Male Genital Emergencies: Part II
Male Genital Emergencies: Part II
Authors: Larry B. Mellick, MD, MS, FAAP, FACEP, Professor of Emergency Medicine and Pediatrics, Medical College of Georgia, Georgia Regents University, Augusta, GA.
Davis L. Mellick, PAC, Doctors Hospital, Emergency Department, Augusta, GA.
Peer Reviewer: Steven M. Winograd, MD, FACEP, Attending Physician, Core Faculty, Emergency Medicine, St. Barnabas Hospital, Bronx, NY; Clinical Assistant Professor, Emergency Medicine, Emergency Medicine Residency, New York College of Osteopathic Medicine, Old Westbury, NY.
In this article, the authors continue their review of common male genital emergencies. Part I covered common scrotal emergencies, and in Part II, common penile emergencies are reviewed.
— J. Stephan Stapczynski, MD, FACEP, Editor
Penile Fracture Injury
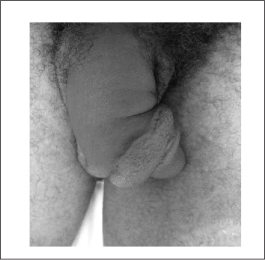
Case. A 44-year-old male presented to the emergency department for evaluation of pain and swelling of his penis. The patient stated that he heard a pop and had sudden pain on the right distal penile shaft while “entertaining” himself that morning. He subsequently noted swelling and deformity of his penis. On initial evaluation, the patient had a swollen and deviated penis. (See Figure 1.) A retrograde urethrogram was performed and demonstrated no evidence for urethral injury. The patient was taken to the OR, where a horizontal tear was found in the ventral right corpora under the spongiosum and repaired.
Figure 1: Fractured Penis with Typical Deformity of Deviation to Side Opposite Tear
Introduction
Despite the embarrassing pain of being hit in the groin, the truth is that male genitalia are fairly resistant to injury. The flaccid, pendulous penis is very capable of absorbing significant amounts of kinetic energy. However, the fixed portion of the genitalia and the erect penis are different situations. The penile and urethral anatomy lie in close relationship to the pubic rami and are vulnerable to blunt trauma. Likewise, the erect penis is more prone to injury because pressures within the rigid penis rise exponentially with bending.1 Penile fracture is considered a rare injury. The 2007 National Inpatient Sample (NIS) reported that a total of 1,043 men were admitted for treatment of a penile fracture.2
Penile fractures have been reported in patients as young as 12 and as old as 82, but the average age of penile fracture is around the third or fourth decade of life.3-7 Victims are often reluctant to seek treatment because of shame over the circumstances surrounding the injury.
Penile Anatomy
The penile shaft is made up of three vascular erectile columns: the two corpora cavernosa that lie next to each other and the ventrally located corpus spongiosum, which contains the urethra. The glans penis is the enlarged and bulbous end of the corpus spongiosum. The foreskin, or prepuce, is a fold of loose skin that exposes the glans when retracted. The two corpora cavernosa are each surrounded by a dense, fibrous sheath of connective tissue, the tunica albuginea. An incomplete midline septum exists between the corpora cavernosa that allows them to communicate freely. Toward the base of the penis, the corpora become divergent, forming the crura, which attach to the ischiopubic rami.
The thick tunica albuginea surrounding the corpora cavernosa is a bi-layered structure with multiple sub-layers.8 Compared to the tunica albuginea, the tunica of the corpus spongiosum is considerably thinner, allowing sufficient distention for the passage of ejaculate through the urethra and a less rigid glans penis to provide a cushioning effect during intercourse.
The erectile tissue is composed of endothelial cell-lined venous sinusoids or cavernous spaces surrounded by sinusoidal trabeculae composed of fibrous tissue and smooth muscle fibers. On cross section, the corpora cavernosa has a sponge-like appearance. Immediately superficial to the penile tunica albuginea is the deep penile or Buck’s fascia, the Dartos fascia, and penile skin. Dartos fascia is the subcutaneous connective tissue of the penis and scrotum and is continuous with the superficial perineal fascia. In the penis, the superficial arteries, veins, and nerves are contained in the Dartos fascia.
Dorsal, cavernosal, and bulbourethral arteries — all branches of internal pudendal artery — provide blood flow to the penis. Paired deep arteries running near the center of each corpora cavernosa (cavernosal arteries) provide blood flow to the corpora cavernosa. The dorsal artery travels with the dorsal nerve and deep dorsal vein on the dorsal aspect of the corpora cavernosa, and its terminal branches are in the glans penis. The skin of the penis receives its arterial blood from the left and right superficial external pudendal arteries.
The dorsal nerve of the penis originates from the pudendal nerve and, at the urogenital diaphragm, provides a branch to the corpus cavernosum. The dorsal penile nerve travels forward on the dorsum of the penis with the penile dorsal artery where it ends at the glans. This nerve supplies somatic motor and sensory innervation. The cavernous nerves supply the erectile tissue and contain parasympathetic and visceral afferent fibers that facilitate penile erection.
Erection Physiology
Erection occurs when nitric oxide, a neurotransmitter that chemically relaxes the smooth muscles of the paired corpora cavernosa, is released when the brain sends a signal via the spinal cord through the parasympathetic nervous system. The relaxed smooth muscle enables enhanced blood flow into the lacunar spaces of the corpora, producing expansion of the erectile tissue with concomitant constriction of venous outflow, furthering more expansion of the erectile tissue.
Mechanisms of Injury
A penile fracture is the traumatic rupture of the tunica albuginea in the tumescent state. Abrupt blunt trauma by forceful bending of the erect penis is the most common mechanism. Most fractures occur during coitus or penile manipulations such as masturbation.3,4,9-14 (See Table 1.) In the Western world, occurrence during sexual intercourse is most commonly reported. Penile fractures appear to occur more commonly with the partner-on-top position from penile impact against the female pelvis bone or perineum or excessive lateral bending. Fractures can also occur from the position that involves the woman on top facing away from the male partner. This position bends the erect penis ventrally beyond the tensile strength of tunica albuginea, and can cause a fracture.
Table 1: Reported Activities Associated with Fractured Penis

In the Middle East, the practice of taqaandan (or taghaandan) is often responsible for penile fractures.9 Taqaandan is a Kurdish work meaning “to click.” Taqaadan involves bending the topmost part of the erect penis while stabilizing the penile shaft, and may be performed to achieve detumescence. This practice has been reported to be a common cause of penile fractures in western Iran.9 A sharp cracking noise, not unlike that of a corn stalk breaking, is often described.3,10
Pathophysiology
The injury is most commonly a rupture of the corpus cavernosum (one or both corporal bodies can be involved), but it may also involve the corpus spongiosum and urethra. The tunica albuginea is approximately 2 mm thick in the flaccid condition, but during erection thins out to 0.25 mm, becoming more vulnerable to injuring forces.15,16 The subsequent tear through the tunica albuginea into the corpus cavernosum, and sometimes the spongiosum, results in hematoma-related swelling and skin ecchymosis. There is a report of structural anomalies of the tunica albuginea, such as fibrosclerosis and phlogistic cellular infiltration, in a majority of penile fracture cases.17
The incidence of urethral injury varies depending on the mechanism. In the United States and Europe, urethral injury is seen in approximately 20% of penile fracture cases because the injury usually occurs during vigorous intercourse. In the Middle East, the incidence is 2-3% due to the less traumatic self-inflicted injury caused by masturbation or taqaandan.3,4,6,18-20
Clinical Presentation
The typical presentation is penile swelling, ecchymosis, penile deviation, and a palpable depression in the penile shaft. The deviation of the penis is away from the side of the injury due to the hematoma. The injuries to the tunica albuginea are usually transverse.7,21 If Buck’s fascia remains intact, the appearance of the fractured penis is often described as that of an eggplant, and the associated sign is the “eggplant sign” or “aubergine sign.”21 If Buck’s fascia is disrupted, the hematoma can be more diffuse and spread to the scrotum and perineum.
Late presenters may have complications associated with this injury that include penile deviation, erectile dysfunction, or plaques similar in appearance to Peyronie’s disease.3,22 Other less common complications include urethrocavernous and urethrocutaneous fistula, as well as a poor urinary stream or urinary retention due to a urethral stricture.22
The diagnosis is primarily clinical based on the characteristic history and penile appearance.7,23 Patients will often report hearing a snapping sound and experience the sudden onset of pain followed by immediate penile detumescence. Urethral bleeding and difficulty with urination is sometimes observed, and usually indicates urethral involvement. However, urethral injuries can occur without the presence of gross blood.18,21,24 So, even if the patient can urinate, urethral injuries cannot be excluded.25
Imaging studies such as cavernosography, ultrasound, and magnetic resonance imaging (MRI) may be useful in selected cases.3,6,23,26 MRI is probably the most sensitive and reliable imaging modality, but access and expense are impediments to its use.23 Cavernosography in the evaluation of penile fractures is discouraged because it is unfamiliar to most urologists and radiologists, and it delays treatment.21,23
Pre-operative urethrography should be strongly considered,6 but intraoperative flexible cystoscopy is now commonly performed when urethral injury is suspected.23
Treatment
There is overwhelming evidence that surgical management of penile fractures results in the best outcomes.27 Furthermore, surgical repair should be performed sooner rather later.3,6,7,16 Various surgical techniques exist; the key is repair of the tunica albuginea.3,28
In the past when conservative management was more common, ice packs, compression, and anti-inflammatory medications were recommended. In fact, conservative management is clearly associated with long-term and undesirable complications such as a curved or painful erection, erectile dysfunction, and a fibrotic plaque that hinders erection.27,29 Conservative management is now widely discouraged.
Even with surgical repair, long-term complications such as deviation of the penis, painful intercourse, painful erection, and erectile dysfunction can occur.11,21 The formation of a fibrous plaque is common and is responsible for pain and penile deviation.
Differential
Other acute conditions may mimic penile fracture. Hematomas may develop from an isolated venous bleeding of the penis, thrombosis of the superficial dorsal vein of the penis (Mondor’s disease), penile lymphangitis, and penile arterial injuries. A false fracture of the penis is described when the tunica is not torn, and only bleeding from a superficial vein is noted at surgery.30-33 In one series, 8.8% of patients with suspected penile fractures had only bleeding from a torn superficial vein.3
Urethral Injuries
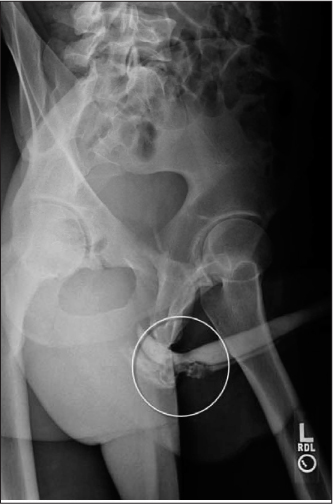
Case. A previously healthy 20-year-old male was performing acrobatic flips in his backyard when in the middle of a flip, the patient’s perineum came into hard contact with a metal post, which he straddled. Upon presentation to the emergency department, there was swelling and marked tenderness of the perineum and obvious bleeding from the urethra. More bleeding occurred with palpation of the perineum. A retrograde urethrogram was performed, and disruption of the ventral aspect of the anterior urethra was documented, along with contrast extravasation. (See Figure 2.)
Figure 2: Urethrogram Showing Evidence of Disruption of the Ventral Aspect of the Posterior Anterior Urethra
Introduction
The male urethra extends proximally from the urinary bladder’s internal urethral orifice to the external urethral orifice of the glans penis. The adult male urethra is approximately 15-25 cm in length and makes an “S” curve in the flaccid position. The urethra is divided into four anatomical segments, primarily determined by the investing structures: the prostatic urethra, membranous urethra, bulbar urethra, and spongy or penile urethra. The membranous urethra is enclosed in the urogenital diaphragm, invested in the external urethral sphincter muscle and the perineal membrane. It extends for approximately 3 cm from the apex of the prostate to the bulb of the penis. The membranous urethra is firmly attached to the ischial and inferior pubic rami via the external sphincter muscle and the perineal membrane. Consequently, it is most susceptible to disruption with pelvic fractures.
Urethral injuries are also classified based on the anatomical site of trauma. The urogenital diaphragm divides the male urethra into the anterior and posterior sections; the prostatic and membranous urethras make up the posterior (or proximal) urethra, while the bulbar and penile urethras define the anterior (or distal) urethra.
Epidemiology
Urethral injuries can occur due to external blunt or penetrating trauma or internal trauma caused by foreign bodies placed in the urethral lumen. Blunt external trauma accounts for more than 90% of urethral injuries.34-36 Posterior urethral injuries occur most commonly in the setting of pelvic fractures. A common teaching is that approximately 10% of pelvic fractures in males are associated with urethral disruption. However, a recent National Trauma Data Bank review reported that urethral disruption occurred in only 1.54% of males with a pelvic fracture.37 Nevertheless, almost all blunt trauma-related membranous urethral disruptions have an associated pelvic fracture or, more specifically, a disruption of the pelvic ring.34,38 Pelvic fractures are more common in men, with an incidence of 2:1 compared to women.34,39 Some urethral injuries occur iatrogenically following surgery or urethral instrumentation.40
Straddle injuries usually involve the anterior urethra, but can involve a section from the penoscrotal junction to the bulbomembranous junction. Anterior urethral injuries occur at one-third of the frequency of posterior urethral injuries.41,42
Urethral injuries range from simple contusions to a partial tear of the urethral epithelium or full transection. Penile fractures associated with accidents during sexual intercourse or masturbation account for the majority of anterior urethral injuries in some series.41,42,43
Clinical Presentation
Certain clinical findings are associated with a urethral injury. (See Table 2.) Nevertheless, the absence of any of these findings does not exclude the possibility of a urethral injury.
Table 2: Urethral Injury Red Flags
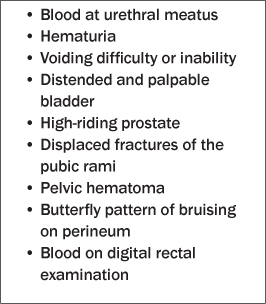
An important concept is that although the classic triad of blood at the external urethral meatus, inability to pass urine, and a distended bladder is strongly suggestive of urethral injuries, the absence of this triad does not exclude the diagnosis.42 Urethral injuries are potentially fraught with long-term and disabling complications such as urethral strictures, urinary incontinence, and erectile dysfunction. Therefore, their diagnosis is essential.
Posterior Urethral Injuries
Certain subtypes of pelvic fractures are more likely associated with urethral disruption. These are straddle fractures when all four pubic rami are fractured, and Malgaigne fractures with disruption through the ischiopubic rami anteriorly as well as through the sacrum or sacroiliac joint posteriorly. While blood at the urethral meatus is a cardinal sign of posterior urethral injury, hematuria can also be evidence of a urethral injury, but the amount of hematuria may not correlate with the injury severity.43 If the patient’s bladder is palpably distended or the inability to urinate is reported along with perineal bruising, consider the possibility of urethral disruption. The digital rectal examination may demonstrate an elevated or displaced prostate gland, but it may not be palpable if a significant hematoma is present with the pelvic fracture. Palpation of the prostate and other physical findings are not reliable in making the diagnosis.
Unfortunately, the diagnosis is sometimes discovered following attempts to place a urinary drainage catheter into the bladder in patients with pelvic fractures. While there is not a uniform consensus, some physicians think that in the absence of these “red-flag” findings, a single gentle attempt at catheterization is not harmful and can be followed by retrograde urethrography if not successful.35,44-46 There is widespread consensus, however, that the diagnostic test of choice is retrograde urethrography.44,47
Anterior Urethral Injuries
Blunt trauma is the most common mechanism causing anterior urethral injuries, and the bulbous urethra is most frequently injured because it is fixed beneath the pubic bone. Since the associated blunt forces are often not of the same caliber as those seen with posterior urethral injuries and the associated pelvic fractures, some anterior urethral injuries may escape initial diagnosis. These patients may present after an interval of months to years with bulbar urethral strictures. Thus, these injuries should be considered when the patient reports perineal pain following a motor vehicle accident, straddle injury following a fall onto a fence, railing, or saddle, or kicks to the perineum. As noted before, anterior urethral injuries occur in the about 20% of patients who have sustained a penile fracture.9 Penetrating trauma to the anterior urethra can be caused by firearms, stab wounds, industrial accidents, self-mutilation, and bites.
The cardinal sign of an anterior urethral injury is blood at the meatus. Other associated symptoms such as dysuria, hematuria, and inability to void urine should also raise the index of suspicion concerning this injury. Evidence of a perineal butterfly hematoma or a hematoma in a sleeve distribution along the penile shaft may also be associated with an anterior urethral injury.
Radiographic Imaging
For suspected urethral injury, retrograde urethrography is considered the gold standard.35,44,45,47,49,50 For suspected urethral stricture, simultaneous cystogram and urethrogram are used, or MRI or endoscopy through the suprapubic tract can help define the anatomy.35,44,45
Urethrography is accomplished with the patient in a 25-35 degree oblique position with the penis straightened as much as possible. A Foley catheter inserted in the fossa navicularis with the balloon inflated using 1-2 mL of saline will occlude the urethra. Approximately 20 to 30 mL of full strength (60%) ionic contrast (water-soluble) medium is gently injected into the urethra.47 (See Figure 2.) Either real-time fluoroscopic imaging or a series of plain radiographs after each 10 mL of contrast medium are used.
Treatment
In the presence of urinary retention, the immediate goal is to attain urinary bladder drainage while simultaneously avoiding further urethral injury. A suprapubic catheter should be inserted or an emergent endoscopic examination should be performed by the urology consultant using a cysto-urethroscope. If a suprapubic catheter is required, simultaneous suprapubic cystography and ascending urethrogram can be performed to assess the location and severity of the urethral injury. If a patient with a suspected urethral injury arrives with a urinary catheter already in place, a peri-catheter urethrogram and/or a voiding urethrogram/urethroscopy should be performed.
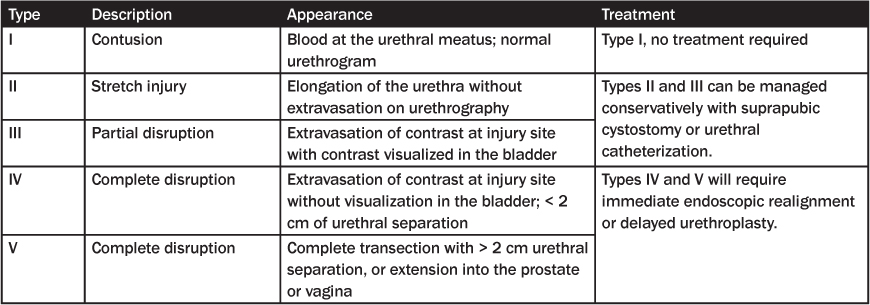
The Committee on Organ Injury Scaling of the American Association for the Surgery of Trauma (AAST) developed a urethral injury scaling system that is used for treatment recommendations.44,48 Timing of definitive surgical interventions is defined as immediate urethroplasty, delayed primary urethroplasty (10 ± 14 days), and late urethroplasty (3 months or more).42
Table 3: AAST Organ Injury Scale for the Urethra44,48
As noted, assuming there is no blood at the meatus, some recommend a single attempt at placement of a Foley catheter and, if successful, a peri-catheter retrograde urethrogram is performed to assess the urethra.35,44-46 If the catheterization fails and depending on the surgeon’s preference, cystoscopy in the operating room (or biplanar fluoroscopy, interlocking sounds, or magnetic catheters) can be used for primary realignment techniques.35,42,45 Immediate realignment of complete disruptions has the best outcomes in all parameters (i.e., strictures, impotence, incontinence, and total number of procedures).35,50,51
Partial tears of the anterior urethra due to blunt trauma can be managed with a suprapubic catheter or with urethral catheterization.42,45,47 The injured anterior urethra can undergo delayed primary repair.
Partial tears of the posterior urethra may be treated with a suprapubic or urethral catheter, followed by repeat retrograde urethrography every several weeks until healing has occurred.42,44,45,47 Immediate or primary open repair of posterior urethral injuries has a high incidence of strictures, re-stenosis, incontinence, and impotence.42,44 Conversely, delayed urethroplasty has a low re-stricture and impotence rate and is the standard of care for posterior urethral distraction defects.42,44,45,53
Infection is the most important early complication of an acute urethral injury.42,45,49 Extravasated urine or blood can produce an inflammatory reaction that can spread infection along fascial planes of the abdomen, perineum, and medial thighs. Significant complications and sequelae that can occur include urethrocutaneous fistulae, periurethral diverticula, and necrotizing fasciitis (Fournier’s gangrene).42,47
Priapism
The term “priapism” is derived from the name of a minor Greek god of fertility, Priapus. Priapism is a persistent penile erection that continues more than four hours beyond sexual stimulation and orgasm or is unrelated to sexual stimulation or desire.
There are two types of priapism: ischemic and non-ischemic. Ischemic priapism (also known as veno-occlusive or low-flow priapism) is characterized by little or no cavernous blood flow and abnormal cavernous blood gases (hypoxic, hypercarbic, acidotic).54 Priapism is associated with a number of drugs and medical conditions. (See Table 4.) The prolonged, painful erection typically involves only the paired corpora cavernosa and not the ventral corpora spongiosum and glans penis, although tricorporal priapism occurs.55 The failure of detumescence in ischemic priapism is an emergency; permanent erectile dysfunction and penile fibrosis and necrosis will occur if untreated. Stuttering or intermittent priapism is a recurrent form of ischemic priapism that presents with repeated unwanted, painful erections that have intervening periods of detumescence.
Table 4: Conditions and Pharmacologic Agents Associated with Ischemic Priapism97,98
Non-ischemic (also known as arterial or high-flow priapism) is caused by unregulated cavernous arterial inflow; penile blood gases are not hypoxic or acidotic. Lack of pain and a partial erection is typical of this form of priapism. Typically, there is a history of antecedent trauma to the penis with an associated arterial injury. Treatment of non-ischemic priapism is not considered an emergency.
Pathophysiology
Ischemic or low-flow priapism is essentially a compartment syndrome localized to the corpora cavernosa. Persistent corporal erection compresses the subtunical veins, preventing any outflow from the sinusoids. When the intracorporeal pressure rises above the mean arterial pressure, the cavernosal arteries running through the middle of each corpus cavernosum are also passively compressed, reducing arterial inflow.56 If prolonged, destruction of sinusoidal endothelium and, eventually, thrombus formation and necrosis will occur within the corporal smooth muscle due to the prolonged ischemia. If untreated, resolution may occur after a period of days, but erectile dysfunction invariably results.
In contrast, non-ischemic priapism features increased vascular flow within the corpora cavernosa without an associated compartment syndrome effect. Rupture of a cavernous artery and unregulated flow into the lacunar spaces is usually the cause for this condition; in effect, an arteriolar-sinusoidal fistula has been created. There is very little, if any, smooth muscle relaxation associated with this condition, and the arterial flow of blood is cleared as rapidly as it can from the sinusoids. Erection occurs when inflow into the sinusoids is greater than outflow.
Management
Current treatment recommendations and guidelines have been developed based on review of the available literature and expert consensus, such as the guidelines created by the American Urological Association (AUA).54 Even though the time urgency for management of ischemic priapism does not compare to a testicular torsion, this is a compartment syndrome, and a timely intervention is important for the future sexual function of the penis.
The primary management of ischemic priapism involves the placement of a large-bore needle into the engorged penis with aspiration and, if necessary, saline irrigation of the corpora cavernosa to remove the stagnant blood. Some resources continue to recommend oral pseudoephedrine or terbutaline in specific settings, such as priapism under four hours or following self-injection of agents for impotence. For these specific conditions, there are anecdotal reports of success.57-59 Nevertheless, most experts and guidelines advise against attempting to manage priapism solely with oral medications.54,60-63 This is especially relevant for patients who present many hours after the onset of prolonged erection.
Injection of sympathomimetic agents (usually phenylephrine) is also recommended, but only after aspiration and irrigation, as there is some evidence that the acidotic, anoxic, hypercarbic, and hypoglycemic condition of the penis will make the injected sympathomimetic agent less effective. Conditions will be much more conducive for success of injected medications after the stagnant blood in the corpora cavernosa is removed.
The aspiration procedure is performed by starting with local anesthesia. A penile dorsal nerve block, ring block, or local injections with lidocaine or bupivacaine can be used. Systemic analgesia with an opioid drug is also recommended. Aspiration and irrigation can be accomplished with a large-bore needle (such as a 19-gauge butterfly or other similar-sized needle) placed using sterile technique at 2 or 3 o’clock or 9 or 10 o’clock (to avoid the neurovascular bundle) into the corpora cavernosa. Generally, only one corpora needs to be accessed because there is communication of blood flow between the two sides. Simultaneous compression of the penile shaft between the thumb and forefinger of the opposite hand will assist with detumescence. While a larger syringe (30 or 60 mL) may seem appropriate for the procedure, it is generally recommended that a smaller syringe be used to prevent excessive suction that can cause collapse of the venous sinusoids. If drainage from the needle is inadequate, gently irrigate with normal saline through the same needle. Alternatively, in that the large-bore needle is typically placed proximal on the penis, place a 21-gauge needle for instilling saline for irrigation into the corpora cavernosa closer to the glans penis and gently irrigate with normal saline through the 21-gauge needle with outflow from the initial needle.
Dorsal Nerve Block A dorsal nerve block can be performed prior to aspiration of blood from the corpora cavernosa. The pudendal nerve (S2-S4) provides sensory innervation to the penis. This nerve passes under the pubis symphysis and divides into the right and left dorsal nerves that travel below the Buck fascia. The right and left dorsal penile nerves are blocked as close to the base of the penis as possible. A 27-gauge needle is used to raise skin wheals at the 2 and 10 o’clock positions with the 1% lidocaine. Next, insert the needle through the center of each skin wheal while directing it toward the center of the shaft. A loss of resistance will occur as the needle penetrates Buck fascia. After aspiration to insure that the needle is not intravascular, slowly inject approximately 2 mL of local anesthetic on each side. Alternatively, simply inject 2 mL of local anesthetic on each side of the midline while avoiding the superficial dorsal penile vein. (See Figure 3.) Figure 3: Schematic Diagram of Penis Anatomy on Cross Section 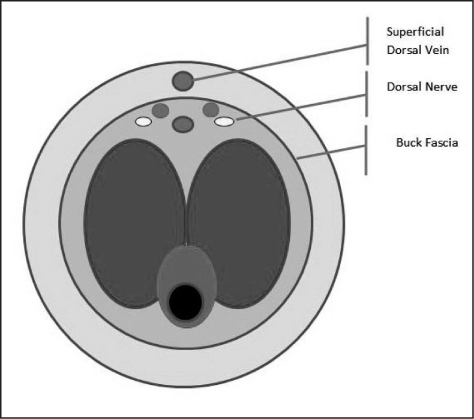 |
Following irrigation, phenylephrine should be injected into the cavernosa. For this purpose, phenylephrine is diluted by mixing the 1 mL vial containing 10 mg with 19 mL of normal saline, resulting in a concentration of 500 micrograms/mL. For healthy adults, inject 0.5 to 1 mL (250 to 500 micrograms of phenylephrine) through the drainage needle. Reassess after 5 to 10 minutes. If the erection does not resolve or returns, repeat the injection. Lower concentrations in smaller volumes should be used in children and patients with severe cardiovascular disease.54
Maximum safe dosing recommendations are not clearly established. Using the AUA guidelines, it is possible to give 6 to 10 mg of phenylephrine within an hour.54 More conservative recommendations advise that phenylephrine be diluted in saline to a 200 micrograms/mL concentration and be administered intermittently as 0.5 to 1.0 mL doses every 5–10 minutes, to a maximum dosage of 1 mg.62 Just as a comparison of another medical condition in which phenylephrine is used, the intravenous pulse dose treatment of shock-related hypotension is 100-500 micrograms/dose every 10 to 15 minutes as needed (initial dose should not exceed 500 micrograms).
The side effects of phenylephrine are similar to other sympathomimetics and include hypertension, tachycardia, seizures, stroke, and myocardial ischemia. These patients should be managed in a monitored room capable of closely tracking the patient’s blood pressure and heart rate. Intracranial bleeding associated with phenylephrine injection during treatment for priapism has been reported.64
When priapism occurs in patients with sickle cell disease or leukemia, it is tempting to think that simply treating the patient’s underlying condition will result in resolution of the priapism. This approach will result in unnecessary delays in the treatment of the priapism; thus, aggressive management of the priapism is recommended, along with appropriate treatment of the medical condition.
Penile Zipper Injuries
Case. A 39-year-old male presented to the emergency department complaining that his zipper was stuck to the skin of his penis. He reported that on the day prior, he caught the skin of his penis in the zipper mechanism of his pants. (See Figure 4.) Because of an urgent need to urinate, he used his knife to cut away the zipper from his pants. Initial attempts to remove the zipper by cutting the connecting bar with metal cutters were unsuccessful. Eventually the zipper was removed by reproducing the injury mechanism and simply “zipping up” the zipper.
Figure 4: Zipper Injury
Discussion
The basic components of a zipper are the stringer or the tape and teeth assembly, the slider that opens and closes the zipper, a tab used to pull or move the slider, and stops that prevent the slider from leaving the chain. The zipper slider is made up of two faceplates connected and held together with a median bar. The metal zipper may be made of stainless steel, aluminum, brass, zinc, or nickel-silver alloy.
Redundant skin of the penile shaft or foreskin can be caught in both the teeth of the zipper stringer and the zipper slider during the process of zipping or unzipping. Most often, this condition is discussed in case reports or small case series.65 In one case series of 30 patients published in 1994, the incidence of zipper entrapment was 1 case per every 4000 emergency department visitors. Most injuries occurred when the zipper was being pulled up, and the uncircumcised foreskin was most commonly entrapped.65 Entrapment of penis skin by the teeth of a zipper or the zipper slider is most commonly described in pre-adolescent males.65,66
Multiple techniques have been described for removal of the zipper mechanism from the entrapped skin. A common recommendation is to cut the bar on the zipper slider with a bone or wire cutter to separate the two faceplates and thereby release the entrapped skin.65,67-70 Unfortunately, this technique is more difficult with the larger zippers found on men’s trousers, and failures are common.71-75
How the skin is entrapped within the zipper mechanism can help guide the release approach. Entrapment of penile skin occurs in at least three ways. Skin can be trapped within the stringer (or the tape and teeth assembly), within the slider next to the open side of the zipper and against the median bar, and within the slider on the closed side of the zipper. If penile skin is entrapped by the slider on the open side of the zipper, access to the median bar is blocked, and another zipper release method is required.
A number of other successful release techniques have been described.76 Use of a flat-head screwdriver to pry the two faceplates apart was described by Raveenthiran in a series of 12 patients.75 Another technique involves the application of mineral oil, cutting the tape and teeth assembly transversely where the skin is entrapped, grasping the cloth portion on opposite sides of the zipper assembly with needle holders, and applying traction.65,77-79 The individual teeth of the stringer can be removed one at a time to release the skin.66,80 The basic technique of simply manipulating the slider by pulling it up or down is favored by other authors.65,71,80 Finally, surgical techniques such as performing a circumcision or the creation of an elliptical incision of the penile skin have been used as last resorts to remove entrapped zippers.66,74,81
Whatever technique is used, the operator should apply local anesthesia around the entrapped skin or use a dorsal nerve block. Procedural sedation may be required when the child’s anxiety level is high or his ability to cooperate is limited.
In summary, the success of bone cutters or wire cutters to release the median bar holding together the two plates of the zipper slider will depend on the material used to construct the zipper slider.82 Awareness of other techniques is useful in the management of this condition. A small flat-head screwdriver to pry apart the zipper slider appears to be the most successful technique.
Paraphimosis
Paraphimosis occurs when the foreskin of the uncircumcised or partially circumcised male is retracted behind the coronal sulcus and entraps the head of the penis. Swelling and edema of the foreskin develop over time and add to the difficulty of foreskin reduction. Lymphatic obstruction is followed by venous and, ultimately, arterial obstruction. The end result of paraphimosis can be loss of blood flow, resulting in infarction, necrosis, and loss of the glans penis.
Phimosis, or a tight foreskin that cannot be retracted to expose the glans penis, will only be mentioned briefly in this review. Phimosis is a physiologic condition in young males, but can be caused by infections or other inflammatory conditions in older males. When this condition is pathologic, a urology referral should be accomplished. Phimosis can be a precursor to paraphimosis when the tight foreskin becomes retracted behind the glans penis.
Pathophysiology
From birth, the foreskin slowly becomes retractable, as stretching occurs during nocturnal erections. However, when the foreskin is forced back prematurely as part of self-manipulation by the child or during attempts at cleaning by the caretaker, paraphimosis can occur in infants and young males. More mature males can develop paraphimosis following urination or sexual intercourse when they fail to reduce the foreskin to its natural condition. Paraphimosis can also occur as an iatrogenic condition when medical personnel fail to reduce the retracted foreskin following catheterization. At presentation, the foreskin is markedly swollen behind the corona of the glans penis and the penis is typically flaccid.
Management
The management of paraphimosis is the reduction of the foreskin in a timely manner. This is accomplished most frequently by manual reduction. The procedure for foreskin reduction includes anesthesia with topical medications or a dorsal nerve block, direct compression of the edematous foreskin to reduce edema, followed by applying direct pressure with the operator’s thumbs on both sides of the urethral meatus and the index and middle fingers proximal to the phimotic ring. Continuous force is applied to pull the foreskin over the head of the penis and push the glans penis through the phimotic ring.83
Other described techniques for reducing the edema of the anesthetized foreskin include the placement of circumferential needle injections to directly drain the edema fluid, the ice glove technique, and the application of granulated sugar. No comparative or randomized trials exist for these techniques, and these recommendations are based on case reports of successful foreskin reductions.84
The puncture technique was first reported in 1990.85,86 After the prepuce is cleaned with an antiseptic, a 25- or 26-gauge needle is used to make approximately 20 puncture holes in the edematous prepuce. Afterward, gentle but firm pressure is applied to express the edema fluid from the foreskin. Once the edema resolves, the prepuce is usually easily reduced.86-88 Edema reduction has also been described using granulated sugar or mannitol.89,90 Injections of hyaluronidase into the edematous foreskin has also been used successfully in reducing edema.91,92 Finally, the application of a glove filled with ice and water is also described as an edema-reducing technique.93
Failure to successfully reduce the foreskin mandates a urology consultation. A dorsal slit of the foreskin is sometimes required as a last resort.94 Circumcision is not performed acutely, but is often recommended for patients who experience paraphimosis. Preventative measures that should be taught to the patient include rapidly returning the foreskin over the head of the penis following cleaning or after sexual activity.
Paraphimosis Mimics
Paraphimosis mimics include angioedema, balanitis, cellulitis, fungal infections (Candida albicans), insect bites (especially chigger bites), various tourniquets (hair, metal, rubber bands), contact dermatitis, and local hypersensitivity reactions to topically applied medications that can cause swelling of the foreskin or its remnants.95 The loose, areolar connective tissue of the foreskin may swell dramatically in response to various stimuli, giving the appearance of a paraphimosis condition. It is obviously critical not to overlook a tourniquet applied to the penis.96
Summary
Despite the relative diminutive size of male genital organs compared to other organs of the body, the functional complexity and the spectrum of associated diseases and vulnerabilities to injury are truly remarkable. Failure to recognize and manage many of these conditions in a timely manner can have lifelong implications to urologic health and normal sexual function.
References
1. Penson DF, Seftel AD, Krane RJ, et al. The hemodynamic pathophysiology of impotence following blunt trauma to the erect penis. J Urol 1992;148:1171–1180.
2. Aaronson DS, Shindel AW. U.S. national statistics on penile fracture. J Sex Med 2010;7(9):3226.
3. el-Assmy A, el-Tholoth HS, Mohsen T, et al. Does timing of presentation of penile fracture affect outcome of surgical intervention? Urology 2011;77(6):1388-1391.
4. Eke N. Fracture of the penis. Br J Surg 2002;89(5):555-565.
5. Ibrahiem el-HI, el-Tholoth HS, Mohsen T, et al. Penile fracture: Long-term outcome of immediate surgical intervention. Urology 2010;75(1):108-111.
6. Moreno Sierra J, Garde Garcia H, Fernandez Perez C, et al. Surgical repair and analysis of penile fracture complications. Urol Int 2011;86(4):439-443.
7. Mazaris EM, Livadas K, Chalikopoulos D, et al. Penile fractures: Immediate surgical approach with a midline ventral incision. BJU Int 2009;104(4):520-523.
8. Brock G, Hsu G, Nunes L, et al. The anatomy of the tunica albuginea in the normal penis and Peyronie’s disease. J Urol 1997;157:276–281.
9. Zargooshi J. Penile fracture in Kermanshah, Iran: Report of 172 cases. J Urol 2000;164(2):364-366.
10. Ghilan AM, Al-Asbahi WA, Ghafour MA, et al. Management of penile fractures. Saudi Med J 2008;29(10):1443-1447.
11. Koifman L, Cavalcanti AG, Manes CH, et al. Penile fracture — experience in 56 cases. Int Braz J Urol 2003;29(1):35-39.
12. Koifman L, Barros R, Júnior RA, et al. Penile fracture: Diagnosis, treatment and outcomes of 150 patients. Urology 2010;76(6):1488-1492.
13. Sawh SL, O’Leary MP, Ferreira MD, et al. Fractured penis: A review. Int J Impot Res 2008;20(4):366-369.
14. El Atat R, Sfaxi M, Benslama MR, et al. Fracture of the penis: Management and long-term results of surgical treatment. Experience in 300 cases. J Trauma 2008;64(1):121-125.
15. Bitsch M, Kromann-Andersen B, Schou J, et al. The elasticity and the tensile strength of tunica albuginea of the corpora cavernosa. J Urol 1990;143(3): 642-645.
16. van der Horst C, Martínez Portillo FJ, Bannowsky A, et al. Penile fractures: Controversy over surgical or conservative treatment. BJU Int 2003;92(4):349-350.
17. De Rose AF, Giglio M, Carmignani G. Traumatic rupture of the corpora cavernosa: New physiopathologic acquisitions. Urology 2001;57:319–322.
18. Jack GS, Garraway I, Reznichek R, et al. Current treatment options for penile fractures. Rev Urol 2004;6:114–120.
19. Zargooshi J. Penile fracture in Kermanshah, Iran: The long-term results of surgical treatment. BJU Int 2002;89:890–894.
20. Derouiche A, Belhaj K, Hentati H, et al. Management of penile fractures complicated by urethral rupture. Int J Impot Res 2008;20:111–114.
21. Mydlo JH, Hayyeri M, Macchia RJ. Urethrography and cavernosography imaging in a small series of penile fractures: A comparison with surgical findings. Urology 1998;51(4):616-619.
22. El-Assmy A, El-Tholoth HS, Abou-El-Ghar ME, et al. False penile fracture: Value of different diagnostic approaches and long-term outcome of conservative and surgical management. Urology 2010;75(6):1353-1356.
23. Morey AF, Metro MJ, Carney KJ, et al. Consensus on genitourinary trauma. BJU Int 2004;94:507–515.
24. Tsang T, Demby AM. Penile fracture with urethral injury. J Urol 1992; 147(2): 466-468.
25. Fergany AF, Angermeier KW, Montague DK. Review of Cleveland Clinic experience with penile fracture. Urology 1999;54(2):352-355.
26. Ishikawa T, Fujisawa M, Tamada H, et al. Fracture of the penis: Nine cases with evaluation of reported cases in Japan. Int J Urol 2003;10(5):257-260.
27. van der Horst C, Martínez Portillo FJ, Bannowsky A, et al. Penile fractures: Controversy over surgical or conservative treatment. BJU Int 2003;92(4):349-350.
28. Shefi S, Pinthus JH, Mor Y, et al. To bury the knot, then, is better than not. Urology 2008;71(6):1206-1208.
29. Gamal WM, Osman MM, Hammady A, et al. Penile fracture: Long-term results of surgical and conservative management. J Trauma 2011;71(2):491-493.
30. Shah DK, Paul EM, Meyersfield SA, et al. False fracture of the penis. Urology 2003;61(6):1259.
31. Nehru-Babu M, Hendry D, Ai-Saffar N. Rupture of the dorsal vein mimicking fracture of the penis. BJU Int 1999;84(1):179-180.
32. Feki W, Derouiche A, Belhaj K, Ouni A, et al. False penile fracture: Report of 16 cases. Int J Impot Res 2007;19(5): 471-473.
33. El-Assmy A, El-Tholoth HS, Abou-El-Ghar ME, et al. False penile fracture: Value of different diagnostic approaches and long-term outcome of conservative and surgical management. Urology 2010;75(6):1353-1356.
34. Rosenstein DI, Alsikafi NF. Diagnosis and classification of urethral injuries. Urol Clin North Am 2006;33(1):73-85, vi-vii.
35. Santucci RA, Bartley JM. Urologic trauma guidelines: a 21st century update. Nat Rev Urol 2010;7(9):510-519.
36. Marks W, Dawid S, Lasek J, et al. Posterior urethra rupture: Contrast-enhanced computed tomography scan and urethrocystography demonstrations. Case Rep Urol 2012;2012:109589.
37. Bjurlin MA, Fantus RJ, Mellett MM, et al. Genitourinary injuries in pelvic fracture morbidity and mortality using the National Trauma Data Bank. J Trauma 2009;67(5):1033-1039.
38. Mundy AR, Andrich DE. Urethral trauma. Part I: Introduction, history, anatomy, pathology, assessment and emergency management. BJU Int 2011;108(3):310-327.
39. Mundy AR. Pelvic fracture injuries of the posterior urethra. World J Urol 1999;17(2):90-95.
40. Kashefi C, Messer K, Barden R, et al. Incidence and prevention of iatrogenic urethral injuries. J Urol 2008;179(6):2254-2257; discussion 2257-2258.
41. Chapple CR. Urethral injury. BJU Int 2000;86(3):318-326.
42. Chapple C, Barbagli G, Jordan G, et al. Consensus statement on urethral trauma. BJU Int 2004;93(9):1195-1202.
43. Cavalcanti AG, Krambeck R, Araújo A, et al. Management of urethral lesions in penile blunt trauma. Int J Urol 2006;13(9):1218-1220.
44. Lynch TH, Martínez-Piñeiro L, Plas E, et al; European Association of Urology. EAU guidelines on urological trauma. Eur Urol 2005;47(1):1-15.
45. Martínez-Piñeiro L, Djakovic N, Plas E, et al; European Association of Urology. EAU Guidelines on Urethral Trauma. Eur Urol 2010;57(5):791-803.
46. Shlamovitz GZ, McCullough L. Blind urethral catheterization in trauma patients suffering from lower urinary tract injuries. J Trauma 2007;62(2):330-335; discussion 334-335.
47. Djakovic N, Plas E, Martínez-Piñeiro L, et al. Guidelines on Urological Trauma. http://www.uroweb.org/fileadmin/guidelines/2012_Guidelines_large_text_print_total_file.pdf.
48. Moore EE, Cogbill TH, Jurkovich GJ, et al. Organ injury scaling. III: Chest wall, abdominal vascular, ureter, bladder, and urethra. J Trauma 1992;33:337.
49. Mundy AR, Andrich DE. Urethral trauma. Part II: Types of injury and their management. BJU Int 2011;108(5): 630-650.
50. Jordan GH, Virasoro R, Eltahawy EA. Reconstruction and management of posterior urethral and straddle injuries of the urethra. Urol Clin North Am 2006;33(1):97-109, vii.
51. Hadjizacharia P, Inaba K, Teixeira PG, et al. Evaluation of immediate endoscopic realignment as a treatment modality for traumatic urethral injuries. J Trauma 2008;64(6):1443-1449; discussion 1449-1450.
52. Mouraviev VB, Coburn M, Santucci RA. The treatment of posterior urethral disruption associated with pelvic fractures: Comparative experience of early realignment versus delayed urethroplasty. J Urol 2005;173(3):873-876.
53. Elgammal MA. Straddle injuries to the bulbar urethra: Management and outcome in 53 patients. Int Braz J Urol 2009;35(4):450-458.
54. Montague DK, Jarow J, Broderick GA, et al; Members of the Erectile Dysfunction Guideline Update Panel; American Urological Association guideline on the management of priapism. J Urol 2003;170(4 Pt 1):1318-1324.
55. Sharpsteen JR Jr, Powars D, Johnson C, et al. Multisystem damage associated with tricorporal priapism in sickle cell disease. Am J Med 1993;94(3):289-295.
56. Bassett J, Rajfer J. Diagnostic and therapeutic options for the management of ischemic and nonischemic priapism. Rev Urol 2010;12(1):56-63.
57. Priyadarshi S. Oral terbutaline in the management of pharmacologically induced prolonged erection. Int J Impot Res 2004;16(5):424-426.
58. Soni BM, Vaidyanathan S, Krishnan KR. Management of pharmacologically induced prolonged penile erection with oral terbutaline in traumatic paraplegics. Paraplegia 1994;32(10):670-674.
59. Placebo-controlled study of oral terbutaline and pseudoephedrine in management of prostaglandin E1-induced prolonged erections. Urology 1993;42(1):51-53; discussion 53-54.
60. Govier FE, Jonsson E, Kramer-Levien D. Oral terbutaline for the treatment of priapism. J Urol 1994;151(4):878-879.
61. Tay YK, Spernat D, Rzetelski-West K, et al. Acute management of priapism in men. BJU Int 2012;109 Suppl 3:15-21.
62. Broderick GA, Kadioglu A, Bivalacqua TJ, et al. Priapism: Pathogenesis, epidemiology, and management. J Sex Med 2010;7:476–500.
63. BestBets: The effectiveness of oral terbutaline in treatment of priapism. http://www.bestbets.org/bets/bet.php?id=1955.
64. Davila HH, Parker J, Webster JC, et al. Subarachnoid hemorrhage as complication of phenylephrine injection for the treatment of ischemic priapism in a sickle cell disease patient. J Sex Med 2008;5(4):1025-1028.
65. Wyatt JP, Scobie WG. The management of penile zip entrapment in children. Injury 1994;25:59-60.
66. Yip A, Ng SK, Wong WC, et al. Injury to the prepuce. Br J Urol 1989;63(5): 535-538.
67. Flowerdew R, Fishman IJ, Churchill BM. Management of penile zipper injury. J Urol 1977;117:671.
68. Saraf P, Rabinowitz R. Zipper injury of the foreskin. Am J Dis Child 1982;136(6):557-558.
69. Nolan JF, Stillwell TJ, Sands JP Jr. Acute management of the zipper-entrapped penis. J Emerg Med 1990;8(3):305-307.
70. Nakagawa T, Toguri AG. Penile zipper injury. Med Princ Pract 2006;15(4): 303-304.
71. Mishra SC. Safe and painless manipulation of penile zipper entrapment. Indian Pediatr 2006;43(3):252-254.
72. Adu-Frimpong J. Genitourinary trauma in boys. Clin Ped Emerg Med 2009;10:45-49.
73. Inoue N, Crook S, Yamamoto L. Comparing two methods of zipper release. Am J Emerg Med 2005;23: 480-482.
74. Mydlo JH. Treatment of a delayed zipper injury. Urol Int 2000;64(1):45-46.
75. Raveenthiran V. Releasing of zipperentrapped foreskin: A novel nonsurgical technique. Pediatr Emerg Care 2007;23(7):463-464.
76. Strait RT. A novel method for removal penile zipper entrapment. Pediatr Emerg Care 1999;15:412-414.
77. Kanegaye JT, Schonfeld N. Penile zipper entrapment: A simple and less threatening approach using mineral oil. Pediatr Emerg Care 1993;9:90–91.
78. Oosterlinck W. Unbloody management of penile zipper injury. Eur Urol 1981;7(6):365-366.
79. McCann PA. Case report: A novel solution to penile zipper injury — the needle holder. Scientific World Journal 2005;5:298-299.
80. Watson CC. Zipper injuries. Clin Pediatr (Phila) 1971;10(3):188.
81. Arrabal-Polo MA, Arias-Santiago SA, Giron-Prieto MS, et al. Foreskin trapped by zipper: A case report. Cases J 200927;2:6345.
82. YouTube. http://www.youtube.com/watch?v=8knytHJYv9s.
83. Vunda A, Lacroix LE, Schneider F, et al. Videos in clinical medicine. Reduction of paraphimosis in boys. N Engl J Med 2013;368(13):e16.
84. Mackway-Jones K, Teece S. Best evidence topic reports. Ice, pins, or sugar to reduce paraphimosis. Emerg Med J 2004;21(1):77-78.
85. Hamdy FC, Hastie KJ. Treatment for paraphimosis: The “puncture” technique. Br J Surg 1990;77(10):1186.
86. Reynard JM, Barua JM. Reduction of paraphimosis the simple way — the Dundee technique. BJU Int 1999;83:859.
87. Barone JG, Fleisher MH. Treatment of paraphimosis using the “puncture” technique. Pediatr Emerg Care 1993;9:298.
88. Kumar V, Javle P. Modified puncture technique for reduction of paraphymosis. Ann R Coll Surg Engl 2001;83(2): 126-127.
89. Cahill D, Rane A. Reduction of paraphimosis with granulated sugar. BJU Int 1999;83(3):362.
90. Anand A, Kapoor S. Mannitol for paraphimosis reduction. Urol Int 2013;90(1):106-108.
91. Doyle JO. Hyaluronidase in treatment of sequela of paraphimosis. Br Med J 1954;2(4901):1401.
92. DeVries CR, Miller AK, Packer MG. Reduction of paraphimosis with hyaluronidase. Urology 1996;48(3):464-465.
93. Houghton GR. The “iced-glove” method of treatment of paraphimosis. Br J Surg 1973;60(11):876-877.
94. Christianakis E. Sutureless prepuceplasty with wound healing by second intention: An alternative surgical approach in children’s phimosis treatment. BMC Urol 2008;8:6.
95. Smith GA, Sharma V, Knapp JF, et al. The summer penile syndrome: Seasonal acute hypersensitivity reaction caused by chigger bites on the penis. Pediatr Emerg Care 1998;14:116.
96. Ng KL, Mahadevan DT, Htun TH, et al. Pseudoparaphimosis: A hairy affair. Med J Malaysia 2011;66(3):268-269.
97. Burnett AL, Bivalacqua TJ. Priapism: New concepts in medical and surgical management. Urol Clin North Am 2011;38(2):185-194.
98. Broderick GA, Kadioglu A, Bivalacqua TJ, et al. Priapism: Pathogenesis, epidemiology, and management. J Sex Med 2010;7:476-500.
In this article, the authors continue their review of common male genital emergencies. Part I covered common scrotal emergencies, and in Part II, common penile emergencies are reviewed.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
