Osteoporosis
Osteoporosis
Author: Priscilla A. Bresler, MD, Clinical Assistant Professor, Division of General Internal Medicine, Center for Women's Health, Ohio State University Wexner Medical Center, Columbus, OH
Peer Reviewer: Udaya Kabadi, MD, Professor, University of Iowa School of Medicine, Iowa City, IA
Author’s Note
Osteoporosis is a common disease that causes significant morbidity and mortality. Its prevalence and costs to society are huge and its incidence is rising as the population ages. Osteoporosis affects people of all races and socioeconomic groups, and the prevalence in men is not far behind that of women, yet it remains underdiagnosed and undertreated. Although osteoporosis is preventable, it is a silent disease that often does not come to medical attention until fracture occurs. A large percentage of osteoporosis is secondary to other disease states or medications, and there are many known modifiable risk factors. Screening to identify individuals at risk and instituting primary and secondary prevention with risk factor reduction and pharmacotherapy provides an opportunity to reduce the incidence of fracture and reduce morbidity and mortality. This review will highlight the salient aspects of screening and treatment of osteoporosis for primary care providers.
Introduction
Osteoporosis is a progressive, systemic skeletal disease characterized by low bone mass and decreased bone strength resulting in increased risk of fracture. Disruption of normal skeletal microarchitecture renders the bones weakened and prone to fracture from stress that ordinarily would not cause fracture, called fragility fracture. Like diabetes and heart disease, osteoporosis is a preventable condition that poses a major health risk in the United States and globally. The adverse health and economic impact of low bone mass is substantial and poised to grow as the population ages. Although we possess the knowledge and tools to identify patients requiring intervention, and studies have demonstrated cost-effectiveness of both screening1 and the use of pharmacologic agents for treatment and prevention,2 the rates of osteoporosis risk assessment and treatment are low.3
Epidemiology
Low bone mass is estimated to affect 200 million people worldwide, approximately one in 10 women older than age 60, and two in five women older than age 80.4 In the United States, approximately 44 million men and women representing 55% of the population older than age 50 are at risk for fracture from low bone mass.4 The lifetime fracture risk for a 50-year-old woman is approximately 40%, similar to the risk of cardiovascular disease, and the lifetime risk for a 50-year-old man is approximately 30%, similar to the risk of prostate cancer.4,5
Age, sex, race, ethnicity, and genetics impact risk and prevalence of osteoporosis. In the United States, prevalence of low bone mass is highest in women and increases with age.6 Approximately 80% of all hip fractures occur in women and 90% of all fractures occur in people age 50 and older.4 Additionally, within a single racial group similar in age and gender, there is a large variation in incidence of low bone mass, of which approximately 84% is attributable to heritable genetics.7 Data from the National Health and Nutrition Examination Survey 2005-2008 demonstrated racial differences for osteoporosis in both men and women.8 In the U.S. population aged 50 and older, approximately half had either osteoporosis or low bone mass at the femur neck or lumbar spine. For both women and men, the age-adjusted lowest rates of osteoporosis and low bone mass were among non-Hispanic blacks. The highest prevalence of osteoporosis and low bone mass was among Mexican Americans and people of “other” races (non-African American, Caucasian, or Mexican American)
Morbidity and Mortality
Osteoporotic fractures incur significant morbidity and cost to the individual and to society. This is confounded by the fact that any individual who has a history of fragility fracture has an 86% increased risk of another fracture at any site.4
The most common sites of fragility fracture are the spine, hip, and wrist. The prevalence of vertebral deformities among postmenopausal white women is estimated to be 20-25%.9 A 65-year-old woman with one vertebral fracture has a one in four chance of sustaining another fracture over 5 years. Treatment can reduce this by 50%.3 Acute vertebral compression fractures may produce significant pain that can last weeks to months. More commonly, multiple vertebral fractures present in a subacute fashion with gradually worsening kyphosis, loss of height, and back pain that is initially attributed to another cause. Ten percent of patients have spinal deformities severe enough to cause chronic disability.9 These skeletal deformities can cause chronic pain, loss of height, immobility, and functional impairment, which may lead to depression and decreased quality of life.9,10 Severe kyphosis may lead to respiratory compromise that can further impact functional status and decrease independence in activities of daily living. In addition, vertebral fractures correlate with increased mortality over time.5,9,10
Hip fractures have the greatest risk of disability, institutionalization, loss of independence, and death. One year after hip fracture, only 40-79% of patients have returned to their prefracture functional status.9 In addition, hip fractures have been associated with > 20% mortality in the first year following fracture, usually due to comorbid conditions.4,11 Meta-analysis has demonstrated that excess mortality after hip fracture persists over time. Compared to the control group, people who experienced hip fracture had higher mortality up to 10 years following the event, and the rate of increased mortality is greater for men than for women in all age groups.10,12 In contrast, distal forearm fractures rarely cause total disability, but do result in pain, loss of function, and increased risk of post-traumatic arthritis.
Definition: Bone Strength = Bone Mineral Density + Bone Quality
Although the term osteoporosis often is used synonymously with low bone mineral density (BMD), other factors contribute to fracture risk. Bone turnover, bone size and shape, microarchitecture, and matrix all play an important role in determining bone strength.13
The World Health Organization (WHO) operating definition of osteoporosis is based on bone density measurements by dual energy x-ray absorptiometry (DXA), and comparing them to a young adult reference population.14,15 These measurements are reported as T-scores representing standard deviations (SD) below or above a projected peak bone mass. Normal is considered one SD below peak bone mass or higher (i.e., ≥ -1). Osteoporosis is defined as 2.5 SD below peak bone mass or lower (i.e., ≤ -2.5), and osteopenia lies between 1.0 and 2.5 SD below peak bone mass (See Table 1). DXA measurements also are reported as Z-scores, which reference a population of age-, ethnicity-, and sex-matched peers. A score ≥ 2 SD below the reference population is considered below expected for age and correlates with a higher risk of secondary causes of osteoporosis. In men younger than 50 years and in premenopausal women, Z-scores are preferred.16
Table 1: World Health Organization T-Score Criteria

Bone density measurements by DXA that define osteoporosis and osteopenia are associated with certain risk categories, but they do not necessarily equate with absolute fracture risk. Fracture rates occur along a continuum as BMD decreases, and with advancing age. Fifty percent of low-trauma fractures occur in people with bone density measurements in the osteopenic or normal range.17 Furthermore, a diagnosis of osteoporosis may be assigned presumptively in anyone who experiences a low-trauma fracture, regardless of his or her bone density measurement. At this time there is no single tool to measure bone strength, so risk factors play an important role in addition to bone density measurements when assessing individual risk. See discussion regarding modifiable risk factors. (See Figure 1.)
Figure 1
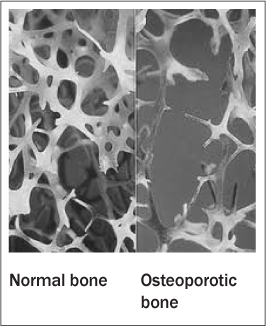
Bone Homeostasis and Pathophysiology
Bones are composed of a strong outer shell known as cortical bone, a spongy inner core of trabecular (cancellous) bone, and the intercellular matrix, which consists of collagen fibers, other proteins, and inorganic salts. Bone modeling occurring during development is the process of bone formation, growth, and changes in bone structure. Bone acquisition during growth years and early adulthood is an important factor in determining bone strength later in life. Skeletal maturation occurs approximately by age 30, at which time people have attained their peak lifetime bone mass. However, bones are dynamic organs that undergo remodeling throughout adult life. Remodeling is the process of coordinated resorption and formation of new bone by osteoclasts and osteoblasts, respectively. In young adults, the amount of bone resorption is equal to the amount of new bone that is formed. In the third and fourth decades, bone resorption and reformation starts to become imbalanced and bone resorption exceeds bone formation. With the onset of menopause in women and with advancing age in men, this imbalance is exaggerated, leading to an increased susceptibility to fracture.
Remodeling is regulated by several circulating hormones, including parathyroid hormone (PTH), vitamin D, estrogens, and androgens, as well as by many locally produced cytokines, prostaglandins, and growth factors. These local factors exert their influence by modulating the activation of new remodeling sites — a process that begins with osteoclast activity causing bone resorption, followed by new bone formation mediated by osteoblasts. Osteoclast activity is triggered by osteoblast secretion of receptor activator for nuclear factor-Kß ligand (RANKL), which binds to the osteoclast receptor RANK in the final path to osteoclast activation. Osteoprotegerin (OPG), which is also produced by osteoblasts, acts as a decoy receptor-binding RANKL to inhibit osteoclastogenesis. The interplay of these three factors modulates most osteoclast activity.
Hormonal Regulation of Bone Remodeling. Calcium-regulating Hormones: The main function of PTH is to maintain serum calcium concentrations, and its primary target is bone. PTH stimulates bone resorption by inducing the production of RANKL by osteoblasts and by inhibiting the production of OPG. PTH also acts on the kidneys to reabsorb calcium; thus, in patients with renal dysfunction, calcium reabsorption may be less efficient, resulting in renal losses of calcium. In addition, PTH stimulates renal hydroxylation of 25 hydroxyvitamin D to 1,25 dihydroxyvitamin D (also known as calcitriol, the hormonally active form of vitamin D). Calcitriol significantly increases intestinal calcium and phosphorous absorption, promoting bone mineralization and increasing availability of calcium to other tissues. Adequate levels of vitamin D enhance calcium and phosphorous absorption by 30-40% and 80%, respectively.18,19 Insufficient vitamin D will result in less calcium and phosphorous absorption, decreased bone mineralization, and a compensatory rise in PTH causing secondary hyperparathyroidism. Vitamin D also has direct effects on the skeleton, binding osteoblast receptors to stimulate the expression of RANKL for normal bone remodeling.
Gonadal Hormones: Bone cells contain receptors for estrogens and androgens, and although sex hormones play a critical role in bone development and maintenance, their direct effects on bone are not well understood. They are crucial for the pubertal growth spurt, and estrogen in particular is needed for epiphyseal closure in both sexes. Estrogen impacts production of RANKL and may also impact cell apoptosis. Estrogen deficiency contributes to bone loss by activation of new bone remodeling sites resulting in increased bone resorption compared to bone formation.
In men, estrogen is at least as important as testosterone in maintaining bone health, and some studies have suggested that estrogen plays the dominant role. Men benefit from the anabolic effects of testosterone, which increases bone mass and muscle strength. However, studies looking at the impact of the natural, age-related decline in serum testosterone concentration on fracture risk have been mixed, and there have been other studies demonstrating a key role for estrogen in regulating bone density in elderly men.20,21
Genetics. Based on twin studies, it has been determined that genetic determinants are responsible for up to 85% of the variation in peak bone mass and that there is a significant heritable component to age-related bone loss.22 The heritability of fracture is less well defined. Family history of fracture is known to be a risk factor for osteoporotic fracture, the genetics of which may be due to factors other than BMD. Determinants such as bone geometry, age at menopause, body mass index, muscle strength, and biochemical markers are likely part of a large, multi-gene inheritance. Hundreds of small genetic variants called polymorphisms have been studied relative to osteoporosis and bone loss. Some have been shown to have small clinically relevant effects, and it is believed that regulation of BMD is likely due to a complex interaction of the effects of multiple polymorphisms in different genes. To date, there have been a few single-gene mutations identified that have profound effects on bone health causing rare diseases, such as osteogenesis imperfecta and hypophosphatasia, but there are no single gene mutations that cause age-related osteoporosis.7,22,23
Risk Assessment
The skeletal health of all patients should be considered, relative to their age and risk factors. Osteoporosis is a painless, silent disease that can go undetected until a fracture occurs, unless people at risk are screened. Approximately 80% of people at high risk who have already had a fracture are not appropriately diagnosed and treated for probable osteoporosis.4 Screening involves risk factor assessment and measurement of bone density when indicated. The presence of risk factors lowers the age threshold for obtaining bone density measurement.
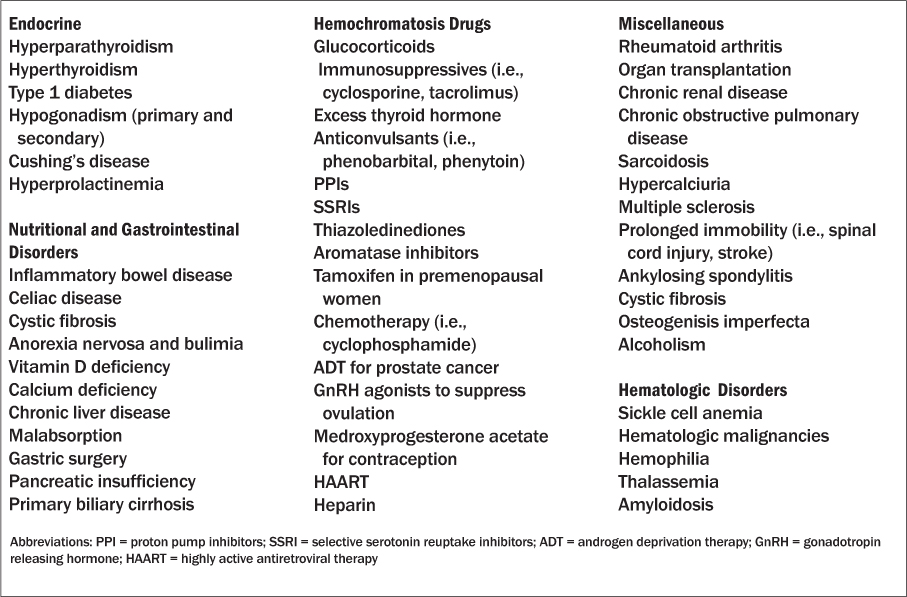
Risk Factors. The risk factors associated with bone loss may be classified into modifiable and non-modifiable factors (see Table 2) and abnormal bone loss may be primary or secondary. See section on osteoporosis prevention and nonpharmacologic management for more about modifiable risk factors. Primary osteoporosis may be idiopathic, a rare condition that usually presents in premenopausal women, or it occurs as a consequence of the natural decrease in estrogen associated with female aging, and later in life in men as sex-hormone levels decline. Osteoporosis secondary to other conditions, disease states, or medications accounts for approximately 30% of all fractures in women and 60-75 % in men.24 Some of the more common causes of secondary osteoporosis include glucocorticoid use, inadequate calcium intake, vitamin D insufficiency, alcoholism, hypogonadism, chronic renal failure, and gastrointestinal and nutritional disorders; frequently, multiple predisposing factors are present. For a more complete list of secondary causes see Table 3.
Table 2: Factors Associated with Increased Risk of Osteoporotic Fracture

Table 3: Secondary Causes of Bone Loss
FRAX Fracture Risk Assessment Tool: The FRAX calculator is a tool that can be used to predict fracture risk (www.shef.ac.uk/FRAX), and is referenced by the National Osteoporosis Foundation (NOF) in its guidelines for osteoporosis treatment. The calculation is country-, gender-, and race-specific, and provides a 10-year projection of probability of hip fracture alone and of any major osteoporotic fracture. The calculation is based on femur neck T-score as measured by DXA, as well as age, gender, body mass index (BMI), personal and family history of fracture, smoking and alcohol use, history of rheumatoid arthritis, and glucocorticoid use. Treatment is recommended when the 10-year probability of hip fracture exceeds 3% or the risk of any major osteoporotic fracture exceeds 20%. The FRAX calculator may be particularly useful when applied to patients with osteopenia and/or risk factors, and the need for pharmacotherapy is uncertain. Limitations of the tool include:
1. It is validated for use only in postmenopausal women and men age 50 years or older.
2. It cannot be used in patients recently on pharmacotherapy (i.e., no bisphosphonate for the previous 2 years, and no estrogen, raloxifene, parathyroid hormone, calcitonin, or denosumab for the previous 1 year.25)
3. It does not consider all known risk factors, nor the dose effects of tobacco, alcohol, and glucocorticoids.
4. It does not incorporate other BMD measurements such as lumbar spine.26
Although the tool can be helpful, it does not replace clinical judgment and individualized treatment decisions.
Bone Density Measurement. Several technologies are available to measure BMD. DXA is favored and the most widely used. Measurements correlate well with fracture risk and radiation exposure is low, similar to daily background radiation. Other available technologies include peripheral DXA (pDXA), quantitative ultrasonography (QUS), and quantitative computerized tomography (QCT), none of which can be used to diagnose osteoporosis because the WHO criteria were established based on DXA measurements. pDXA units are portable and easily accessible, and low T-scores correlate with increased fracture risk. However, their use is limited by technical differences between devices and lack of standardization. pDXA results should not be used for routine monitoring or to make treatment decisions. Rather the results of pDXA should prompt a standard DXA scan.
QUS is portable, free of ionizing radiation, less costly than DXA, and has been shown to accurately predict fracture risk. It measures properties of bone at peripheral sites; the heel has been most tested. However, QUS use is also limited by significant technological diversity between instruments, as well as different measurement and reporting parameters that make application to clinical practice difficult. T-score measurements are not equivalent to DXA T-scores so they cannot be used to diagnose osteoporosis according to the WHO classification, and QUS has not been shown to be reliable for monitoring bone changes over time. However, according to the International Society of Clinical Densitometry Official Positions, QUS may be used to identify patients at low and high risk for fracture, and it may be used in conjunction with risk factor assessment to initiate therapy when DXA is not available.27,28
The main advantage of QCT is that it can isolate trabecular bone from cortical bone, and might have an advantage over DXA in predicting fracture risk at the spine. It is associated with a higher radiation exposure than DXA and presently is used primarily as a research tool.
DXA is the gold standard and may be used for screening and diagnosis, as well as for prognosis and to monitor therapeutic response. However, it also has pitfalls:
1. There are several different types of machines available utilizing different x-ray sources, different calibration, and different reference databases. These variables preclude comparison of bone density readings across instruments. Whenever possible, repeat DXA scans should be performed on the same machine.
2. Disease states such as osteoarthritis and scoliosis may falsely elevate bone density measurements.
3. Correct interpretation depends on precise anatomical accuracy. Lumbar vertebrae must be correctly identified, and total hip and femur neck measurements require proper positioning.
DXA Screening Interval: Whereas general guidelines exist for the timing of bone density measurements in people on pharmacotherapy, data guiding the frequency of screening recommendations are limited. A recent study addressing this question in untreated women ages 67 years and older looked at the time it took from baseline BMD measurement to development of osteoporosis before fracture occurred.29 They found that the time varied widely depending on baseline T-score. In women with normal bone density, the transition time was significantly longer than in women with advanced osteopenia, and this held true when adjusted for age, BMI, estrogen use, any fracture after age 50, current smoking, glucocorticoid use, and rheumatoid arthritis. For example, among women with normal bone density and women with osteopenia at baseline, less than 1% and 5%, respectively, transitioned to osteoporosis during the 15-year study period. This suggests that baseline T-score may be an important factor in determining BMD screening interval.30 (See Table 4 and Table 5.)
Table 4: Transition Time to Osteoporosis Before Fracture Occurred in 10% of Study Patients29

Table 5: Guidelines for Osteoporosis Screening and Treatment

Prevention and Nonpharmacologic Management of Low Bone Mass
Osteoporosis is a complex disease that can be significantly impacted by certain modifiable risk factors. It is important to consider these risk factors when counseling about preventive care as well as in the management of low bone mass.
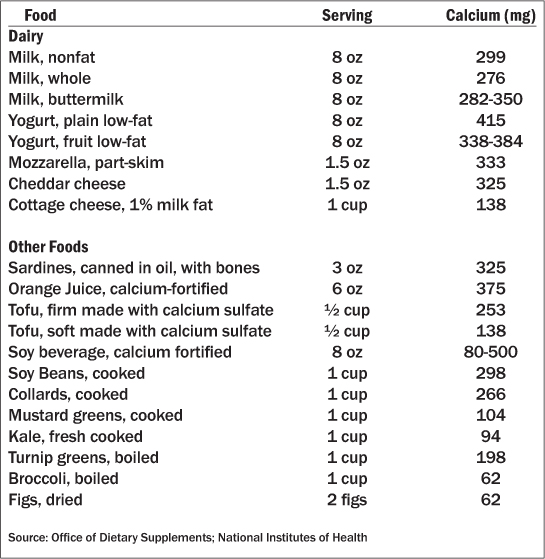
Nutritional Factors. Calcium: Most Americans do not meet the recommended daily intake of calcium, whose primary function is in the formation of bones and teeth (See Table 6). When dietary calcium intake is insufficient, supplementation is recommended, although absorption of dietary calcium is superior and is therefore preferred (See Table 7). Among supplements, calcium carbonate and citrate are the most widely available and the most concentrated. Calcium carbonate is the least expensive, but requires an acidic environment and needs to be taken with a meal to ensure optimal absorption. Calcium citrate may be taken without food, and is better absorbed in patients with achlorhydria and in those taking proton pump inhibitors or histamine-2 blockers. Potential side effects of calcium supplements are primarily gastrointestinal, including constipation and dyspepsia, and they are contraindicated in individuals with hypercalcemia. In addition, calcium supplements interfere with iron and thyroid hormone absorption and therefore should be taken separately. Supplementation should not exceed 500 mg of elemental calcium at one time due to limits of intestinal absorption and because of concerns regarding potential hypercalcemia, nephrolithiasis, and vascular or soft tissue deposition of excess calcium — concerns that do not apply to dietary calcium.38 There is a 20% increased risk of renal calculi associated with calcium supplementation,39,40 and there is mounting concern that calcium supplementation may be associated with adverse cardiovascular events.40-42
Table 7: Calcium Content of Certain Foods37
Vitamin D: Vitamin D is a fatsoluble vitamin that is essential to bone health. Subtherapeutic levels have an adverse effect on skeletal health and persistent deficiency results in rickets (children) and osteomalacia (adults). Vitamin D aids intestinal calcium and phosphorous absorption promoting bone mineralization and is needed by osteoblasts and osteoclasts for normal bone remodeling. Whereas osteoporosis is caused by bone matrix disintegration and is generally painless, osteomalacia and rickets are caused by defective matrix mineralization. Symptoms of vitamin D deficiency include muscle weakness and increased falls in the elderly. Osteomalacia causes decreased BMD and may cause generalized myalgias and bone pain.18 Low serum calcium and phosphorous, elevated bone-specific serum alkaline phosphatase, and decreased 24-hour urine calcium excretion may be observed in patients with osteomalacia whereas they are generally normal in osteoporosis.
The primary source of vitamin D is endogenous skin production triggered by exposure to the sun’s ultraviolet (UV) rays. Direct sun exposure for as little as 10-15 minutes per day is generally considered sufficient,43 though vitamin D insufficiency is common particularly in the elderly and people living in northern latitudes. Three to five times longer sun exposure may be needed to attain the same amount of vitamin D in people with darker skin complexions.19 Sunscreen worn to protect the skin from harmful effects of UV rays also blocks vitamin D synthesis. In addition, adipose tissue sequesters vitamin D and alters its release into the circulation, resulting in lower serum vitamin D levels in obese patients. Few foods are naturally high in vitamin D; examples include oil-rich fish (swordfish, tuna, mackerel, salmon, sardines, herring), cod liver oil, and egg yolks.
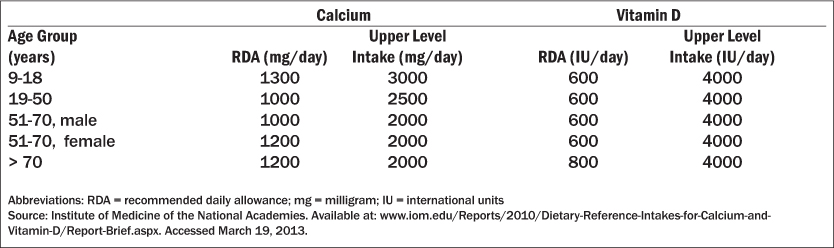
After comprehensive review, the Institute of Medicine Food and Nutrition Board issued recommended daily allowances (RDAs) for vitamin D assuming minimal sun exposure. It concluded that 600 IU daily up to age 70 and 800 IU daily for ages ≥ 71 was adequate for most adults, and that serum 25-hydroxyvitamin D of 20 ng/mL is sufficient to maintain adequate bone health.43 (See Table 6.) Endocrine Society Guidelines recommend similar intakes, but add that individuals with a BMI > 30 kg/m2 and patients on medications that increase the catabolism of vitamin D, including anticonvulsants, glucocorticoids, antifungals such as ketoconazole, and medications for AIDS take two to three times more vitamin D for their age group to meet their body’s vitamin D requirement.19
Table 6: Dietary Reference Intakes for Calcium and Vitamin D36
Good nutrition also includes maintaining normal weight, treating eating disorders such as anorexia nervosa and bulimia, and attending to potential deficiencies associated with conditions such as inflammatory bowel disease, celiac disease, and history of gastric surgery.
Health Habits. Maximizing peak bone mass through physical activity and adequate calcium intake during development is a key factor in determining bone health in later years. In fact, the beneficial impact of athletic activity on bone mass is most marked during growth years, though regular exercise is recommended for people of all ages. Tobacco use and excess alcohol intake both adversely impact bone health. Smokers are at increased risk of fracture compared to non-smokers, and intake of more than two drinks per day is associated with a 40% increased risk of osteoporotic fracture.4
Other Lifestyle Factors. Prevention of falls in the elderly: 90% of hip fractures result from falls.4 Physical activity during adulthood improves strength and balance, reduces falls, and may increase BMD.44 Factors that increase falls include visual impairments, balance problems, neuromuscular weakness, dementia, and use of sleeping pills. The surgeon general recommends 30 minutes of physical activity on most days for all adults.45 The U.S. Preventive Services Task Force recommends exercise or physical therapy and vitamin D supplementation to all adults aged 65 and older who are at increased risk of falls.46 The Endocrine Society recommends vitamin D supplementation for all, in usual doses, for fall prevention.19 In addition, the Centers for Disease Control and Prevention and the National Institute on Aging recommend medication management and monitoring of environmental hazards in the home that may increase falls,46 such as the presence of throw rugs or loose electric cords.
Other considerations include avoidance of drugs that increase bone loss whenever possible, and use of preventive pharmacologic therapy to reduce the risk of developing osteoporosis in selected patients. See Table 8 for medications approved for this purpose.
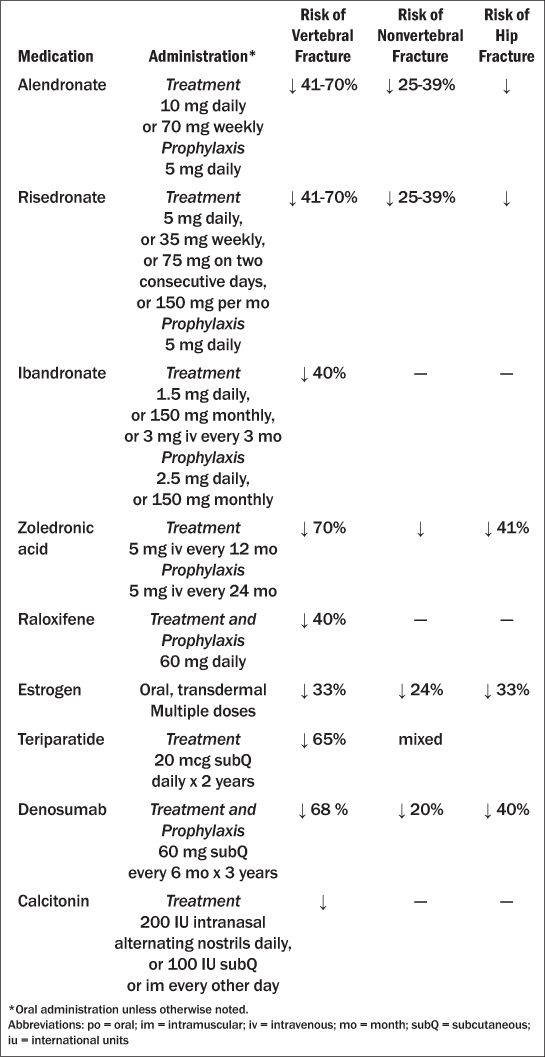
Table 8: Bone Preserving Medications: Doses and Effects33,47-52
Osteopenia, Osteoporosis Work-up
Once osteopenia or osteoporosis is diagnosed, initial evaluation should include a detailed history and physical examination with attention to potential risk factors and secondary causes. History should include details regarding chronic diseases, medications, alcohol and tobacco use, history of falls, and family history of osteoporosis. Physical exam should include assessment of height, kyphosis, balance, and mobility, in addition to a search for findings suggestive of a secondary cause. Initial laboratory evaluation should include a serum biochemistry profile (calcium, phosphorous, albumin, total protein, creatinine, liver enzymes, alkaline phosphatase, and electrolytes), 25-hydroxyvitamin D, 1,25 dihydroxyvitamin D in patients with renal failure, thyroid stimulating hormone, complete blood count, total or free serum testosterone in men, and 24-hour urine calcium and creatinine. Abnormal findings on the initial laboratory tests should guide a second tier of testing in search of secondary cause or alternative etiology. For example, a finding of elevated serum calcium suggests hyperparathyroidism or malignancy; obtaining a serum PTH in most cases will distinguish between them. Elevated urinary calcium (> 300 mg/24 hours) may be due to excessive bone turnover from hematologic malignancy, Paget’s disease, hyperparathyroidism, hyperthyroidism, or it may be due to renal causes. The differential diagnosis of low-trauma fracture includes malignancy, osteomalacia, osteogenesis imperfect, and Paget’s disease (See Table 9).
Table 9: Differential Diagnosis of Low-trauma Fracture

Pharmacotherapy
Medications to treat osteoporosis may be classified into two groups: antiresorptive and anabolic agents. Bisphosphonates (alendronate, risedronate, ibandronate, and zoledronic acid), raloxifene, estrogen, calcitonin, and denosumab all decrease bone resorption. Teriparatide is the only anabolic agent available in the United States. Strontium ranelate, which is available outside North America, has both antiresorptive and anabolic properties (See Table 8).
Bisphosphonates.
Bisphosphonates are generally considered first-line treatment for osteoporosis and are available in both oral and intravenous formulations; intravenous administration provides superior bioavailability. They act by inhibiting bone resorption by binding to calcium hydroxyapatite and inhibiting osteoclast function and survival. They are approved for both treatment and prevention of osteoporosis. The most common side effects of oral bisphosphonates are gastrointestinal, and they are contraindicated in patients with esophageal dysmotility and esophageal strictures or varicies. They should be used with caution in patients with gastroesophageal reflux disease or Barrett’s esophagus. Bone pain is also sometimes reported. To maximize absorption and minimize the risk of esophageal side effects, administration guidelines recommend dosing oral bisphosphonates in the morning on an empty stomach with 8 ounces of water and to wait at least 30 minutes (60 minutes for ibandronate) before eating, drinking, taking other medications, or reclining. Risedronate is also available in a delayed-release formulation that should be dosed immediately after breakfast. The main side effects of intravenous bisphosphonates are flu-like symptoms and arthralgias. Prior to infusion 25-hydroxy vitamin D level should be checked and corrected if less than 15 ng/mL (37 nmol/L) and adequate calcium intake should be ensured to reduce the risk of hypocalcemia. All bisphosphonates should be avoided in patients with creatinine clearance below 30-35 mL per minute.
Complications: There are potentially serious complications of bisphosphonate use that frequently concern patients and deserve mention. Atypical hip fractures and osteonecrosis of the jaw (ONJ) both occur rarely and may relate to the marked suppression of bone turnover associated with bisphosphonate use. Older people treated for more than 3-5 years with oral or intravenous bisphosphonate have a small increased risk of low-trauma fractures that are “atypical” in their location and radiographic appearance. Although there remains some controversy over causality, there appears to be an association, and bisphosphonates carry an FDA-issued warning label about atypical fractures.53 Whereas classic osteoporotic hip fractures usually occur at the femoral neck or intertrochanteric region, atypical fractures occur in the subtrochanteric region or femoral shaft above the distal metaphysis. The mechanism of these atypical fractures is thought to be inhibition of the normal healing process that is engaged when physiologic microfractures occur, leading to stress fractures.54,55 Atypical hip fractures may be further distinguished from classic fractures by the frequent occurrence of weeks to months of prodromal pain. Deep thigh pain associated with weight-bearing, which is often bilateral, should alert the clinician to consider atypical fracture. The absolute risk of atypical fractures is low, 32 cases per million person-years,55 and should not impact the decision to initiate bisphosphonate therapy in individuals at high risk for osteoporotic fracture. However, the selection of patients should be individualized with assessment of risk:benefit ratio, and bisphosphonates should be avoided in patients at low risk. The use of a drug holiday after 3-5 years of treatment in patients with stable bone density and no fractures has been suggested as a potential mechanism to prevent oversuppression of bone turnover.54-56
ONJ is another potential complication of long-term bisphosphonate use. It is rare in osteoporosis patients, one case per 10,000-50,000 person years, although it occurs in up to 5% of cancer patients with skeletal metastases treated for 2-3 years or longer with high-dose intravenous therapy.50 When possible, invasive dental procedures should be completed before initiation of therapy to minimize risk of ONJ.
Other very rare side effects of bisphosphonates include uveitis and scleritis, which are generally reversible after discontinuation of the drug. Eye pain shortly after initiation of the medication should alert the prescriber to a possible causal relationship.57
Studies have shown mixed results regarding an association between atrial fibrillation and the use of bisphosphonates in postmenopausal women with osteoporosis.58 FDA recently conducted its own review of the cardiovascular safety data for alendronate, zoledronic acid, risedronate, and ibandronate and concluded that the current data do not support an association. FDA stated, “Health care providers should not alter their prescribing patterns for bisphosphonates and patients should not stop taking their bisphosphonate medication.”59
Duration of Treatment. Currently, there is no consensus on how long to treat with bisphosphonates. Alendronate has been shown to be effective for at least 10 years60 and to have a residual benefit on BMD for up to 5 years after discontinuation;61 however, the beneficial effects of bisphosphonates start to wane as soon as the drug is discontinued. A drug holiday may be considered after 3-5 years in people with stable bone mass who are not at high risk. Although there are no formal guidelines regarding drug holiday with bisphosphonate use, McClung et al proposed the following:58
1. In high-risk patients (T-score still ≤ -2.5 at the hip, previous fracture of hip or spine, or ongoing high-dose glucocorticoid therapy), a drug holiday is not justified. Re-assessment of the need for treatment should be conducted at regular intervals.
2. In moderate-risk patients (T-score now > 2.5 at the hip and no prior hip or spine fracture), a drug holiday may be considered after 3-5 years of alendronate, risedronate, or zoledronic acid (no information is available for ibandronate and drug holidays). The decision to take a drug holiday should be individualized and the potential risks and benefits discussed with the patient.
3. In low-risk patients (those who did not meet current treatment criteria at the time of treatment initiation), therapy should be discontinued. Treatment should be re-started when indications for therapy are met.
Compliance: Compliance with oral bisphosphonates has been suboptimal with up to 75% discontinuance during the first year of treatment with daily therapy. Weekly dosing has been associated with only 35-45% persistence at 1 year, and compliance with monthly dosing has not been much better. These findings are consistent with compliance rates for oral medications taken for other chronic, asymptomatic conditions.62 Reasons for noncompliance are varied, with a significant proportion due to medication side effects. Other reasons include patients’ perceived risk of side effects, perceived lack of medication benefit, concerns related to cost and convenience, and lack of belief that they are at risk for fracture.62 Poor compliance increases fracture rate. A recent meta-analysis determined that the risk of fracture was 46% greater in patients with poor bisphosphonate compliance compared to patients who were highly compliant, and that the impact of noncompliance was highest for vertebral fractures.63 The most effective compliance intervention may be increased time with a health care provider; however, research has shown that compliance behavior is generally difficult to impact.63 Parenteral administration of bisphosphonates, first approved for postmenopausal osteoporosis in 2006, provides an alternative strategy to improve compliance and, thus, treatment outcomes. Ibandronate is administered as a quarterly intravenous injection and zoledronic acid is administered yearly as an intravenous infusion over 15 minutes or longer to minimize renal toxicity. Both medications are approved for prevention and treatment of osteoporosis, both are highly efficacious, and both have similar side effect profiles.49,64
Comparative Efficacy: All bisphosphonates reviewed in this article have demonstrated efficacy in preventing vertebral fracture and are appropriate first-line treatment agents. However, only alendronate, risedronate, and zoledronic acid also decrease the incidence of hip and non-vertebral fractures.47,65 Mortality benefit is consistent among the bisphosphonates.65 Side effect profiles differ with respect to oral vs intravenously administered bisphosphonates as described above. When choosing bisphosphonate therapy, decision-making should be individualized based on the patient’s risks, medical comorbidities, and personal preferences regarding dosing strategies. For example, initiating therapy with ibandronate is not appropriate in a patient with a history of hip fracture as there is no clinical evidence of its efficacy with hip fracture. Intravenously administered bisphosphonates have superior bioavailability and may be useful in a patient who fractures while on oral bisphosphonate or in a patient who has gastrointestinal side effects with oral medication.
Selective Estrogen Receptor Modulators. Selective estrogen receptor modulators (SERMs) bind estrogen receptors (ER) and have both estrogen agonist and antagonist properties. Raloxifene demonstrates estrogen activity in bone and prevents bone loss. It is approved for prevention and treatment of vertebral fractures in postmenopausal women, though it has not been shown to decrease non-vertebral fractures.44 Although raloxifene is less potent than other agents, it has the unique property of decreasing the incidence of ER-positive breast cancer by approximately 50-60% and is FDA-approved for chemoprevention of breast cancer in high risk women,66 which may be an important consideration in some women when choosing among antiresorptive treatments. Potential adverse effects include vasomotor symptoms, increased risk of venous thromboembolism, and fatal stroke. Tamoxifen, another SERM that is used in the treatment of ER-positive breast cancer, provides some protection against postmenopausal bone loss,67 although it is not approved for the treatment or prevention of osteoporosis.
Estrogen. Estrogen, with or without progesterone, is effective at decreasing the risk of both vertebral and non-vertebral osteoporotic fractures, even at very low doses.51,68 Estrogen is FDA-approved for treatment and prevention of osteoporosis. However, the risks of combination estrogen and progesterone therapy, including breast cancer, stroke, venous thromboembolism, and possible coronary artery disease, are believed to outweigh the benefits, and the U.S. Preventive Services Task Force does not recommended hormone therapy for treatment or prevention of osteoporosis.69 In contrast, the North American Menopause Society recommendations support the use of hormone therapy in women with low bone mass regardless of menopausal symptoms for fracture prevention when alternative therapies are not appropriate. Preferably, therapy is initiated early in menopause when the risk:benefit ratio for menopausal hormone therapy is more favorable.70
Teriparatide. Teriparatide, recombinant human 1-34 PTH peptide, is the only anabolic agent available in the United States for treatment of osteoporosis. Although hyperparathyroidism is associated with bone loss, teriparatide, when administered intermittently, stimulates bone formation more than resorption, increasing bone mass and improving bone microarchitecture. It dramatically decreases the risk of vertebral fractures after at least 8 months of therapy, although evidence of its impact on non-vertebral fractures has been mixed. Teriparatide is approved for use in men and women and is often effective in patients who fail treatment with bisphosphonates. Treatment termination is followed by rapid bone loss that may be mitigated by bisphosphonates, resulting in the frequent practice of administering a bisphosphonate following completion of a course of teriparatide.50 It is administered as a daily subcutaneous injection during which patients should be monitored for hypercalcemia and hypercalciuria. Other side effects include nausea, flushing, headache, and dizziness. An FDA black box warning is issued with teriparatide because of its association with osteosarcoma in rats. Although there is no evidence of increased risk of osteosarcoma in humans taking teriparatide, its use is limited to a maximum of 2 years, and the drug is contraindicated in people with certain conditions including Paget’s disease, patients with a history of skeletal radiation, unexplained increased serum alkaline phosphatase, hypercalcemia, bone metastases, open epiphyses, and other people at increased risk for osteosarcoma.
Denosumab. Denosumab is a human monoclonal antibody that targets RANK ligand and acts by reducing osteoclastogenesis. It binds RANKL, preventing activation of its receptor RANK, thereby inhibiting osteoclast formation and decreasing bone resorption. Denosumab improves bone density and decreases fracture risk, and is indicated for the treatment of postmenopausal women at high fracture risk (history of fragility fracture or having multiple risk factors) who have failed other therapy and in men on androgen deprivation therapy. It is administered as a subcutaneous injection every 6 months for 36 months, and substantially reduces the incidence of vertebral, nonvertebral, and hip fractures. The most common side effect is musculoskeletal pain, and serious skin infections have been reported, though denosumab is generally well-tolerated. It is contraindicated in patients with hypocalcemia. Long-term safety data are not available.52
Calcitonin. Calcitonin is a synthetic hormone that decreases bone turnover by inhibiting osteoclasts. It is approved for use in postmenopausal osteoporosis though it is less effective than bisphosphonates and other available agents and is not considered first-line therapy. It has been shown to decrease vertebral fracture rates, but data on its effect on nonvertebral bone are inconsistent.27 It has the unique benefit of decreasing acute back pain associated with vertebral fracture. The main side effect is rhinitis when it is administered by the intranasal route and nausea when administered subcutaneously.
Strontium ranelate. Strontium ranelate is approved for treatment of postmenopausal osteoporosis outside the United States. Its mechanism of action is not fully understood but appears to have antiresorptive effects while maintaining bone formation.71
Monitoring Response to Therapy
Monitoring response to pharmacotherapy is usually accomplished by serial DXA measurements. Typically, measurements are taken 1-2 years after initiation of or change in therapy, and periodically thereafter depending on clinical indications. Another tool with limited use is measurement of markers of bone turnover (MBT). Certain collagen breakdown products that are released by osteoclasts and osteoblasts during bone remodeling can be measured, permitting assessment of bone turnover. Whereas they are not useful in diagnosing osteoporosis, measurements taken at baseline and again at 3 to 6 months may provide an early assessment of response to treatment. Early decreases in MBT correlate with anti-fracture efficacy of pharmacotherapy. Their clinical usefulness is limited by biologic and laboratory variability and by lack of standardized guidelines. The most clinically relevant bone resorption markers are urinary collagen type 1 cross-linked N-telopeptide and serum collage type 1 cross-linked C-telopeptide, while bone-specific alkaline phosphatase, serum osteocalcin, and the amino terminal propeptides of type 1 pro-collagen are used as markers of bone formation.
Special Populations
Premenopausal Women. Low bone density in premenopausal women is defined as Z score ≤ 2 on DXA15 or having a fragility fracture. Screening is not routinely performed in these women, so they come to medical attention after a fracture or when screening is done due to the presence of risk factors. Initial evaluation is the same as in a postmenopausal woman with attention to a careful search for secondary causes. If none is found, it is possible she never reached optimal peak bone mass either due to adverse conditions during adolescence, a genetically determined low bone mass, or a secondary condition that contributes to ongoing risk. The correlation between low bone density measurement and fracture is weaker than in postmenopausal women,16 and the focus of management is with non-pharmacologic intervention and treatments geared toward the underlying cause. Antiresorptive therapy in premenopausal women has not been well studied. Bisphosphonates carry an FDA-category C pregnancy rating (i.e., animal studies have shown an adverse effect on the fetus and there are no well-controlled studies in humans), and the decision to treat with medication should be made only after careful consideration of individual risks. Referral to a specialist may be considered.
Men. Although we tend to think of osteoporosis as women’s disease, men account for approximately one-third of all osteoporotic fractures worldwide,24 and men have higher rates of fracture-related mortality.12 Yet, osteoporosis remains underdiagnosed and undertreated in men.20,72 The most common fracture sites in men are the distal radius, proximal humerus, hip, and vertebral body. Prevalent risk factors include age ≥ 70, low body weight, previous low-trauma fracture, hypercalciuria, glucocorticoids, primary and secondary hypogonadism, vitamin D insufficiency, current smoking, excess alcohol use, diabetes, hyperparathyroidism, and subclinical hyperthyroidism. Compared to women, men have a higher rate of secondary osteoporosis. In one recent study, secondary osteoporosis accounted for 75% of all men with fractures, many of whom had ≥ 4 risk factors.24 The diagnosis of osteoporosis in men is based on the same WHO T-score criteria applied to women, or when there is a history of low-trauma fracture.20
Drugs FDA-approved for treatment of osteoporosis in men include alendronate, risedronate, zoledronic acid, and teriparatide, as well as denosumab in men on androgen deprivation therapy (ADT) for prostate cancer. The Endocrine Society recommendations for screening, treatment, and monitoring of osteoporosis in men is similar to the NOF and the International Society for Clinical Densitometry. (See Table 5.) Additional treatment recommendations include testosterone therapy in men with serum testosterone levels below 200 ng/dL and symptoms of hypogonadism. For men at high fracture risk on testosterone therapy, bisphosphonate or teriparatide should be added. The guidelines recommend monitoring therapy with DXA every 1-2 years or less frequently if BMD stabilizes and to consider use of bone turnover markers.21
Secondary Causes
Many disease states and medications have been implicated as potentially detrimental to skeletal health. For a complete listing see Table 3.
Glucocorticoids. Glucocorticoids at a daily dose of ≥ 5 mg prednisone-equivalent for ≥ 3 months is the most common cause of secondary osteoporosis.73 Glucocorticoids exert both direct and indirect skeletal effects. There is an early transient increase in bone resorption followed by decreased bone formation due to direct inhibitory effects on osteoblasts, which continues for the duration of treatment.74 Indirect effects include decreased intestinal calcium absorption, increased urinary calcium excretion, and hypogonadism. Glucocorticoids alter parathyroid hormone dynamics and oppose vitamin D activity. Fracture risk is dose-dependent: the higher the dose and duration of therapy, the greater the risk of fracture. Between 30-50% of these patients will experience a fracture, often at higher BMDs than in postmenopausal osteoporosis.73 Prevention and treatment guidelines recommend calcium and vitamin D supplementation, fall risk-assessment, and lifestyle modification for all patients, in addition to risk stratification based on baseline BMD measurement and risk factors. In high-risk individuals, guidelines recommend initiation of pharmacotherapy at BMD T-scores of ≤ -1.0 to -1.5, and preventive therapy when treatment is anticipated for ≥ 3 months.75 Agents approved for prevention and treatment of glucocorticoid-induced osteoporosis include alendronate, risedronate, zoledronic acid, and teriparatide.
Cancer. As cancer has become more treatable, there are more long-term survivors and people living with cancer as a chronic disease. Consequently, medical concerns related to survivorship are more common in primary care practice. There are several different mechanisms for cancer-treatment-associated bone loss. Chemotherapy-induced ovarian failure (CIOF), defined as loss of ovarian function in premenopausal women, and testicular failure in men are known risks of certain chemotherapy drugs. Risk varies with specific drugs used, cumulative doses, and increased age at the time of administration. Several classes of chemotherapy drugs can cause premature gonadal failure; most common are the alkylating agents such as cyclophosphamide. Tamoxifen given to premenopausal women either for treatment or prevention of breast cancer may cause modest bone loss, although when administered to postmenopausal women, tamoxifen can favorably impact bone density. Aromatase inhibitors — such as exemestane, anastrozole, and letrozole, which are commonly used in postmenopausal women with hormone-sensitive breast cancer — cause bone loss and modestly increase the risk of osteoporotic fracture. The degree of risk may be confounded by prior breast cancer therapies that also increase risk. Orchiectomy and ADT used to treat prostate cancer increase the risk of osteoporotic fracture in men. Glucocorticoids, which are widely used in chemotherapy regimens to treat a variety of malignancies, also adversely impact bone health. Another at-risk group is adult survivors of childhood malignancy, usually acute lymphoblastic leukemia, who have a higher incidence of osteoporosis. There are several causative factors: chemotherapy regimens that include corticosteroids, aklylating agents, and radiation to reproductive organs. Survivors of bone marrow transplant for Hodgkin’s and non-Hodgkin’s lymphomas constitute another group at increased risk of osteoporosis due to similar mechanisms. Guidelines for screening and treatment in cancer patients are similar to noncancer patients, although recommendations for initiation of pharmacotherapy tend to occur at higher T-score thresholds. As in noncancer patients, calcium, vitamin D, and exercise recommendations should be encouraged and other modifiable risk factors addressed.76
Costs
The number of hospitalizations involving an injury due to osteoporosis increased 55% from 1995 to 2006, when there were more than 254,000 hospital admissions; 90% were for patients 65 years and older, with the largest proportion 85 years and older.11 The precise cost is difficult to assess due to the multiple indirect costs involved. A conservative estimate of the direct and indirect costs worldwide due to hip fracture alone in 1990 was $34.8 billion. This number is projected to increase to $131.5 billion by 2050 ($21,000 per patient) and does not include costs related to other types of fractures.10
Cost-effectiveness of pharmacologic treatment has been demonstrated in a broad range of at-risk patients: those with fragility fractures, those with osteoporosis by WHO criteria, older people at average risk, and osteopenic patients with additional risk factors including certain high-risk conditions.77 Treatment of patients at increased risk of fracture with pharmacotherapy improves fracture rate, decreases morbidity and mortality, and decreases health care costs.58 Consider prophylactic treatment of patients at increased risk due to the presence of other high-risk conditions including patients on long-term glucocorticoids, men with osteopenia on ADT, patients with inflammatory bowel disease, organ transplant, hypogonadism, CIOF, osteopenia secondary to hormone therapy for breast cancer, gastric bypass surgery, cystic fibrosis, growth hormone deficiency, and others.
Improving the Osteoporosis Care Gap
Despite evidence that instituting secondary prevention and treatment of osteoporosis can reduce the incidence of future fractures, intervention rates are low in both hospital and office-based settings.3,78 In one study including 51,346 patients admitted to 313 U.S. hospitals for hip fracture, only 6.6% received calcium and vitamin D supplements and 7.3% received pharmacotherapy. This compares to 84% of patients who received antibiotic prophylaxis before surgery and 74% who received prophylaxis for deep venous thrombosis. In much the same way that secondary prevention management is initiated in cardiac patients before hospital discharge, appropriate evaluation and management may be started before a patient leaves the hospital after hip fracture.78 Similarly, these tasks may be undertaken in the outpatient setting, and include BMD testing, fall risk assessment, calcium and vitamin D recommendations, and pharmacotherapy.
Initiatives are underway to improve both primary and secondary prevention in osteoporosis management. Several models favor a coordinator-based “fracture-liaison service” incorporating an interdisciplinary approach to management. Programs that are already operational have proven to be both fracture-reducing and cost-saving.79-81
References
1. Nayak S, et al. Ann Int Med 2011;155; 751-761.
2. Dawson-Hughes B, et al. Osteoporos Int 2008;19:449-458.
3. Giangregorio L, et al. Semin Arthritis Rheum 2006;35:293-305.
4. International Osteoporosis Foundation. Available at: www.iofbonehealth.org/facts-statistics. Accessed March 19, 2013.
5. Johnell O, Kanis JA. Osteoporos Int 2006;17:1726-1733.
6. International Osteoporosis Foundation. Available at: www.iofbonehealth.org/frax-information-and-resources. Accessed March 19, 2013.
7. Richards JB, et al. Lancet 2008;371: 1505-1512.
8. Looker AC, et al. NCHS data brief no.93. Hyattsville, MD: National Center for Health Statistics; 2012.
9. Melton III LJ. J Bone Min Res 2003; 18:1139-1141.
10. Harvey N, et al. Nat Rev Rheumatol 2010;6:99-105.
11. Russo A, et al. Available at: www.hcup-us.ahrq.gov/reports/statbriefs/sb76.pdf. Accessed March 19, 2013.
12. Haentjens P, et al. Ann Intern Med 2010;152:380-390.
13. Osteoporosis Prevention, Diagnosis, and Therapy. Available at: http://consensus.nih.gov/2000/2000Osteoporosis111PDF.pdf. Accessed Dec. 22, 2012.
14. World Health Organization (WHO) Scientific Group on the Assessment of Osteoporosis at Primary Health Care Level. Summary Meeting Report. Brussels, Belgium, 5-7 May 2004.
15. Lewiecki EM, et al. Bone 2008;43:1115-1121.
16. The Writing Group for the ISCD Position Development Conference. J Clin Densitom 2004;7:17-26.
17. Wainwright SA, et al. J Clin Endocrinol Metab 2005;90:2787-2793.
18. Holick MF. N Engl J Med 2007;357:266-280.
19. Holick MF, et al. J Clin Endocrinol Metab 2011;96:1911-1930.
20. Khosla S. J Clin Endocrinol Metab 2010; 95:3-10.
21. Watts NB, et al. Endocr Pract 2010;16: 1016-1019.
22. Ralston SH, Uitterlinden AG. Endocr Rev 2010;31:629-662.
23. Richards JB, et al. Ann Intern Med 2009;151:528-537.
24. Ryan CS, et al. Osteoporos Int 2011;22:1845-1853.
25. National Osteoporosis Foundation (NOF)/International Society for Clinical Densitometry (ISCD) FRAX Implementation Guide. Available at: http://www.iscd.org/wp-content/uploads/2012/10/FRAXImplementationGuide_000.pdf. Accessed Nov. 30, 2012.
26. Kanis JA, et al. Osteoporos Int 2011; 22:2395-2411.
27. Krieg MA, et al. J Clin Densitom: Assess Skelet Health 2008;11:163-187.
28. Hans D, Krieg MA. IEEE Trans Ultrason Ferroelectr Freq Control 2008;55:1529-1538.
29. Gourlay ML, et al. N Engl J Med 2012;366:225-232.
30. Yu EW, Finkelstein JS. JAMA 2012;307:2591-2592.
31. National Osteoporosis Foundation Clinician’s Guide to Prevention and Treatment of Osteoporosis. National Osteoporosis Foundation, Washington, DC; 2008: 1-36.
32. U.S. Preventive Services Task Force. Ann Intern Med 2011;154:356-364.
33. Qaseem A, et al. Ann Intern Med 2008;149:404-415.
34. Qaseem A, et al. Ann Intern Med 2008;148:680-684.
35. Lim LS, et al; ACPM Prevention Practice Committee. Am J Prev Med 2009;36:366-375.
36. Institute of Medicine of the National Academies. Available at: www.iom.edu/Reports/2010/Dietary-Reference-Intakes-for-Calcium-and-Vitamin-D/Report-Brief.aspx. Accessed March 19, 2013.
37. National Institutes of Health. Office of Dietary Supplements. Available at: http://ods.od.nih.gov/factsheets/Calcium-HealthProfessional. Accessed March 19, 2013.
38. Straub DA. Nutr Clin Prac 2007;22:286-296.
39. Jackson RD, et al. N Engl J Med 2006;354:669-683.
40. Reid IR, et al. Osteoporosis Int 2011;22:1649-1658.
41. Reid IR, Bolland MJ. Heart 2012;98:895-896.
42. Li K, et al. Heart 2012;98:920-925.
43. Ross AC, et al. J Clin Endocrinol Metab 2011;96:53-58.
44. Kemmler W, et al. Arch Intern Med 2010;170:179-185.
45. U.S. Department of Health and Human Services. Available at: http://www.surgeongeneral.gov/initiatives/healthy-fitnation/obesityvision2010.pdf. Accessed March 19, 2013.
46. Moyer VA. Ann Intern Med 2012;157:197-204.
47. Reginster JY. Drugs 2011;71:65-78.
48. Cadarette SM, et al. Ann Intern Med 2008;148:637-646.
49. Black DM, et al. N Engl J Med 2007;356:1809-1822.
50. Body JJ. Acta Clin Belg 2011;66:443-447.
51. Rossouw JE, et al. JAMA 2002;288:321-333.
52. Cummings SR, et al. N Engl J Med 2009;361:756-765.
53. U.S. Food and Drug Administration. Available at www.fda.gov/Drugs/DrugSafety/ucm229009.htm. Accessed March 19, 2013.
54. Park-Wyllie LY, et al. JAMA 2011;305: 783-789.
55. Meier RPH, et al. Arch Intern Med 2012;172:930-935.
56. Sellmeyer DE. JAMA 2010;304:1480-1484.
57. Etminan M, et al. CMAJ 2012;184:E431-E434.
58. McClung M, et al. Am J Med 2013;126:13-20.
59. U.S. Food and Drug Administration. Available at: http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/DrugSafetyInformationforHeathcareProfessionals/ucm136201.htm. Accessed March 15, 2013.
60. Bone HG, et al. N Engl J Med 2004;350:1189-1199.
61. Black DM, et al. JAMA 2006;296:2927-2938.
62. Silverman SL, et al. Osteoporosis Int 2011;22:21-26.
63. Imaz I, et al. Osteoporosis Int 2010;21: 1943-1951.
64. Emky RD. Women’s Health 2008;43:219-228.
65. Rizzoli R. Q J Med 2011;104:281-300.
66. Visvanathan K, et al. J Clin Oncol 2009;27:3235-3258.
67. Love RR, et al. N Engl J Med 1992; 326:852-856.
68. Khosla S. J Clin Endocrinol Metab 2010;95:3569-3577.
69. Nelson HD, et al. Ann Intern Med 2012;157:104-113.
70. North American Menopause Society. Menopause 2008;15:584-602.
71. National Osteoporosis Guidelines Group. Available at: www.shef.ac.uk/NOGG. Accessed March 19, 2013.
72. Feldstein AC, et al. Osteoporos Int 2005;16:953-962.
73. Mazziotti GE, et al. Am J Med 2010;123:877-884.
74. Pitts CJD, Kearns AE. Mayo Clin Proc 2011;86:338-343.
75. Grossman JM, et al. Arthritis Care Res 2010;62:1515-1526.
76. Lustberg MB, et al. J Clin Oncol 2012;30:3665-3674.
77. Dawson-Hughes B, et al. Osteoporos Int 2008;19:449-458.
78. Jennings LA, et al. J Am Geriatr Soc 2010;58:650-657.
79. Marsh D, et al. Osteoporos Int 2011;22:2051-2065.
80. International Osteoporosis Foundation. Available at: http://www.iofbonehealth.org/capture-fracture-report-2012. Accessed Jan. 7, 2013.
81. National Bone Health Alliance. Eye on bone health: Secondary fracture prevention initiative. Volume 1. Washington, DC; 2011.
Osteoporosis is a common disease that causes significant morbidity and mortality. Its prevalence and costs to society are huge and its incidence is rising as the population ages. Osteoporosis affects people of all races and socioeconomic groups, and the prevalence in men is not far behind that of women, yet it remains underdiagnosed and undertreated.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
