Effects of Conjugated Equine Estrogen in Postmenopausal Women with Hysterectomy: The Women’s Health Initiative Randomized Controlled Trial
Effects of Conjugated Equine Estrogen in Postmenopausal Women with Hysterectomy: The Women’s Health Initiative Randomized Controlled Trial
Abstract & Commentary
Synopsis: The use of CEE conferred protection against fracture without an increase in breast cancer but with an increased risk of hypertension, stroke, and venous thromboembolism.
Source: The WHI Steering Committee. JAMA. 2004;291:1701-1712.
The goal of this arm of the whi trial was to determine whether a standard oral 0.625 mg dose of conjugated equine estrogen (CEE) would confer cardioprotection when given to hysterectomized women with common health burdens typical for their age. A total of 10739 women were enrolled between 1993 and 1998. At the time of study termination, about half of the women in each arm (placebo or CEE) had already stopped taking their assigned medication. At enrollment, more than 80% of the women had had a hysterectomy before age 50, but only 40% of the enrollees had had a bilateral oophorectomy. About 45% of the women were in the age range 60-69 years and 25% were between 70-79 years old at the time of enrollment. The oldest women were enrolled first and thus were followed the longest. In the survival curves, the age of the cohort in the last years was most likely older than the mean age of the entire study population. The authors do not
comment of how this aspect of the study design might have skewed the results. Because the data were analyzed by intention-to-treat, the survival curves also include those who were noncompliant. Interestingly, while CEE appeared to reduce cardiovascular risk in women between ages 50-59 years, this is the group with the shortest duration of follow-up and least number of events, so therefore the group for which there was the least power.
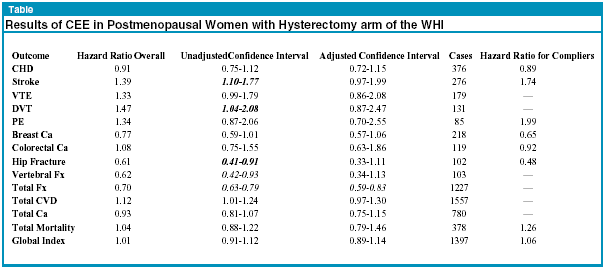
For quick reference, the overall findings are summarized in the Table below. The hazard ratios for the compliant group are also included when given in the text. The most robust finding is that CEE provide significant protection against fractures. An increase in the risk of stroke and VTE was noted. Breast cancer risk was not increased and could be interpreted as decreased.
Comment by Sarah L. Berga, MD
Those who enjoy ambiguity and complexity will certainly be rewarded by the release of the most recent WHI data. However, this ambiguity presents an additional challenge to those whose job it is to explain the results to already bewildered women. Where do these latest data leave us as practitioners?
Taking the results of the estrogen-only arm of the WHI at face value and assuming that they are generally applicable to unselected women using an oral estrogen product, one can feel confident that CEE protects against fractures. However, one will be left having to explain why the breast cancer results differed from those in the combined estrogen-progestin arm of the WHI that received so much attention when it was released about 2 years ago. One wonders if the difference is explained by the use of progestagens in general or medroxyprogesterone acetate in particular. However, it is true that the 2 populations were somewhat different in health burden and thus technically it is not proper to directly compare the results of the 2 arms. For breast cancer risk, the negative impact of progestins has been shown in other studies, including the controversial Million Woman Study published in the Lancet in August of 2003. Although not widely acknowledged, several studies have shown a trend for reduced breast cancer risk in estrogen-only users.
The question about the impact of progestagens upon various tissues and disease processes remains largely unanswered by the trial, although an original aim of the study was to determine if progestagens counteracted the benefits of estrogen upon cardiovascular risk. The PEPI trial attempted to determine the impact of different progestagens, including micronized progesterone delivered orally, but the trial size was much smaller and had minimal power to detect meaningful differences among progestagens. So the question about how different progestagens impact overall risk, especially for cardiovascular events and breast cancer, remains open. There is evidence to suggest that progesterone causes vasodilation whereas other progestagens cause modest vasoconstriction. One can imagine a scenario whereby small risks (such as the combination of mild hypertension, mild hypercoagulability, and mild vasoconstriction) synergistically amplify the impact of each other, especially in older women with pre-existing cardiovascular burden, but this notion has received minimal direct investigative attention.
A finding from the present study that has not received much attention is that systolic blood pressure at 1 year was higher by a mean of 1.1 mg Hg in women taking CEE and remained similarly elevated throughout follow-up. Diastolic blood pressure did not differ between the two groups. The authors note in the discussion that the small but persistent increase in systolic blood pressure is one possible contributor to the increased risk of stroke and lack of cardiovascular benefit in the older age groups. The risk of stroke would have likely been further increased by the pro-coagulant effect of oral CEE, which is amply demonstrated by the increased risks of deep-vein thrombosis (DVT) and pulmonary embolism(PE). One is tempted to conclude that the present study design reveals the worst case scenario, that is, what happens when an oral estrogen is given without titration to women regardless of age, years since menopause, symptomatology, or overall health burden.
Like all good studies, this one raises more questions than it answers. Such is the piecemeal pace of progress. One cannot help wonder what would happen if the VTE risk were eliminated or reduced by using a transdermal approach (Scarabin P, et al. Lancet. 2003;362:428-32) and titrating the dose to the lowest level needed to control symptoms. A nonoral route would also be less likely to raise systolic blood pressure. Might the benefits be preserved and the risks ameliorated or eliminated to move the overall global index in favor of benefit by use of a low dose of estradiol delivered nonorally? Total mortality was similar in the placebo and CEE arms in the present study. Is there a way to improve mortality by reducing untoward consequences, such as eliminating stroke and VTE events? Are there particular women who might especially benefit from the use of estrogens postmenopausally and, if so, what are their characteristics?
The hypothesis that estrogen use would synergize with a healthy lifestyle to confer long-term health benefits has not been directly tested in this or any other randomized trial, although earlier observational studies suggested that this might be the case. While the question about neuroprotective effects of exogenous estrogen and progestagens remains open, one would predict that any preparation that causes hypercoagulability and increases the rate of stroke would lead to increased rates of dementia. As noted recently, it is practically impossible to separate vascular and other forms of dementia (Casserly I, Topol E. Convergence of atherosclerosis and Alzheimer’s disease: inflammation, cholesterol, and misfolded proteins. Lancet. 2004;363:1139-1146). Given these considerations, one would predict that neuroprotection most likely might be seen with a nonoral delivery of estradiol.
Thoughts About the WHI
Leon Speroff, MD, adds additional commentary.
The published results of the women’s health Initiative agree with more than 20 years of case-control and cohort data with the exception of the cardiovascular results. Clinicians and patients should regard this as good news. With the exception of breast cancer, the results from the estrogen-only arm of the clinical trial are essentially identical to those in the estrogen-progestin arm.
The report of neither a beneficial effect nor an adverse effect on coronary heart disease gives the impression that the estrogen-only arm represents an improvement over the estrogen-progestin arm. However, the updated results, after central adjudication of diagnoses, indicating an increased risk of coronary heart disease from the canceled estrogen-progestin arm of the WHI did not achieve statistical significance.1 Indeed, only the first year results were statistically significant in the year-by-year analysis, a conclusion based on a difference of only 19 cases. In the subgroup analysis, only the women who were 20 or more years distant from menopause when they started treatment had a statistically significant increased risk of coronary heart disease (1.71; 95% CI, 1.20-2.50). Subtracting this group from the rest of the participants, coronary heart disease now was observed in an identical prevalence comparing the treated and placebo groups.
In both clinical arms of the WHI, there was an indication that younger postmenopausal women treated with hormones had a decreasing risk of coronary heart disease over time. The test for trend was significant in both the estrogen-progestin and estrogen-only arms. However, the high dropout rate of about 50% in both arms hampered this analysis because of decreasing numbers over time.
Therefore the 2 arms of the WHI are not so dissimilar in regard to coronary heart disease. Like coronary heart disease, it is reasonable to expect the stroke and vascular dementia (not Alzheimer’s disease) data to reflect the effect of hormone therapy given to an older group of women many years distant from menopause. In the canceled estrogen-progestin arm, the only increase in dementia was in the women who were 75 years and older when they started treatment. It is not appropriate to conclude that hormone therapy increases the risk of coronary heart disease, and perhaps stroke, in all post-menopausal women; this conclusion applies only to a specific older group of women.
The WHI concluded (as did many individuals and organizations) that postmenopausal hormone therapy is not a viable intervention for primary prevention of coronary heart disease. We cannot quarrel with the uniform conclusions in a series of secondary prevention trials that hormone therapy does not reduce or slow the progression of established coronary heart disease. The results provide a reasonably solid basis not to recommend postmenopausal hormone therapy for women with existing atherosclerosis in the anticipation of preventing future cardiovascular events. The results also indicate that there is no need to avoid the use of medroxyprogesterone acetate, because there is no clinical difference observed comparing women treated only with estrogen to those treated with estrogen and progestin. Certainly the case for primary prevention of coronary heart disease merits controversy; however, at the same time, the issue is not settled. In my view, there continues to be good reason to believe that hormone therapy can have a beneficial role in the primary prevention of coronary heart disease.
The most important unanswered question in regard to breast cancer is whether postmenopausal hormone therapy initiates the growth of new breast cancers or whether the epidemiologic results reflect an impact on preexisting tumors. Observations that favor an impact on preexisting tumors include: 1) in the studies reporting an increase in risk, the evidence is apparent relatively rapidly, within a few years; 2) the return of the risk ratio in the WHI estrogen-progestin arm almost to 1.0 in year 6, and in the observational data, a return to baseline immediately after discontinuing therapy; 3) no difference in noninvasive breast cancers in the WHI; and 4) the large body of literature documenting lower grade and stage disease in hormone users, resulting in better survival rates.
In contrast, the WHI results in the estrogen-progestin arm indicated an earlier appearance of worse tumors than previously reported in case-control and cohort studies. And of course, the estrogen-only arm of the WHI indicated a reduced risk of breast cancer, although it failed to reach statistical significance. It is important to keep in mind that the participants in the 2 arms of the WHI were not identical.2 In regard to risk factors for breast cancer, the women in the estrogen-only arm experienced more births and bilateral oophorectomy and more and longer duration of previous hormone therapy. It is possible that earlier and greater use of hormone therapy before participation in the study identified those individuals with preexisting tumors who were then excluded from participation, accounting for the lower incidence of breast cancer in the treated group. Postmenopausal hormone therapy is either associated with a small increase in the risk of breast cancer or it affects preexisting tumors. The different results reported by the WHI in regard to tumor characteristics are a puzzle and may reflect the older age of the participants or variations in diagnosis and management.
It is tempting to compare the results obtained in the two arms of the WHI. But there are important differences.2 The estrogen-only arm had 5859 fewer participants, making it more susceptible to a loss of statistical power with the increasing dropout rate that reached 53.8% over time. In addition to the differences in breast cancer risk factors already mentioned, the women in the estrogen-only arm were more obese, less active, and had more preexisting cardiovascular disease. Therefore, these were 2 different trials with two different populations and treatments, making direct comparisons inappropriate.
The overall news from the WHI has been presented in many forums and in the media in a pessimistic, exaggerated fashion. There are critical questions still unanswered, explanations for adverse results that disagree with previous reports, and impressive good news (protection against fractures and colorectal cancer). In my view, the results of the WHI do not preclude clinicians and patients from making individualized decisions that support postmenopausal hormone therapy.
References
1. Manson JE, et al. N Eng J Med. 2003;349:523-534.
2. Stefanick ML, et al. Ann Epidemiol. 2003;13:S78-S86.
Sarah L. Berga, MD, James Robert McCord Professor and Chair, Department of Gynecology and Obstetrics, Emory University School of Medicine, and Leon Speroff, MD, Professor of Obstetrics and Gynecology, Oregon Health, Sciences University, Portland, are both Associate Editors of OB/GYN Clinical Alert.
The use of CEE conferred protection against fracture without an increase in breast cancer but with an increased risk of hypertension, stroke, and venous thromboembolism.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
