Minor Trauma Management: Part I
Minor Trauma Management: Part I
Authors: Justin L. Weppner, DO, Lieutenant, Medical Corps, USN, Naval Medical Center Portsmouth, Portsmouth, VA.
James A. McCombs, DO, Lieutenant, Medical Corps, USN, Naval Medical Center Portsmouth, Portsmouth, VA.
Peer Reviewer: Michael C. Bond, MD, FACEP, FAAEM, Assistant Professor, Residency Program Director, Department of Emergency Medicine, University of Maryland School of Medicine
Disclaimer: The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the United States Government.
Over the past few years, we have learned that seemingly minor injuries can cause significant morbidity and, in some cases, even contribute to mortality. Astute emergency providers have learned to look beyond minor complaints and watch for signs of significant problems. Mild traumatic brain injury or concussion is one of the best examples of an injury that has been often dismissed. Post-concussive syndrome was overlooked for many years. Athletes in particular are prone to repeated injury, which we now know contributes to later cognitive impairment and even in some cases death.
This is a two-part series covering minor injuries that are, in fact, not minor. In this first part, the authors discuss mild traumatic brain injury with special emphasis on the judicious use of testing. Part two will concentrate on neck and thoracic injuries.
— Sandra M. Schneider, MD, Editor
Introduction
Determining how to manage minor injuries such as mild traumatic brain injury and minor neck and chest trauma can be difficult due to the trivial outward appearance of such injuries. In cases of minor traumatic injuries, emergency physicians must consider injuries that do not seem apparent upon first glance and must proactively identify and treat unseen injuries before they become catastrophic. Emergency physicians should be cognizant of the potential pitfalls in order to safely and effectively manage patients with minor trauma. This evidence-based review assesses the literature and reviews current guidelines and decision rules on mild traumatic brain injury.
Mild Traumatic Brain Injury
Traumatic brain injury (TBI) is defined as the impairment of brain function due to mechanical force. TBI is an important public health problem in the United States and is frequently referred to as the “silent epidemic” because the complications from TBI, such as changes affecting thought, sensation, language, or emotions, may not be readily apparent.1 The clinical approach to these patients varies widely, and despite the availability of clinical guidelines, many patients undergo computed tomography (CT) imaging, with the majority being interpreted as normal. The challenge for emergency physicians is to quickly screen for the small subset of patients who have potentially lethal intracranial lesions while minimizing excessive cost and radiation.
Each year, an estimated 1.7 million people sustain a head injury in the United States, with 1.3 million of those undergoing emergency evaluation, of which 80% are considered mild injury.1,2 Men are overrepresented at a ratio of 3:1 in all subgroups of TBI, but in some comparable sports, the rate of mild TBI is higher in women.3,4 The four leading causes of TBI treated in the emergency department (ED) are: falls, motor vehicle–related injury, unintentional strike by or against an object (which includes sports and recreational injury), and assaults.3
Mild TBI may result in a predominantly metabolic insult, as opposed to a structural one.5-7 The neuropathology involved in producing the signs and symptoms of minor TBI may remain at the neurobiochemical level, without damage to the microstructure.8 Disruption of enzymatic pathways and accumulation of lactate and nitric oxide in the brain tissue have been reported after experimental minor TBI.9 Approximately 6–8% of patients with mild TBI will have specific injuries detectable on CT.10-12 These injuries include subarachnoid hemorrhage, subdural hematomas, epidural hematomas, cerebral contusions, intraparenchymal hemorrhage, and edema and petechial hemorrhage consistent with axonal injury.
History
From a practical standpoint, minor TBI is a clinical diagnosis.13 The American Congress of Rehabilitation Medicine defines mild TBI based on the following inclusion criteria: 1) any period of loss of consciousness of less than 30 minutes and a Glasgow Coma Scale (GCS) score of 13–15 after this period of unconsciousness (see Table 1); 2) any loss of memory of the event immediately before or after the accident, with post-traumatic amnesia spanning no more than 24 hours; 3) any alteration in mental state at the time of the accident, such as feeling dazed, disoriented, or confused.14
Table 1: Glasgow Coma Scale Scoring
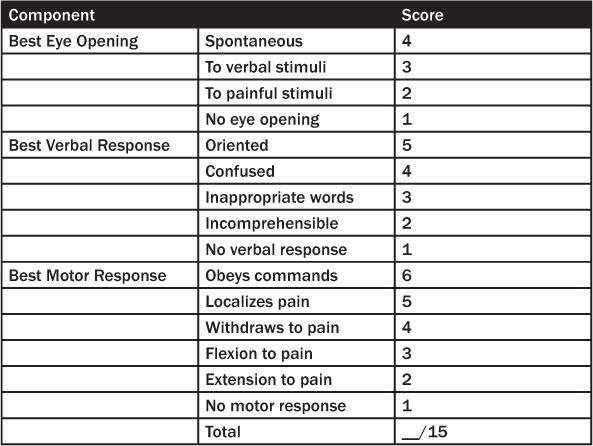
By the time most patients with minor TBI reach the ED, their symptoms are resolving or have completely resolved.13 In the ED, the diagnosis of mild TBI relies on the history surrounding the injury, such as any alteration in mental state at the time of the event or subsequent to the event. This includes the layman’s account of “getting your bell rung,” “seeing stars,” or being dazed and confused because of the injury. The presence of amnesia further supports the diagnosis and may be associated with injury that is more significant.
The mechanism of injury is a key component of the history and provides information regarding associated injuries. Mechanisms of injury that are associated with an increased risk of intracranial injuries include pedestrians being struck by a motor vehicle, falls from a height greater than 3 feet, or falls from more than five stairs.11 The presence of loss of consciousness has been shown to increase the likelihood of intracranial injury, but its absence is only useful as a negative predictor if there are no associated symptoms.12 Signs and symptoms such as seizure, GCS score less than 14, repeated vomiting, and focal neurological deficit have a significantly high positive likelihood of intracranial injury and should be gleaned from the patient.15,16 Drug and alcohol use is associated with intracranial injury in patients with TBI and is an important factor in the history to help determine management, but it does not have a clear role as an independent indicator of outcome.17,18 In sports, past history of concussions, time since last concussion, and severity and duration of past concussion symptoms are all important factors and can assist in predicting outcomes after mild TBI.19
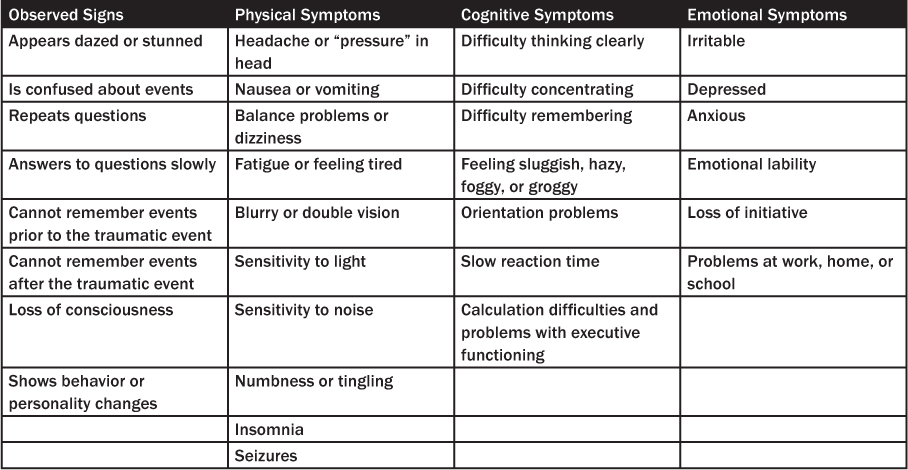
Many patients with mild TBI experience initial objective neuropsychological difficulties involving memory, attention, and executive functioning.20 The most common complaint after minor TBI is headache. A complicating factor is that many of the signs and symptoms of concussion are non-specific and overlap those of other conditions. (See Table 2.) Clinical symptoms may begin immediately after the injury or be delayed for days to weeks. The lack of signs and symptoms at the time of evaluation does not exclude mild TBI if the history is consistent with the diagnosis.
Table 2: Signs and Symptoms Associated with Mild Traumatic Brain Injury
Physical Examination
The physical findings of isolated mild TBI are often normal, but it is important to document a full neurologic examination and look for clues to the severity of the mechanism of injury and any neurologic impairment. Focal neurologic findings are rare and suggest potential significant intracranial pathology. Abnormal pupillary reflexes indicate underlying pathology and should be monitored serially with neurologic examinations.21
Although it is insensitive in mild TBI, it is important to document the GCS score in the patient’s chart. Deficits in the motor component have the strongest correlation with poor outcome in patients with TBI.21,22 Assess the patient for signs of global impairment, such as confusion and amnesia. An examination of gait is important to identify possible balance impairments. In addition, coordination can be assessed using finger-nose-finger testing, heel-to-shin testing, and rapid alternating movements.
The most common findings in mild TBI are subtle impairments in cognitive function. A prospective study conducted on 1,262 subjects revealed that intracranial injury on the CT scan does not predict cognitive deficits.23 Additionally, cognitive tests have not been shown to predict abnormalities on head CT.24 However, several prospective studies have demonstrated that memory tests can be used to predict post-concussive syndrome (PCS).25-27 Assessing cognitive function in the ED is challenging. A patient with mild TBI can be assessed for cognitive deficits in the ED by testing short-term memory (using 3-item recall) and concentration by asking the patient to count serial sevens backward from 100, spelling the word “world” backward, or reciting the months of the year backward. Emergency physicians should be aware that no isolated test can exclude or confirm cognitive deficits. The neuropsychological examination administered by a trained neuropsychologist is the gold standard for evaluation, but it is time-consuming and not practical in the ED setting.19,28,29
Radiographic Indications
An emergency physician faces the challenge of identifying patients with acute intracranial injury from a spectrum of injuries that includes isolated cranial fractures, subarachnoid hemorrhage, subdural and epidural hematomas, and contusions; the emergency physician also has to identify those who can be safely sent home. Up to 15% of mild TBI patients with a GCS score of 14 or 15 will have an acute intracranial injury on non-contrast CT; 1% will have a lesion requiring neurosurgical intervention.30-36
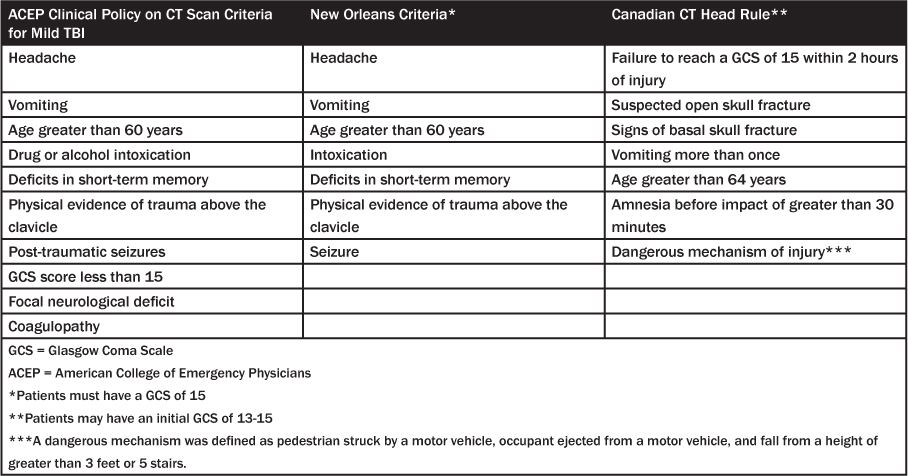
In 2008, the American College of Emergency Physicians (ACEP) published its clinical policy on CT scan criteria for mild TBI. The level A recommendation is for a non-contrast head CT in head trauma patients with loss of consciousness or post-traumatic amnesia only if one or more of the following is present: headache; vomiting; age greater than 60 years; drug or alcohol intoxication; short-term memory loss; physical evidence of trauma above the clavicle; post-traumatic seizure; GCS score less than 15; focal neurologic deficit; coagulopathy. A level B recommendation is for a non-contrast head CT in head trauma patients with no loss of consciousness or post-traumatic amnesia if one or more of the following are present: focal neurologic deficit; vomiting; severe headache; age 65 years or older; physical finding of basilar skull fracture; GCS score less than 15; coagulopathy; a dangerous mechanism of injury such as ejection from a motor vehicle, a pedestrian struck by a motor vehicle, or a fall from a height of more than 3 feet or five stairs.37 However, it is important to note that these are recommendations for when to obtain a CT. They are not guidelines for when it is safe to forego a CT.
Haydel et al10 prospectively enrolled 1,429 patients with a GCS score of 15 in the ED and a history of loss of consciousness or amnesia after the traumatic event. The study had two phases: an initial phase in which predictors from intracranial injury were identified (n = 520) and a validation phase to validate the predictors (n = 909). The study reported that 93 (6.5%) patients had an intracranial lesion and that six (0.4%) required neurosurgical intervention. The New Orleans Criteria derived from the study consisted of the following seven criteria: headache; vomiting; age greater than 60 years; intoxication; deficit in short-term memory; physical evidence of trauma above the clavicle; seizure. Absence of all seven findings had 100% negative predictive value (95% confidence interval [CI], 99–100%).
Steill et al11 evaluated 3,121 patients prospectively, 2,489 of whom had a GCS score of 15. Patients attended a follow-up interview at 14 days to assess outcome. The primary outcome measure was the need for neurosurgical intervention, and the secondary outcome was a clinically important brain injury, defined by expert consensus. Clinically unimportant lesions were defined as solitary contusions less than 5 mm in diameter, smear subdurals less than 4 mm thick, isolated pneumocephalus, and closed depressed skull fractures not through the inner table. The Canadian CT Head Rule derived from the study consisted of five criteria that were considered high-risk features: failure to reach a GCS score of 15 within 2 hours of injury; suspected open skull fracture; signs of basal skull fracture; vomiting more than once; and age greater than 64 years. In addition, there were two criteria that were considered medium-risk features: amnesia of events before impact greater than 30 minutes and a dangerous mechanism of injury such as a pedestrian struck by a motor vehicle, occupant ejected from a motor vehicle, or fall from a height greater than 3 feet or five stairs. (See Table 3.)
Table 3: ACEP Clinical Policy, New Orleans, and Canadian CT Clinical Decision Rules
Multiple studies have compared the performance of the New Orleans Criteria and the Canadian CT Head Rule.38-41 Smits et al39 applied the two decision rules at four university hospitals in the Netherlands to 3,181 consecutive adult patients with a GCS score of 13 or 14 or a GCS score of 15 and one risk factor, as identified by the decision rules. The New Orleans Criteria had 100% sensitivity for identifying a neurosurgical lesion (95% CI, 34.2–100%) and 5.3% specificity (95% CI, 2.5–8.3%). In addition, the New Orleans Criteria had 98.3% sensitivity for identifying an intracranial lesion (95% CI, 94–99.5%) and 5.6% specificity (95% CI, 2.7–8.8%). The Canadian CT Head Rule had 100% sensitivity for identifying a neurosurgical lesion (95% CI, 64.6–100%) and 37.2% specificity (95% CI, 34.1– 40.4%). Furthermore, the Canadian CT Head Rule had 83.4% sensitivity (95% CI, 77.7–87.9%) and 39.4% specificity (95% CI, 36–42.8%) for identifying an intracranial lesion. The study validated the high sensitivity of both rules for identifying lesions requiring neurosurgical intervention. It also demonstrated the superiority of the New Orleans Criteria over the Canadian CT Head Rule for identifying acute intracranial lesions. However, the higher sensitivity of the New Orleans Criteria came at the expense of a significantly lower specificity.
Papa et al40 consecutively enrolled 431 patients at a United States level I trauma center who presented with mild head injury, who experienced loss of consciousness, disorientation, or amnesia, and had a GCS score of 13–15. Both the New Orleans Criteria and the Canadian CT Head Rule had 100% sensitivity for detecting traumatic intracranial lesions (95% CI, 82–100%), but the Canadian CT Head Rule was more specific, with a specificity of 36.3% (95% CI, 31–42%) vs. the specificity of 10.2% (95% CI, 7–14%) of the New Orleans Criteria. The Canadian CT Head Rule and New Orleans Criteria both had 100% sensitivity for detecting clinically important brain lesions (95% CI, 68–100%), but specificity was 35% (95% CI, 30–41%) for the Canadian CT Head Rule and 9.9% (95% CI, 7–14%) for the New Orleans Criteria. When the rules were compared for predicting the need for neurosurgical intervention, their sensitivity was equivalent at 100% (95% CI, 31–100%), but the Canadian CT Head Rule had higher specificity at 80.7% (95% CI, 76–85%) vs. the 9.6% (95% CI, 7–14%) specificity of the New Orleans Criteria. Again, the New Orleans Criteria had equivalently high sensitivities for detecting a traumatic intracranial lesion on CT, clinically important brain injury, and neurosurgical intervention, but the Canadian CT Head Rule was more specific.
Although both the New Orleans Criteria and the Canadian CT Head Rule have been validated, it is very important to ensure that they are applied within the limits of their inclusion criteria, and the clinician should understand the above sensitivity and specificity both for neurosurgical lesions and for intracranial injury. These rules are valid when applied to patients who have experienced loss of consciousness or amnesia and who are not on anticoagulants.
A combination of these rules provides a basis for identifying patients at risk for underlying intracranial injury and for determining the need for head CT. Decision rules can guide clinical practice, but each patient must be assessed individually, and none of these rules address short- or long-term non-neurosurgical sequelae.28
Laboratory Evaluation
Currently, no reliable tests can confirm the diagnosis of concussion. A number of serum biomarkers such as S100B, neuron-specific enolase, myelin basic protein, cleaved tau, and creatine kinase isoenzyme BB have all been investigated in minor TBI. At this time, only S100B predicts abnormal CT findings in minor TBI. However, S100B appears to lack central nervous system specificity and is often elevated in multisystem trauma patients with no head injury.9 The development of serum markers is evolving and may play a new role in diagnosis and management in the future.42-55
Patients with known or suspected coagulation disorders, liver disease, or those taking anticoagulants should have coagulation studies performed as soon as possible.56
Management
There are no pharmacologic treatments for mild TBI.28 The management decisions faced by the emergency physician are the same as those addressed when evaluating all patients with mild TBI, such as the extent of the workup to initiate in the ED and whether the patient can be discharged safely. The main objectives are to identify patients who have intracranial lesions requiring neurosurgical intervention, identify patients whose condition might deteriorate, and guide their return to normal activities.
Anticoagulants and Anti-platelet Agents with Mild TBI. The use of anticoagulation or anti-platelet agents is common for the treatment of various conditions, ranging from warfarin for atrial fibrillation to clopidogrel for cardiac stents. The prevalence of immediate traumatic intracranial hemorrhage in patients with pre-injury warfarin use ranges from 0% to 65%,56-65 and that in patients with pre-injury clopidogrel use, prevalence ranges from 36% to 71%.58,59,66 The risk of intracranial injury increases with increased international normalization ratios (INR). An INR of 2.4 or more increases the risk of immediate intracranial injury.56,60,67 Regrettably, no specific INR can be used to exclude the risk of intracranial injury in patients with a sub-therapeutic INR.68 Current guidelines recommend that patients with head trauma and pre-injury warfarin use undergo routine cranial CT scanning.37,69-71 A recent retrospective study by Moore et al2 found that failing to perform a CT in patients presenting with a GCS score of 15 and no history of loss of consciousness would have led to a missed injury rate of 66% (43 of 65 patients).
A prospective, observational, multicenter study evaluated the incidence and prevalence of immediate and delayed traumatic intracranial hemorrhage in patients with blunt trauma who were receiving clopidogrel and warfarin. Seventy of the 1,000 patients had immediate traumatic intracranial hemorrhage on CT in the ED. The prevalence of immediate traumatic intracranial hemorrhage was higher in patients receiving clopidogrel (33/276; 12%; 95% CI, 8.4–16.4%) than in those receiving warfarin (37/724; 5.1%; 95% CI, 3.6–7%). Delayed traumatic intracranial hemorrhage was identified in four of 687 (0.6%; 95% CI, 0.2–1.5%) patients receiving warfarin and none of 243 (0%; 95% CI, 0–1.5%) patients receiving clopidogrel.72 The prevalence of immediate traumatic intracranial hemorrhage in well-appearing patients is a matter of great concern. In the immediate traumatic intracranial hemorrhage study, more than 60% of both the warfarin and clopidogrel cohorts had normal mental status and a GCS score of 15. In addition, a significant proportion of patients experienced no loss of consciousness and exhibited no physical evidence of trauma above the clavicle. Thus, the authors recommended routine urgent CT imaging in head-injured patients with previous warfarin or clopidogrel use, even in well-appearing patients without a history of loss of consciousness or amnesia.
The concern for delayed intracranial hemorrhage in patients with warfarin use is based on several case reports and case series.73-75 This has led to published guidelines recommending routine admission for all head-injured patients receiving warfarin despite a normal CT scan result.76 Nevertheless, the results of the prospective, multicenter study by Nishijima et al indicate that delayed traumatic intracranial hemorrhage occurs infrequently in less than 1% of both the warfarin and clopidogrel populations. According to that study, patients receiving warfarin or clopidogrel who have a normal cranial CT scan result and no other indications for admission may be discharged home with explicit discharge instructions for close follow-up.72
Peck et al77 retrospectively reviewed 500 patients taking prescription anti-platelet and anticoagulant agents. They found a 1% incidence of delayed intracranial hemorrhage following a repeat CT of the patients. Of four patients identified in the study with delayed bleeding, none had focal findings on examination and none required any intervention. These findings suggest that clinically significant delayed intracranial hemorrhage is rare.
Overall, patients taking anticoagulation agents with suspected head injury should undergo an initial head CT and an observation period for signs and symptoms of neurologic deterioration. If the initial head CT is negative, and the neurologic examination is normal or unchanged, these patients can be safely discharged without a second head CT and with close outpatient follow-up. This approach should be sufficiently sensitive to detect significant intracranial hemorrhage while avoiding unnecessary CTs. Patients who are on anti-platelet therapy or who are anticoagulated with a normal CT and continued symptoms of TBI or a supratherapeutic INR should be admitted for 24 hours.67,78
Emergency physicians should have a low threshold for factor replacement or reversal agents in patients taking anticoagulants or anti-platelet agents.61,79 Patients on warfarin with intracranial injury on CT should undergo rapid reversal using fresh frozen plasma or prothrombin complex concentrates.80 It should be noted that the standard dose of 10-20 mg/kg of fresh frozen plasma may far exceed the initial two units that are ordered by most clinicians.81 Prothrombin complex concentrates have replaced fresh frozen plasma at many institutions due the considerable volume of fresh frozen plasma required to lower the INR. Yasaka et al82 found that a dose of 500 IU of prothrombin complex concentrates was optimal for a rapid reversal for an INR less than 5.0, but that higher doses might be needed for higher INRs. Vitamin K should be initiated in the ED, but emergency clinicians must be aware that full reversal using vitamin K may take up to 24 hours. Recombinant factor VIIa (rFVIIa) may also be administered to anticoagulated patients who require urgent craniotomy. Administering rFVIIa to reverse anticoagulation is an off-label use, and further research is needed to better define its role in patients with traumatic intracranial bleeding.83,84 In the case of anti-platelet agents, platelet transfusions in patients taking aspirin or clopidogrel have not been shown to influence outcomes after TBI.85 Reversal of newer anticoagulants is more difficult, as they do not respond to vitamin K or fresh frozen plasma.
Post-concussive Syndrome
The symptoms of almost all patients with minor TBI will undergo rapid and complete resolution,86 and there is currently no good evidence that uncomplicated minor TBI leads to long-term sequelae.87 However, post-concussive syndrome affects a subset of patients that report persistent symptoms for prolonged periods after injury. The incidence of post-concussive syndrome is reported to be 10–25%, but these estimates are based on inpatient studies and are likely to be an overestimation of the incidence in mild TBI patients.9,87,88 In EDs, there has been little focus on the potential development of post-concussive syndrome.89 The most common delayed or persistent post-concussive complaints are headache, sensory sensitivity, memory or concentration difficulties, irritability, sleep disturbances, and depression. Studies of concussed athletes who undergo pre-concussion testing show that the cognitive domain most frequently involved in post-concussive syndrome is memory.9 Dizziness that occurs early after trauma is associated with prolonged post-concussive syndrome.11
Resuming Sports Activities
It is imperative to emphasize the need for competent follow-up and clearance before resuming normal activities, especially contact sports.90 Repeated concussions can lead to long-term cognitive deficits and structural damage to the brain.20,91 Ensure that patients with mild TBI avoid any unnecessary risk of a “second impact” during the symptomatic period, which can result in devastating brain edema. Rapid onset of cerebral edema and death can occur when a second concussion occurs prior to recovery from the first, particularly in athletes returning prematurely to sports.91 Decisions on return to play should be managed very carefully by health care providers trained specifically in concussion management. In view of the concerns for second impact syndrome, a graded activity program should be instituted until athletes can resume full activities.20,92-94 Recent return to play guidelines no longer advocate return to play the same day of the traumatic event.4 The assessment for return to play is not a decision made by the emergency physician, but is managed by primary health care providers and specialists with consideration of the severity of the injury, past injuries, and the possibility of future sports-related injuries.95
Admission Criteria
While most patients with mild TBI are safe to discharge, a brief inpatient observation period of 12–24 hours is advisable if any doubt exists regarding the safety of the discharged patient with minor TBI.13 Other criteria for admission are mass lesions associated with head trauma requiring intensive monitoring or surgery, intracranial bleeding, and patients with ongoing symptoms that include repetitive questioning, anterograde amnesia, or disorientation.96 Patients who are on anti-platelet therapy or who are anticoagulated with a normal CT and continued symptoms of TBI or a supratherapeutic INR should be observed for 24 hours.67,78
Discharge Criteria
Patients with isolated mild TBI who have a normal head CT scan and are at minimal risk for developing an intracranial lesion may be safely discharged from the ED.37 A prospective study of 1,170 mild TBI patients with a GCS score of 15 and who underwent CT and 24-h admission found that no patient with a negative CT scan result later deteriorated.97 A retrospective study of 2,252 trauma center admissions who had a head CT scan found that no patient with an initial negative head CT scan required subsequent neurosurgical intervention.98 A comprehensive literature review involving 2,187 abstracts and 410 articles indicated a total mild TBI population of 62,000 subjects who presented with a GCS score of 15. Only three (0.005%) cases of an adverse outcome in the first 48 hours were reported in the literature. Eight additional patients with possible early adverse outcomes were also observed. The conclusion based on the literature review was that in patients meeting guidelines for non-contrast head CT, the evidence supports the CT strategy as a safe way to triage mild TBI patients for admission versus discharge.99
Patients with minor head trauma not meeting criteria for head CT and normal neurologic findings may be discharged home with a friend or family member and with head injury discharge instructions.96
Discharge Instructions
Several studies have documented poor understanding of discharge instructions in patients with mild TBI.100,101 The decision to discharge a mild TBI patient from the ED must be coupled with appropriate discharge instructions. It is imperative to discharge the patient to the care of a responsible individual and to provide instructions to both the patient and the individual. If CT is not clinically indicated, the patient should receive careful instructions about delayed symptoms and circumstances for return to the ED. It is also very important to provide instructions regarding physical activity and primary care follow-up.
Bazarian et al100 evaluated abstracted records from the National Hospital Ambulatory Medical Care Survey for 306 patients with isolated mild TBI. Of these 306 patients, 9% were discharged without any recommendations for follow-up, and 28% had only “return to the ED as needed” as a follow-up recommendation. Another study reviewed mild TBI discharge instruction sheets from 15 institutions for the presence of six factors that were deemed significant for post-discharge patient monitoring based on their literature review: signs and symptoms of declining GCS score; amnesia; headache; vomiting; neurologic deficit; seizure. Of the 15 institutions, the discharge instructions of only one institution contained all six factors. They also found that most of the discharge instructions were at a high-grade reading level.101 Discharge instructions approved by ACEP and Centers for Disease Control and Prevention (CDC) are available on the CDC website at http://www.cdc.gov/concussion/pdf/TBI_Patient_Instructions-a.pdf.102
Summary
The ACEP clinical policy on CT scan criteria for mild TBI provides clearly defined guidelines for patients who should undergo non-contrast CT scanning. Although both the New Orleans Criteria and the Canadian CT Head Rule are validated decision rules, it is very important to ensure that they are applied within the limits of their inclusion criteria, and the clinician should be cognizant of their sensitivity and specificity both for neurosurgical lesions and for intracranial injury. It is imperative to emphasize the need for competent follow-up and clearance before resuming normal activities, especially contact sports. Repeated concussions can lead to long-term cognitive deficits and structural damage to the brain. The assessment for return to play is not a decision made by the emergency physician, but is managed by primary providers and specialists with consideration of the severity of the injury, past injuries, and the possibility of future sports-related injuries.95 The decision to discharge a mild TBI patient from the ED must be coupled with appropriate discharge instructions, and the patient should be discharged to the care of a responsible individual.
The authors are military service members who prepared this work as part of their official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the United States Government.” Title 17 U.S.C. 101 defines a United States Government work as a work prepared by a military service member or employee of the United States Government as part of that person’s official duties.
References
1. Faul M XL, Wald MM, Coronado VG. Traumatic brain injury in the United States: Emergency department visits, hospitalizations and deaths 2002–2006. Atlanta (GA): Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. Vol March 2010: CDC; 2010: http://www.cdc.gov/traumaticbraininjury/pdf/blue_book.pdf. Accessed 3/15/2013.
2. Moore MM, Pasquale MD, Badellino M. Impact of age and anticoagulation: Need for neurosurgical intervention in trauma patients with mild traumatic brain injury. J Trauma Acute Care Surg 2012;73(1):126-130.
3. Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain injury: A brief overview. J Head Trauma Rehabil 2006;21(5):375-378.
4. Herring SA, Cantu RC, Guskiewicz KM, et al. Concussion (mild traumatic brain injury) and the team physician: A consensus statement — 2011 update. Med Sci Sports Exerc 2011;43(12):2412-2422.
5. Hovda DA. Oxidative need and oxidative capacity following traumatic brain injury. Crit Care Med 2007;35(2):663-664.
6. Lifshitz J, Sullivan PG, Hovda DA, et al. Mitochondrial damage and dysfunction in traumatic brain injury. Mitochondrion 2004;4(5-6):705-713.
7. Cimatti M. Assessment of metabolic cerebral damage using proton magnetic resonance spectroscopy in mild traumatic brain injury. J Neurosurg Sci 2006;50(4):83-88.
8. Biasca N, Maxwell WL. Minor traumatic brain injury in sports: A review in order to prevent neurological sequelae. Prog Brain Res 2007;161:263-291.
9. Bazarian JJ, Blyth B, Cimpello L. Bench to bedside: Evidence for brain injury after concussion — looking beyond the computed tomography scan. Acad Emerg Med 2006;13(2):199-214.
10. Haydel MJ, Preston CA, Mills TJ, et al. Indications for computed tomography in patients with minor head injury. N Engl J Med 2000;343(2):100-105.
11. Stiell IG, Wells GA, Vandemheen K, et al. The Canadian CT Head Rule for patients with minor head injury. Lancet 2001;357(9266):1391-1396.
12. Mower WR, Hoffman JR, Herbert M, et al. Developing a decision instrument to guide computed tomographic imaging of blunt head injury patients. J Trauma 2005;59(4):954-959.
13. Marx JA, Hockberger RS, Walls RM, et al. Rosen’s Emergency Medicine: Concepts and Clinical Practice, 7th ed. Philadelphia: Mosby/Elsevier; 2010.
14. Kay T, Harrington DE, Adams, R, et al. Definition of mild traumatic brain injury. J Head Trauma Rehabil 1993;8(3): 86-87.
15. Pandor A, Goodacre S, Harnan S, et al. Diagnostic management strategies for adults and children with minor head injury: A systematic review and an economic evaluation. Health Technol Assess 2011;15(27):1-202.
16. Bainbridge J, Khirwadkar H, Hourihan MD. Vomiting — is this a good indication for CT head scans in patients with minor head injury? Br J Radiol 2012;85(1010):183-186.
17. Godbout BJ, Lee J, Newman DH, et al. Yield of head CT in the alcohol-intoxicated patient in the emergency department. Emerg Radiol 2011;18(5):381-384.
18. Jacobs B, Beems T, Stulemeijer M, et al. Outcome prediction in mild traumatic brain injury: Age and clinical variables are stronger predictors than CT abnormalities. J Neurotrauma 2010;27(4):655-668.
19. McCrory P, Meeuwisse W, Johnston K, et al. Consensus Statement on Concussion in Sport: The 3rd International Conference on Concussion in Sport held in Zurich, November 2008. Br J Sports Med 2009;43 Suppl 1:i76-90.
20. Iverson GL, Gaetz M, Lovell MR, et al. Cumulative effects of concussion in amateur athletes. Brain Inj 2004;18(5): 433-443.
21. Hoffmann M, Lefering R, Rueger JM, et al. Pupil evaluation in addition to Glasgow Coma Scale components in prediction of traumatic brain injury and mortality. Br J Surg 2012;99 Suppl 1:122-130.
22. Compagnone C, d’Avella D, Servadei F, et al. Patients with moderate head injury: A prospective multicenter study of 315 patients. Neurosurgery 2009;64(4):690-696; discussion 696-697.
23. Lannsjo M, Backheden M, Johansson U, et al. Does head CT scan pathology predict outcome after mild traumatic brain injury? Eur J Neurol 2013;20(1): 124-129.
24. Stein SC, Spettell C, Young G, et al. Limitations of neurological assessment in mild head injury. Brain Inj 1993;7(5):425-430.
25. Faux S, Sheedy J, Delaney R, et al. Emergency department prediction of post-concussive syndrome following mild traumatic brain injury — an international cross-validation study. Brain Inj 2011;25(1):14-22.
26. Bazarian J, Hartman M, Delahunta E. Minor head injury: Predicting follow-up after discharge from the emergency department. Brain Inj 2000;14(3): 285-294.
27. Sheedy J, Harvey E, Faux S, et al. Emergency department assessment of mild traumatic brain injury and the prediction of postconcussive symptoms: A 3-month prospective study. J Head Trauma Rehabil 2009;24(5):333-343.
28. Tintinalli JE, Stapczynski JS. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 7th ed. New York: McGraw-Hill; 2011.
29. Register-Mihalik JK, Guskiewicz KM, Mihalik JP, et al. Reliable change, sensitivity, and specificity of a multidimensional concussion assessment battery: Implications for caution in clinical practice. J Head Trauma Rehabil Jun 9 2012.
30. Stein SC, Ross SE. The value of computed tomographic scans in patients with low-risk head injuries. Neurosurgery 1990;26(4):638-640.
31. Jennett B, Teasdale G. Aspects of coma after severe head injury. Lancet 1977;1(8017):878-881.
32. Harad FT, Kerstein MD. Inadequacy of bedside clinical indicators in identifying significant intracranial injury in trauma patients. J Trauma 1992;32(3):359-361; discussion 361-353.
33. Miller EC, Holmes JF, Derlet RW. Utilizing clinical factors to reduce head CT scan ordering for minor head trauma patients. J Emerg Med 1997;15(4): 453-457.
34. Borczuk P. Predictors of intracranial injury in patients with mild head trauma. Ann Emerg Med 1995;25(6):731-736.
35. Jeret JS, Mandell M, Anziska B, et al. Clinical predictors of abnormality disclosed by computed tomography after mild head trauma. Neurosurgery 1993;32(1):9-15; discussion 15-16.
36. Nagurney JT, Borczuk P, Thomas SH. Elder patients with closed head trauma: A comparison with nonelder patients. Acad Emerg Med 1998;5(7):678-684.
37. Jagoda AS, Bazarian JJ, Bruns JJ, Jr, et al. Clinical policy: Neuroimaging and decisionmaking in adult mild traumatic brain injury in the acute setting. Ann Emerg Med 2008;52(6):714-748.
38. Stiell IG, Clement CM, Rowe BH, et al. Comparison of the Canadian CT Head Rule and the New Orleans Criteria in patients with minor head injury. JAMA 2005;294(12):1511-1518.
39. Smits M, Dippel DW, de Haan GG, et al. External validation of the Canadian CT Head Rule and the New Orleans Criteria for CT scanning in patients with minor head injury. JAMA 2005;294(12): 1519-1525.
40. Papa L, Stiell IG, Clement CM, et al. Performance of the Canadian CT Head Rule and the New Orleans Criteria for predicting any traumatic intracranial injury on computed tomography in a United States Level I trauma center. Acad Emerg Med 2012;19(1):2-10.
41. Ro YS, Shin SD, Holmes JF, et al. Comparison of clinical performance of cranial computed tomography rules in patients with minor head injury: A multicenter prospective study. Acad Emerg Med 2011;18(6):597-604.
42. Townend W, Dibble C, Abid K, et al. Rapid elimination of protein S-100B from serum after minor head trauma. J Neurotrauma 2006;23(2):149-155.
43. Bazarian JJ, Beck C, Blyth B, et al. Impact of creatine kinase correction on the predictive value of S-100B after mild traumatic brain injury. Restor Neurol Neurosci 2006;24(3):163-172.
44. Biberthaler P, Linsenmeier U, Pfeifer KJ, et al. Serum S-100B concentration provides additional information fot the indication of computed tomography in patients after minor head injury: A prospective multicenter study. Shock 2006;25(5):446-453.
45. Biberthaler P, Mussack T, Wiedemann E, et al. Rapid identification of high-risk patients after minor head trauma (MHT) by assessment of S-100B: Ascertainment of a cut-off level. Eur J Med Res 2002;7(4):164-170.
46. Biberthaler P, Mussack T, Wiedemann E, et al. Elevated serum levels of S-100B reflect the extent of brain injury in alcohol intoxicated patients after mild head trauma. Shock 2001;16(2):97-101.
47. Ingebrigtsen T, Romner B, Marup-Jensen S, et al. The clinical value of serum S-100 protein measurements in minor head injury: A Scandinavian multicentre study. Brain Inj 2000;14(12):1047-1055.
48. Muller K, Townend W, Biasca N, et al. S100B serum level predicts computed tomography findings after minor head injury. J Trauma 2007;62(6):1452-1456.
49. Mussack T, Biberthaler P, Kanz KG, et al. Immediate S-100B and neuron-specific enolase plasma measurements for rapid evaluation of primary brain damage in alcohol-intoxicated, minor head-injured patients. Shock 2002;18(5):395-400.
50. Poli-de-Figueiredo LF, Biberthaler P, Simao Filho C, et al. Measurement of S-100B for risk classification of victims sustaining minor head injury — first pilot study in Brazil. Clinics (Sao Paulo) 2006;61(1):41-46.
51. Anderson RE, Hansson LO, Nilsson O, et al. High serum S100B levels for trauma patients without head injuries. Neurosurgery 2001;48(6):1255-1258; discussion 1258-1260.
52. Levitt MA, Cook LA, Simon BC, et al. Biochemical markers of cerebral injury in patients with minor head trauma and ethanol intoxication. Acad Emerg Med 1995;2(8):675-680.
53. Defazio MV, Rammo RA, Robles JR, et al. The potential utility of blood-derived biochemical markers as indicators of early clinical trends following severe traumatic brain injury. World Neurosurg Jan. 8, 2013.
54. Muller B, Evangelopoulos DS, Bias K, et al. Can S-100B serum protein help to save cranial CT resources in a peripheral trauma centre? A study and consensus paper. Emerg Med J 2011;28(11): 938-940.
55. Unden J, Romner B. Can low serum levels of S100B predict normal CT findings after minor head injury in adults?: An evidence-based review and meta-analysis. J Head Trauma Rehabil 2010;25(4): 228-240.
56. Claudia C, Claudia R, Agostino O, et al. Minor head injury in warfarinized patients: Indicators of risk for intracranial hemorrhage. J Trauma 2011;70(4): 906-909.
57. Li J, Brown J, Levine M. Mild head injury, anticoagulants, and risk of intracranial injury. Lancet 2001;357(9258): 771-772.
58. Brewer ES, Reznikov B, Liberman RF, et al. Incidence and predictors of intracranial hemorrhage after minor head trauma in patients taking anticoagulant and antiplatelet medication. J Trauma 2011;70(1):E1-E5.
59. Pieracci FM, Eachempati SR, Shou J, et al. Degree of anticoagulation, but not warfarin use itself, predicts adverse outcomes after traumatic brain injury in elderly trauma patients. J Trauma 2007;63(3):525-530.
60. Franko J, Kish KJ, O’Connell BG, et al. Advanced age and preinjury warfarin anticoagulation increase the risk of mortality after head trauma. J Trauma 2006;61(1):107-110.
61. Ivascu FA, Howells GA, Junn FS, et al. Rapid warfarin reversal in anticoagulated patients with traumatic intracranial hemorrhage reduces hemorrhage progression and mortality. J Trauma 2005;59(5):1131-1137; discussion 1137-1139.
62. Gittleman AM, Ortiz AO, Keating DP, et al. Indications for CT in patients receiving anticoagulation after head trauma. AJNR Am J Neuroradiol 2005;26(3):603-606.
63. Mina AA, Bair HA, Howells GA, et al. Complications of preinjury warfarin use in the trauma patient. J Trauma 2003;54(5):842-847.
64. Garra G, Nashed AH, Capobianco L. Minor head trauma in anticoagulated patients. Acad Emerg Med 1999;6(2):121-124.
65. Major J, Reed MJ. A retrospective review of patients with head injury with coexistent anticoagulant and antiplatelet use admitted from a UK emergency department. Emerg Med J 2009;26(12): 871-876.
66. Jones K, Sharp C, Mangram AJ, et al. The effects of preinjury clopidogrel use on older trauma patients with head injuries. Am J Surg 2006;192(6):743-745.
67. Menditto VG, Lucci M, Polonara S, et al. Management of minor head injury in patients receiving oral anticoagulant therapy: A prospective study of a 24-hour observation protocol. Ann Emerg Med 2012;59(6):451-455.
68. Rendell S, Batchelor JS. An analysis of predictive markers for intracranial haemorrhage in warfarinised head injury patients. Emerg Med J 2013;30(1):28-31.
69. Servadei F, Teasdale G, Merry G. Defining acute mild head injury in adults: A proposal based on prognostic factors, diagnosis, and management. J Neurotrauma 2001;18(7):657-664.
70. National Collaborative Centre for Acute Care and National Institute for Health and Clinical Excellence. Head injury: triage, assessment, investigation, and early management of head injury in infants, children, and adults. 2007; http://www.nice.org.uk/nicemedia/live/11836/36257/36257.pdf. Accessed 3/19/2013, 2013.
71. Fortuna GR, Mueller EW, James LE, et al. The impact of preinjury antiplatelet and anticoagulant pharmacotherapy on outcomes in elderly patients with hemorrhagic brain injury. Surgery 2008;144(4):598-603; discussion 603-595.
72. Nishijima DK, Offerman SR, Ballard DW, et al. Immediate and delayed traumatic intracranial hemorrhage in patients with head trauma and preinjury warfarin or clopidogrel use. Ann Emerg Med 2012;59(6):460-468 e461-e467.
73. Cohen DB, Rinker C, Wilberger JE. Traumatic brain injury in anticoagulated patients. J Trauma 2006;60(3):553-557.
74. Itshayek E, Rosenthal G, Fraifeld S, et al. Delayed posttraumatic acute subdural hematoma in elderly patients on anticoagulation. Neurosurgery 2006;58(5):E851-E856; discussion E851-E856.
75. Reynolds FD, Dietz PA, Higgins D, et al. Time to deterioration of the elderly, anticoagulated, minor head injury patient who presents without evidence of neurologic abnormality. J Trauma 2003;54(3):492-496.
76. Vos PE, Battistin L, Birbamer G, et al. EFNS guideline on mild traumatic brain injury: Report of an EFNS task force. Eur J Neurol 2002;9(3):207-219.
77. Peck KA, Sise CB, Shackford SR, et al. Delayed intracranial hemorrhage after blunt trauma: Are patients on preinjury anticoagulants and prescription antiplatelet agents at risk? J Trauma 2011;71(6):1600-1604.
78. Li J. Admit all anticoagulated head-injured patients? A million dollars versus your dime. You make the call. Ann Emerg Med 2012;59(6):457-459.
79. Brown CV, Sowery L, Curry E, et al. Recombinant factor VIIa to correct coagulopathy in patients with traumatic brain injury presenting to outlying facilities before transfer to the regional trauma center. Am Surg 2012;78(1):57-60.
80. Goodnough LT, Shander A. How I treat warfarin-associated coagulopathy in patients with intracerebral hemorrhage. Blood 2011;117(23):6091-6099.
81. Ansell J, Hirsh J, Hylek E, et al. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008;133(6 Suppl):160S-198S.
82. Yasaka M, Sakata T, Naritomi H, et al. Optimal dose of prothrombin complex concentrate for acute reversal of oral anticoagulation. Thromb Res 2005;115(6):455-459.
83. Bartal C, Freedman J, Bowman K, et al. Coagulopathic patients with traumatic intracranial bleeding: Defining the role of recombinant factor VIIa. J Trauma 2007;63(4):725-732.
84. Stein DM, Dutton RP, Kramer ME, et al. Reversal of coagulopathy in critically ill patients with traumatic brain injury: Recombinant factor VIIa is more cost-effective than plasma. J Trauma 2009;66(1):63-72; discussion 73-65.
85. Downey DM, Monson B, Butler KL, et al. Does platelet administration affect mortality in elderly head-injured patients taking antiplatelet medications? Am Surg 2009;75(11):1100-1103.
86. Carroll LJ, Cassidy JD, Peloso PM, et al. Prognosis for mild traumatic brain injury: Results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. J Rehabil Med 2004(43 Suppl):84-105.
87. Rees PM. Contemporary issues in mild traumatic brain injury. Arch Phys Med Rehabil 2003;84(12):1885-1894.
88. Chamelian L, Feinstein A. Outcome after mild to moderate traumatic brain injury: The role of dizziness. Arch Phys Med Rehabil 2004;85(10):1662-1666.
89. Reuben A, Sampson P, Harris AR, et al. Postconcussion syndrome (PCS) in the emergency department: Predicting and pre-empting persistent symptoms following a mild traumatic brain injury. Emerg Med J Mar. 6, 2013.
90. Advanced trauma life support for doctors ATLS: Manuals for coordinators and faculty. 8th ed. Chicago, IL: American College of Surgeons; 2008.
91. Lovell M. The neurophysiology and assessment of sports-related head injuries. Neurol Clin 2008;26(1):45-62; viii.
92. Collins MW, Lovell MR, Iverson GL, et al. Cumulative effects of concussion in high school athletes. Neurosurgery 2002;51(5):1175-1179; discussion 1180-1171.
93. Guskiewicz KM, Bruce SL, Cantu RC, et al. Research based recommendations on management of sport related concussion: Summary of the National Athletic Trainers’ Association position statement. Br J Sports Med 2006;40(1):6-10.
94. Covassin T, Elbin R, 3rd, Stiller-Ostrowski JL. Current sport-related concussion teaching and clinical practices of sports medicine professionals. J Athl Train 2009;44(4):400-404.
95. Cantu RC, Register-Mihalik JK. Considerations for return-to-play and retirement decisions after concussion. PM R 2011;3(10 Suppl 2):S440-S444.
96. Schaider J, Barkin RM, Rosen P. Rosen & Barkin’s 5-minute Emergency Medicine Consult, 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2011.
97. Nagy KK, Joseph KT, Krosner SM, et al. The utility of head computed tomography after minimal head injury. J Trauma 1999;46(2):268-270.
98. Dunham CM, Coates S, Cooper C. Compelling evidence for discretionary brain computed tomographic imaging in those patients with mild cognitive impairment after blunt trauma. J Trauma 1996;41(4):679-686.
99. af Geijerstam JL, Britton M. Mild head injury: Reliability of early computed tomographic findings in triage for admission. Emerg Med J 2005;22(2):103-107.
100. Bazarian JJ, McClung J, Cheng YT, et al. Emergency department management of mild traumatic brain injury in the USA. Emerg Med J 2005;22(7):473-477.
101. Fung M, Willer B, Moreland D, et al. A proposal for an evidenced-based emergency department discharge form for mild traumatic brain injury. Brain Inj 2006;20(9):889-894.
102. CDC. What to expect after a concussion. 2008. http://www.cdc.gov/concussion/pdf/TBI_Patient_Instructions-a.pdf. Accessed 3/11/2013.
Over the past few years, we have learned that seemingly minor injuries can cause significant morbidity and, in some cases, even contribute to mortality. Astute emergency providers have learned to look beyond minor complaints and watch for signs of significant problems. Mild traumatic brain injury or concussion is one of the best examples of an injury that has been often dismissed. Post-concussive syndrome was overlooked for many years. Athletes in particular are prone to repeated injury, which we now know contributes to later cognitive impairment and even in some cases death.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
