Polyunsaturated Fatty Acids and Skin Cancer: Part I
Polyunsaturated Fatty Acids and Skin Cancer: Part I
By Anthony P. Albino, PhD and Leonard A. Cohen, PhD
In many cases, the long-term prognosis for individuals with advanced cancers is dismal because of the lack of effective treatment options. New approaches are needed to control and suppress tumor progression. The stimulus for studies on the anticancer role of n-3 polyunsaturated fatty acids (PUFAs) comes from epidemiological studies conducted in the early 1980s of Greenland Eskimos and Japanese farmers, that confirm an association between low cancer incidence rates and a high intake of n-3 PUFAs.1,2 These initial epidemiological observations, which suggested that dietary n-3 PUFAs play important roles in inhibiting the evolution and/or progression of a broad range of human cancers, simply tallied the total fish oil intake of individuals.3 Subsequent studies using controlled model systems provided tantalizing evidence that n-3 PUFAs can moderate or reverse a diverse set of molecular mechanisms that are deranged during tumor progression of many cancer types and can increase the efficacy of various therapeutic modalities. Using dietary intervention to modify both the relative consumption of essential PUFAs and the n-3:n-6 ratio, PUFAs may be a potentially potent chemopreventive and adjuvant antitumor modality.4
Background
Among the four families of PUFAs (n-9, n-7, n-6, n-3; so termed because of the position of the first double bond from the methyl end of the molecules), the n-6 and n-3 (also called omega-6 and omega-3) are essential fatty acids that cannot be synthesized by humans and therefore, must be obtained from the diet. In Western diets the 18-carbon linoleic acid (c18:2, n-6) is the most abundant n-6 PUFA. Vegetable oils (e.g., sunflower, safflower, and corn oil) are particularly rich sources of n-6 PUFAs. In contrast, the most common n-3 PUFAs in the human diet are a-linolenic acid (aLA; c18:3, n-3; found in canola and soybean oils, some nuts, and flaxseed), and eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) (both found in high levels in cold water fish such as mackerel, menhaden, salmon, and tuna). In addition, aLA also can be desaturated and elongated to form EPA (20:5, n-3) and DHA (22:6, n-3) in the body (see Figure 1).
 |
The rationale for using n-3 PUFAs as antitumor agents is based on epidemiological and experimental data that support both a suppressive role of dietary lipids in all phases of tumor progression, and a therapeutic role when used in combination with conventional treatment modalities. Epidemiological studies show that the stage-for-stage survival of patients with one of several different common cancers, including those of the skin, is consistently greater in countries with low dietary fat intake but high intake of n-3 PUFAs (e.g., Japan) vs. Western countries in which there is high fat intake but low n-3 PUFA intake.2,5 A wide range of studies also support the conclusion that diets high in n-6 PUFAs play a key role in the growth and/or progression of many common human cancers, including breast, prostate, and skin (i.e., non-melanoma skin cancers), whereas diets high in n-3 PUFAs attenuate various aspects of these cancers.6,7 Animal experiments clearly demonstrate the enhancing effects of diets high in n-6 PUFAs on ultraviolet radiation (UVR)-induced skin neoplasms and the inhibitory effects of diets high in n-3 PUFA.8
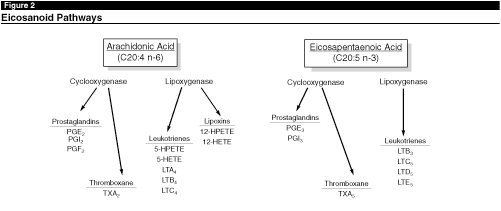
Both in vivo and in vitro experiments with a range of human cancers have demonstrated that specific n-3 PUFAs can increase the efficacy of chemotherapeutic treatment and radiation therapy.9,10 In addition to non-melanoma skin cancers, a growing body of data indicate that dietary fat influences the development and progression of cutaneous malignant melanoma, with the n-6 PUFAs exerting stimulatory effects and n-3 PUFAs having inhibitory activity.11-13 For example, a diet supplemented with DHA, or the closely related EPA, inhibits the cyclooxygenase- and lipoxygenase-catalyzed formation of downstream eicosanoids (see Figures 1 and 2) and can suppress the growth, invasiveness, and metastatic potential of melanoma cells in a dose-dependent manner.14,15 Thus, n-3 PUFAs have the potential to attenuate several of the most relevant and aberrant characteristics of tumor cells (i.e., deregulated cell-cycle control, development of invasive and metastatic potential, and inhibition of apoptotic circuits).
 |
Role of Nutrition in Non-Melanoma Skin Cancers
A direct relationship between UVR-induced cancers of the skin and diet has long been recognized. In 1939, Baumann and Rusch first observed that rodents fed high levels of dietary fat formed UVR-induced tumors more rapidly than animals receiving less fat.16 More recently, it has been shown that high dietary fat influences both the initiation and promotion stages of UVR-induced tumors. Using UV-irradiated hairless mice, Black and colleagues observed a convincing linear relationship between increased lipid level intake and the numbers of UVR-induced skin tumors per mouse.17 Increased lipid intake also resulted in a decrease in the tumor latency in these mice. Moreover, after UVR-initiation, switching to a low-fat diet abrogated the exacerbating effects seen with high dietary fat intake.8
The observation that dietary fatty acids can directly influence the development and progression of UVR-induced skin cancers provided a strong rationale for a clinical intervention trial to determine the potential of dietary fat modification as a prevention strategy for both skin cancers and clinically identifiable precursor lesions.6,8 Several clinical trials have confirmed that a reduction in fat intake plays an important role in reducing the incidence of premalignant and malignant skin lesions. For example, in a two-year clinical intervention trial, 76 patients were randomized to either a control group (approximately 38% of calories from fat) or to a low-fat dietary intervention group (20% calories from fat).18 The patients on the low-fat diet had a 66% reduction in actinic keratosis, a premalignant lesion with a high conversion rate to squamous cell carcinoma. In a second trial, 101 patients with either squamous or basal cell carcinoma experienced a large decrease in occurrence of new skin cancers in the low-fat intervention group (20% of calories from fat) as compared to the control group which remained on a high-fat diet (35-40% of calories from fat).6 Thus, these clinical studies support the conclusion that a reduction in premalignant and malignant skin lesions can be achieved by reducing dietary fat, and that dietary manipulation can play an important role in the management and prevention of the most common forms of skin cancers (i.e., basal and squamous cell carcinomas).
Role of Nutrition in Cutaneous Malignant Melanoma
Similar to non-melanoma skin cancers, there is a growing body of data indicating that nutrition, dietary fat, and specific PUFAs influence the development, progression, and treatment of cutaneous malignant melanoma. In 1987, Mackie et al analyzed 100 melanoma patients and matched controls for constituent fatty acids in samples of subcutaneous adipose tissue.12 Compared to the controls, melanoma patients had substantially increased percentages of n-6 PUFAs. The supposition of this study was that increased consumption of dietary n-6 PUFAs had a contributory effect in the etiology of melanoma. A recent epidemiology study of more than 50,000 men and women with cutaneous malignant melanoma indicated that n-6 PUFA intake was associated with a significantly increased risk of melanoma in women.11 A case-control study showed that obesity was significantly related to melanoma risk, indicating that a high proportion of calories from fat may facilitate melanoma development.19 A population-based control study suggested that high intake of fish oils and n-3 fatty acids reduces the risk of melanoma.13 A study comparing five-year survival rates of patients with stage I, II, III, or IV melanoma who consumed a low-fat diet (in addition to other nutrients) to those on a standard diet found a considerable stage-for-stage survival increase for melanoma patients on a low-fat diet.20
Conclusion
These studies, though not dismissing excessive UVR exposure as the major risk factor, suggest a complicated and, as yet unclear, contributory role for dietary fat in the evolution and progression of melanoma. Moreover, these studies also suggest that dietary fat can play a significant role in the clinical management of melanoma. To date, however, no studies similar to those examining the role of dietary fat and specific PUFAs in non-melanoma skin cancers have been performed in individuals with mela-noma or at high risk for developing melanoma by virtue of having increased numbers of normal or atypical nevi. (Dr. Albino is the Director of Research and Dr. Cohen is the Section Head, Nutrition and Endocrinology, The American Health Institute, Valhalla, NY.)
References
1. Sinclair HM. The relative importance of essential fatty acids of the linoleic and linolenic families: Studies with an Eskimo diet. Prog Lipid Res 1981;20:897-899.
2. Iso H, Sato S, Folsom AR, et al. Serum fatty acids and fish intake in rural Japanese, urban Japanese, Japanese Americans, and Caucasian American men. Int J Epidemiol 1989;18:374-381.
3. Ackman RG. The absorption of fish oils and concentrates. Lipids 1992;27:858-862.
4. Budowski P, Crawford MA. a-Linolenic acid as a regulator of the metabolism of arachidonic acid: Dietary implications of the ratio n-6:n-3 fatty acids. Proc Nutr Soc 1985;44:221-229.
5. Wynder EL, Fujita Y, Harris S, et al. Comparative epidemiology of cancer between the United States and Japan. A second look. Cancer 1991;67:746-763.
6. Black HS, Thornby JI, Wolf JE Jr, et al. Evidence that a low-fat diet reduces the occurrence of non-melanoma skin cancer. Int J Cancer 1995;62:165-169.
7. Rose DP. Effects of dietary fatty acids on breast and prostate cancers: Evidence from in vitro experiments and animal studies. Am J Clin Nutr 1997;66(suppl): 1513S-1522S.
8. Black HS, Thornby JI, Gerguis J, et al. Influence of dietary omega-6, -3 fatty acid sources on the initiation and promotion stages of photocarcinogenesis. Photo-chem Photobiol 1992;56:195-199.
9. Burns CP, Wagner BA. Effects of exogenous lipids on cancer and cancer chemotherapy. Implications for treatment. Drug Saf 1993;8:57-68.
10. Jiang WG, Bryce RP, Horrobin DF. Essential fatty acids: Molecular and cellular basis of their anti-cancer action and clinical implications. Crit Rev Oncol Hematol 1998;27:179-209.
11. Veierod MB, Thelle DS, Laake P. Diet and risk of cutaneous malignant melanoma: A prospective study of 50,757 Norwegian men and women. Int J Cancer 1997;71:600-604.
12. Mackie BS, Mackie LE, Curtin LD, et al. Melanoma and dietary lipids. Nutr Cancer 1987;9:219-226.
13. Bain C, Green A, Siskind V, et al. Diet and melanoma: An exploratory case-control study. Ann Epidemiol 1993;3:235-238.
14. Reich R, Royce L, Martin GR. Eicosapentaenoic acid reduces the invasive and metastatic activities of malignant tumor cells. Biochem Biophys Res Commun 1989;160:559-564.
15. Abbott WG, Tezabwala B, Bennett M, et al. Melanoma lung metastases and cytolytic effector cells in mice fed antioxidant-balanced corn oil or fish oil diets. Nat Immun 1994;13:15-28.
16. Baumann CA, Rusch HP. Effect of diet on tumors induced by ultraviolet light. Amer J Cancer 1939;35: 213-221.
17. Black HS, Lenger W, Phelps A, et al. Influence of dietary lipid upon ultraviolet light-carcinogenesis. J Environ Pathol Toxicol Oncol 1984;5:271-282.
18. Black HS, Herd JA, Goldberg LH, et al. Effect of a low-fat diet on the incidence of actinic keratosis. N Engl J Med 1994;330:1272-1275.
19. Kirkpatrick CS, White E, Lee JA. Case-control study of malignant melanoma in Washington State: Diet, alcohol, and obesity. Am J Epidemiol 1994;139: 869-880.
20. Hildenbrand GL, Hildenbrand LC, Bradford K, et al. Five-year survival rates of melanoma patients treated by diet therapy after the manner of Gerson: A retrospective review. Altern Ther Health Med 1995;1:29-37.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
