Treating Pain in Pediatric Patients
Treating Pain in Pediatric Patients
Authors:
John Cheng, MD, Assistant Professor, Division of Pediatric Emergency Medicine, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA.
Michael Greenwald, MD, Emory University School of Medicine, Atlanta, GA.
Peer Reviewer:
Christopher J. Haines, DO, Associate Professor of Pediatrics and Emergency Medicine, Drexel University College of Medicine, Director, Department of Emergency Medicine, Medical Director, Critical Care Transport Team, St. Christopher's Hospital for Children, Philadelphia, PA.
It has been 13 years since the Joint Commission on Accreditation of Healthcare Organizations directed hospitals to treat pain as a "fifth vital sign." Since that time, a great deal has been written about pain physiology and treatment, and stories of grossly inadequate pain control have caught the attention of the lay press. National guidelines have detailed standards of care and recommended a variety of interventions to promote better pain control, and dozens of organizations have formed to champion better pain management for patients. Nevertheless, studies continue to show that, despite these developments, patients still report inadequate pain control during medical care encounters.1,2
This article will examine some of the impediments to better pain management in the ED. We will describe strategies to improve pain control using guidelines, non-pharmacologic, and pharmacologic approaches. While no single approach can effectively eliminate pain in all cases, the ED can greatly improve its overall pain management of pediatric patients through this multi-faceted approach.
Challenges in Pain Assessment
Knowing the severity of pain is critical in determining appropriate treatment strategies and measuring their effectiveness. While this may seem intuitive, pain assessment (or lack thereof) is often the underlying impediment to better pain control. The reasons for this include choosing a suboptimal tool for measuring pain, disputing the validity of the pain score, and inadequate reassessment of pain.
The gold standard of pain measurement is the patient's subjective report. There is no objective tool that accurately and reliably measures pain. Biochemical markers, behavioral cues, and physiologic responses, when used alone, have failed to provide reliable measures of acute or chronic pain. Another important idea is that the best tools for subjective pain measurement must take into account the patient's developmental and verbal abilities. For example, in general, patients who have a developmental level of at least a 7-year-old can use a numerical pain rating system, most commonly defined as a 0-10 numerical score, with 0 equal to "no pain" and 10 equal to "the worst possible pain." Using such a rating system requires the patient to be able to grasp several ideas: the concept of increasing numbers in a series, how to correlate the severity of pain to a numerical system, and what the "worst possible pain" actually means. Alternatively, the "Faces" pain scale is typically used for patients who function at a level of a 3-year-old. This correlates symbolic facial expressions with pain severity. This is an exercise in analogy that is beyond the ability of those who function below a 3-year-old's developmental level. There are many variations on the traditional "Faces" pain scale(e.g., the Wong Baker Face scale and the Oucher). Each of these variations attempts to control different aspects inherent in the tools (real vs. cartoon faces, ethnicity of the faces in the pictures, severity of expressions, etc.), in hopes of getting a more reliable scoring tool.
Below this age level there are no standardized tools for measuring pain by self-report. As a proxy, measurement of physical responses to pain is often used. There are several scales designed for this group, each geared toward a particular patient population. One of the better-known scales is the FLACC (faces, legs, activity, cry, consolability) scale.3
The next challenge is interpreting the pain score. It is not uncommon for children to report pain scores out of proportion to their affect. When a child reports that pain from an ankle injury is a "10" while completely engrossed in a video game, it is not surprising that clinicians disbelieve the report and treat the pain more to the level of the child's affect rather than the reported score. This common experience undermines the credibility of the pain score for many clinicians, as well as some parents.
One possible explanation of the "exaggerated" pain score is that it is based on the limitation of personal experiences. It is not surprising that a usually healthy child with acute pharyngitis might describe the level of pain as a "10." The throat pain is most likely the worst pain he can recall. In contrast, a patient whose past experiences include fractures or kidney stones will have a very different perspective on pain severity. The issue is not whether the patient's pain score is "believable," but rather what to do with that information. The primary purpose of the pain score is as a reference point for effectiveness of treatment.4 A child may not have a mature perspective on pain severity in general, but he can usually reference different levels of pain within a given acute illness.
Rejecting or verbalizing disbelief about a patient's pain report is an unfortunately common event. When it happens, it undermines the confidence a patient has in the clinician and potentially sets up an adversarial relationship.5 In short, no good can come of arguing with a patient's pain report. That by no means obligates the clinician to aggressively treat pain reports that are inconsistent with a patient's affect. The inconsistency should prompt the clinician to explore carefully for any contributing factors such as secondary gain and anxiety, while carefully titrating pain treatment. When dealing with chronic pain (e.g., sickle cell pain crisis), the ED physician should defer to the primary care provider, if available, or pain specialist who has an established relationship with the patient and family.
Finally, perhaps the biggest systemic failure in pain assessment stems from a lack of follow-up. Many institutions effectively record pain scores only to subsequently ignore them. Abnormal findings such as an elevated pain score require not only treatment but also continued reassessment. The clinician should make every effort to safely "normalize" the finding to the patient's baseline or acceptable levels. Simply put, pay attention to pain; assess and reassess after interventions.
Institutional Approaches
Pain medicine is now an internationally recognized medical specialty and is supported by hundreds of specialists, many texts, and a broad array of research. Clearly there is no lack of information on why pain occurs, how it is harmful, and how it is treated. Why, then, do clinicians provide inconsistent care in this area? There are many postulated reasons, including competing demands for time and resources, suspicions about addiction, and discomfort with different treatment approaches. The challenge is not only how to educate but also how to modify clinician attitudes and behavior.
The first avenue for improving pain management through policy is to focus on the standard of care. Clinicians have a variety of approaches to similar conditions based on their training and personal experiences. The same patient with a gluteal abscess may have an incision and drainage with local anesthesia from one physician and deep sedation from another. One physician may provide a sickle cell pain crisis patient with a dose of ketorolac, while another orders morphine. While it is important to allow for some variations in practice, the goals should remain consistent. This is where policy can play a productive role.
Institutions should clearly state their position on pain management, taking into account established standards of care. This statement should include a goal of quickly and safely administering pain relief for all patients. The ED should emphasize that the priority in choosing a treatment approach to painful procedures is based on subjecting the patient to the least amount of discomfort in the safest possible manner. While such statements cannot address every single case, they can be used to change the culture so that pain relief is valued over expediency.
A more tangible way to use policy is through the implementation of standing orders. Standing orders must follow CMS guidelines. In 2008, CMS restricted the use of standing orders. The American College of Emergency Medicine was successful in convincing CMS to allow standing orders, provided the physician later sign them so as not to delay emergency treatment. However, several states do restrict standing orders, and individual hospitals may have policies as well.
One area of concern with respect to pain management of children in the ED is the time it takes to receive any treatment for pain, let alone effective reduction in pain. Numerous studies have demonstrated that it is common for pediatric patients to wait for hours before receiving medication for severe pain from fractures, burns, and even more common conditions such as otalgia. Standing orders will allow these patients to receive something for their pain quickly, without having to wait for a full evaluation by a physician. Similarly, nurse-initiated guidelines can help identify patients who will likely require placement of a peripheral intravenous catheter and apply topical anesthetics such as lidocaine creams before the physician sees the patient. By empowering pre-hospital providers and ED nurses with standing orders for analgesics and nurse-initiated protocols, the time to first analgesic dose is reduced dramatically, analgesic adjuncts are used to their full extent, and overall pain scores improve.
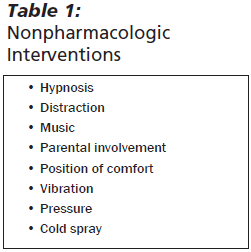
Non-pharmacologic Approaches
As the saying goes, "If you have a hammer, all the world's a nail." This is quite true when it comes to pain management. A child with back pain will likely get a very different recommendation depending on whether the patient sees a surgeon, anesthesiologist, neurologist, psychologist, chiropractor, or acupuncturist. Because pain is a complex bio-psycho-social experience, the most effective approaches address several different aspects of pain, not just one. In addition, by using non-pharmacologic interventions to attenuate anxiety, the clinician can effectively treat pain with less medication and, therefore, with fewer medication side effects.
Going to the ED is an anxiety-provoking event for most people, especially children. Such anxiety would understandably be influenced by any prior experiences in the ED or other medical facilities, as well as by what the child is expecting to happen (whether imaginary or real). Several studies have looked at pain responses in children and have found that poorly controlled pain from a variety of procedures (e.g., heel sticks) led to an almost hyperalgesic response to painful procedures later in life.6,7 In addition to the anxiety fostered by memories of previous experiences, real or imagined anxiety can also modulate the pain response.8 Rajasagaram et al looked at pain scores in children presenting to the ED, comparing those obtained in the waiting room to those obtained in a triage room.9 They found that the self-reported scores were lower in the triage room, as opposed to in the waiting room, and concluded that the anxiety level in the waiting room adversely affected the perceived amount of pain the patient was reported.
The Family Factor
A family arrives at the ED with their ill or injured child, often after struggling to manage the symptoms at home. Their decision to accept the cost and inconvenience of the visit is, in a sense, an admission of failure and desperation. The expectation is that their child will feel better once they get to the hospital. The unfortunate reality, however, is that their arrival to the hospital is just the first step in a process of getting pain relief. First comes triage and registration. Then they may see several staff members who can neither diagnose nor begin treatment while waiting for the physician. Even after the physician's interview, relief from the symptoms may take a significant amount of additional time. It should be no surprise that the family and patient feel (and express) increasing frustration and anxiety. This, in turn, can fuel the pain experience, setting up a vicious cycle of distress.
During the physical examination, it helps to demonstrate care in not causing the child any unnecessary discomfort. Something simple such as the child's position during the exam can be a source of anxiety.10 Encourage a small child to remain in the parent's lap for the exam. Examine the most sensitive areas last. Engage and distract the child with age-appropriate conversation. To round out the interview, explain your recommendations and the process while the family is in the ED and, of course, invite questions. Children in pain are naturally fearful. Anything you can do to alleviate their anxiety (and their parents') can make a large impact on the success of your treatment.
One specific example of how a child's anxiety can be modulated is the child's response to being separated from a parent during the different parts of the examination and treatment. The issue of family presence during procedures has been the focus of considerable attention in medical literature and hospital policy. Traditional assumptions that most parents and children will be better off if separated during procedures have been rejected by research results; however, this belief persists in many EDs.11 Research in this area has shown that most parents have more anxiety when separated from their children during common procedures such as placement of a peripheral intravenous catheter or lumbar puncture. Studies of pediatric patients have shown no evidence that parental presence adversely affects the success of these procedures.12
The keys to success with family presence during a procedure are preparation and education. Explain to the parents what to expect and the possibility of unsuccessful attempts. Point out that some parents may feel ill during a procedure and need to sit or leave the room. Give them specific things to do: Instruct them on where to sit or stand, what kinds of things to say, and what not to say. Common mistakes parents make in trying to console their child include minimizing the child's pain or calling attention to it. The emphasis should be on positive reinforcement of good coping skills and voluntary distraction. With practice and a small amount of preparation, parents who wish to stay and help their child through a brief procedure can provide unique and invaluable assistance in coping with the situation.
There are times when families may refuse analgesia for the child. They may worry about addiction or other consequences of medication. When dealing with males, families may project their cultural stereotypes to "take it like a man." Careful intervention is needed in these cases to both win over the parent as well as treat the child. Parents need to know that brief treatment with pain medication does not lead to addiction.
Voluntary Distraction (Hypnosis)
Many readers will associate hypnosis with theatrical settings and have serious doubts about its authenticity and/or efficacy in a clinical setting. In fact, hypnosis predates much of what is regarded as modern medicine. It was developed by Dr. Mesmer in the late 1700s to help patients cope with painful procedures long before the advent of purified morphine and other anesthetics. Today, hypnosis is used in a variety of clinical settings, including, yes, the emergency department.
There are a variety of skills involved with hypnosis, but at its most basic level, it is a form of voluntary distraction. Most people have observed a child (or adult for that matter) who is so engrossed by a television show that even loud sounds are seemingly unnoticed. Researchers have demonstrated that this effect is powerful enough to obscure the experience of an intramuscular injection in young children. This is also effective for other procedures, particularly those in which fear is the main cause of distress. Puzzles, books, games, flashing and moving lights, a movie or cartoon, and singing/music can all be used to refocus attention.14
The effectiveness of the distraction relies on the skill of the distracter and the age and interests of the patient. For example, flashing and moving lights will work better for an infant, while singing or a TV program will work better for toddlers and school-age children. It should be noted that any successful distraction relies on the willingness of the subject to be distracted. Insisting that a child "look away" from the procedure only calls attention to it. In fact, some patients are far more stressed at having their view blocked than by seeing what is being done. In these cases, the following approaches are often helpful: Explain the procedure step by step using age-appropriate language; allow the child to see and hold some of the equipment (e.g., an IV catheter without the needle, or an empty staple gun); and answer questions truthfully. Decide what to say to the question, "Will it hurt?"15 Child life specialists are tremendous assets in these situations, as they are trained in both child development and distraction techniques.16 Individuals skilled in child behavior can reduce the anxiety of a child without medications.
Gate Theory Pain Blockade
Another inexpensive and rapid intervention to blunt some forms of needle pain relies on what is known as the "gate theory." Originally proposed by Melzack and Wall, and later modified by Melzack and Casey, this theory postulates that the dorsal root ganglion acts as a gate that is modulated by previous pain experiences and that modifies both the excitatory and the inhibitory pain impulses coming from the body, as well as those coming from the brain.17 Simply put, the impulses of a noxious stimulus such as cold or pressure can prevent or mask the impulses of sharp pain from a percutaneous needle. For example, several studies have looked at the use of pressure to reduce the pain of IM injections. In these cases, the application of pressure to the site of an injection for several seconds prior to the administration of the injection seems to diminish the pain of the injection itself.18 The idea is that applying pressure will send impulses up the dorsal root ganglion. When the injection is given and causes more pain fibers to be fired, the impulses cannot get through the dorsal root ganglion very well, and, thus, there is less perceived pain. This theory may explain the efficacy of other "shot blocker" instruments, including vapocoolant sprays (or "cold sprays"). While not extensively studied, these interventions have varying degrees of effectiveness, at times limited by how noxious the intervention itself is perceived to be. In general, interventions such as cold spray are reserved for older children who can make a choice in terms of analgesia and who can understand the purpose of the intervention.
Medications
Anxiolysis through non-pharmacologic means provides a safe and effective way to reduce the need for medication in many patients. Unfortunately, not all patients are amenable to these approaches, and many procedures and conditions found in the ED are simply too noxious for most patients to tolerate. It is therefore essential for any ED physician to become expert at using medications. This requires a comprehensive understanding of the drugs' pharmacologic properties, contraindications, and side effects. This section will review medications used primarily for the treatment of acute pain, as chronic pain is beyond the scope of this article.
As with any medication, any one of multiple routes may be used. When choosing a route of administration, one has to assess the degree of pain (usually based on pain scores), the onset and duration of analgesia, and whether the benefits of a given plan outweigh the risks, particularly in the clinical setting (e.g., past medical history, allergies, etc.). The following sections will discuss some of more salient points of commonly used medications. Specific details about dosing and pharmacokinetics can be found in Tables 3-6.
Non-Opioids
Acetaminophen is a commonly used pain medication; although it works well for mild pain, its mechanism of effect is not well understood. Nonsteroidal anti-inflammatory drugs (NSAIDs) may provide effective analgesia for mild to moderate pain without the opioid side effects of somnolence or respiratory depression. Ibuprofen and naproxen, both NSAIDs, are commonly used medications. Both of these are well-tolerated and widely available. Aspirin is not recommended due to its association with Reye syndrome. The most common IV NSAID is ketorolac (Toradol). Overall well-tolerated, it is often used to treat migraine headaches and sickle cell pain crises (in combination with an opiate). Like all NSAIDs, it has the potential to cause gastric upset. It is generally not used in its IV formulation for more than 2-3 days in a row, as the incidence of GI ulceration, bleeding, and perforation increases dramatically with prolonged use. Given its renal clearance, it should be used with caution in children with impaired renal function. Any oral analgesic is limited in its onset, typically 30 minutes in patients with a healthy GI tract. (See Tables 2 and 3.)
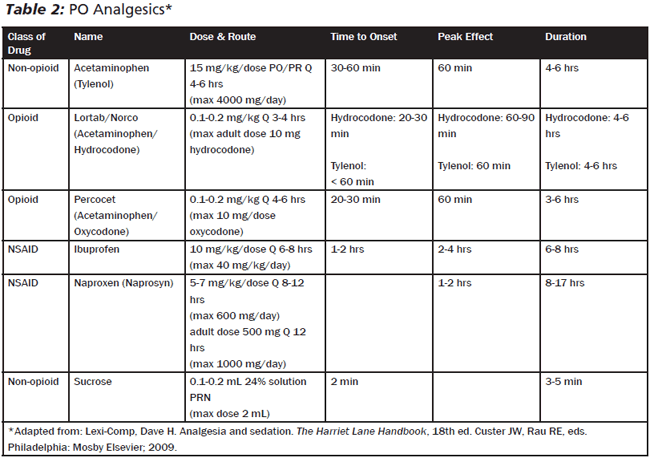
Another analgesic option for infants younger than 3 months of age is a 24% sucrose solution. When given during common medical procedures (e.g., heel lancing, bladder catheterization, IM vaccination, IV placement, phlebotomy), application of this solution orally reduces pain scores for infants when compared with placebo. The exact mechanism of how sucrose works remains elusive. The analgesic effect is not produced with artificial sweetener. Sucrose solution produces EEG changes similar to that found from opioids and is reversible by Naloxone. No efficacy has been demonstrated for patients older than 3 months of age. While these findings are intriguing, sucrose solution should be considered an adjunct for painful procedures rather than a primary intervention.19
Opioids
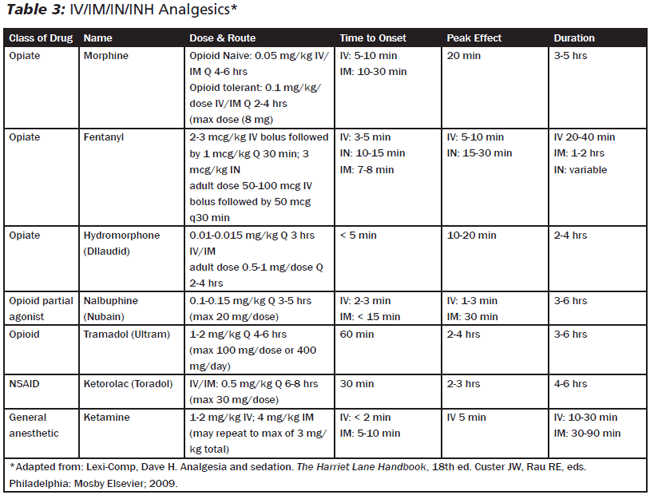
Opioids (specifically morphine) are the gold standard for analgesics. Morphine is the oldest medication, derived centuries ago from the seeds and, later, from the extract of the poppy plant (Papaver somniferum). From this original alkaloid, the rest of the opiates were derived and are more potent, each to a varying degree. Their unique features include effectiveness for most forms of pain and no ceiling effect (i.e., no additional effects with higher dosing). The most common side effects of opioids include nausea, respiratory depression, and somnolence. However, when dosed appropriately, such adverse events are unlikely (and reversible with naloxone, see below). Commonly used oral opioids include hydrocodone, oxycodone, and hydromorphone. (Codeine has fallen out of favor due to its higher rate of GI side effects and limited effect in people who lack the enzyme [CYP2D6] to convert codeine to hydrocodone. It is important to note that Tylenol with codeine and other codeine-containing drugs carry a black box warning. Children receiving these drugs, even in normal doses, have died when given these drugs after tonsillectomy/adnoidectomy.) Hydrocodone has a liquid formulation that is more widely available than oxycodone. Hydromorphone (Dilaudid) is more potent than the opioids listed above and generally reserved for patients with very high degrees of pain, opioid tolerance, or allergic reactions to other opioid formulations. Long-acting formulations of opioids (e.g., Oxycontin, Morphine IR, Methadone, etc.) are not commonly used in the acute setting of the ED visit. (See Table 2.) Many analgesics and anxiolytics are effective via multiple routes: PO, SC, IM, IV, IN, transdermal, even nebulized. (See Tables 2 and 3.)
Fentanyl has many advantages for ED purposes: potency, rapid onset, and short duration. Intranasal fentanyl is less potent than IV administration but may be effective to help sedate and provide a modest degree of analgesia. All opioids tend to cause pruritis, most notably morphine (urticaria) and fentanyl (nasal pruritis). It is thought to be the result of a release of histamine and usually responds well to a dose of diphenhydramine. Opiates will also slow gut motility, resulting in nausea and vomiting, as well as constipation with chronic use. Fentanyl is thought to have less of a hemodynamic effect and cause less dysmotility of the GI tract. It is associated with a unique paradoxical effect on ventilation known as "rigid chest syndrome," typically reported in infants. These cases may require either reversal with Naloxone or paralysis to facilitate intubation. Demerol should be avoided due to its relatively poor side-effect profile and risk of seizures in patients with decreased renal function.
Nalbuphine (Nubain) is a mixed agonist-antagonist opioid that may have a beneficial role in patients with particular sensitivity to common opioid effects such as constipation and respiratory depression. Care should be taken when using this in a patient who is on chronic opiates, given its antagonistic effects. Tramadol (Ultram) is an analgesic that is not technically an opiate but binds opioid receptors, resulting in similar effects to morphine. It is known to cause dizziness, but not as much respiratory depression or somnolence as other opioids. Of note, it has been associated with seizures, so care must be taken when there is a history of seizures or head injury.
One of the benefits of using opioids for analgesia is the availability of a reversal agent: naloxone (Narcan). It works quickly to block opioid receptors, but typically lasts for less than 30 minutes. Care must be exercised when dosing the naloxone, as it might reverse all analgesia (both from recent or chronic doses), resulting in severe pain. In some cases, a constant infusion is needed at a lower dose to balance between sufficient analgesia and opiate side effects.
There are two common misconceptions about opiate use in children. The first concerns the confusion between tolerance, dependence, and addiction. Tolerance to the effects of opioids, like that of other medications that act on receptors, will occur in most patients after regular use for approximately one week. Some opioids tend to lead to tolerance more quickly (e.g., fentanyl). Opioid tolerance can be addressed by either increasing the dose, changing the opioid, or using non-opioid adjuncts. Opioid dependence may occur after regular use of an opioid for more than one week. This refers to the withdrawal symptoms experienced when opioids are sharply reduced or discontinued. This is treated or prevented by slowly reducing the dose (tapering) and with the use of non-opioid adjuncts. Opioid addiction is an extremely uncommon occurrence in pediatric patients. This condition refers to the psychological craving for the euphoria from opioids that can lead a patient to do harmful things to themselves and others in pursuit of more medication. None of these issues — opioid tolerance, dependence, or addiction — are an expected occurrence in the acute setting of an opioid naïve patient in the ED. Unfortunately, some parents and providers fear that a few doses of opioid can lead to dependence or addiction and may attempt to block their use for pediatric patients. These issues must be understood and confronted openly as they occur.
The second opioid myth is that these medications will delay diagnosis and lead to increased morbidity by masking tenderness in the acute abdomen. Multiple studies of patients with appendicitis have shown this not to be the case.20,21,22 While many centers have relaxed these concerns, the overall fear of opioids masking pain and delaying diagnosis persists for other conditions without evidence.
Local and Topical Anesthetics
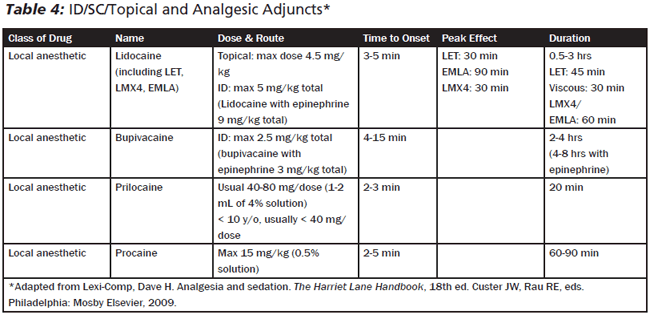
In order to control pain with the least amount of systemic medication possible, clinicians should be adept with the use of local and regional anesthetics. The main limitations to the use of these drugs include allergic reactions (uncommon), pain from injection, localization of the nerves for blockade, limits of duration, and toxicity in high doses.
There are two families of injected anesthetics: esters (e.g., tetracaine, benzocaine, procaine) and amides (e.g., lidocaine, bupivacaine, mepivacaine). Patients allergic to one family of anesthetics may tolerate anesthetics from the other family. Short-acting anesthetics have varying times to onset and durations. (See Table 4.) One effective approach for procedural pain is to mix medications (e.g., lidocaine and bupivacaine) in a 1:1 ratio when quick onset and longer duration of analgesia are desired. Of note, the acidity of the medication solution causes additional pain with injection. This effect is blunted by the addition of sodium bicarbonate (9:1 ratio).23 The maximum dose for lidocaine without epinephrine is 4 mg/kg, up to a maximum of 300 mg for child older than 8 years. The maximum dose of lidocaine with epinephrine is 7 mg/kg up to a maximum of 500 mg/kg for child older than 8 years. For children younger than 8 years, the dose is 80% of the doses above. To reduce the risk of toxicity (tinnitus, arrhythmias, seizures), the clinician may choose to use lidocaine with epinephrine, which slows the release of the medication and may allow for higher doses. Alternatively, a smaller amount of medication is required if a regional block is used instead of local infiltration. Finally, the pain from injection may be blunted (via the gate theory as described above) with the concurrent application of cold spray at the site of injection. For most children younger than 8 years of age, the concept that a brief painful procedure (e.g., lidocaine injection) will prevent subsequent pain (e.g., laceration repair) is too abstract. They will only focus on their fear of anesthetic injection. For age-appropriate patients, a clear explanation of the injection can help reduce fear of the procedure. Attention must be paid to the fact that these medications, while they do work quickly, they do not work immediately.
There are several specific applications of local and regional anesthesia that are particularly useful in the ED. The "Bier block" is often used in upper extremity fractures. Lidocaine is instilled intravenously after a specialized cuff isolates the affected extremity from the general circulation. Once the painful manipulations to set the fracture are done, the cuff releases the lidocaine in controlled bursts into the general circulation so as not to cause arrhythmias or seizures. Please note that standard blood pressure cuffs will not provide the same control and may allow a rapid release of lidocaine into the circulation, causing toxic and even fatal results.
Another useful approach is the use of topical anesthetics. These are particularly useful interventions for needle phobic patients. The three areas of application are broken skin, intact skin, and mucosal surfaces. When the skin surface is already broken, local anesthesia may be achieved using products such as LET (lidocaine, epinephrine, and tetracaine) or TAC (tetracaine, adrenaline, and cocaine). These medications are most effective in the face and scalp, but less reliable on other parts of the body. Another use of topical lidocaine is the use of lidocaine jelly for second-degree burns and large areas of abrasions. When applied five minutes prior to wound cleaning, it is extremely effective for analgesia.
Anesthesia through intact skin offers a different challenge: getting a water-soluble anesthetic through the phospholipid bilayer of the skin. One approach is to embed the water-based drug within a lipid. Two products are commonly used: EMLA (eutectic mixture of lidocaine and prilocaine) and LMX4 (lidocaine embedded within liposomes). These are effective dermal anesthetics for procedures such as IV placement and port-a-cath access. Their limitations include their onset time and duration. EMLA is effective to a depth of approximately 3 mm after an hour. LMX4 appears to be effective within 30 minutes, but no depth studies are available. In contrast to some reports on EMLA, LMX4 does not obscure visualization of veins and is therefore often preferred for placement of peripheral IV catheters. It is important to remember that lidocaine is absorbed from topical applications. Applying these products to a large surface area can cause toxicity. Further, their absorption needs to be calculated into the maximum dose of local anesthetic administered.
There are additional products available that also provide effective local anesthesia for percutaneous needle pain. The J-tip® is a needle-less auto-injector system that uses CO2 to propel a drug (e.g., lidocaine) into the skin. Its advantage over the topical creams is its more rapid onset (2-5 min vs. 30 min). Its drawbacks include noise and cost. When this system is used, a loud noise is heard on injection, which may startle young patients. Its cost is slightly more than the topic creams; however, its rapid onset and painless application make it favored in many EDs.24 A newer product named "Buzzy"® uses a dual approach for local anesthesia ("gate theory"): vibration and cold. This product is effective, drug-free, and visually appealing (which may offer challenges in keeping it in stock in the ED). The cost is similar to vapocoolant spray (< $0.50/use) with greater effectiveness in at least one study.25
Finally, topical anesthetics alone are used effectively on mucosal surfaces such as the oral cavity (e.g., viscous lidocaine for mouth ulcers, nasogastric tube placement, esophagitis, or gastritis), the sclera and cornea, the tympanic membrane (e.g., for otitis media), and the urethra (e.g., lidocaine jelly for urethral catheterization). There are a few precautions with the use of these medications. These medications should be avoided when surfaces such as the eye or eardrum are not intact. Benzocaine (Hurricaine spray) may cause local edema of mucosal surfaces. Benzocaine and prilocaine are associated with cases of idiosyncratic methemoglobinemia, particularly in younger children. As with all short-acting anesthetics, it is important to remember that the onset of action requires at least a few minutes.
Pharmacologic Anxiolysis
As mentioned above, more often than not, there are two issues working synergistically when children present to the ED: pain and anxiety. While distraction and pain medication can frequently help control, or at least blunt, the child's anxiety, anxiolytic medications are sometimes needed. The most common anxiolytics are benzodiazepines: midazolam (Versed), diazepam (Valium), and lorazepam (Ativan). They produce sedation and amnesia, in addition to anxiolysis. In high doses, or if a child is particularly sensitive to the medication, these drugs can cause profound hypoventilation and even apnea, requiring either the use of a reversal agent or ventilatory support (e.g., intubation). The reversal agent for benzodiazepines is flumazenil (Romazicon). Like naloxone, it works well, but has a short half-life. It, too, must be used with caution (if at all) in a patient who is on chronic benzodiazepines; its administration in this setting is likely to cause a seizure.
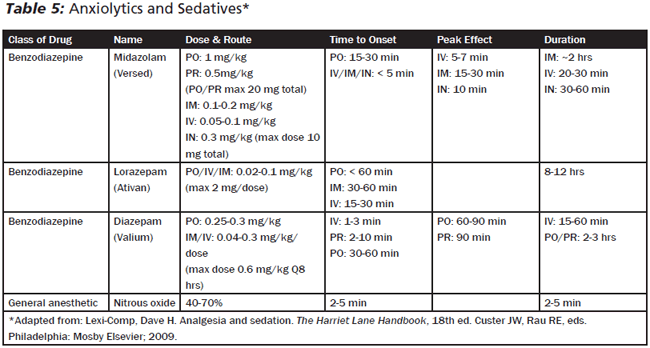
While lorazepam is commonly used for agitated patients, midazolam's shorter onset and duration makes it preferable for brief procedures. It can be given via several routes: IV, IN, IM, PR, PO. The onset of action is 15-25 minutes for non-injection (i.e., non-painful) routes. They may be safely combined with oral opioids. On rare occasions, paradoxical excitement may occur; these symptoms typically resolve without intervention but may be reversed with flumazenil, if needed.26 Of note, when giving midazolam via the IN route, it does cause a burning sensation in the nasal mucosa. The maximum dose is about 5 mg per nostril; this is a function of the concentration of the midazolam, since more than 5 mg would require too large a volume to be effective with an atomizer. What ends up happening is that the dose becomes a PO dose, rather than an IN dose. Even when midazolam has the desired effect, it is not a bad idea to let parents know that, as it starts to wear off, their child may go from being sleepy and happy to dysphoric and agitated. These effects wear off without any intervention.
Nitrous oxide (N2O) has a long track record for minor procedures. Its advantages include rapid onset and offset, no incidence of allergic reactions, easy titratability to individual needs, and safe effect. Dysphoric reactions are far less common than with midazolam. Contraindications to its use include any pathologic gas trapping (e.g., pneumothorax, recent body cavity surgery, acute lung disease, otitis media, or sinusitis) since N2O can diffuse into these spaces and expand, creating further problems. N2O works very well to provide anxiolysis and mild analgesia for brief procedures such as foreign body removal, relocation of joints, IV placement, bladder catheterization, and lumbar puncture. Keys to success include allowing the child some time to play with the mask prior to sedation, engaging them in the titration, and praising patients for cooperation. On occasion, during prolonged exposure at higher concentrations, some children will become nauseated and vomit. In addition, dizziness and headaches are other occasional complaints. These symptoms resolve quickly as the N2O is washed out of their system and may be prevented by administering 100% oxygen for several minutes after the N2O is turned off.
Deep Sedation for Painful Procedures
The ED is unique in its frequent need to perform painful procedures. Some procedures such as abscess incision and drainage, as well as some major joint dislocations and fractures, require a greater degree of analgesia than one can safely provide with local anesthetics or systemic opioids. One of the most useful drugs available for this situation is ketamine. Ketamine is a dissociative anesthetic that acts on opioid receptors, resulting in analgesic, anxiolytic, and amnestic effects. In doses of 1-2 mg/kg IV, it reliably causes deep sedation without respiratory depression. It has a stimulatory effect on the cardiovascular and respiratory systems; it also increases cerebral blood flow and bronchodilates. As a result, ketamine causes tachycardia and hypertension, but does not typically cause respiratory depression like the opiates. Because of the hypertension and increased cerebral blood flow, it is theoretically not a good choice in the setting of head injuries. Given its bronchodilatory effects, it is useful for severe asthma attacks in intubated patients or in those who are not tolerating ventilatory support such as CPAP or BiPAP. Its onset of effect is typically heralded by nystagmus and tachycardia. In addition, it does increase secretions, particularly saliva and tears. Often it is helpful to let the parents know this ahead of time so that they are not alarmed when they see these effects of the drug. These sialogogue effects can be minimized with pretreatment with atropine or glycopyrrolate. The most severe side effects of ketamine include laryngospasm and the emergence reaction. In rare cases, obstruction from laryngospasm requires intubation, but more often it improves with deep pressure applied behind the angle of the mandible. Older children (pre-teen and teenager) may experience an emergence reaction in which the child wakes up from the ketamine violently flailing and vocalizing. This reaction is idiosyncratic and rare in younger children. Benzodiazepines such as midazolam have been reported to help with ameliorating this reaction. A more common side effect is nausea and vomiting, usually seen as the child wakes up after a higher dose (> 1.5 mg/kg IV) of ketamine. This can be preempted by using adjuncts such as propofol in conjunction with ketamine (typically 1 mg/kg each) that facilitate effective sedation with a lower dose of ketamine.
The discussion and fine points of procedural sedation are beyond the scope of this review article. However, being familiar with the requirements for procedural sedation and being comfortable with the use of these medications, either alone or in combination, is an essential skill set for the c emergency physician.
Discharge/Homecare Instructions
Once a child has been treated with analgesics (and/or anxiolytics) in the ED, it is important to have a discussion with the family about care after discharge. There are two parts to this issue. The more obvious issue is that of what to expect at home as the medications given in the ED wear off. Will there be any dysphoria or agitation. Will the pain return? The second issue that is not quite as obvious is the importance of continuing with analgesics at home. There are actually two components to this. The first is dependent on the ED providers: It is imperative to remember that the pain of a given pathology does not end at the ED doors. Prescriptions and instructions for appropriate doses of oral analgesics for home should be given to these patients and their families. While this point would seem obvious, oligoanalgesia with subtherapeutic dose of analgesics for home has been a persistent problem over time.27 The second component is the assumption that the parents will be diligent about giving analgesics at home, and that they do not want to have their child in pain. Unfortunately, parents also tend to underestimate their child's pain level, much like healthcare professionals.9 One study reported that, when surveyed, parents did not continue pain medications at home as we would have expected them to, often waiting until the child is in extreme distress before giving the medication.28 Stressing to parents the need for continued reassessment and administration of analgesics at home will help control the child's pain and hopefully speed recovery.
In the ED, almost every child presenting for care has some component of pain and anxiety. Despite an increasing understanding of how it works and ways to treat it, effective pain control remains a challenge. This is most likely due to a lack of attention to pain and discomfort with analgesic approaches. We can overcome these issues with improved education, guidelines, and policies. The regular use of age-appropriate pain scales creates an environment where pain is regarded as an important problem. Nurse-initiated protocols and standing orders provide patients with prompt analgesia. Treatment of pain can range from simple non-pharmacologic interventions such as distraction, hypnosis, and the use of other sensory input (e.g., pressure or cold application) to minimize pain sensations and, of course, to medications. There is a whole panoply of medications, each with its own pharmacokinetics, benefits, and risks. The ED physician should become familiar with most, if not all, of these medications to help create an "ouchless" ED as best as possible for the child's current and future health. The challenge lies not in more effective treatments, for we have plenty of effective interventions at our disposal. The key is recognizing which treatment approach best addresses the clinical situation, i.e., which is the right tool for the right job. As systems change the culture of tolerance to needless suffering of patients regardless of age, consistent effective pain control will occur, and the "ouchless" ED will become the new norm.
References
- Probst BD, Lyons E, Leponard D, et al. Factors affecting emergency department assessment and management of pain in children. Ped Emerg Care 2005;21:298-305.
- Cimpello LB, Khine H, Avner J. Practice patterns of pediatric versus general emergency physicians for pain management of fractures in pediatric patients Ped Emerg Care 2004;20:228-232.
- Beyer JE, Wells N. The assessment of pain in children. Ped Clin North Am 1989;36:837-853.
- Scott J, Huskisson EC. Graphic representation of pain. Pain 1976;2:175-184.
- Miner J, et al. Patient and physician perceptions as risk factors for oligoanalgesia: A prospective observational study of the relief of pain in the emergency department. Acad Emerg Med 2006;13:140-146.
- Kennedy RM, et al. Clinical implications of unmanaged needle-insertion pain and distress in children. Pediatrics 2008;122:S130-S133
- Weisman SJ, et al. Consequences of inadequate analgesia during painful procedures in children. Arch Pediatr Adoles Med 1998;152:147-149.
- Walco GA. Needle pain in children: Contextual factors. Pediatrics 2008;122:S125-S129.
- Rajasagaram U, et al. Paediatric pain assessment: Differences between triage nurse, child, and parent. J Paediatrics Child Health 2009;45:199-203.
- Ali S, et al. Pain management of musculoskeletal injuries in children: Current state and future directions. Pediatr Emerg Care 2010;26(7):518-524.
- Henderson DP, Knapp JF. Report of the National Consensus Conference on Family Presence During Pediatric Cardiopulmonary Resuscitation and Procedures. Ped Emerg Care 2005:21:787-791.
- Bauchner H, Vinci R, Bak S, et al. Parents and procedures: A randomized controlled trial. Pedatrics 1996;98:861-867.
- Eppich W, Arnold L. Family member presence in the pediatric emergency department. Curr Opin Ped 2003;15:294-298.
- Cohen L. Behavioral approaches to anxiety and pain management for pediatric venous access. Pediatrics 2008;122:S134-9.
- Brown JL. Imagination training: A toll with many uses. Cont Peds 1995;12:22-36.
- Stevenson MD, Bivins CM, O'Brien K, et al. Child life intervention during angiocatheter placement in the pediatric emergency department. Ped Emerg Care 2005;21:712-718.
- Lee JS. Pain measurement: Understanding existing tools and their application in the emergency department. Emerg Med 2001;13(3):279-287.
- Chung JWY et al. An experimental study on the use of manual pressure to reduce pain in intramuscular injections. J Clin Nurs 2002;1(4):457-461.
- Harrison D, et al. Analgesic effects of sweet-tasting solutions for infants: Current state of equipoise. Pediatrics 2010;126:894-902.
- Green R, et al. Early analgesia for children with acute abdominal pain. Pediatrics 2005;116:978-983.
- Pace S, Burke TF. Intravenous morphine for early pain relief in patients with acute abdominal pain. Acad Emerg Med 1996;3(12):1086-1092.
- LoVecchia F, et al. The use of analgesics in patients with abdominal pain. J Emerg Med 1997;15(6):775-779.
- Scarfone RJ, et al. Pain of local anesthetics: Rate of administration and buffering. Ann Emerg Med 1998;31:36-40.
- Lysakowsli C, et al. A needle-free jet injection system with lidocaine for peripheral intravenous cannula insertion: A randomized controlled trial with cost effectiveness analysis. Anesth Analg 2003;96(1):215-9.
- Baxter AL, et al. An integration of vibration and cold relieves venipuncture pain in a pediatric emergency department. Ped Emerg Care 2011;(27) 12:1151-56.
- Thurston TA, et al. Reversal of a paradoxical reaction to midazolam with flumazenil. Anesth Anal 1996;83(1):192-193.
- Petrack EM, et al. Pain management in the emergency department: Patterns of analgesic utilization. Pediatrics 1997;99:711-714.
- Zisk RY, et al. The squeaky wheel gets the grease: Parental pain management of children treated for bone fractures. Pediatr Emerg Care2008;24(2):89-96.
- Cohen, et al. Evidence-based assessment of pediatric pain. J Pediatr Psychol 2008;33(9):939-955.
- Dave H. Analgesia and sedation. The Harriet Lane Handbook, 18th ed. Custer JW, Rau RE, eds. Philadelphia: Mosby Elsevier; 2009: 153-154.
- LexiComp. http://www.crlonline.com.
- Atkinson P, et al. Pain management and sedation of children in the emergency department. BMJ 2009;339:1075.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
