fluorescent antibody tests are highly specific for influenza and may be performed within less than an hour of the collection of the sample. However, the interpretation of the DFA assay requires highly experienced laboratory technicians. In addition, hospitals commonly batch samples for DFA testing and perform the test only intermittently. For both these reasons, it is not a practical test for most clinics or EDs.
Assays based on ELISA have the greatest potential to aid in the diagnosis and management of acute influenza because they can be performed in as little as 15-20 minutes by clinicians in the outpatient setting. There are at least five rapid tests now available for this purpose.15 (See Table 4.) Sensitivities range from about 60-90%. Specificities are generally higher, in the range of 90-99%, although reported specificity for the Flu OIA from BioStar is substantially lower. The rapid test with the best combination of sensitivity and specificity is the Directigen Flu A + B from Becton Dickinson; this assay also can distinguish influenza A from influenza B; most of the other available assays detect influenza A or influenza B but do not distinguish between the two.15
Table 4. Rapid Diagnostic Tests for Influenza

A limited number of studies have been performed on the utility of the influenza rapid test in children.16-19 Two have been performed specifically in an ED setting. Noyola et al reported reduced antibiotic use in both inpatients and outpatients whose diagnosis was confirmed by the rapid test.16 However, antibiotics were prescribed to 53% of febrile children in the control group who were discharged from the ED. It is not clear why the rate of antibiotic use was so high in a population with typical symptoms of influenza.
Bonner et al reported significant reductions in orders for blood cultures, CBCs, urine cultures, chest radiographs, and antibiotics prescribed among children whose ED physician knew that their rapid test for flu was positive.19 However, the data in this study were analyzed by comparing the management of patients who had a positive test with the management of those who had a negative test. This analysis is relevant for patients with a known positive test, but it is not relevant for determining the overall value of performing the assay itself. If the data are analyzed more appropriately by comparing the management of those patients who had a known test result (test performed) with that of those who did not (test not performed), no statistically significant difference can be shown in any of the above categories, except antibiotic use (author’s own unpublished analysis). Furthermore, close inspection of the data presented in Bonner’s report shows a clear trend toward more tests ordered if the patient had a known negative rapid flu test.
In a recent review article, the clinical usefulness of rapid tests for influenza in children managed in the ED has been questioned.15 Rapid tests for flu may be of value in alerting physicians to the presence of influenza in their community, and for tracking the activity of influenza A versus influenza B. This may be particularly important in deciding which antiviral medication to prescribe. However, the routine use of rapid tests in otherwise healthy children with symptoms compatible with uncomplicated flu does not appear to be warranted.
Therapy
The Committee on Infectious Diseases of the AAP does not recommend antiviral therapy for uncomplicated influenza in otherwise healthy children.20 This recommendation from the AAP is central to the argument against the routine use of rapid tests for the diagnosis of influenza in the ED. If specific antiviral therapy is not warranted, in most cases a diagnostic test is not required; the diagnosis can be made on clinical grounds alone. The Committee on Infectious Diseases does suggest the use of antiviral therapy for several categories of patients: children at high risk of complications from acute influenza (Table 5); healthy children with severe illness; and patients with special environmental, family, or social situations in whom a shortened illness would be helpful (i.e., important school examination, sports event). In these patients, the use of a rapid test may help to guide therapeutic decisions, although considering that the sensitivities of the rapid tests are less than 100%, a better strategy may be to treat empirically with antiviral therapy based on symptoms and local influenza activity.
Table 5.
Persons Recommended to Receive
Influenza Vaccine: Interim Recommendations

There are two classes of antiviral medications available for the treatment of influenza. None of these medications have been studied extensively in children, although limited data are available.13,21-24 Treatment must be begun within 48 hours of symptom onset for significant benefit. The Food and Drug Administration (FDA) has not approved the use of any of these medications for children younger than 1 year. Clinicians also should be aware that studies on the effectiveness of antiviral medications largely have been conducted in healthy patients with uncomplicated flu. According to the CDC, "Data are limited and inconclusive concerning the effectiveness of amantadine, rimantadine, zanamivir, and oseltamivir for treatment of influenza among persons at high risk for serious complications of influenza".7
Amantadine and rimantadine are closely related tricyclic amines that inhibit the replication of the influenza A virus. They have no effect on influenza B. They are useful for treatment of active disease and for prophylaxis. Amantadine is approved for both treatment and prophylaxis in children older than 1 year, whereas rimantadine is approved only for prophylaxis in this age group. Treatment with rimantadine is approved for children older than 13 years. Limited data show that treatment with either medication will reduce the duration of uncomplicated influenza illness by approximately one day. The principal side effect of these drugs is a reversible neurotoxicity that is seen most commonly with amantadine. Another concern about the use of these agents is that rapid emergence of resistance has been well documented with both. Studies have shown that up to 30% of isolates are resistant by the third to fifth day of therapy.24,25 Several case reports have demonstrated transmission of resistant viruses, but the frequency of this is unclear.5,26 There is no evidence to indicate that the resistant viruses are more virulent.
Zanamivir and oseltamivir are neuraminidase inhibitors that are active against both influenza A and influenza B. Zanamivir is rarely used in children or adults primarily due to bronchospasm induced after its administration by inhalation. Oseltamivir is approved for treatment of influenza in children older than 1 year, and for prophylaxis in children older than 13 years. Side effects with oseltamivir are less severe than with the tricyclic amines and relate mostly to gastrointestinal symptoms. Resistance has been reported but is much less common than with amantadine or rimantadine. Limited data indicate that treatment with oseltamivir may reduce the duration of symptoms of flu in children by up to 1.25 days.13,27 Treatment with oseltamivir also may reduce the incidence of concomitant acute otitis media by up to 44%.13
Control of Influenza Through Vaccination
Influenza vaccine has been available for decades and has been shown to be of varying degrees of benefit in all age groups, including children older than 6 months. Vaccine efficacy for preventing influenza respiratory illness in children ranges from 50-95%, depending on the closeness of match between the vaccine strain and the circulating wild strain.20 In general, influenza vaccines are less effective in children than in adults in preventing ILI because of the higher frequency of ILI caused by other respiratory pathogens in children. Influenza vaccine is not approved for children younger than 6 months.
Currently, there are two types of vaccine products available for prevention of influenza: inactivated vaccine and live, attenuated influenza vaccine (LAIV). The inactivated vaccines have a long history of use, while the LAIV was licensed for use in 2003. The inactivated vaccines are trivalent, and carry proteins specific for three strains of influenza, including two strains of influenza A and one strain of influenza B. Because the components are inactivated, they cannot cause flu upon administration, although transient side effects such as fever and myalgia may mimic some of the symptoms of influenza infection. Inactivated influenza vaccine is administered as an intramuscular injection. Children younger than 9 years require two doses if they have not been vaccinated previously.
The LAIV product is also trivalent but is administered as an intranasal spray. The vaccine strains contained in LAIV are cold adapted so that they replicate easily in the nasopharynx but are unable to replicate efficiently in the warmer environment of the lower respiratory tract. Studies have shown that although the vaccine contains live virus that replicates in the nasopharynx, transmission can be demonstrated in less than 1% of patients.28 A recent study showed vaccine efficacy of 86% among children 26 to 85 months of age, even during an epidemic caused by a variant strain not contained in the vaccine.29
There are two products containing inactivated influenza virus in the United States: Fluzone from Aventis Pasteur and Fluvirin from Chiron Corp. Fluvirin is not approved for children younger than 4 years. The sole LAIV product on the market is FluMist from Medimmune, which is approved only for healthy persons from 5 years to 49 years of age. Fluzone, therefore, is the only influenza vaccine of any kind that is available and approved for children younger than 4 years.
The Advisory Committee on Immunization Practices (ACIP) recommends influenza vaccine for persons who are at increased risk for complications from the flu. Vaccine also is recommended for other groups such as health care providers involved in direct patient care.7 As of 2000, more than 73 million persons in the United States were included in one of the ACIP high-risk target groups. In 2004, the AAP, for the first time, recommended influenza vaccine for children 6 months to 2 years of age, largely due to data showing increased rates of hospitalization and morbidity in this age group relative to other children.30 These children can receive only inactivated influenza vaccine.
On Oct. 5, 2004, the British government suspended Chiron’s license to manufacture Fluvirin in its Liverpool factory for at least three months.31 This decision was made due to concerns about contamination. The company also was unable to release any existing vaccine, thereby reducing the availability of inactivated vaccine in the United States from approximately 100 million doses to 54 million doses; at the time of the announcement, 30 million doses of Fluzone already had been distributed. Only 1.1 million doses of LAIV were prepared for the U.S. market this season. The CDC and ACIP quickly issued interim recommendations in an attempt to focus vaccine efforts and supply on those most in need. (See Table 5.)
As a result of this interruption in the vaccine supply, the CDC decided to direct the available vaccine to persons who need it the most.32 Working collaboratively with Aventis Pasteur, it chose to distribute the remaining 22.4 million doses of unshipped vaccine to identified areas of need in the United States. In phase one, 14.2 million doses were allocated directly to high-priority providers, including hospitals, long-term care facilities, nursing homes, and private providers who care for young children. The remaining 8.2 million doses are to be shipped to other high-need areas after completion of phase one. On Oct. 5, 2004, the AAP Executive Committee issued a statement recommending that available vaccine should be used to vaccinate high-risk children on a first-come, first-served basis; doses should not be held in reserve to ensure that two doses will be available.33
Considering the number of doses of influenza vaccine available for the 2004-2005 influenza season and the number of persons who are included in the priority list, many high-risk patients will not receive vaccine. However, vaccination coverage has never approached 100% in any of these groups. In 2001-2002, vaccine coverage for adults older than 65 years was only 66%; estimated vaccine coverage among adults with high-risk conditions aged 18-49 years was 23%; and for those age 50-64 years, it was 44%.6 Results from a 2001 study showed that in a clinic using a computerized reminder system, only 32% of children with asthma received influenza vaccination.34 In 2002, just 38% of health care workers reported receiving influenza vaccine.7
The implications of the vaccine shortage for the 2004-2005 flu season are not entirely clear. There is no doubt that many U.S. citizens who desire the vaccine will be unable to obtain it. However, given the historically low rates of influenza vaccination even within high-risk groups, the anxiety and publicity surrounding the current vaccine shortage actually may increase the rate of vaccination among high-risk patients. In addition, there was a poor match between the strains contained in the 2003-2004 vaccine and the circulating wild-type strains due to the late emergence of a drifted strain. The vaccine contained A/Panama/2007/99 (H3N2), while 82% of all viruses collected by U.S. laboratories were characterized as A/Fujian/411/2002 (H3N2).35 The vaccine for the 2004-2005 flu season does contain the Fujian strain. Thus, despite the vaccine shortage, there may be a paradoxical decrease in P&I mortality in the coming season due to more focus on high-risk groups, a better vaccine match to the circulating strain, and large numbers of people with natural immunity to the Fujian strain because of infection last year.
Synopsis of 2003-2004 Influenza Season
The first state to report widespread flu activity during the 2003-2004 influenza season was Texas on Oct. 18, 2003.35 The weekly percentage of patient visits to sentinel physicians for ILI exceeded the baseline from Nov. 15, 2003, until Jan. 10, 2004, a total of 8 weeks. (See Figure 2 and Figure 3.) The highest proportion of ILI visits for the 2003-2004 flu season occurred during the week ending Dec. 27, 2003, when 7.6% of patients who presented to sentinel providers had symptoms. In contrast, the highest proportion of ILI visits for any single week during the previous four flu seasons ranged from 3.3% to 7.1%; peaks in those years occurred during late January and early February.35
The proportion of all deaths in the United States attributed to P&I exceeded the epidemic threshold for nine consecutive weeks from Dec. 20, 2003, to Feb. 14, 2004. (See Figure 3.) The highest weekly proportion of deaths due to P&I during that period was 10.3% between Jan. 10 and Jan. 17, 2004, compared with peaks ranging from 8.1% to 11.2% during the previous four flu seasons.35
According to the CDC, the 2003-2004 season was "moderately severe in terms of its impact on mortality."35 This conclusion may come as a surprise to physicians and many in the general public who believe that last season was a very severe one. There are two primary reasons for this misperception: Last season’s vaccine shortage and mismatch, and the widely disseminated reports in medical and popular forums about an alarming number of pediatric deaths due to flu.
The appearance of the drifted Fujian strain of influenza was detected before the beginning of the 2003-2004 season but too late for inclusion in that season’s influenza vaccine. Due to evidence from prior epidemics that seasons dominated by A (H3N2) viruses are associated with high levels of severe illness and death, public health authorities began to warn the public about the potential for a severe epidemic well before the peak of the flu season.36 Partly as a result of these warnings, by the first week of December 2003, the manufacturers had sold all 83 million doses of the available inactivated vaccine. In response, at least 28 states redistributed influenza vaccine from providers and public health clinics with excess supplies to those that needed vaccine.37 These shortages and redistribution efforts received widespread coverage in print and broadcast media.
In addition to the vaccine shortage and mismatch, there also were early reports of severe illness and deaths in the pediatric population,37 including deaths in previously healthy children due to secondary pneumonia caused by community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA).38,39 However, a proper frame of reference on these pediatric deaths was difficult to obtain because neither the CDC nor any other public health agency had any definitive statistics on the number of pediatric deaths in previous epidemics. The case fatality rate in children had been estimated to be 3.8 per 100,000 in a publication from 1982.6 Another study that modeled influenza-related deaths estimated that an average of 92 deaths per year occurred among children younger than 5 years during the 1990s.36
As of June 2004, a total of 152 laboratory-confirmed pediatric deaths had been reported voluntarily to the CDC from the 2003-2004 season.40 A particularly disturbing finding is that 40% of those deaths occurred in previously healthy children. (See Table 6.) Only 27% of deaths were among children with ACIP high-risk conditions. Shortly before the beginning of the 2004-2005 influenza season, the Council of State and Territorial Epidemiologists (CSTE) approved an initiative to add pediatric influenza-associated deaths to the list of nationally notifiable conditions in the United States, permitting better recognition of excess mortality in children in future flu epidemics.
Table 6. Pediatric Influenza Mortality, 2003-2004

Avian Influenza
The last major influenza pandemic was in 1968. In that year, influenza A isolates first identified in Hong Kong demonstrated an antigenic shift to the H3N2 subtype. For the past 27 years, this subtype has co-circulated with an H1N1 subtype that re-emerged in 1977. The world has not experienced another major pandemic since the Hong Kong flu of 1968, but speculation abounds about when the next one will occur. As one author put it, "There is no reason to believe there will not be another one."41 For a number of reasons, public health officials have focused their surveillance efforts in Southeast Asia, where many experts expect the next pandemic strain to emerge.
The natural reservoirs for all known influenza subtypes are birds and waterfowl, including poultry. Fifteen hemagglutinin (H1-H15) and nine neuraminidase (N1-N9) subtypes have been identified thus far, but only H1, H2, H3 and N1, N2 are known to be established firmly in humans.42 Due to the close association between humans and poultry, the potential exists for subtypes to cross the species barrier from poultry to humans through genetic re-assortment. This re-assortment may occur if two different viruses infect the same cell simultaneously. Because influenza has a segmented genome, the two viral strains replicating within the infected cell can exchange genetic material between each other, resulting in a novel virus and subtype.43 Conditions for this cross-species transfer are greatest in Southeast Asia, where dense human populations live in close contact with farm animals, particularly chickens and ducks.
In 1997, an outbreak of influenza A (H5N1) in humans was reported in Hong Kong.44 Between May and December, a total of 18 cases and six deaths were confirmed for a case-fatality rate of 33%. Half of the cases occurred in children 10 years of age or younger. Avian flu is a strain of influenza that had been known to infect birds, but it had not been reported previously in humans. Cohort studies suggested that human-to-human transmission might have occurred through close physical contact with infected patients (i.e., within households), but there was no evidence that social exposure was associated with any of the infections.45 Another cohort study of medical personnel documented seropositivity to the virus in 3.7% of exposed health care workers and 0.7% of non-exposed health care workers, again indicating a low rate of transmission between humans.46
The source of the outbreak was identified as poultry; up to 20% of the chickens in the Hong Kong market were discovered to be infected. In a vigorous response, the government slaughtered all the poultry in farms and markets in Hong Kong. Millions of chickens were destroyed to contain the spread of the virus. In addition, import of live poultry from mainland China was halted temporarily. These efforts appeared to stop the further spread of the virus, and the local outbreak came to an end for several years. In May 2001, a cluster of chicken deaths due to the H5N1 subtype again was identified. A second territory-wide slaughter of poultry was undertaken in response, and once again it appeared that the virus was contained. No human infections were reported during the 2001 outbreak.
In December 2003, the outbreak became more widespread. Eight Asian countries from Thailand and Indonesia to Japan and Korea reported widespread H5N1 activity in the largest outbreak of avian influenza in poultry ever reported. At least 100 million domestic poultry died or were culled in an effort to contain the epidemic. Although widespread infection in humans was not found, 34 confirmed cases were reported in Vietnam and Thailand in 2004. The case fatality rate was 68%.43,47,48 As of 2004, H5N1 strains have been resistant to amantadine and rimantadine but are susceptible to oseltamivir.43
Efforts to contain influenza A (H5N1) have been fairly successful; a feared pandemic has not occurred to date. These efforts have been aided because this strain of avian influenza appears to have a very low rate of transmissibility between humans; most cases resulted from close, intense exposure to infected poultry. However, an ominous point was raised about these outbreaks in a recent publication: Two of the three key criteria that characterized the pandemic of 1918-1919 have already been fulfilled in the current epidemic: 1) the ability of the virus to infect humans resulting in high mortality, and 2) a global immunologically naïve human population. The third criterion, efficient human-to-human transmission, thus far, has not been observed.48
So far, all of the genes involved in the strains producing avian flu have been of avian origin. No genetic re-assortment with human influenza viruses has occurred. If it does occur, human-to-human transmission may be facilitated, which could usher in the long-awaited next pandemic. If mortality rates reach the level seen during the 1918 pandemic, more than 100 million people could perish worldwide.
Conclusions
Influenza exacts a heavy toll on the United States and the rest of the world. Billions of dollars are spent each year for office and ED visits for flu symptoms, and billions more are spent for hospitalizations. Vaccination remains the main line of defense against the disease, but doses for the coming season already are being rationed due to an interruption in the supply. As a result, both antiviral medications and influenza rapid tests may play a larger role in the coming epidemic than they have in the past. Although the rapid tests for influenza may be valuable for alerting physicians to the presence of influenza in the community and for distinguishing influenza A from B, in the majority of otherwise healthy children who present with typical flu symptoms during the influenza season, the diagnosis may be established on clinical grounds alone. Once diagnosed, antiviral therapy for uncomplicated influenza in a healthy child is not recommended.
EPs and primary care physicians will be on the front lines of the battle once again. It is important for these physicians to keep themselves informed about influenza and new recommendations as the season progresses. It is also important for these physicians to protect themselves and their patients by rolling up their sleeves for the influenza vaccine as soon as possible. Further information may be obtained by telephone by calling the CDC Voice Information System at 888-232-3228 or by visiting the CDC web site at www.cdc.gov/flu/weekly/fluactivity.htm.
References
1. Glezen WP. Influenza viruses. In: Feigin RD, ed. Textbook of Pediatric Infectious Diseases. Philadelphia:Elsevier Inc, 2004:2252-2269.
2 Betts RF. Influenza. In: Mandel Gl, ed. Principles and Practice of Infectious Diseases. Philadelphia: Churchill Livingstone Inc;1995: 1546-1567.
3. Kobasa D, Takada A, Shinya K, et al. Enhanced virulence of influenza A viruses with the hemagglutinin of the 1918 pandemic virus. Nature 2004;431:703-707.
4. Katz SL. Preventing Influenza: Vaccination Guidelines. In: Importance of vaccinating health care workers against influenza. Infect Dis Children, Feb 2004:8-9.
5. Cox NJ, Subbarao K. Influenza. Lancet 1999;354:1277-1282.
6 . Glezen WP. Serious morbidity and mortality associated with influenza epidemics. Epidemiol Rev 1982;4:25-44.
7. Centers for Disease Control and Prevention. Prevention and Control of Influenza: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR May 2004;53(RR-6).
8. Wilde JA, McMillan JA, Serwint J, et al. Effectiveness of influenza vaccine in health care professionals: A randomized trial. JAMA 1999;281:908-913.
9 . Neuzil KM, Zhu Y, Griffin MR, et al. Burden of interpandemic influenza in children younger than 5 years: A 25-year prospective study. J Infect Dis 2002;185:147-152.
10. Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004;292:1333-1340.
11. Boivin G, Hardy I, Tellier G, et al. Predicting influenza infections during epidemics with the use of clinical case definition. Clin Infect Dis 2000;31:1166-1169.
12. Monto AS, Gravenstein S, Elliott M, et al. Clinical signs and symptoms predicting influenza infection. Arch Intern Med 2000;160: 3243-3247.
13. Whitley RJ, Hayden FG, Reisinger KS, et al. Oral oseltamivir treatment of influenza in children. Pediatr Infect Dis J 2001;20:127-133.
14. Peltola V, Ziegler T, Ruuskanen O. Influenza A and B virus infections in children. Clin Infect Dis 2003;36:299-305.
15. Wilde JA. Rapid diagnostic testing for the identification of respiratory agents in the emergency department. Clin Ped Emerg Med 2002;3: 181-190.
16. Noyola DE, Demmler GJ. Effect of rapid diagnosis on management of influenza A infections. Pediatr Infect Dis J 2000;19:303-307.
17. Woo PCY, Chiu SS, Wing-Hong S, et al. Cost-effectiveness of rapid diagnosis of viral respiratory tract infections in pediatric patients. J Clin Micro 1997;35:1579-1581.
18. Adcock PM, Stout GG, Hauck MA, et al. Effect of rapid viral diagnosis on the management of children hospitalized with lower respiratory tract infection. Pediatr Infect Dis J 1997;16:842-846.
19. Bonner AB, Monroe KW, Talley LI, et al. Impact of the rapid diagnosis of influenza on physician decision-making and patient management in the pediatric emergency department: Results of a randomized, prospective, controlled trial. Pediatrics 2003;112:363-367.
20. American Academy of Pediatrics. Influenza. In: Pickering LK, ed. 2003 Red Book: Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics, 2003:351-359.
21. Tominack RL, Hayden FG. Rimantadine hydrochloride and amantadine hydrochloride use in influenza A virus infections. Infect Dis Clin North Am 1987;1:459-478.
22. Wintermeyer SM, Nahata MC. Rimantadine: A clinical perspective. Ann Pharmacother 1995;29:299-310.
23. Thompson J, Fleet W, Lawrence E, et al. Comparison of acetaminophen and rimantadine in the treatment of influenza A infection in children. J Med Virol 1987;21:249-255.
24. Hall CB, Dolin R, Gala CL, et al. Children with influenza A infection: Treatment with rimantadine. Pediatrics 1987;80:275-282.
25. Hayden FG, Sperber SJ, Belshe BR, et al. Recovery of drug-resistant influenza A virus during therapeutic use of rimantadine. Antimicrob Agents Chemother 1991;35:1741-1747.
26. Hayden FG, Belshe RB, Clover RD, et al. Emergence and apparent transmission of rimantadine-resistant influenza A virus in families. New Engl J Med 1989;321:1696-1702.
27. Uyeki TM. Influenza diagnosis and treatment in children: A review of studies on clinically useful tests and antiviral treatment for influenza. Pediatr Infect Dis J 2003; 22:164-177.
28. Subbarao K. As good as the real thing (commentary). J Peds 2000; 136: 139-141.
29. Belshe RB, Gruber WC, Mendelman PM, et al. Efficacy of vaccination with live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine against a variant (A/Sydney) not contained in the vaccine. J Peds 2000;136:168-175.
30. American Academy of Pediatrics, Committee on Infectious Diseases. Recommendations for influenza immunization of children. Pediatrics 2004;113:1441-1447.
31 Centers for Disease Control and Prevention. Interim Influenza vaccination recommendations, 2004-2005 influenza season. MMWR Oct 2004;53:923-924.
32. Centers for Disease Control and Prevention. Official CDC Health Advisory. Health Alert Network, Oct 12 2004. Available at: www.phppo.cdc.gov/HAN/ArchiveSys/.
33. American Academy of Pediatrics, Executive Committee statement Oct. 5, 2004.
34. Gaglani M, Riggs M, Jamenicky C, et al. Computerized reminder strategy is effective for annual influenza immunization of children with asthma or reactive airway disease. Pediatr Infect Dis J 2001;20: 1155-1160.
35. Centers for Disease Control and Prevention. Update: Influenza activity US and worldwide 2003-2004 season and composition of the 2004-2005 influenza vaccine. MMWR July 2004;53:547-552.
36. Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncitial virus in the United States. JAMA 2003;289:179-186.
37. Centers for Disease Control and Prevention. Update: Influenza activity United States 2003-2004 season. MMWR Dec 2003;52:1197-1202.
38. Martinez-Aguilar G, Hammerman WA, Mason EO, et al. Clindamycin treatment of invasive infections caused by community-acquired, methicillin-resistant and methicillin-susceptible Staphylococcus aureus in children. Pediatr Infect Dis J 2003;22:593-598.
39. Barrett TW, Moran GJ. Update on emerging infections: News from the Centers for Disease Control and Prevention. Ann Emerg Med 2004;43:43-47.
40. Bhat N. Preliminary Update: Influenza-associated deaths and encephalopathy among children <18 years — United States, 2003-04 influenza season. Oral presentation. Meeting of the Advisory Committee on Immunization Practices; 2004 June 23-24; Atlanta, GA.
41. Shortridge KF. The next pandemic influenza virus? Lancet 1995; 346:1210-1212.
42. Nicholson KG, Wood JM, Zambon M. Influenza. Lancet 2003;362: 1733-1745.
43. Trampuz A, Prabhu RM, Smith TF, et al. Avian Influenza: A new pandemic threat? Mayo Clin Proc 2004;79:523-530.
44. Chan PK. Outbreak of avian influenza A (H5N1) virus infection in Hong Kong in 1997. Clin Infect Dis 2002;34:S58-64.
45. Katz JM, Lim W, Bridges CB, et al. Antibody response in individuals infected with avian influenza A (H5N1) viruses and detection of anti-H5 antibody among household and social contacts. J Infect Dis 1999;180:1763-1770.
46. Bridges CB, Katz JM, Seto WH, et al. Risk of influenza A (H5N1)infection among health care workers exposed to patients with influenza A (H5N1), Hong Kong. J Infect Dis 2000;181: 344-348.
47. Tran TH, Nguyen T, Nguyen TD, et al. Avian influenza A (H5N1) in 10 patients in Vietnam. N Engl J Med 2004;350:1179-1188.
48. Chotpitayasunondh T, Lochindarat S, Srisan P, et al. Cases of influenza A (H5N1) in Thailand. MMWR 2004;53:100-103.
With the media currently focused on vaccine shortages, the emergency physician must be prepared to rationally and scientifically explain diagnostic and therapeutic approaches in children with influenza. The author of this article prepares the ED physician to confidently face the 2004-2005 influenza season.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
