Lung Recruitment Maneuvers in Acute Respiratory Distress Syndrome
By Richard H. Kallet, MS, RRT, FAARC, FCCM, Director of Quality Assurance, Respiratory Care Services, San Francisco General Hospital. Mr. Kallet reports no financial relationships relevant to this field of study.
A recruitment maneuver (RM) is the technique of briefly increasing alveolar pressure to levels in excess of what normally is recommended to reopen collapsed peripheral airways and alveoli so that both resting lung volume, or functional residual capacity (FRC), and oxygenation are restored.1 Moreover, amelioration of sheer injury through reversal of atelectasis is a major component in preventing ventilator-induced lung injury (VILI) in patients with acute respiratory distress syndrome (ARDS). This special feature will provide a brief overview of the physiologic foundation and history of RMs as well as discuss its clinical application.
Physics and Physiology of the Recruitment Maneuver
The RM is inextricably related to the pulmonary pressure volume (compliance) curve and the concept of hysteresis. Moving the lungs from a relatively collapsed to a fully inflated state produces a leftward shift in the pressure-volume relationship (that is, improved compliance).1 During subsequent deflation to FRC, tidal ventilation requires a lower driving pressure that lessens the risk for VILI. In essence, the RM is the same phenomenon that occurs with the first postnatal breath. At birth, the initial recruitment of the gasless lung requires a critical opening pressure of -40 cm H2O to begin inflation and pressures approaching -80 cm H2O to achieve full inflation.2 Similarly, inflation of a collapsed, excised lung (at residual volume) requires a lower critical opening pressure of 20 cm H2O; thereafter, recruitment proceeds unevenly until full inflation is achieved at a transpulmonary pressure of 40 cm H2O.2
Reversal of significant lung collapse not only requires high inflation pressures, but those pressures must also be sustained for a period of time. This temporal component is particularly important in ARDS. Lung recruitment is determined by numerous factors, including the surface tension, viscosity and film thickness of the airway lining fluid, airway radius, axial wall traction exerted by the surrounding alveoli, and the presence of surfactant.3 Increased lining fluid surface tension raises the applied airway pressure necessary to reopen collapsed airways, while an increased viscosity (as with protein-rich pulmonary edema fluid) prolongs the time necessary to reopen sequentially collapsed airways.3 The opening pressure must also counter the superimposed weight of the edematous lung itself, the mediastinum and chest wall, as well as disperse edema fluid and cellular debris residing in the small airways and alveoli.1
Brief History
In the early 1960s, the term “pulmonary hyperinflation” was used to describe the reopening of collapsed air spaces to reverse intraoperative atelectasis and hypoxemia.4 This was achieved with sustained (15 seconds) inflation pressures of 30-40 cm H2O. The term “recruitment” was introduced in the 1970s to describe how positive end-expiratory pressure (PEEP) improved pulmonary function.5,6 The fact that recruitment occurred because the concomitant increase in inspiratory pressure at a constant tidal volume (Vt) exceeded the critical opening pressure was only alluded to and not emphasized. It was only in 1992 with the advocacy for “open-lung ventilation” that the concepts of recruitment/derecruitment and the attendant roles of inspiratory plateau (i.e., alveolar) pressure (Pplat) and PEEP were clearly delineated.7 By the end of the 1990s, publications focusing on RMs began to appear regularly in the medical literature, such that now several hundred publications on the topic are listed in Pubmed.
Methodology
Two common methods for lung recruitment are 1) applying continuous positive airway pressures (CPAP) of 35-40 cm H2O for 40 seconds8 and 2) a “maximum recruitment” strategy of stepwise super-PEEP using pressure control ventilation. In this approach, an initial PEEP of 25 cm H2O is set with a driving pressure of 15 cm H2O to achieve a Pplat of 40 cm H2O for 4 minutes.9 Typically, a low respiratory frequency and prolonged inspiratory time (e.g., 10 and 3 seconds, respectively) are set to enhance recruitment. If cardiorespiratory stability is maintained, then PEEP is titrated upwards in 5 cm H2O increments at the same driving pressure for 2 minutes. Then the ventilator is returned to the initial RM settings for evaluation either by arterial blood gases or pulse oximetry. Full recruitment has been defined as the achievement of a PaO2 + PaCO2 ≥ 400 mmHg (using an FIO2 of 1.0). If pulse oximetry is used, then the FIO2 is titrated to achieve an SpO2 of 90-92% so that improvements or deteriorations in oxygenation are readily apparent. If recruitment goals are not met, then the RM is repeated at the next highest pressure increment. The maximum RM settings with this technique are a PEEP of 45 cm H2O and a Pplat of 60 cm H2O.
Both the maximum recruitment and high CPAP methods incorporate a decremental PEEP trial following the RM because many studies found that the positive effects of an RM dissipate within a few minutes to hours.1 In the decremental PEEP method, optimal PEEP is determined empirically by a quick (e.g., 5-20 minutes) stepwise PEEP reduction of 2 cm H2O until a mild deterioration in either oxygenation or compliance is detected.10 This signifies the beginning of derecruitment and the RM then is repeated. Afterwards, PEEP is set 2 cm H2O above the level at which derecruitment was detected. This procedure can be streamlined as several studies have shown that measuring pulmonary dead-space fraction is a more sensitive indicator of derecruitment that occurs prior to deterioration in oxygenation.11
Of note, in a heterogeneously injured lung, delayed or progressive derecruitment often occurs whereby oxygenation and compliance deteriorate over a matter of hours, not minutes. This may be more prevalent when chest wall compliance is low and a constant external force acts to compress the lungs. Therefore, clinicians should anticipate that determining “optimal PEEP” by the decremental PEEP method may be biased toward underestimating the actual PEEP requirements post RM.
In Whom, When, and How?
While clearly beneficial in restoring lung volume and improving gas exchange, RMs create radical changes in intrathoracic pressures and volumes that can result in hemodynamic instability, cardiac arrhythmias, acute hypoxemia, and pulmonary barotrauma.1 In a recent study of 366 patients receiving up to four RMs daily using the CPAP method, 22% experienced complications, and the latter were significantly related to the number of RMs attempted and to the patients’ having a “direct” or pulmonary form of ARDS (e.g., pneumonia, aspiration, pulmonary contusion).12 The most common problems were transient hypotention (11%), oxygen desaturation (10%), and tachycardia/bradycardia (5%). Both arrhymias and barotrauma were rare (1% each). Moreover, there is no high-level evidence that incorporation of RMs improves clinically significant outcomes in ARDS so that the risk/benefit ratio of RMs may seem unattractive.
Patient selection and timing of the RM are also important factors as RMs are not effective in all patients with ARDS. The best indicator for using an RM is the same as that for escalating PEEP therapy: patients with low FRC tend to have the greatest potential for lung recruitment.6 FRC essentially represents the alveolar volume and is a major determinant of both compliance and PaO2. Although the potential for lung recruitment varies widely in ARDS, patients with poorer oxygenation, lower compliance, and higher dead-space fraction tend to exhibit a higher degree of lung recruitment.13 RMs tend to be more effective if instituted during the early (exudative) phase of the syndrome.1
However, this is not always true, and a recent study of patients with early ARDS and relatively profound hypoxemia reported that only half of them responded positively to a maximum recruitment strategy.14 As in other studies, nonresponders tended to be those with direct lung injury, which classically is associated with both consolidation and limited recruitable lung tissue. Responders tended to have “non-pulmonary” etiologies of ARDS (e.g., sepsis, pancreatitis). And yet, even these general tendencies do not always hold true. Others have found that some patients with direct pulmonary injury do respond positively to RMs.10 Therefore, clinicians ultimately must evaluate RMs in individual patients empirically and not be overly swayed by tendencies reported in the literature as it currently exists.
But the most important unaddressed issue pertains to the temporal aspects of lung recruitment. It is clear that in the absence of elevated PEEP, the benefits of RMs are ephemeral. Therefore, could similar improvements in pulmonary gas exchange and mechanics be achieved if higher PEEP levels simply were used for longer durations? The fear of high PEEP partly reflects the findings of studies establishing PEEP’s adverse effects back in the early 1970s. These were done with large Vts (often 15-20 mL/kg) that contributed significantly to the incidence of hypotension and barotrauma. For example, even at modest levels of PEEP, Vts between 10-20 mL/kg caused abrupt deterioration in compliance. Yet in these same patients, compliance continued to improve when physiologic Vts were used (5-7 mL/kg), even at 15 cm H2O of PEEP.15
Recent studies suggest that lower levels of super-PEEP (i.e., 20-25 cm H2O) with physiologic Vts appear to be well tolerated.9,10 Over an extended period of time, such an approach might be sufficiently effective as an RM and may carry less risk of adverse effects. Incorporating prone positioning may further enhance lung recruitment by favorably altering the dorsal-ventral pleural pressure gradient while preventing overdistension of the non-dependent lung.16
In the context of severe ARDS, the risk/benefit ratio of using RMs as an adjunctive therapy appears reasonable. However, appropriate patient selection, meticulous evaluation, preparation, and monitoring are key to maximizing the benefits of RMs while minimizing adverse effects (see Table).
Table. A Practical Guide for Considering Recruitment Maneuvers in ARDS
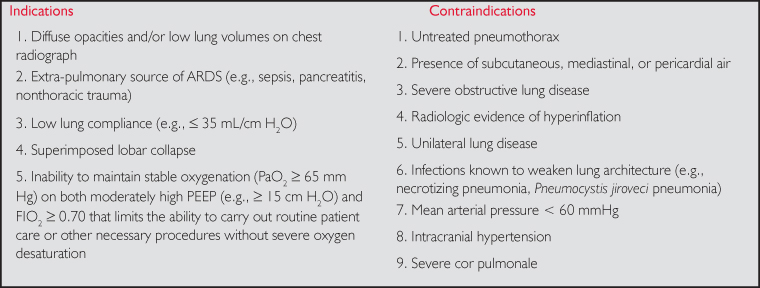
Assuring normovolemia and correction of hypotension are essential. Keeping in mind that the RM reduces/redistributes pulmonary perfusion, hypotension will obscure the detection of alveolar recruitment. In these situations, titrating vasopressor support to achieve and maintain a mean arterial pressure of at least 70 mmHg is useful in facilitating real-time evaluation of lung recruitment. In situations where hypotension is refractory to therapy, then the RM should only be attempted using intermittent sighs on the ventilator. Some ventilators do this with an “intermittent PEEP” function, whereby PEEP is raised transiently to a clinician-selected level for two breaths every few minutes. This method has been shown to improve oxygenation and lung volumes.17 However, this approach may be less effective than common RM methods.
In patients with severe acidosis and hypermetabolism, it is probably better to choose the maximum recruitment method as even relatively brief periods of apnea can increase the risk of instability. Careful analysis of minute ventilation demand, PaCO2, and pH prior to the RM is useful in setting the baseline minute ventilation during the RM and to evaluate the potential need for buffer therapy. THAM (tris buffer) is particularly useful in this regard, as it does not produce CO2 and has been used to achieve apneic oxygenation.18 Finally, assuring passive ventilation with a neuromuscular blocking agent is important to prevent active expiratory efforts that promote derecruitment and reduce the risk of barotrauma.
Summary
RMs are beneficial in restoring lung volume and improving gas exchange in patients with ARDS, but as yet no evidence exists to suggest that they improve outcomes. Adverse effects are relatively frequent but usually are transient and resolve with termination of the procedure. When its use is restricted to the early phase of severe ARDS, particularly in those with an “extra-pulmonary etiology,” RMs tend to be more effective in stabilizing pulmonary function and reasonably justify potential risks. Several methods of RMs are available and should be chosen after careful evaluation of individual patients.
References
1. Kacmarek RM, Kallet RH. Respir Care 2007;52:622-631.
2. Cherniack RM, et al. Respiration in Health and Disease. 2nd ed. Philadelphia: WB Saunders; 1972.
3. Gaver DP, et al. J Appl Physiol 1990;69:74-85.
4. Bendixen HH, et al. N Engl J Med 1963;269:991-996.
5. Falke KJ, et al. J Clin Invest 1972;51:2315-2323.
6. Suter PM, et al. N Engl J Med 1975;292:284-289.
7. Lachmann B. Intensive Care Med 1992;18:319-321.
8. Amato MBP, et al. N Engl J Med 1998;338:347-354.
9. Borges JB, et al. Am J Respir Crit Care Med 2006;174:268-278.
10. Tugrul S, et al. Crit Care Med 2003;31:738-744.
11. Kallet RH. Minerva Anesthesiol 2012;78:1297-1305.
12. Fan E, et al. Respir Care 2012;57:1842-1849.
13. Gattinoni L, et al. N Engl J Med 2006;354:1775-1786.
14. Moran I, et al. Minerva Anesthesiol 2011;77:1167-1175.
15. Suter PM. Chest 1978; 73:158-162.
16. Cornejo RA, et al. Am J Respir Crit Care Med 2013; Jan. 24. [Epub ahead of print.]
17. Foti G, et al. Intensive Care Med 2000;26:501-507.
18. Nahas GG, et al. Drugs 1998;55:191-224.
A recruitment maneuver (RM) is the technique of briefly increasing alveolar pressure to levels in excess of what normally is recommended to reopen collapsed peripheral airways and alveoli so that both resting lung volume, or functional residual capacity (FRC), and oxygenation are restored.1Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
