Pulmonary Embolism: Recent Advances in Diagnosis and Treatment Modalities, Part I
Part I — Epidemiology, Risk Factors, and Pathophysiology
Author: Marianne Haughey, MD, Assistant Professor of Emergency Medicine, Jacobi Hospital, Albert Einstein College of Medicine, Bronx, NY.
Peer Reviewers: Brian K. Snyder, MD, Assistant Clinical Professor of Medicine, Department of Emergency Medicine, University of California, San Diego; and Albert C. Weihl, MD, FACEP, Assistant Professor of Surgery-Emergency Medicine, Department of Surgery, Yale University School of Medicine, New Haven, CT.
Pulmonary embolism (PE) is an illness that frequently presents with nonspecific symptoms, that affects people of all ages and stages of life, and that is difficult to diagnose with available tests. Unfortunately, it also is the third most common cause of death in the United States,1 and many patients with PE present to emergency departments (EDs) across the country each day.
The diagnosis of PE rarely is made in a straightforward manner, and when the diagnosis is clear, the patient usually is extremely ill. In diagnosing PE, emergency physicians must consider a variety of factors, all of which must be weighed carefully to determine whether the patient has PE, a potentially fatal illness for which effective treatments are available.
A number of advances in both the treatment and diagnosis of thromboembolic disease have been made during the past decade. Several diagnostic tests commonly are used today that were not available even five years ago. The first article in this two-part series will cover the epidemiology of PE, the factors that increase a patient’s risk for the disease, and the pathophysiology and clinical features of PE. In addition, complicated issues regarding the diagnosis of PE and the controversies involved will be addressed. The second part of the series will examine the differential diagnosis of PE, its treatment, and prevention of the disease.—The Editor
Epidemiology
PE is a relatively common and potentially life-threatening disease. The diagnosis of PE is missed more than 400,000 times per year, and 100,000 of those patients may have lived if they had received the appropriate treatment.1 PE is the third most common cause of death in the United States, and at least 650,000 cases of the disease occur each year.2-5
Ten percent of patients with PE die within the first hour.4 One-third of the patients who survive that first hour eventually will be diagnosed and treated, and two-thirds will remain undiagnosed.2 One-third of the undiagnosed group will die, usually due to recurrent embolism and right heart failure.4 Identification of patients with PE is the essential first step in improving the survival statistics. In one study, 45% of patients who died of PE had the correct diagnosis suspected premortem.6 The correct clinical diagnosis was made even less often in those with a diagnosis of myocardial infarction (MI), coronary artery disease (CAD), or chronic obstructive pulmonary disease (COPD). The same study found that only 47% of the patients with a suspected diagnosis of PE were anticoagulated with proper dosages.6 Of the group that is diagnosed and treated, only one in 12 will die of massive PE or PE complications.4,7
Venothromboembolic disease, which includes deep vein thrombosis (DVT) as well as PE, accounts for 250,000 hospitalizations per year and about 50,000 deaths from recognized events.8-11 DVT and PE are two points on the continuum of thromboembolic disease. In fact, there is much more crossover than previously thought. Asymptomatic PE occurs in 50% or more of patients with DVT, and DVT is found in 80% of patients with PE at autopsy.12-14
One study found a positive trend in the prevention and treatment of thromboembolic disease. The age-adjusted mortality rates for PE in Minneapolis/St. Paul decreased 50% for adults ages 30-84 during the period of 1980-1995.15 This trend was paralleled by declining hospital discharge rates for phlebitis and thrombophlebitis. It also is possible that this may reflect a changing natural history of the disease.
Pathophysiology
The basic pathophysiology of PE involves material that becomes lodged in the pulmonary vasculature. This material, most commonly blood thrombus, does not permit normal pulmonary blood flow into an area of the lung. This causes the ventilated area of the lung to become essentially ventilatory dead space.2 A blockage of one part of the pulmonary vasculature results in an increase in pulmonary vascular resistance, which increases pulmonary arterial pressures and right ventricular pressures.2 Pulmonary hypertension occurs, and the patient develops a ventilation perfusion mismatch. If the obstruction is large enough, the patient also can become hypoxic.16 The hemodynamic effects of embolism depend on the amount of pulmonary vasculature that is occluded.
The pO2 and pCO2 may vary during different stages of PE severity. Increased ventilation rate actually can increase the PO2 and decrease the PCO2 in smaller pulmonary emboli. Larger emboli can cause enough of a shunt that a significant amount of blood is unable to exchange oxygen or carbon dioxide, and the PO2 can decrease while the PCO2 increases.2
PE does not always present in a uniform manner. Patients with a large clot load may present in shock with hemodynamic collapse. On the other end of the spectrum, some patients may present more subtly with a smaller clot load and normal vital signs. Between these two extremes of presentation is a group of patients that presents with normal blood pressure, but if an echocardiogram is done, it can show the right ventricle under strain.17 If 50% of the pulmonary vasculature is blocked, significant pulmonary hypertension and acute cor pulmonale can occur, which, if not detected, can cause long-term morbidity or death.2
Thrombus is the most common cause of pulmonary emboli, and the most common sources of these emboli are clots in other parts of the deep venous system. Often, the main concern for most patients is not that the clot already in the pulmonary circulation will be fatal, but rather that the possible next clot could pose a danger.
Clots most commonly originate in the deep venous system of the legs, as evidenced by the fact that 70% of patients with PE are found by venography to have clot in the lower extremity.2 Clots can embolize from the iliac, common femoral, and popliteal veins. The significant risk of clot embolizing from the veins in the calf often is discounted; however, about one-third of those with pulmonary emboli have a calf thrombus as the source for the emboli.2,18 In addition, thrombus in the calf is likely to grow and extend, therefore becoming a larger thrombus that may be more dangerous to the patient if it embolizes.
Upper extremity DVT can embolize to become a PE. This type of DVT forms more often in those with malignancy or with central venous lines.19 Pelvic veins also can be a source for thrombus, and may be missed in a search for a lower extremity DVT.
While thrombus is the most common material to embolize, air embolisms, amniotic fluid embolisms, and fat embolisms also have occurred. Other material, such as bullets and tumors, embolizes even more rarely. These nonthrombotic emboli require the usual supportive and interventional care, but they do not benefit from the anticoagulation treatment given to patients with thrombotic emboli.2
Risk Factors
In 1858, Virchow established that there were three main reasons for the pathogenesis of thrombus: venous stasis, intimal injury, and hypercoagulability.2 The risk factors for thrombus formation, and hence PE, still may be categorized neatly within the umbrella of Virchow’s triad. These risk factors may occur alone or in combination. (See Table 1.)
|
Venous stasis is the reason for the increase in thromboembolic disease among patients with prolonged immobility as a result of medical illness, burns, trauma, or surgery.20 Patients who have experienced strokes or paralysis also are at high risk. Congestive heart failure (CHF) is another significant risk factor. Venous stasis may be the reason for the increased thromboembolic events in people who travel long distances, especially on long airline flights.21,22 The hyperviscosity syndromes, varicose veins, and pelvic tumors may cause increased venous stasis and are risk factors for thromboembolism as well.20,22 Obesity has been found to be an independent risk factor for VTE.23-25 History of prior DVT increases venous stasis, and patients with proximal DVT who receive inadequate anticoagulation treatment have a 20-50% risk of recurrent VTE.20
Intimal injury can occur as a result of trauma or as an iatrogenic by-product of care. Central lines or intravenous drug abuse can contribute to a thromboembolic event. Injury to the vessel may result from orthopedic surgery, especially hip or knee surgery, or vascular surgery. Chemotherapeutic agents also have been shown to cause intimal damage.20,22
Hypercoagulability results from alteration of the clotting mechanisms. Malignancy, trauma, burns, and hormone replacement therapy are acquired causes of hypercoagulability.20,26 The duration of pregnancy and the postpartum period are times of increased risk of thrombosis, and this is thought to be due to a state of hypercoagulability.22,27,28
In addition to these acquired states of hypercoagulability, there are inherited hypercoaguable states. These are rare causes for VTE but, if present, they are concerning as the patient is at risk for multiple recurrences. The more common of these rare, inherited states include protein C deficiency, protein S deficiency, antithrombin III deficiency, Factor V Leiden mutation, antiphospholipid antibodies, hyperhomocysteinemia, and high factor VII.
The risk of VTE also increases with age.20 The reasons in this heterogeneous group are probably multiple, including a higher incidence of co-morbid conditions. Morbidity and mortality also increase with age.20 The higher percentage of patients with underlying cardiopulmonary disease makes it more difficult to exclude those illnesses from the differential diagnosis list.20
Finally, the most significant risk factor for PE is the presence of a current DVT.
Clinical Manifestations
The history and physical exam in a patient with PE do not confirm the diagnosis; they merely raise the suspicion of such a diagnosis, triggering further investigation. A patient with PE may present as severely ill or in cardiac arrest;29 those who present in this manner may be among the 10% of patients with PE who will die within the first hour.2 The other 90% of patients may present more subtly, with nonspecific symptoms.
The history may help elucidate whether the patient has any of the risk factors listed above, which may be the strongest indicator that the clinician should proceed with additional examinations. Other findings in the history may include dyspnea, pleuritic chest pain, cough, leg swelling, hemoptysis, wheezing, palpitations, or anxiety.2,14,20 (See Table 2.)
|
Dyspnea is the most common symptom and is found in 70-90% of those found to have PE.14 Although dyspnea is a sensitive sign for PE, it clearly is not specific. Sudden onset dyspnea may be more specific for PE.30,31
Chest pain is the second most common symptom of PE and is present in 49-85% of patients, with pleuritic pain being more common.31-33 Pulmonary infarction near the innervation of the pleural nerves is thought to be the reason for pleuritic chest pain in a patient with PE.14 Massive emboli and centrally lodged emboli are less likely to cause pleuritic chest pain.14
Cough, hemoptysis, sweating, and syncope are less common than the above symptoms. Hemoptysis occurs when a small embolus travels to the periphery of the pulmonary vasculature, resulting in infarction and hemorrhage. Syncope can result when a very large embolus increases the pulmonary resistance and, therefore, the afterload in the right ventricle, thus decreasing the blood flow return to the left ventricle. The right ventricle also increases in size somewhat, impinging on the left ventricle. Cardiac output decreases, and the result is syncope. Syncope occurs more commonly in massive PE, from 17% to 35% of the time.32-34
Signs of PE commonly include tachycardia, which occurs up to 71% of the time, and tachypnea, which occurs 92% of the time.14 Rales occur in about 50% of patients, and a low-grade fever can be found in 30-54% of patients.14 Because many patients with PE have a DVT at autopsy, the signs of leg swelling also may be found in those with PE. (In fact, only 17-35% of patients with non-massive PE are found to have leg swelling.)14 Other signs demonstrate the right heart strain that occurs in large or massive PEs. These signs include jugular venous distension, right ventricle heave, circulatory collapse, and an increase in the intensity of the pulmonic component of the second heart sound. (See Table 3.)
|
Diagnosis
The diagnosis of PE frequently remains elusive despite the advances in testing that have been developed during the past decade. Emergency physicians have stated that this is one of the diagnoses for which they would most like to see a decision rule created.35 There is no single test with adequate sensitivity and specificity that can be provided for the patient in a minimally invasive manner and that is available to the ED physician 24 hours per day.
A multitude of exams has been studied and used in the diagnosis of PE. Some of these exams have been used to evaluate other diagnoses on the differential diagnosis list. Others are an attempt to screen for PE. Still others are used to confirm a highly suspected PE. These tests include electrocardiograms (ECGs), arterial blood gases, chest radiographs, computed tomography (CT) scans, ventilation/perfusion scans, lower extremity or upper extremity ultrasound, pulmonary angiograms, and D-dimer products. Pretest clinical suspicion also has been accorded a significant weight by many authors in their studies. Unfortunately, the fact that there are a vast number of exams in use demonstrates that each has a significant weakness. There is no one test with adequate sensitivities or specificities for absolute inclusion or exclusion from treatment. Yet, each also has strengths in certain clinical situations. Multiple authors now have suggested that these tests may be used in combination, with a more powerful result than may be achieved by each individual exam.36-40 In combination, the tests can be used very constructively to establish who should be treated and who may have alternate diagnoses.
Exams for PE may be divided into screening tests and confirmatory tests, as well as tests that are helpful in either excluding or confirming alternate diagnoses. Screening tests ideally have a high rate of sensitivity, and confirmatory tests ideally have a high rate of specificity.
Clinical Evaluation
Multiple authors now suggest that the pursuit of a diagnosis of PE utilize a decision tree system. The first step in each decision tree is the clinician’s pretest evaluation of the probability of PE as the diagnosis.41 PE is not a clinical diagnosis, yet the degree of suspicion held by the clinician has been shown to be significant clinically. The majority of authors, however, have not specified what criteria clinicians should use to come up with their pretest probability. Wells and Wicki each have developed separate systems for objectively evaluating the clinical probability of PE.42,43 (See Table 4 for Wells’ criteria.)
|
Each of the scoring systems places the patient into a low, intermediate, or high probability group. Both sets of criteria include risks such as recent surgery, previous thromboembolic event, and tachycardia. Wicki additionally considers age, hypocapnia, hypoxemia, and chest x-ray findings. Wells scores factors such as signs of DVT, immobilization, hemoptysis, malignancy, and whether PE is considered as likely or more likely than an alternate diagnosis by the treating physician. Wicki’s score found that 10.3% of those considered to be low probability had PE. Of those found to be at intermediate risk, 38% had PE, and of those scored to be high probability, 81% had PE. A total of 49% of the patients were in the low probability group, thus permitting a large portion of those suspected to proceed through a less intensive arm of a diagnostic decision tree.43
D-Dimer. D-dimer is a fibrin degradation product that circulates in the bloodstream of patients with a dissolving fibrin clot.44-46 D-dimer usually begins to circulate within an hour of thrombus formation and can be found to circulate for up to about a week after thrombus formation, as the continued fibrinolysis of clot continues to elevate the D-dimer level.47-51 D-dimer has a circulating half-life of 4-6 hours, so the levels decrease over time.45 After seven days, false-negative test results are more likely as the D-dimer normalizes.50,52,53
D-dimer, when used well, is a wonderful tool in helping the physician determine whether to treat a patient suspected of having a thromboembolic event. As with all of the other exams available, it is essential to understand the strengths and limitations of the exam. The initial point to understand is that the best use of any D-dimer is to exclude the disease in low probability patients. There is not adequate evidence to use a negative result to exclude the disease in patients whose clinical probability is intermediate or high.36 In addition, a positive test has low specificity, with the most specific test only having a 20-50% specificity.46,54,55
A significant limitation of the D-dimer exam is that there are a heterogeneous collection of exams with varied sensitivities and specificities. It is essential for emergency physicians to understand which exam is available at their institution. There now are multiple suggested protocols for diagnosing PE via a decision tree.36-39 They almost universally use a negative D-dimer test as a method to screen out patients who clinically are considered to have a low probability of PE. The D-dimer test used in these studies is the enzyme-linked immunoabsorbent assay (ELISA) technique. ELISA tests that show a normal D-dimer level have excellent negative predictive values of 91-98%.51,56-58 A D-dimer level less than 500 ng/mL on an ELISA test has been found to have a 93% sensitivity in predicting a normal angiogram result.51 Unfortunately, the ELISA test is not the more commonly available exam, and in fact it has other limitations that make it less attractive for rapid diagnosis in the ED. The drawbacks to the ELISA D-dimer test are that it is technician-dependent, expensive, run in batches, and inefficient for clinical use.45,59
The semiquantitative latex agglutination (LA) technique is a more commonly found and rapid technique. The LA assays are more widely available but they are not as sensitive as the ELISA assays. A normal LA D-dimer level alone or with V/Q scan results is not recommended to preclude the treatment of possible PE.44,45 The LA exam has been found to have a sensitivity of 70%.41 The SimpliRED exam is another commonly studied D-dimer exam. It is a whole blood agglutination assay and is easier and faster to perform than the ELISA-based tests. It is a bedside exam that can be performed in two minutes.44 The sensitivity of SimpliRED was 68-85%, and the negative predictive value was 81% for VTE.41,60,61 But the SimpliRED test may have more of a role in those with a low pretest probability of PE or a nondiagnostic V/Q scan. The sensitivity of this exam in low probability PE patients is 99-100%.36,44,60
There is an interesting problem in introducing a highly sensitive yet poorly specific screening exam into the diagnostic decision tree. When a rapid D-dimer (SimpliRED) test was introduced to a study ward, the number of V/Q scans done increased because the number of patients screened for PE increased. There also was an increase in those who were diagnosed as having thromboembolic disease.46 While more VTE was diagnosed, there was no change in any three-month outcomes.46
Several second-generation rapid D-dimer tests are available. They work via an erythrocyte agglutination assay, a turbidimetric assay, the rapid ELISA test, and an immunofiltration assay. It is incumbent upon emergency physicians to determine which exam with which level of sensitivity is available at their institution to frame the results of the exam in the proper context. COPD does not affect the accuracy of the D-dimer test, although many other illnesses lead to false positives.62
Arterial Blood Gases. Arterial blood gases (ABG) and the A-a gradient have been used in the past in the attempt to diagnose pulmonary emboli. Unfortunately, the sensitivity and specificity of the exam are both too low to make it a useful test for diagnosis.44,63 Many patients who are suspected of having a PE have other pulmonary pathology, which, therefore, decreases the specificity of the exam. The sensitivity of the exam also is poor, as it takes only one or two extra breaths per minute to make the PaO2 normal even when the pulmonary gas exchange is significantly impaired.2 In a population of classically symptomatic patients with angiographically proven PE, at least 17% will have a normal PO2, and one in 20 will have a PaO2 greater than 100 mmhg on room air.64,65 A completely normal ABG may be found in up to 23% of patients with known PE. There is no role for pulse oximetry in the diagnosis of PE.2
Certainly, an ABG may provide useful information about a patient who is critically ill the need for additional oxygenation, intubation, or choices of treatment. A large PE may cause a significant A-a gradient; however, A-a gradient is nonspecific and insensitive.
Electrocardiogram. A massive PE that causes severe pulmonary hypertension can produce specific ECG abnormalities. Sub-massive PE manifests a wide spectrum of nonspecific changes on surface ECG, ranging from no abnormality to multiple disturbances of rate, rhythm, and conduction pattern.66,67 Common ECG findings in those with PE include tachycardia, nonspecific ST-T wave abnormalities, atrial fibrillation, right axis deviation, left axis deviation, S1-S2-S3, S1-Qlll-Tlll, and p-pulmonale.2 ECG frequently is relegated to screening for other cardiac abnormalities in patients with PE.68 One study found that inverted t waves in leads V1-V4 was the most common finding on ECG, and was found in 68% of patients with angiographically confirmed PE. The study also found 9% of those with PE had normal ECGs.69 Another study looked at multiple studies and found that normal ECGs were noted in 10-46% of patients with angiographically proven PE.70 New-onset atrial fibrillation must encourage the physician to consider PE as the cause.
Right ventricular strain on ECG may have clinical importance in determining the severity of the strain and, hence, the severity of pulmonary hypertension. In a patient who has PE, the severity of strain may correspond with the presence of the right ventricular dysfunction. One study produced a scoring system to grade the severity of pulmonary hypertension from PE.71 (See Table 5.) A score of 10 or greater was found to be highly suggestive of severe pulmonary hypertension from PE.
|
Sreeram et al examined ECGs of patients with and without PE using the criteria in Table 6. The authors suspected PE if three or more of the listed findings were present. The sensitivity of their study was 82%.72 The specificity of ECG is poor for PE, and its sensitivity certainly is not adequate. The purpose of an ECG is to evaluate for other causes of the patient’s symptoms, especially myocardial infarction ischemia and pericarditis.
|
Chest Radiographs. Chest radiographs are important to obtain in the patient thought to have PE. Chest x-rays are essential in making the diagnosis of other respiratory illnesses that may masquerade as PE.
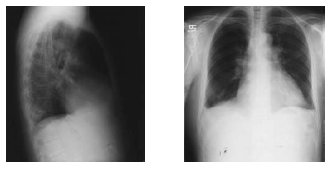
There are nonspecific findings that are associated with PE on chest x-ray, including an elevated hemidiaphragm, focal infiltrates, pleural effusions, or atelectasis. In addition, there are some eponymous signs for PE that can be found on chest x-ray, including Hampton’s hump and Westermark’s sign. Hampton’s hump is a triangular pleural-based infiltrate with the apex toward the hilum, commonly adjacent to the diaphragm. Westermark’s sign shows up as a dilation of the pulmonary vessels proximal to the embolism along with the collapse of more distal vessels. This also is called oligemia or regional pulmonary paucity. Unfortunately, all of these signs are nonspecific and insensitive.73 In addition, 30% of those with PE have a normal chest x-ray.74 (See Figure 1.)
|
Another important issue regarding the chest x-ray is that it is helpful in interpreting the V/Q scan.63,75 In subjects investigated for PE, an abnormal chest x-ray increases the prevalence of non-diagnostic lung scans. A normal pre-test chest x-ray more often is associated with a definitive (normal or high) probability lung scan result. The chest radiograph may be useful in deciding the optimum sequence of investigations.75
Echocardiography. Transesophageal echocardiography (TEE) is a bedside test that has been used to evaluate right heart strain and to attempt to identify clots in those suspected of having PE. TEE has been found to be more sensitive than transthoracic echocardography for the diagnosis of PE.
TEE fails to identify some 50% of patients with angiographically proven PE.76 Although echo findings of right ventricular strain, paired with a high clinical suspicion, support a diagnosis of PE, TEE has to have a better sensitivity to be used as a screening tool to rule out PE.
The role of echocardiography in PE may be more appropriately directed to evaluating the patient with known PE. Patients can present in hemodynamic embarrassment or with an apparently stable exam. There seems to be a group of patients who present with stable vital signs, yet who already have increased right ventricular strain and may be on the verge of decompensation. An echo may be used to identify this deceptively relatively well-appearing group before their clinical decompensation occurs.
DVT Evaluation. Because about 80% of PEs originate in the lower extremities, an evaluation for DVT can be an extremely useful test in the diagnostic workup for PE.63,77 It is significant to realize that thromboembolism is a continuum of illness, and that the important issue first may be to diagnose the presence of a clot and then its location. This becomes even more important as the outpatient treatment of DVT with low molecular weight heparins (LMWHs) becomes more common. It must be recognized that only 42% of patients with PE and DVT have any symptoms of DVT.77
Venous ultrasound (US) has a sensitivity of about 95% and a specificity of about 96% in patients with an initial episode of DVT. In isolated calf vein DVT, the sensitivity is about 73%. About one-fourth of isolated distal DVTs will extend proximally, so repeating the US after about one week is indicated after a single normal proximal venous US study. The risk of subsequent venous thromboembolism is very low (< 2% during six months of follow-up), provided that US of the proximal veins remains normal for one week in a patient with suspected DVT or for two weeks in someone with suspected PE (in a patient who also has a nondiagnostic lung scan).78
A negative single bilateral lower extremity venous ultrasound exam is not a reliable method of excluding PE in outpatients with nondiagnostic V/Q scans.79
One study has examined the use of serial impedance plethysmography in those who have nondiagnostic lung scans and adequate cardiopulmonary reserve to diagnose PE. The leg study was done on presentation and, if negative, was repeated on days 2, 3, and 5 or 7, 10, and 14. The study found this to be a safe manner of evaluating for PE in stable patients.38
CT Scans. In 1992, the first study was published comparing CT angiogram (CTa) to a pulmonary angiogram.80 The use of helical CT has risen dramatically as more EDs have gained easier access to this diagnostic tool than to angiograms. Helical CT scans are fast, with the ability to scan from the aortic knob to the diaphragm in 20-30 seconds.81,82 They have the potential to show images of the pulmonary vasculature from the right and left main pulmonary arteries to at least the segmental and possibly subsegmental arteries.83 Using 2- to 3-mm sections is superior to using 5-mm sections. The study requires 120 mL of ionic or nonionic contrast.44 An intraluminal filling defect or vascular occlusion characterizes a positive result.44 False positives are possible with tortuous or oblique segmental arteries and kinetic effects from breathing.83,84 Specificity ranges from 89-100% and sensitivity is 64-93%.85,86 The CTa can be viewed as having an overall test accuracy that is equal to or slightly superior to that of the V/Q scan, but a CTa scan that is read as negative for PE is not as sensitive as a V/Q scan read as normal.44
Subsegmental vessels are difficult to identify and evaluate for PE.44 This consistently has been presented as a significant disadvantage of CTa. It has been suggested that it may not be necessary to diagnose PE with the certainty of a pulmonary angiogram. Isolated subsegmental PEs appear to be uncommon, representing only 6% of all PEs.52
CTa is recommended for those who have a positive screening test or as an initial test in those who have conditions that cause nondiagnostic V/Qs, such as heavy smoking, chronic pulmonary disease, or pulmonary infiltrate.87 It also can be useful in those with a high pretest clinical probability but indeterminate V/Q scans.88
The sensitivity of CTa for PE is too low to use it to rule out PE without other tests. One study looked at the rates of PE in those patients who had a negative CTa after a non-diagnostic V/Q and negative leg ultrasounds. The researchers found an overall false-negative rate for PE of 5% in the three months following their exams.81 The false-negative rate included patients who had syndromes meriting a high clinical suspicion but negative CTa, but who had an angiography that was diagnostic for PE at initial presentation. Also included in the false negative rate were patients who only had a low or moderate clinical suspicion, and therefore no initial angiography, but who were diagnosed with PE in the three months following the initial presentation.81 Other studies have found a 2-5% follow-up thrombosis rate.89-91 The interobserver agreement on the CTa readings is high,86 and is much higher than the interobserver agreement found in the Prospective Investigation of Pulmonary Embolus Diagnosis (PIOPED) study in reading V/Qs.92 It has been suggested that CTa might replace pulmonary angiogram in the combined strategies that use US and V/Q scanning.86,93 CTa can be found to confirm an alternate diagnosis in 11-67% of patients.86 A meta-analysis of 277 patients showed only 4% with inconclusive results from CTa.36 Others show up to a 10% indeterminate reading on the CTa.2 This percentage still is far less than the 60-75% of indeterminate readings obtained with a V/Q scan.92 Thrombi in the main pulmonary arteries mostly were true positives, while 15% of those at the lobar and 38% of those at the segmental level were false positives.86 There also is the possibility, not yet widely practiced, of using the CT to evaluate the pelvic and thigh veins as well as the pulmonary vasculature.94 The true clinical utility of this approach has yet to be evaluated.
The advantages of CTa are that it is fast, frequently available, relatively specific, and can be helpful in demonstrating other lung pathology. Disadvantages include the fact that the patient may have to leave the department for the test, there is a significant dye load, and the test is not sensitive enough to use alone. CTa is a powerful tool in the diagnosis of PE, but only when used with an understanding of its limitations.
V/Q. Ventilation-perfusion (V/Q) studies are among the most commonly ordered exams used to diagnose PE. A V/Q test requires injection of a gamma radioactive isotope into the patient. The patient must leave the ED for the test, which may be impossible for a critically ill patient. V/Q examines only the lungs and usually is not helpful in evaluating other diagnoses.
V/Q scans in PIOPED (in which they were compared against the findings of pulmonary angiograms) had variable rates of sensitivity and specificity, depending on their reading. The scans were read as normal, very low probability, low probability, intermediate probability, and high probability. The sensitivity of a high probability scan was only 47%, but the specificity was 97%. The negative predictive value of a near normal/normal scan is 91%, and that of a low probability scan is 84%.92 Other authors have recommended that all other scan readings be considered indeterminate.75 An abnormal chest x-ray increases the prevalence of non-diagnostic scans. A normal pretest chest radiograph more often is associated with a normal or high probability scan (definitive scan).75
Lung scanning is diagnostic in only 25-40% of patients with a reading of normal/near normal or of high probability.38,39,92,95 This is a serious limitation of the exam. It is imperative to remember to pursue other diagnostic routes when a non-diagnostic scan is obtained. Another drawback is that V/Q scans may be difficult if not impossible to obtain in some institutions during off-hours.
It also has been shown that when the readings are done, they are most clear and accurate when a standardized conclusion is included. Examples included high probability, no conclusion, or diagnosis excluded.22 Given the difficulty of diagnosing PE, physicians should make it as clear as possible when results are obtained.
Angiography. Pulmonary angiography is considered to be the definitive gold standard in the evaluation for PE. Dye is injected into the pulmonary vascular tree (which has 26 branchings of the pulmonary vessels). If a defect is seen intraluminally, or if the dye abruptly is cut off, the presumption is that PE is occluding the vessel. This is not an infallible exam. Objects other than thrombus, including extraluminal tumor, can cause the vessel to appear occluded. Even a well-performed pulmonary angiogram cannot detect material in vessels below the third branch point. This corresponds with lobular arteries. As we have discussed, small emboli may herald the imminent arrival of the larger, life-threatening emboli.
Angiography is a highly accurate tool in the diagnosis of PE. Interobserver agreement for positive angiograms is 89%, and it is 83% for negative studies. Agreement was found to be more concordant as the size of the vessels involved increased.96 In fact, the agreement between readers for the main or lobar arteries is the best, with 98% agreement. The agreement for readers of clots involving the segmental arteries (and not larger vessels) was 90%, and the agreement level was 68% in those clots that involved only the subsegmental arteries.97
In 1990, the PIOPED study used the pulmonary angiogram as the gold standard in its classic comparison of V/Q scans to angiograms, yet even a patient with a normal pulmonary angiogram can expect a 2.2% chance of a venous thromboembolic event within a year.96 An angiogram also is invasive and frequently difficult to arrange in some institutions on a 24-hour basis. Some studies cannot be completed for technical reasons. In addition, if lytic therapy is used, the angiogram may be normal within 90 minutes of therapy.
Risks for the procedure include allergic reaction to the contrast, vascular injury, cardiac dysrythmias, endocardial injury, or perforation. Clearly, some of these injuries can lead to significant morbidity or death. Even given the multiple manners in which the risk expresses itself, the PIOPED data demonstrate a mortality rate of 0.5%.2
Combination Strategies
It is apparent that any individual test other than a pulmonary angiogram lacks either the sensitivity or specificity to use as a single instrument in determining whether to treat a patient for PE. It also is important to note that treatment for PE is not benign, and that the risk of bleeding from treatment with unfractionated heparin, LMWH, or thrombolysis is significant and cannot be dismissed. Likewise, the surgical placement of a filter has its own inherent risks.
To diagnose PE more accurately, multiple authors have begun to suggest using a decision tree approach rather than a single exam. The initial exams used are those that have a high sensitivity, while the later exams test a smaller group with more specific tests. Pulmonary angiogram is performed on fewer people, but is available if the rest of the analysis does not provide an adequate answer.
Some authors begin their decision trees with an interpretation of the clinician’s pretest clinical probability. (See Figures 2a and 2b for two strategies.) In those patients considered to have a low pretest clinical probability of PE, a D-dimer (an ELISA D-dimer) is used and, if negative, precludes further testing for PE. If positive, or if the pretest clinical probability is moderate or high, further testing is pursued.
In one strategy for evaluating PE, lower extremity ultrasound is the next level of testing which, depending on the results, might be followed by a V/Q scan, then a spiral CT angiography with contrast (not requiring catheterization), and then, if needed, angiography.36 Another approach includes duplex ultrasound if there is high clinical probability for DVT, but otherwise uses a V/Q or spiral CT as the next testing level for either moderate or higher clinical probability of PE or a positive D-dimer (an ELISA D-dimer test) in a patient considered to have a low pretest clinical probability.37 Further testing may require a determination of the patient’s cardiac reserve in evaluating treatment or follow-up. It has been found that in patients who are clinically suspicious for PE yet have nondiagnostic workups, including negative DVT studies of the legs, who also have good cardiac reserve, it is safe to follow them with outpatient duplexes of the legs during the next 14 days.38 This is not to say that they do not have PE, because as many as 18% of patients with a high clinical suspicion of PE but a low probability lung scan and negative compression ultrasonography of the lower extremities will be found to have PE.40 Wolfe uses further outpatient testing in those determined to be stable with a nondiagnostic V/Q scan, a positive D-dimer, and a negative duplex at the time of presentation.
Each of these algorithms attempts to determine which patients need treatment as early in the strategy as possible while minimizing the need for pulmonary angiograms. They are complete and attempt to address all possible presentations of PE.
Another study looked at those patients with nondiagnostic lung scans and negative leg DVT exams on the day of presentation and found that they may be followed safely as outpatients with serial impedance plethysmography studies on days 2, 3, 5, 7, and 10. Anticoagulation was withheld in those whose exams remained negative. The author found this to be a safe method of evaluation, but clearly it is limited in the ED setting if the patient lacks access to ongoing care.38
Summary
PE is a common, potentially lethal disease that is under-diagnosed and under-treated. The second part of this two-part series will address treatment methods and the differential diagnosis that should be considered.
References
1. Kim V, Spandorfer J. Epidemiology of venous thromboembolic disease. Emerg Med Clin 2001;19:839-859.
2. Fried C. Pulmonary embolism. In: Rosen P, et al, eds. Emergency Medicine, 4th ed. St. Louis: Mosby; 1998:1770-1805.
3. Freiman DG, Suyemoto J, Wessler S. Frequency of pulmonary thromboembolism in man. N Engl J Med 1965;272:1278.
4. Dalen JE, Alpert JS. Natural history of pulmonary embolism. Prog Cardiovasc Dis 1975;17:257.
5. Ginsberg JS. Management of venous thromboembolism. N Engl J Med 1996;335:1816-1828.
6. Pineda LA, Hathwar VS, Grant BJ. Clinical suspicion of fatal pulmonary embolism. Chest 2001;120:791-795.
7. Carson JL, Kelley MA, Duff A, et al. The clinical course of pulmonary embolism. N Engl J Med 1992;326:1240-1245.
8. Kniffin WD, Baron JA, Barrett J, et al. The epidemiology of diagnosed pulmonary embolism and deep venous thrombosis in the elderly. Arch Inter Med 1994;154:861-866.
9. Anderson FA, Wheeler HB, Goldberg RJ, et al. A population based perspective of the hospital incidence and case fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study. Arch Intern Med 1991;151:933-938.
10. Silverstein MD, Heit JA, Mohr DN, et al. Trends in the incidence of deep vein thrombosis and pulmonary embolism: A 25-year population-based study. Arch Intern Med 1998;158:585-593.
11. Hirsh J, Hoak J. Management of deep vein thrombosis and pulmonary embolism. A statement for health care professionals. Council on Thrombosis (in consultation with the Council on Cardiovascular Radiology). American Heart Association. Circulation 1996;93: 2212-2245.
12. Moser KM, LeMoine JR. Is embolic risk conditioned by location of deep venous thrombosis? Ann Intern Med 1981;94:439-444.
13. Huisman MV, Buller HR, ten Cate JW, et al. Unexpected high prevalence of silent pulmonary embolism in patients with deep venous thrombosis. Chest 1989;95:498-502.
14. Lee LC, Shah K. Clinical manifestations of pulmonary embolism. Emerg Med Clin 2001;19:925-942.
15. Janke RM, McGovern PG, Folsom AR. Mortality, hospital discharges, and case fatality for pulmonary embolism in the Twin Cities: 1980-1995. J Clin Epidemiol 2000;53:103-109.
16. Santolicandro A, Prediletto R, Fornai E, et al. Mechanisms of hypoxemia and hypocapnia in pulmonary embolism. Am J Respir Crit Care Med 1995; 152:336-347.
17. Ribeiro A, Lindmarker P, Juhlin-Dannfelt A, et al. Echocardiography Doppler in pulmonary embolism: Right ventricular dysfunction as a predictor of mortality rate. Am Heart J 1997;134:479-487.
18. Havig O. Deep vein thrombosis and pulmonary embolism: An autopsy study with multiple regression analysis of possible risk factors. Acta Chir Scand 1997;478:1-120.
19. Prandoni P, Polistena P, Bernardi E, et al. Upper-extremity deep vein thrombosis. Arch Intern Med 1997;157:57-56.
20. Berman AR. Pulmonary embolism in the elderly. Clin Geriatric Med 2001; 17:107-130.
21. Lapostolle F, Surget V, Borron S, et al. Severe pulmonary embolism associated with air travel. N Engl J Med 2001;345:779-783.
22. Ferrari E, Chevallier, T, Chapelier, A, et al. Travel as a risk factor for venous thromboembolic disease: A case-controlled study. Chest 1999;115:440-444.
23. Goldhaber SZ, Grodstein F, Stampfer MJ, et al. A prospective study of risk factors for pulmonary embolism in women. JAMA 1997;277:642-645.
24. Goldhaber SZ, Savage D, Garrison RJ, et al. Risk factors for pulmonary embolism: The Framingham study. Am J Med 1983;74:1023-1028.
25. Hansson P, Eriksson H, Welin L, et al. Smoking and abdominal obesity: Risk factors for venous thromboembolism among middle-aged men, "The Study of Men Born in 1913." Arch Intern Med 1999;159:1886-1890.
26. Douketis JD, Ginsberg JS, Holbrook A, et al. A reevaluation of the risk for venous thromboembolism with the use of oral contraceptives and hormone replacement therapy. Arch Intern Med 1997;157:1522-1529.
27. Coon WW. Epidemiology of venous thromboembolism. Ann Surg 1977;187: 149-164.
28. James KV, Lohr JM, Deshmukh RM, et al. Venous thrombotic complications of pregnancy. Cardiovasc Surg 1996;4:777-782.
29. Kurkciyan I, Meron G, Sterz F, et al. Pulmonary embolism as cause of cardiac arrest. Arch Intern Med 2000;160:1529-1535.
30. Manganelli D, Palla A, Donnamaria V, et al. Clinical features of pulmonary embolism. Chest 1995;107( 1 suppl):25S-32S.
31. Miniati M, Prediletto R, Formichi B, et al. Accuracy of clinical assessment in the diagnosis of pulmonary embolism. Am J Respir Crit Care Med 1999; 159:864-871.
32. Bell WR, Simon TL, Demets DL. The clinical features of submassive and massive pulmonary emboli. Am J Med 1977;62:355-360.
33. Stein PD, Willis PW, Demets DL. History and physical examination in acute pulmonary embolism in patients without preexisting cardiac or pulmonary disease. Am J Cardiol 1981;47:218-223.
34. Janata-Schwatczek K, Weiss K, Riezinger I, et al. Pulmonary embolism: II. Diagnosis and treatment. Semin Thromb Hemost 1996;22:33-52.
35. Grahm I, Steill IG, McAuley L, et al. Potential areas for new clinical decision rules: Comparison of North America and Europe. Acad Emerg Med 1999;6:433.
36. Gallagher EJ. Clots in the lung. Ann Emerg Med 2000;35;181-187.
37. Wolfe TR, Hartswell SC. Pulmonary embolism: Making sense of the diagnostic evaluation. Ann Emerg Med 2001;37:504-514.
38. Hull RD, Raskob GE, Ginsberg JS, et al. A noninvasive strategy for the treatment of patients with suspected pulmonary embolism. Arch Int Med 1994; 154:289-297.
39. Wells PS, Ginsberg JS, Anderson DR, et al. Use of a clinical model for safe management of patients with suspected pulmonary emboli. Ann Int Med 1998;129:997-1005.
40. Meyerovitz MF, Mannting F, Polak JF, et al. Frequency of pulmonary embolism in patients with low probability lung scan and negative lower extremity venous ultrasound. Chest 1999;115:980-982.
41. Rodger M, Jones G, Rasuli P, et. al. Steady-state end-tidal alveolar dead space fraction and D-dimer. Bedside tests to exclude pulmonary embolism. Chest 2001;120:115-119.
42. Wells PS, Anderson DR, Rodger M, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: Management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and D-dimer. Ann Int Med 2001;135:98-107.
43. Wicki J, Perneger T, Junod A. et al. Assessing clinical probability of pulmonary embolism in the emergency ward. Arch Int Med 2001:161;92-97.
44. Kline JA, Johns KL, Colucciello SA, et al. New diagnostic tests for pulmonary embolism. Ann Emerg Med 2000;35:168-180.
45. Kutinsky I, Blakley S, Roche V. Normal D-dimer levels in patients with pulmonary embolism. Arch Intern Med 1999;159:1569-1572.
46. Goldstein NM, Kollef MH, Ward S, et al. The impact of the introduction of a rapid D-dimer assay on the diagnostic evaluation of suspected pulmonary embolism. Arch Int Med 2001;161:567-571.
47. Devine DV, Greenberg CS. Monoclonal antibody to fibrin D-dimer (DD-386) recognizes an epitope on the gamma chain of fragment D. Am J Clin Pathol 1988;89:663-666.
48. Greenberg CS, Devine DV, McCrae KM. Measurement of plasma fibrin D-dimer levels with the use of a monoclonal antibody coupled to latex beads. Am J Clin Pathol 1987;87:94-100.
49. Rowbotham BJ, Vernon JE, Whitaker AN, et al. Plasma cross-linked fibrin degradation products in pulmonary embolism. Thorax 1990;45:684-687.
50. Oger E, Leroyer C, Bressollette L, et al. Evaluation of a new, rapid and quantitative D-dimer test in patients with suspected pulmonary embolism. Am J Respir Crit Care Med 1998;158:65-70.
51. Goldhaber SZ, Simons GR, Elliot G, et al. Quantitative plasma D-dimer levels among patients undergoing pulmonary angiography for suspected pulmonary embolism. JAMA 1993;270:2819-2822.
52. Elms MJ, Bunce IH, Bundensen PG, et al. Rapid detection of cross-linked fibrin degradation products in plasma using monoclonal antibody-coated latex particles. J Clin Pathol 1986;85:360-364.
53. Whitaker AN, Elms MJ, Masci PP, et al. Measurement of cross-linked fibrin derivatives in plasma: An immunoassay using monoclonal antibodies. J Clin Pathol 1984;37:882-887.
54. Bounameaux H, Schneider PA, Reber G, et al. Measurement of Plasma D-dimer for diagnosis of deep vein thrombosis. Am J Clin Pathol 1989;91:82-85.
55. Ginsberg JS, Brill-Edwards PA, Demers C, et al. D-dimer in patients with clinically suspected pulmonary embolism. Chest 1993;104:1679-1684.
56. Hillyard CJ, Blake AS, Wilson K, et al. A latex agglutination assay for D-dimer: evaluation and application to the diagnosis of thrombotic disease. Clin Chem 1987;33:1837-1840.
57. Bounameaux H, Slosman S, de Moerloose P, et al. Diagnostic value of plasma D-dimer in suspected pulmonary embolism. Lancet 1988;2:628-629.
58. Perrier A, Bounameaux H, Morabia A, et al. Diagnosis of pulmonary embolism by a decision tree analysis-based strategy including clinical probability, D-dimer levels, and ultrasonography: A management study. Arch Inter Med 1996;156:531-536.
59. Van Beek EJR, van den Ende B, BerckmansR, et al. A comparative analysis of D-dimer assays in patients with clinically suspect pulmonary embolism. Thromb Haemost 1993;70:408-413.
60. Farrell S, Hayes T, Shaw M. A Negative SimpliRED D-dimer assay result does not exclude the diagnosis of deep vein thrombosis or pulmonary embolism in emergency department patients. Ann Emerg Med 2000;35:121-125.
61. Ginsberg JS, Wells PS, Kearon C, et.al. Sensitivity and specificity of a rapid whole-blood assay for D-dimer in the diagnosis of pulmonary embolism. Ann Int Med 1998;129:1006-1011.
62. Hartmann IJ, Hagen PJ, Melissant CF, et al. Diagnosing acute pulmonary embolism: Effect of chronic obstructive pulmonary disease on the performance of D-dimer testing, ventilation-perfusion scintigraphy, spiral computed tomographic angiography, and conventional angiography. Am J Resp Crit Care Med 2000;62:2232-2237.
63. DA Weiner, SG, Burstein, JL. Nonspecific tests for pulmonary embolism. Emerg Med Clin North Am 2001;19:943-955.
64. UPET: The Urokinase Pulmonary Embolism Trial: A national cooperative study. Circulation 1973;47( Suppl 11):1.
65. USPET: Urokinase Streptokinase Pulmonary Embolism Trial: Phase II results. JAMA 1964;229:1606.
66. Stein PD, Dalen JE, McIntyre KM, et al. The electrocardiogram in acute pulmonary embolism. Prog Cardiovasc Dis 1975;17:247-257.
67. Stein PD, Terrin ML, Hales CA. Clinical, laboratory, roentgenographic and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest 1991;100:598-603.
68. McIntyre K, Sasahara A, Littman D. Relation of the electrocardiogram to hemodynamic alterations in pulmonary embolism. Am J Cardiol 1972;30:205-210.
69. Ferrari E, Imbert A, Chevalier T, et al. The ECG in pulmonary embolism: Prediction value of negative T waves in precordial leads—80 case reports. Chest 1997;111:537-543.
70. Stein PD, Henry JW. Clinical characteristics of patients with acute pulmonary embolism stratified according to their presenting syndromes. Chest 1997;112:974- 979.
71. Daniel KR, Courtney DM, Kline JA. Assessment of cardiac stress from massive pulmonary embolism with 12 lead ECG. Chest 120;474-481.
72. Sreeram N, Cheriex EC, Smeets JL, et al. Value of the 12-lead electrocardiogram at hospital admission in the diagnosis of pulmonary embolism. Am J Cardiol 1994;73;298-303.
73. Greenspan RH, Ravin CE, Polansky SM. Accuracy of the chest radiograph in the diagnosis of pulmonary embolism. Invest Radiol 1984;17:539-543.
74. Simon M. Plain film and angiographic aspects of pulmonary embolism. In: Moser K, Stein, M, eds. Pulmonary Thromboembolism, St Louis: Mosby;1973.
75. Forbes KP, Reid JH, Murchison JT. Do preliminary chest x-ray findings define the optimum role of pulmonary scintigraphy in suspected pulmonary embolism? Clin Radiol 2001;56:397-400.
76. Miniati M, Monti S, Pratali L, et al. Value of transthoracic echocardiography in the diagnosis of pulmonary embolism: Results of a prospective study in unselected patients. Am J Med 2001;110:528-535.
77. Girad P, Musset D, Parent F, et al. High prevalence of detectable deep venous thrombosis in patients with acute PE. Chest 1999;116:903-908.
78. Kearon C, Ginsberg JS, Hirsh J. The role of venous ultrasonography in the diagnosis of deep venous thrombosis and pulmonary embolism. Ann Intern Med 1998;129:1044-1049.
79. Daniel KR, Jackson RE, Kline JA. Utility of lower extremity venous ultrasound scanning in the diagnosis and exclusion of pulmonary embolism in outpatients. Ann Emerg Med 2000;35:547-554.
80. Remy-Jardin M, Remy J, Wattine L, et al. Central pulmonary thromboembolism: Diagnosis with spiral volumetric CT with single breath hold technique: Comparison with pulmonary angiography. Radiology 1992;185:381-387.
81. Ferretti GRF, Bosson JL, Buffaz PD, et al. Acute pulmonary embolism: Role of helical CT in 164 patients with intermediate probability at ventilation-perfusion scintigraphy and normal results at duplex US of the legs. Radiology 1997;205:453-458.
82. Remy-Jardin M, Remy J, Deschildre F, et al. Diagnosis of pulmonary embolism with spiral CT in comparison with pulmonary angiography and scintigraphy. Radiology 1996;200:699-706.
83. Goodman LR, Curtin JJ, Mewissen MW, et al. Detection of pulmonary embolism in patients with unresolved clinical and scintigraphic diagnosis: Helical CT versus angiography. AJR Am J Roentgenol 1995;164:1369-1374.
84. Beigelman C, Chartrand-Lefebure C, Howarth N, et al. Pitfalls in diagnosis of pulmonary embolism with helical angiography. AJR AM J Roentgenol 1998;171:579-585.
85. Mullins MD, Becker DM, Hagspiel KD, et al. The role of spiral volumetric computed tomography in the diagnosis of pulmonary embolism. Arch Intern Med 2000;160:293-298.
86. Perrier A, Howarth N, Didler D, et al. Performance of helical computed tomography in unselected outpatients with suspected pulmonary embolism. Ann Int Med 2001;135 :88-97.
87. Goldberg SN, Palmer EL, Scott JA, et al. Pulmonary embolism: Prediction of the usefulness of initial ventilation-perfusion scanning with chest radiographic findings. Radiology 1994;193:801-805.
88. Ost D, Rozenshtein A, Saffran L, et al. The negative predictive value of spiral CT for the dignosis of PE in patients with nondiagnostic V/Q scans. Am J Med 2001:110;16-21.
89. vanStrijen MJ, de Monye W, Schiereck J, et al. Single-detector helical computed tomography as the primary diagnostic test in suspected pulmonary embolism: A multicenter clinical management study of 510 patients. Ann Int Med 2003;138:307-314.
90. Bourriot K, Couffinhal T, Bernard V, et al. Clinical outcome after a negative spiral CT pulmonary angiographic finding in an inpatient population from cardiology and pneumonology wards. Chest 2003;123:359-365.
91. Musset D, Parent F, Meyer G, et al. Evaluation du Scanner Spirale dans L’Embolie Pulmonaire study group. Diagnostic strategy for patients with suspected pulmonary embolism: A prospective multicenter outcome study. Lancet 2002;14:1914-1920.
92. PIOPED. Value of the ventilation/perfusion scan in acute pulmonary embolism: Results of the prospective investigation of pulmonary embolism diagnosis. JAMA1990;263:2753-2759.
93. Paterson DI, Schwartzman K. Strategies incorporating spiral CT for the diagnosis of acute pulmonary embolism. A cost effective analysis. Chest 2001;119:1791-1800.
94. Bruce D, Loud PA, Klippenstein DL, et al. Combined CT venography and pulmonary angiography: How much venous enhancement is routinely obtained? Am J Roent 2001;176:1281.
95. Perrier A, Bounameaux H, Miron MJ, et al. Using clinical evaluation and lung scan to rule out suspected pulmonary embolism: Is it a valid option in patients with normal results of lower-limb venous compression ultrasonography? Arch Intern Med 2000;160:512-516.
96. Nilsson T, Turen J, Billstrom A, et al. Validity of pulmonary cine arteriography for the diagnosis of pulmonary embolism. Eur Rad 1999;9:276-280.
97. Stein PD, Henry JW, Gottschalk A. Reassessment of pulmonary angiography for the diagnosis of pulmonary embolism: Relation of interpreter agreement to the order of the involved pulmonary arterial branch. Radiology 1999;210: 689-691.
Pulmonary embolism (PE) is an illness that frequently presents with nonspecific symptoms, that affects people of all ages and stages of life, and that is difficult to diagnose with available tests. The first article in this two-part series will cover the epidemiology of PE, the factors that increase a patients risk for the disease, and the pathophysiology and clinical features of PE. In addition, complicated issues regarding the diagnosis of PE and the controversies involved will be addressed.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.