An Update on Pediatric Toxicology
Author: Mark S. Mannenbach, MD, Division Head, Pediatric Emergency Medicine; Assistant Professor, Department of Pediatric and Adolescent Medicine, Mayo Medical Center, Rochester, MN.
Peer Reviewer: Milton Tenenbein, MD, FRCPC, FAAP, FAACT, FACMT, Professor of Pediatrics, Medicine and Pharmacology, University of Manitoba, Winnipeg.
The recent release of the American Academy of Pediatrics (AAP) policy statement regarding the use of ipecac has illustrated the need to continually assess and reevaluate the standard of care to ensure quality care for pediatric patients with a potential ingestion.1 During a 30-year time period, recommendations regarding the treatment of the potentially poisoned pediatric patient have come full circle. The routine use of ipecac, once considered the standard of care, is no longer considered the best approach. The care of the poisoned patient continues to evolve.
What is a clinician to do when faced with a pediatric patient who may have ingested a toxin of some type? Should every patient undergo gastric lavage? Should every patient receive charcoal? Is there a time frame beyond which none of these measures will be helpful? Information on POISINDEX® often seems to suggest the same approach to every patient who may have the potential for harm from a poison exposure. The clinician is left with the challenge of evaluating recommendations and developing a treatment plan for each individual patient. The information to make the right decision for a particular patient often does not exist in written form.
This article provides a review of the most current recommendations for an approach to the potentially poisoned pediatric patient and the latest recommendations for gastrointestinal decontamination. The utility of routine toxicologic testing is discussed also. Each of these areas will be highlighted in case studies to make the information as clinically relevant as possible. —The Editor
Introduction
A 2-year-old boy was brought to an emergency department (ED) in respiratory distress after ingesting an unknown quantity of lamp oil. The child progressively decompensated to hypotension and respiratory failure. He underwent endotracheal intubation and was transferred to a tertiary care center. At the accepting facility, his chest x-ray showed diffuse pulmonary disease (a white out). The child also required vasopressor support and gradually showed improvement in both his blood pressure and his respiratory status. However, by 36 hours after his ingestion, he had fixed pupils and no spontaneous respiratory efforts. Supportive care was withdrawn on the sixth hospital day.
Although an increasingly uncommon scenario, pediatric patients continue to suffer fatal consequences of poisonings in the United States each year. Individuals providing care for the pediatric population continue in their efforts not only to find ways to treat exposed children, but also to avoid exposures from occurring at all. The goal of reducing morbidity and mortality from poisonings in ever-changing environments, both in the home and at the hospital, remains a challenge to all caregivers.
In addition, medical providers are beginning to critically evaluate what therapeutics are available to reduce the harm to children exposed to an ever-increasing variety of toxins. Often medical providers have intervened because they felt they had to do something. But did their intervention have a positive effect, or did they potentially expose a child to further harm?
This article is a review of the current epidemiology of pediatric poisonings in the United States and of the most recent recommendations regarding general evaluation and treatment principles for the poisoned pediatric patient. Finally, some prevention measures are included to encourage medical providers to be involved in reducing the risks to the pediatric patient.
Epidemiology
Data from the annual report of the American Association of Poison Control Centers (AAPCC) show 2,380,028 human exposures during 2002.2 The 64 participating poison control centers reported that 65.7% of exposures involved patients younger than 20 years; 51.6% of exposures involved patients younger than 6 years; and 39.1% involved patients younger than 3 years. These percentages have been relatively stable during the past decade, with 66% of reported exposures in 1990 involving patients younger than 17 years.3 For the pediatric population, a single substance was implicated in 91.9% of reports, and only 2.7% of patients were exposed to more than two possibly poisonous drugs or products. In contrast, 53% of fatal ingestions involved two or more drugs or products.
Fortunately, although children younger than 6 years of age account for the majority of toxic ingestions reported to the AAPCC, children in this age group composed only 2.0% (23) of the fatalities. The percentage of pediatric fatalities related to total pediatric calls has remained unchanged during the last five years at approximately 0.002%. By comparison, in 2002, 0.13% of all adult exposures reported resulted in death.2
However, there have been changes in the predominant cause of pediatric fatalities. The curious toddler who unintentionally ingested a household poison is no longer the primary scenario for a fatal exposure. The deaths in 2002 involved four therapeutic errors, three environmental exposures, three adverse drug reactions, three malicious exposures, and two envenomations.2
The more likely fatal exposure involves the teenage patient. In the 13- to 19-year-old age group, there were 76 fatalities with the most common reasons being suicide (37) and intentional abuse (21). Only four fatalities in this age range were unintentional.
The number of human exposures continues to increase each year along with the potential for both morbidity and mortality. The total number of human exposures has nearly doubled since 1987. The estimated number of exposures per thousand population has, however, remained fairly steady with the estimated number of exposures of 8.5 per thousand population in 1987 and 8.2 per thousand population in 2002. 2
Assessment of a Child with a Potential Ingestion
A father brought his 20-month-old daughter to the ED after she reportedly ingested an unknown amount of an over-the-counter product containing ibuprofen and pseudoephedrine. The father had given her a dose of the medication that morning for her upper respiratory tract infection. He found her with the bottle in her mouth and some of the contents on her clothes as well as on the floor. He brought her to the ED within 35 minutes of the ingestion. She has had no vomiting, agitation, or restlessness prior to arrival. Her vital signs are: temperature 36.8º C; heart rate 112 beats per minute; respiratory rate 24; and blood pressure 98/58 mmHg. She is awake, alert, and cooperative with the exam. Her pupils are 4 mm in size and react well to light. Her mucous membranes are moist. She has some nasal congestion with clear rhinorrhea. Her skin is warm and dry with no rashes. Her lungs are clear. Her pulses are strong and equal throughout with brisk capillary refill time.
What should be done at this point? This child has taken an unknown amount of a combination medication. She is currently without symptoms and has a normal physical exam. In addition, she has come to care promptly.
The ingredient of concern in the medication is pseudoephedrine. A review of information on POISINDEX® regarding the management of pseudoephedrine ingestions shows a familiar approach including gastric lavage and administration of activated charcoal.4 However, a closer look shows some important information regarding the kinetics and range of toxicity for this medication—the majority of cold medications containing this product cause no toxicity. The time to onset of symptoms is reported to be 15-30 minutes with peak effect 30- 45 minutes after ingestion.
This child is without any symptoms after at least 35 minutes. The risks of gastric lavage and administration of charcoal outweigh the benefit in this case. The child could be discharged to home safely with instructions to the father to observe for any symptoms. The father also should be reminded that child-resistant caps should be closed firmly after each use to be effective. He also should be given the phone number for the Poison Control Center for future use. (The national number is (800) 222-1222. A parent’s or caregiver’s call would be routed to the local poison control center.)
In estimating the amount of material ingested, the physician must assume the worst possible scenario, as the exact amount most likely will never be known. If necessary, asking a relative, a prehospital care provider, or the police to bring in the remainder of the medication will allow for a more informed estimation of the amount potentially ingested. This also may allow the providers to verify the active ingredients of the medication, including concentrations. Other medication containers accessible to the child also may be discovered with this approach.
Close attention must be paid to the signs and symptoms of possible ingestion. Direct questioning regarding any change in level of alertness, occurrence of vomiting, and skin changes are some of the detailed information the provider must ask to make an accurate assessment regarding any possible toxicity. During the physical exam, attention must focus upon early identification of any toxidrome. (See Table 1.) Pupillary size, skin temperature, presence or absence of bowel sounds, and hydration status must be monitored closely during observation. By specifically looking at those signs and symptoms, the providers may recognize subtle changes that could prompt intervention or longer periods of observation. Cardiac monitoring should be considered as a routine part of the physical exam of the child with a potential ingestion. Although dysrhythmias generally are uncommon in the pediatric patient population, they may be present without the clinician being readily aware based upon physical exam alone. Certainly, any child with altered mental status requires cardiac monitoring.
Table 1. Toxidromes

Similar to the patient who has suffered a traumatic event, a patient with suspected ingestion should be completely undressed. Attention to skin color, presence or absence of diaphoresis, and presence of skin rashes should be noted in any child who may have been exposed to a toxin.
A case of toxic poisoning from the application of transdermal clonidine was reported in one study.5 A 6-year-old was difficult to arouse after a nap while staying at her grandparents’ house earlier in the day. The child was arousable to verbal stimuli only. The child initially had a heart rate of 54 beats per minute, respiratory rate of 18, blood pressure of 120/72 mmHg, and oxygen saturation of 100% on blow-by oxygen. Her pupils were only 2 mm in size. Her electrolyte and blood glucose levels were within normal limits. She became progressively less interactive, re-sponding only to painful stimuli. She received naloxone without any apparent effect. Gastric lavage was completed with saline, and no pill fragments were returned. A urine drug screen was positive for clonidine and caffeine. She improved during an 18-hour hospitalization in the pediatric intensive care unit. The grandfather later reported that recently he was prescribed clonidine patches for treatment of hypertension. Upon further questioning, the child reported that she had been using the patches as adhesive bandages earlier that day. She had been applying and removing one of them from her extremities during the afternoon. She was discharged without any apparent residual effect.
Any child with altered mental status after a suspected ingestion requires supplemental oxygen and anticipation of definitive airway management. Adequate oxygen supply, suction equipment, oropharyngeal or nasopharyngeal airways, and intubation equipment should be readily available. Bedside measurement of glucose levels and appropriate correction of deficiencies should be readily available as well. (See Table 2.) The administration of naloxone should be considered for any patient with altered mental status and evidence of opioid toxicity. A clinical response to naloxone may allow the patient to maintain his own airway independently and may provide some information regarding the etiology of the patient’s altered mental status as well. Naloxone is given at a dose of 0.1 mg/kg and may need to be repeated several times to see any effect. If a response to naloxone is seen, further doses or a constant infusion of naloxone may need to follow as the effect of the opioid ingested may have a longer half-life than naloxone.
Table 2. Dextrose Administration (0.5 to 1g/kg)
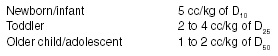
Gastrointestinal Decontamination
A 3-year-old male ingested an unknown quantity of acetaminophen pills. The babysitter found the child with an opened bottle of 160-mg chewable tablets. Apparently, the child-resistant cap had not been secured tightly after a dose had been given to his older sister earlier in the day. The child was found with several pills in his mouth and only two pills remaining in the bottle. The pills had been purchased that morning; only three pills had been given to the sister that afternoon. No pills or pill fragments were found on the floor. The child swallowed the pills that were in his mouth before the babysitter realized what had happened. The child had no vomiting or change in his mental status. There were no other medication containers found open. The babysitter has a supply of syrup of ipecac in the home and wonders if it would be beneficial to the child. The babysitter calls the poison control center for advice. What would you advise the babysitter to do at this point?
The Use of Ipecac
This child has ingested a potentially significant amount of acetaminophen. Assuming there were 30 pills in the original package, there would have been up to 27 tablets available to the child. With only two pills now accounted for, he could have ingested up to 25 tablets or 4,000 mg of acetaminophen. If he weighs 18 kg, he could have ingested more than 200 mg/kg. Clearly, this child is at risk for the toxic effects of acetaminophen.
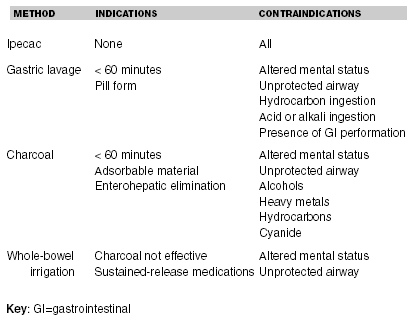
Would the child benefit from ipecac? According to the recent policy statement of the AAP, current advice should be: Tell the babysitter not to administer ipecac.1 In fact, the statement goes one step further and states that ipecac should not be made available routinely in homes. The statement reports that the recommendation for routine availability of ipecac in the home was made as early as 1983, but was not published until 1989 due to the lack of clear efficacy. Recommended as part of an office-based injury prevention strategy in an 1994 AAP statement,6 the use of ipecac in the home was questioned based upon the lack of proven efficacy.
A sample study involving 13 children who were given ipecac as a component of treatment for the ingestion of a potentially toxic amount of a drug found a mean amount of recovery of only 28% with a range of 0% to 78%.7 Several studies evaluated the potential of ipecac removal of a poisonous substance from adult volunteers and showed removal of 51-83% when ipecac was given at 5 minutes and removal of 2-59% when it was given 30 minutes after ingestion.8-12 Results from another study demonstrated no benefit on patient outcome from the administration of ipecac before activated charcoal vs activated charcoal alone, regardless of the time of ipecac administration13
Due to the limited value of ipecac, the American Academy of Clinical Toxicology and the European Association of Poisons Centres and Clinical Toxicologists have issued a position statement regarding its use. A portion of the statement is as follows: "There is no evidence from clinical studies that ipecac improves the outcome of poisoned patients and its routine administration in the emergency department should be abandoned. There are insufficient data to support or exclude ipecac administration soon after poison ingestion."14 This recommendation already appears to be part of routine practice as ipecac was administered in only 0.6% of exposures according to the 2002 AAPCC annual report.2
An argument could be made to use ipecac in the case of potentially serious ingestions such as iron. However, several case reports involving children who have ingested iron tablets point against the value of its use. Tenenbein reported a case of a 17-month-old child who ingested 10 ferrous sulfate tablets. Nine of the tablets were found in the stomach on x-ray. After the child was given ipecac, three episodes of vomiting followed, but a follow-up x-ray revealed that all 10 tablets remained in the stomach.15 A second patient, a 15-month-old child ingested 10-15 ferrous sulfate 300 mg tablets and then had ipecac-induced emesis. Several tablet fragments were recovered in the emesis, but an abdominal x-ray revealed that a large number of iron tablets remained in the stomach. The tablets were found imbedded in the gastric mucosa at the time of an emergency gastrotomy.16
Other concerns about the use of ipecac include its potential for adverse effects including vomiting, lethargy, and diarrhea, making ongoing assessment of patients difficult. The 3-year-old child in the case presented who has ingested a significant amount of acetaminophen clearly requires an evaluation in an ED. Administration of ipecac would delay his transport to the ED and might interfere with the use of charcoal, N-acetylcysteine, or whole-bowel irrigation. The child who has ingested a caustic substance or a hydrocarbon certainly should not receive an emetic due to the potential for further exposure of the esophagus or lungs to the toxin involved. The use of an emetic is certainly contraindicated in any child who is somnolent, or could become somnolent, secondary to the increased risk for aspiration of toxin, gastric contents, or charcoal. More serious complications have been reported after ipecac has been used, including Mallory-Weiss tears, pneumomediastinum, and aspiration pneumonia.
Viewing the potential benefit of the use of ipecac to pediatric patients as a whole, some might argue that ED visits would be decreased with the use of ipecac at home. Investigators who evaluated this issue, found neither improved outcome nor re-duced utilization of emergency services when ipecac was given selectively in the home of the population served by poison control centers.17
A major concern about the availability of ipecac in the home is its use when no benefit would occur. The use of ipecac in the home was intended only after receiving the recommendation of a health care professional. In one study, 61% of children were given ipecac for a nontoxic ingestion.18 Misuse of ipecac by patients with eating disorders is another disadvantage of widespread availability of ipecac in the home.19
Parents or caregivers who suffer from Munchausen-by-proxy syndrome (MBPS) also have been reported to use ipecac to harm children.20 With no clear benefit and with the potential for harm, routine availability and use of ipecac seems to be contraindicated.
Would gastric lavage be beneficial?
Gastric Lavage
Depending upon the length of time required to get a child to the ED following an ingestion, gastric lavage may result in removal of some pill fragments. Results from studies involving adult volunteers have shown limited efficacy of gastric lavage more than 60 minutes after the time of ingestion. Results from one study found that the mean reduction of area under the curve was 32% when lavage was performed at 60 minutes after dosing.21 Results from another study showed the mean reduction of salicylate excretion was only 8%.22 Even when performed fewer than 60 minutes after ingestion, gastric lavage has not been shown to be consistently beneficial. Results from several studies have even suggested that the process of gastric lavage actually may increase drug absorption.23-25 Again, the return of an apparently large amount of pills or pill fragments may be reassuring, but the benefit to the particular patient is questionable. The yield after lavage of liquid preparations is less likely to be of clinical benefit to any patient.
The risk of complications also must be considered before attempting gastric lavage in any individual patient. Aspiration pneumonia can occur, especially in the case of hydrocarbon ingestions. The patient with altered mental status and loss of protective reflexes is at a higher risk of aspiration, and airway protection should be considered prior to lavage attempts. Aspiration has been reported in patients who were alert on presentation.24.26 Other potential complications include laryngospasm,27 hypoxia,28 esophageal perforation,29 and fluid or electrolyte imbalance.30 Absolute contraindications for gastric lavage include patients with loss of airway protective reflexes; ingestion of a hydrocarbon; risk of gastrointestinal perforation; or ingestion of corrosive substances, such as acids or alkalis.
The American Academy of Clinical Toxicology and the European Association of Poisons Centres and Clinical Toxicologists also have issued a position statement regarding the use of gastric lavage. Their statement includes the following recommendation: "Based on experimental and clinical studies, gastric lavage should not be considered unless a patient has ingested a potentially life-threatening amount of a poison and the procedure can be undertaken within 60 minutes of ingestion. Even then, clinical benefit has not been confirmed in controlled studies."31 If attempted, gastric lavage should be performed by an experienced clinician who is able to handle all possible complications, in-cluding the airway management of the pediatric patient. Hegenbarth, et al provide an excellent review of the process of successful and safe gastric lavage.32 Would this child benefit from the use of activated charcoal? If so, how should it be administered? Should a cathartic be given along with the activated charcoal?
Activated Charcoal and Cathartics
The use of a specific treatment for any patient who has ingested a substance should always rest upon whether there is the potential for toxicity. The other question to be answered is: Will activated charcoal adsorb the ingested material? The routine administration of charcoal to all patients who have ingested a substance should be discouraged. The administration of charcoal is not without its risks and potential complications.
Many substances are not adsorbed by charcoal. Alcohol, heavy metals and minerals, and hydrocarbons, are not affected by the use of charcoal. The potential for aspiration is increased when charcoal is given to a patient who has ingested a hydrocarbon. Any patient who has ingested a substance that has the potential for decreasing his/her level of alertness or ability to protect his/her airway from aspiration, probably should not re-ceive activated charcoal.
Givens et al33 report a case of a 14-year-old female who attempted suicide by ingesting a tricyclic antidepressant and a phenothiazine. They report that the patient was "alert but sleepy" prior to undergoing gastric lavage. Multiple pill fragments were returned. The patient then had a grand mal seizure, became apneic, and required endotracheal intubation. She was given 50 grams of activated charcoal down a nasogastric tube and then transferred to another institution. She erroneously received 50 grams of charcoal every hour for the first six hours of her hospital stay. She showed steady improvement during the first 12 hours and was extubated showing a good cough reflex. She then repeatedly vomited several large amounts of charcoal followed by decreasing oxygen saturation levels, wheezing, cyanosis, and agitation. Large amounts of charcoal were suctioned from her endotracheal tube for several days after she was reintubated. She was hospitalized for 22 days and, at the time of discharge, had permanent neurologic deficits with generalized weakness, poor muscle coordination, slow movements, and dysarthric speech. A magnetic resonance image (MRI) showed signal abnormalities in the basal ganglia bilaterally consistent with a hypoxic or ischemic insult. The patient’s initial pH was 7.05 at presentation to the first institution, and she did require defibrillation for repeated episodes of ventricular tachycardia and fibrillation. Those factors certainly could have contributed to her outcome, but the morbidity that patient suffered may have been at least partially avoidable.
Complications, such as those outlined in the case above, are not common when a single dose of charcoal has been used for the poisoned patient. The incidence of emesis appears to be greater when activated charcoal is administered with sorbitol.34 Effectiveness—rather than potential complications—is more important when considering the administration of charcoal. In several studies, when charcoal was administered 30 minutes or less after drug administration to volunteers, the mean bioavailabilitiy of the drug was reduced by 69.1%. But when activated charcoal was given at 60 minutes after drug administration, the mean reduction in bioavailability was only 34.4%. In multiple studies involving 26 drugs, the mean reduction in drug absorption was 88.6% when charcoal was administered up to 30 minutes after dosing and only 37.3% at 60 minutes.35 One study did find that symptomatic patients who received activated charcoal and some form of gastric emptying were less likely to be admitted to an intensive care unit.25
The American Academy of Clinical Toxicology and European Association of Poisons Centres and Clinical Toxicologists recommend that "single-dose activated charcoal should not be administered routinely in the management of poisoned patients." They further recommend administration of activated charcoal may be considered if a patient has ingested a potentially toxic amount of a poison that is known to be adsorbed to charcoal if this can occur within one hour of the ingestion. An emphasis is placed on the fact that they could not find sufficient data to support or exclude the use of charcoal after one hour of ingestion. Finally, they conclude that there is "no evidence that the administration of activated charcoal improves clinical outcome." 35
Charcoal may be indicated for multiple-dose use in patients who have ingested substances that undergo significant enterohepatic circulation. A form of gastrointestinal dialysis can be used to enhance elimination of drugs like theophylline, phenobarbital, or salicylates. Ingestion of drugs that have substantial elimination of a parent compound or an active metabolite through the bile, such as a tricyclic antidepressant or carbamazepine, also may be indications for repeated doses of charcoal. Oral charcoal can be used to enhance the elimination of drugs that have been administered intravenously, such as theophylline or phenobarbital.36
Considering the case of the child who ingested acetaminophen, some would advocate for the use of charcoal in the home of this 3-year-old, if it were available there. If timeliness is the limiting factor to the effectiveness of charcoal, why not make it part of every family’s medicine cabinet? Three articles have addressed the use of charcoal in the home. In each of these articles, the main obstacle to the success of administration rests with the difficulty of giving the recommended dose of charcoal, with lesser amounts being given from 40% to 80% of the time.37-39 Results from one of these studies stated, "activated charcoal can be administered successfully by the lay public in the home" but the mean dose accepted by the study children was less than the target dose.39 In those studies in which home and ED administration times were compared, the mean times were 35 and 65 minutes after drug ingestion, respectively. A recent review on this approach concluded that there should be more clear evidence of patient benefit before its use is recommended routinely.40
One of the ways to overcome the barriers to effective administration of charcoal is through the development of more palatable means of consumption. Dagnone et al41 reported that 30 healthy children felt the palatability of charcoal was improved when a cola drink was added. The children in the study volunteered to participate and did not ingest any medications or toxins. The children rated the taste on a modified visual analog scale after being given charcoal mixed with a cola drink, water, orange juice, and chocolate milk. The difference was significant among the cola drink and the other three beverages. In a similar study, Skokan et al42 found that children preferred either a cola drink or chocolate milk when compared with charcoal without any additive or charcoal with cherry-flavoring added. Again, this difference was found to be statistically significant. The morbidities associated with the placement of a nasogastric tube can be completely avoided if the child is willing to drink charcoal with one of these more palatable additives. Recommendations regarding dosage of charcoal are listed in Table 3.
Table 3. Charcoal Administration
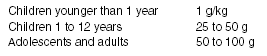
The use of cathartics should be limited to a single dose, if used at all. There are no studies evaluating the ability of a cathartic—with or without activated charcoal—to improve the outcome for poisoned patients. An argument favoring the use of cathartics is based upon the theory that they will decrease the absorption of substances by decreasing the transit time of the poison from the gastrointestinal tract. Another argument supports the use of sorbitol because of its potential to increase the palatability of charcoal by its sweet taste. The use of cathartics can lead to complications, but these usually are limited to patients who receive multiple or excessive doses. Dehydration, transient hypotension, hypernatremia, and hypermagnesemia are potential complications. These complications are especially important to consider in the patient with possible renal dysfunction.43,44 The American Academy of Clinical Toxicology stated that "based on available data, the routine use of a cathartic in combination with activated charcoal is not endorsed."45
Whole-Bowel Irrigation
Another technique for gastrointestinal decontamination is whole-bowel irrigation (WBI). The process involves the rapid administration of a balanced electrolyte solution in large amounts during several hours. The administration is continued until the effluent is clear of any evidence of further ingested material or medication. The solution used is polyethylene glycol electrolyte solution, which often is used as part of a colonoscopy preparation procedure. This solution was designed specifically to prevent fluid or electrolyte changes across the gastrointestinal epithelium.46 The theory behind its use is based upon the idea of increasing the transit of the poison beyond the area of the proximal small intestine where most toxins are absorbed.47 The volume of solution needed for different age groups is outlined in Table 4.
Table 4. Whole-bowel Irrigation Solution
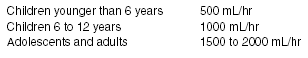
The main indication would be the case of a significant ingestion when charcoal would not be expected to be of any benefit. Situations such as the ingestion of sustained release medications or substances not adsorbed by charcoal may warrant the use of whole-bowel irrigation. The poisons often cited as a prime indication for the use of whole-bowel irrigation are iron48 and lithium.49 The process has been utilized in the treatment of lead50 and zinc51 ingestions. Human studies have shown 67-73% decreases in drug absorption, which is far better than that seen with the use of ipecac, gastric lavage, or single-dose charcoal. 49,52,53
Potential adverse effects of whole-bowel irrigation include nausea, vomiting, or abdominal cramps. Pulmonary aspiration also could occur during whole-bowel irrigation in the patient with a compromised or unprotected airway. Contraindications to the use of whole-bowel irrigation include bowel perforation, bowel obstruction, gastrointestinal hemorrhage, and ileus.
The American Academy of Clinical Toxicology and European Association of Poisons Centres and Clinical Toxicologists also have issued a position statement regarding the use of whole-bowel irrigation.54 The statement is based upon the fact that no controlled clinical trials have been performed and "there is no conclusive evidence that WBI improves the outcome of the poisoned patient." The statement continues to say that "there are insufficient data to support or exclude the use of WBI" and that "WBI remains a theoretical option for these ingestions."
Table 5. Summary of GI Decontamination Methods
Laboratory Testing of the Child with a Potential Ingestion
An 8-month-old male was brought to the ED by his mother because the child has refused to sleep for the past three hours. The mother was concerned because the child usually sleeps without any difficulty. The child has no history of fever or recent illness. On exam, the child is crying and is very difficult to console. The child breast feeds only for short periods during his time in the ED. His vital signs are: temperature 38.5º C; heart rate 164 beats per minute; respiratory rate 48; blood pressure 128/88 mmHG; and his O2 saturation was 98% on room air. His pupils are equal and measure 6 mm each. He is markedly diaphoretic. He has no external signs of trauma. His lungs are clear with good aeration, and he has no retractions, nasal flaring, or grunting. His heart exam shows no murmurs, rubs, or thrills. His color is pink, and he has no skin rashes.
An extensive evaluation was done in the ED including a complete blood count, blood culture, electrolyte levels measurement, serum glucose measurement, urinalysis and urine culture, a computerized tomography scan of the head, and a lumbar puncture. All of the test results were unremarkable. The father came to the ED after finishing his overnight shift as a firefighter. After hearing the results of all the testing and seeing his son, the father asked if his older son’s methylphenidate may have caused the symptoms. The father stated that earlier in the day, the older son had spilled his medication on the floor and he wondered if his infant son may have ingested a pill left on the floor.
The patient was given a dose of midazolam intravenously with normalization of his vital signs and his demeanor. A serum drug screen was sent and was found to be positive for amphetamines. The patient was admitted to the hospital for observation and received several more doses of midazolam during the next 24 hours. He was free of symptoms 30 hours after his initial refusal to sleep and discharged to home. He suffered no known adverse sequelae.
The Role of Toxicologic Testing
In the case of an irritable, fussy child above, the differential diagnosis is fairly extensive at the time of the child’s presentation. Trauma, sepsis, meningitis, pyelonephritis, or ingestion may each have explained the child’s symptoms. Only when the father raised the question about his older son’s methylphenidate could the diagnosis become more certain. In this case, drug testing confirmed the suspected diagnosis. Given the inherent delays in obtaining drug screen results, the previously described patient still warranted the complete evaluation. A positive drug-screening test should not rule out the presence of other concerning, and potentially treatable, causes for a patient’s altered mental status.
Under what circumstances is toxicology testing warranted for the pediatric patient with suspected ingestion? Do all children require toxicologic testing when ingestion is suspected?
Less direct laboratory testing may lead to identification of the agent ingested. As mentioned earlier, any child with an altered mental status warrants measurement of his/her blood glucose level. Serum electrolyte measurement and calculation of the anion gap can be useful in cases of poisonings by those agents known to cause a high anion gap metabolic acidosis. Methanol, salicylates, ethanol, isoniazid, iron, and ethylene glycol can all lead to high anion gap metabolic acidosis, and the results of the anion gap calculation will be obtained sooner than specific drug level results. Serum osmolality can be used to calculate the osmolality gap when a toxic alcohol ingestion is possible. Specific drug levels after acetaminophen ingestions certainly are indicated, as the potential for toxicity is predictable using the Rumack-Matthew nomogram. An acetaminophen level should be done routinely in the course of the assessment of any patient with polydrug ingestion, altered mental status, or ingestion with attempt to do harm.
The toxicology screen often implies a qualitative or quantitative detection of specific drugs in the urine, blood, or both. The possibilities of detection are immense with great variability among facilities offering drug testing. Those drugs included in a drug screen test and the cost of the testing should be understood by those health care providers ordering these tests. The comprehensive toxicology screening of potentially poisoned patients rarely influences the management of most patients.
Several studies have looked specifically at the issue of drug testing for the pediatric population presenting for acute care. Sugarman et al55 reviewed the records of all children younger than 18 years who had a toxicology screen performed and who were seen in a children’s hospital ED during a one-year period. Of the 338 charts reviewed, 195 patients (57%) had a positive toxicologic screen for at least one item, and only 22 (7%) had a positive screen for an unexpected item. The authors report that only three patients with unexpected positive toxicologic screens had a change in medical management as a result of the testing.
Belson et al6 reviewed cases of children younger than 18 years of age presenting with a suspected ingestion to one of two tertiary care children’s hospital EDs during a six-month period. This prospective study included a questionnaire completed by pediatric emergency physicians on each patient; the questionnaire asked about signs and symptoms as well as their assessment of the ability of positive or negative drug analyses to affect patient management. Drug analysis was ordered in 72% of cases with 59% of those tests obtained for a history of ingestion and 27% obtained for altered mental status. Seventy-eight (49%) of 158 patients had positive toxicology tests with 17 (11%) showing unexpected results. Unsuspected results that affected management were found in only four of the 158 (3%) tests done.
Finally, a retrospective study by Belson et al57 of 463 pediatric patients found that in 234 positive screening tests, only 7 (3%) were positive without a documented suspicion of an exposure to the agent. Again, in these cases, detection of these drugs did not result in any change in medical management. The costs of these tests reached $88,433 or $2,315 per patient.
An indication for routine drug screening should include cases of suspected child abuse or neglect. For those children who cannot intentionally use drugs of abuse or who are suspected of being poisoned intentionally, comprehensive drug testing is very important. For children with altered mental status that is not explainable by the parent or caregiver, toxicologic testing must be done to make the diagnosis and document what specific agent has been used to harm the child. Langlois et al58 also highlights this aspect of toxicologic testing for the infant with possible sudden infant death syndrome (SIDS). The authors found three children who had methadone detected at the time of their autopsy, pointing against the diagnosis of SIDS.
One of the practical limitations of toxicology screening is that many of the techniques for detection may not reveal all drugs in specific drug classes. Screening tests for amphetamines may not detect more commonly abused forms, such as methamphetamine or ecstasy. Screening for opiates may not detect synthetic forms, such as methadone or fentanyl. Drugs such as gamma-hydroxybutyrate (GHB), ketamine, flunitrazepam (Rohypnol), and lysergic acid diethylamide (LSD) are not included routinely in drug-of-abuse testing.
There are also problems with false-positive tests when toxicologic screens are used indiscriminately. Over-the-counter cough and cold medicines cause positive results for amphetamines and phencyclidine. Poppy seeds may lead to a positive test for opioids.59
Specific drug levels are sometimes indicated in the patient with suspected ingestion. Specific drug levels also may not be part of routine drug screens and may not include acetaminophen, lithium, digoxin, or salicylate levels. Familiarity with what is included in a particular institution’s comprehensive drug screen is again important for health care providers who order those tests.
Often, pregnancy testing for females of childbearing age who are cared for after suspected ingestion is done routinely. A prospective study60 examined women between the ages of 12 and 30 years who had overdosed intentionally and found that 11.6% of them were pregnant. Another prospective study61 found that of more than 20,000 women between 16 and 50 years hospitalized after attempted suicide by ingestion, only 2.8% were pregnant. Consideration of risks to an unborn child must be made in the management of the pregnant patient who has ingested a potentially harmful substance.
Prevention
The reduction in morbidity and mortality for pediatric patients after ingestion has not necessarily been the result of improvements in treatment or development of newer antidotes. The use of child-resistant packaging certainly has reduced the exposure of children to medications and cleaning products.62 Manufacturers have helped decrease the morbidity of third-degree esophageal burns and the resulting stricture formation by significantly reducing the concentrations of alkali in cleaning products from 30% to 5%.62
The use of packaging products such as the blister pack has been shown to reduce the incidence and severity of some overdoses. Results from one study63 showed that in the United Kingdom in September 1998, paracetamol became available almost exclusively in blister-pack form. (A pack contains 16 500-mg tablets and a pharmacy pack contains 32 tablets. A prescription is required to obtain more than 100 tablets.) A review of all paracetamol overdose cases presenting to the Royal Free Hospital during a four-year period showed a 21% reduction in all paracetamol overdoses and a 64% reduction in severe overdoses in the year since the compulsory introduction of blister packs. Clinicians may be the best voices to suggest such changes in packaging elsewhere in the hope of reducing the exposure to the suicidal patient who acts upon impulse.
As the average age of the U. S. population increases, more medications continue to be developed and prescribed. The need for easy access to medications for the elderly population will be in direct opposition to the need to maintain safe packaging for the curious pediatric population. While child-resistant containers are helpful to reduce the risk of ingestion for young children, these containers may interfere with the elderly population maintaining compliance with prescribed medications. In a survey of senior citizens, 56% of respondents reported transferring their medications to other containers or simply leaving child-resistant containers open.64 The need for creative packaging solutions to meet the needs of all patient populations will need to be investigated.
The value of poison control centers cannot be overstated in terms of the prevention of morbidity and mortality for the pediatric patient after ingestion. Most of the phone calls to poison control centers result in management at home, reducing the costs of ED visits, ambulance use, hospital admissions, and treatment delays. Poison control center services reduced the number of patients who were medically treated but not hospitalized for poisoning by 350,000 and the number of hospitalizations by 40,000 in 1992.65 Those delivering care to poisoned patients should make others aware of the value provided by the poison control center system.
Summary
Pediatric patients who have ingested a medication or product commonly are cared for in the EDs of the United States. Thankfully, most of these children do not require interventions and often are managed at home with the help of the national poison control center system. The mortality from poisonings remains low for the pediatric population with the exception of the adolescent age group.
Those who provide care for children must be aware of the varied presentations of the pediatric patient with an ingestion. The toddler who has ingested a known product is not the only scenario that the EP will face. The child with altered mental status always must be considered as a patient with a potential ingestion; the patient may be suffering the effects of an ingestion because of exploration or because of intentional harm by a caregiver. A high index of suspicion and close attention to reported symptoms, signs of ingestion, and vital signs for all pediatric patients should uncover the child without a clear-cut presentation for ingestion.
The EP always must consider the risks and benefits of interventions and testing before ordering them for the pediatric patient. Knowing that any intervention can lead to complications or morbidities of its own, the EP must resist a cookbook approach and make decisions that result in the best care for each individual patient.
References
1. American Academy of Pediatrics, Committee on Injury, Violence, and Poison Prevention. Poison treatment in the home. Pediatrics 2003;112:1182-1185.
2. Watson WA, Litovitz TL, Rodgers GC, et al. 2002 Annual report of the American Association of Poison Control Centers toxic exposure surveillance system. Am J Emerg Med 2003;21:353-421.
3. Litovitz TL, Bailey KM, Schmitz BF, et al. 1990 Annual report of the American Association of Poison Control Centers national data collection system. Am J Emerg Med 1991;9:461-509.
4. Poisindex Toxicologic Managements for pseudoephedrine. Available at: www.micromedex.com/. Accessed on Jan. 15, 2004.
5. Killian CA, Roberge RJ, Krenzelok EP, et al. "Cloniderm" toxicity: Another manifestation of clonidine overdose. Ped Emerg Care 1997;13:340-341.
6. American Academy of Pediatrics, Committee on Injury and Poison Prevention. Office-based counseling for injury prevention. Pediatrics 1994;94:566-567.
7. Corby DG, Decker WJ, Moran MJ, et al. Clinical comparison of pharmacologic emetics in children. Pediatrics 1968;42:361-364.
8. Saincher A, Sitar DS, Tenenbein M. Efficacy of ipecac during the first hour after drug ingestion in human volunteers. J Toxicol Clin Toxicol 1997;35:609-615.
9. Neuvonen PJ, Vartiainen M, Tokola O. Comparison of activated charcoal and ipecac syrup in prevention of drug absorption. Eur J Clin Pharmacol 1983;24:557-562.
10. Vasquez TE, Evans DG, Ashburn WL. Efficacy of syrup of ipecac-induced emesis for emptying gastric contents. Clin Nucl Med 1988; 13:638-639.
11. Neuvonen PJ, Olkkola KT. Activated charcoal and syrup of ipecac in prevention of cimetidine and pindolol absorption in man after administration of metoclopramide as an antiemetic agent. J Toxicol Clin Toxicol 1984;22:103-114.
12. Young WF Jr, Bivins GH. Evaluation of gastric emptying using radionuclides: Gastric lavage versus ipecac-induced emesis. Ann Emerg Med 1993;22:1423-1427.
13. Kulig K, Bar-Or D, Cantrill SV, et al. Management of acutely poisoned patients without gastric emptying. Ann Emerg Med 1985:14; 562-567.
14. American Academy of Clinical Toxicology, European Association of Poisons Centres and Clinical Toxicologists. Position statement: Ipecac syrup. J Toxicol Clin Toxicol 1997;35:699-709.
15. Tenenbein M. Inefficacy of gastric emptying procedures. J Emerg Med 1985;3:133-136.
16. Peterson CD, Fifield GC. Emergency gastrotomy for acute iron poisoning. Ann Emerg Med 1980;9:262-264.
17. Bond GR. Home syrup of ipecac use does not reduce emergency department use or improve outcome. Pediatrics 2003;112:1061-1064.
18. Chaffee-Bahamon C, Lacouture PJ, Lovejoy FH Jr. Risk assessment of ipecac in the home. Pediatrics 1985;75:1105-1109.
19. Schiff RJ, Wurzel CL, Brunson SC, et al. Death due to chronic syrup of ipecac use in a patient with bulimia. Pediatrics 1986;78:412-416.
20. McClung HJ, Murray R, Braden NJ, et al. Intentional ipecac poisoning in children. Am J Dis Child 1988;142:637-639.
21. Tenenbein M, Cohen S, Sitar DS. Efficacy of ipecac-induced emesis, orogastric lavage, and activated charcoal for acute drug overdose. Ann Emerg Med 1987;16:838-841.
22. Danel V, Henry JA, Glucksman E. Activated charcoal, emesis, and gastric lavage in aspirin overdose. BMJ 1988;296:1507.
23. Comstock EG, Faulkner TP, Boisaubin EV, et al. Studies on the efficacy of gastric lavage as practiced in a large metropolitan hospital. Clin Toxicol 1981;18:581-597.
24. Matthew H, Mackintosh TF, Tompsett SL, et al. Gastric aspiration and lavage in acute poisoning. BMJ 1966;1:1333-1337.
25. Merigian KS, Woodard M, Hedges JR, et al. Prospective evaluation of gastric emptying in the self-poisoned patient. Am J Emerg Med 1990;8:479-483.
26. Boxer L, Anderson FP, Rowe DS. Comparison of ipecac-induced emesis with gastric lavage in the treatment of acute salicylate ingestion. J Pediatr 1969;74:800-803.
27. Allan BC. The role of gastric lavage in the treatment of patients suffering from barbiturate overdose. Med J Aust 1961;2:513-514.
28. Thompson AM, Robins JB, Prescott LF. Changes in cardiorespiratory function during gastric lavage for drug overdose. Human Toxicol 1987;6:215-218.
29. Mariani PJ. Gastrointestinal tract perforation with charcoal peritoneum complicating orogastric intubation and lavage. Ann Emerg Med 1993;22:606-609.
30. Leclerc F, Martin V, Gaudier B. Intoxication par l’eau secondaire au lavage d’estomac. Presse Med 1981;10:1149-1150.
31. American Academy of Clinical Toxicology, European Association of Poisons Centres and Clinical Toxicologists. Position statement: Gastric lavage. J Toxicol Clin Toxicol 1997;35:711-719.
32. Hegenbarth MA, Wasserman GS. Gastric Emptying. In: Henretig FH, King C, eds. Textbook of Pediatric Emergency Procedures. Baltimore: Williams and Wilkins;1997:1297-1304.
33. Givens T, Holloway M, Wason S. Pulmonary aspiration of activated charcoal: A complication of its misuse in overdose management. Pediatr Emerg Care 1992;8:137-140.
34. Kornberg AE, Dolgin J. Pediatric ingestions: Charcoal alone versus ipecac and charcoal. Ann Emerg Med 1991;20:648-651.
35. American Academy of Clinical Toxicology, European Association of Poisons Centres and Clinical Toxicologists. Position statement: Single-dose activated charcoal. J Toxicol Clin Toxicol 1997;35:721-741.
36. Pond SM. Role of repeated oral doses of activated charcoal in clinical toxicology. Med Toxicol 1986;1:3-11.
37. Lamminpaa A, Vilska J, Hoppu K. Medical charcoal for a child’s poisoning at home: Availability and success of administration in Finland. Hum Exp Toxicol 1993;12:29-32.
38. Scharman EJ, Cloonan HA, Durback-Morris LF. Home administration of charcoal: Can mothers administer a therapeutic dose? J Emerg Med 2001;21:357-361.
39. Spiller HA, Rodgers GC Jr. Evaluation of administration of charcoal in the home. Pediatrics 2001;108:E100. Available at: www.pediatrics.org/cgi/content/full/108/6/e100.
40. McGuigan MA. Activated charcoal in the home. Clin Pediatr Emerg Med 2000;1:191-194.
41. Dagnone D, Matsui D, Rieder MJ. Assessment of the palatability of vehicles for activated charcoal in pediatric volunteers. Ped Emerg Care 2002:18:19-21.
42 Skokan EG, Junkins EP, Schunk J. Palatability of activated charcoal in the pediatric population: A prospective study. Ped Emerg Care 2000;16:71-72.
43. James LP, Nichols MH, King WD. A comparison of cathartics in pediatric ingestions. Pediatrics 1995;96:235-238.
44. Weber CA, Santiago RM. Hypermagnesemia: A potential complication during treatment of theophylline intoxication with oral activated charcoal and magnesium-containing cathartics. Chest 1989;95:56-59.
45. American Academy of Clinical Toxicology and European Association of Poison Centres and Clinical Toxicologists. Position statement: Cathartics. J Toxicol Clin Toxicol 1997:35:743-752.
46. Davis GR, Santa Ana CA, Morawski SG, et al. Development of a lavage solution associated with minimal water and electrolyte absorption or secretion. Gastroenterology 1980;78:991-995.
47. Tenebein M. Whole-bowel irrigation as a gastrointestinal decontamination procedure after acute poisoning. Med Toxicol 1988:3:77-84.
48. Tenebein M. Whole-bowel irrigation in iron poisoning. J Pediatr 1987;111:142-145.
49. Smith SW, Ling LJ, Halstenson CE. Whole-bowel irrigation as a treatment for acute lithium overdose. Ann Emerg Med 1991;20: 536-539.
50. Roberge RJ, Martin TG. Whole-bowel irrigation in an acute lead intoxication. Am J Emerg Med 1992;10:577-583.
51. Burkhart KK, Kulig KK, Rumack B. Whole-bowel irrigation as treatment for zinc sulfate overdose. Ann Emerg Med 1990:19:1167-1170.
52. Tenebein M, Cohen S, Sitar DS. Whole-bowel irrigation as a decontamination procedure after acute drug overdose. Arch Int Med 1987; 147:905-907.
53. Kirshenbaum LA, Mathews SC, Sitar DS, et al. Whole-bowel irrigation versus activated charcoal in sorbitol for the ingestion of modified release pharmaceuticals. Clin Pharmacol Ther 1989;46:264-271.
54. American Academy of Clinical Toxicology and European Association of Poison Centres and Clinical Toxicologists. Position statement: Whole bowel irrigation. J Toxicol Clin Toxicol 1997;35:753-762.
55. Sugarman JM, Rodgers GC, Paul RI. Utility of toxicology screening in a pediatric emergency department. Pediatr Emerg Care 1997;13: 194-197.
56. Belson MG, Simon HK, Sullivan K, Geller RJ. The utility of toxicologic analysis in children with suspected ingestions. Pediatr Emerg Care1999;15:383-387.
57. Belson MG, Simon HK, Sullivan K. Utility of comprehensive toxicologic screens in children. Am J Emerg Med 1999;17:221-224.
58. Langlois NE, Ellis PS, Little D, et al. Toxicologic analysis in cases of possible sudden infant death syndrome: A worthwhile exercise? Amer J Forensic Med Path 2002;23:162-166.
59. Hoffman RJ, Nelson L. Rational use of toxicology testing in children. Curr Opin Pediatr 2001;13:183-188.
60. Perrone J, Hoffman RS. Toxic ingestion in pregnancy: Abortifacient use in a case series of pregnant overdose patients. Acad Emerg Med 1997:4:206-209.
61. Czeizel AE, Timar L, Susanszky E. Timing of suicide attempts by self-poisoning during pregnancy and pregnancy outcomes. Intl J Gyn Ob 1999;65:39-45.
62. Walton WW. An evaluation of the Poison Prevention Packaging Act. Pediatrics 1982;89:999-1006.
63. Turvill JL, Burroughs AK, Moore KP. Change in occurrence of paracetamol overdose in UK after introduction of blister packs. Lancet 2000;355:2048-2049.
64. Khanderia M, Clausing M, Robertson WO. The safety cap and the senior citizen. Vet Hum Toxicol 1980;22:239-240.
65. Miller TR, Lestina D. Costs of poisoning in the United States and savings from poison control centers: A benefit-cost analysis. Ann Emerg Med 1997;29:239-245.
Although an increasingly uncommon scenario, pediatric patients continue to suffer fatal consequences of poisonings in the United States each year. Individuals providing care for the pediatric population continue in their efforts not only to find ways to treat exposed children, but also to avoid exposures from occurring at all. The goal of reducing morbidity and mortality from poisonings in ever-changing environments, both in the home and at the hospital, remains a challenge to all caregivers.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
