Complications of Supplements and Herbal Medications
December 1, 2013
Reprints
Complications of Supplements and Herbal Medications
This article originally appeared in the Sept. 22, 2013, issue of Emergency Medicine Reports.
Executive Summary
Polypharmacy is a huge problem in U.S. health care and is compounded by the increasing use of supplements and herbal medications. Many patients don't even tell their doctors that they are taking these agents. Physicians can be unaware of the complications that the use of these agents can cause in their patients.
- Compared to pharmaceutical manufacturers, marketers of supplements and herbal medications are not required by the FDA to demonstrate that their products are safe or effective for their intended use.
- Doses of active ingredients can vary widely and can contain contaminants such as lead and mercury.
- The most commonly used supplements in the United States are fish oil, glucosamine, echinacea, flaxseed, and ginseng.
- Herbals can have significant potential for serious toxicity involving multiple organ systems such as gastrointestinal, cardiovascular, dermatologic, neurologic, and renal complications.
- The side effects of vitamins and minerals vary widely in significance but many of the hydrophilic vitamins, such as thiamin and pantothenic acid, are relatively non-toxic even in high doses. Selenium, however, has a complex role and overdoses have had serious side effects including death.
Introduction
Supplements and herbal medications have extensive worldwide use; in the United States alone, more than $5 billion was spent on herbal supplements in 2012.1 This number is almost certainly an underestimate, as at least one major retailer of herbal supplements is not counted in this statistic. Outside the United States, use of herbal and supplemental medications is even more widespread. In China, $14 billion was spent on herbal remedies in 2005.2 In parts of Asia and Africa, the vast majority of the population uses traditional medicine for primary health care.2 In Western Europe, use of herbal supplements is also widespread. Sixty-five percent of Germans have used herbal supplements, and the vast majority of German physicians regularly prescribe herbal supplements.3 Vitamins, minerals, and herbal supplements are frequently pharmacologically active and are taken for a broad range of conditions. Herbal and nutritional supplements can cause medical complications through direct toxicity and interaction with other drugs. Previously well-controlled medical conditions may also be exacerbated if an ineffective herbal or supplemental medication replaces an efficacious medical regimen. The complications of herbal and nutritional supplements can range from mildly irritating to life-threatening.
This article will describe the use and potential toxicity of supplemental and herbal medications. All of the applications for supplemental and herbal medications described in this paper should be considered "off label," as they have not been approved by the Food and Drug Administration (FDA) for the treatment or prevention of diseases.
Regulation of Herbal and Supplemental Medications
Herbal and supplemental medicines do not have the same regulatory oversight as drugs do in the United States. Drugs are regulated by the Food, Drug, and Cosmetic Act of 1938. In 1962, the Kefauver-Harris Drug Amendments expanded the FDA’s oversight of drugs.4 The Food, Drug, and Cosmetic Act of 1938 required manufacturers to show that a drug was safe and to include directions for safe use. Among other things, the 1962 amendments required that prior to marketing a drug in the United States, a manufacturer had to show evidence that it was effective for its intended use. The Kefauver-Harris Amendments also required the reporting of adverse reactions and minimum standards for the manufacture of drugs. Compared to pharmaceutical manufacturers, marketers of supplements and herbal medications are not required by the Food, Drug, and Cosmetic Act of 1938 or the Kefauver-Harris Drug Amendments to demonstrate that their products are safe or effective for their intended use.
The Food, Drug, and Cosmetic Act of 1938 and the Kefauver-Harris Drug Amendments both arose in the aftermath of drug tragedies. In 1937, Elixir Sulfanilamide killed 107 people in the United States.4 This product contained sulfanilamide that was dissolved in diethylene glycol. Both of these agents were relatively new, and the human toxicity of diethylene glycol was poorly understood. Elixir Sulfanilamide was marketed without prior safety testing. The Kefauver-Harris Drug Amendments were passed, in part, in response to the catastrophic birth defects caused by thalidomide.
In the 1990s, the FDA was granted a larger role in the regulation of herbal and supplemental medications.3 Efforts to increase the regulation of the herbal and supplemental medicine industry were strongly opposed by industry and consumers. Supplements and herbal medications are now regulated under the Dietary Supplement Health and Education Act of 1994 (DSHEA). This law amended the Federal Food, Drug, and Cosmetic Act to include oversight of supplements. Prior to that time, supplements were under the same regulatory requirements as foods. Under this act, the manufacturer of a supplement is responsible for making sure the product is safe, and empowers the FDA to take action against unsafe dietary supplements. Importantly, unless a supplement contains a new dietary ingredient, a dietary supplement does not require FDA approval prior to marketing; only new dietary ingredients require safety data prior to being marketed in the United States.
Supplements can be labeled with structure or function claims. These claims refer to general statements like "fiber maintains bowel regularity."5 The supplement must then contain the disclaimer that the statement has not been evaluated by the FDA. If a specific health claim is made with regard to treating nutritional deficiency, such as "vitamin D prevents rickets" or "niacin prevents pellagra," the label must also state how common the disease is in the United States.5 Manufacturers of supplements are responsible for the truthfulness of their claims. Supplement labels that make health claims also must carry the disclaimer that the product is not designed to "diagnose, treat, cure, or prevent any disease." The FDA can monitor reports of adverse reactions to supplements and take action to address dangerous supplements.
The FDA did take action against a supplement, L-tryptophan, an amino acid that was promoted to treat insomnia. In 1989, a cluster of patients taking L-tryptophan developed myalgias, weakness, shortness of breath, rash, and multiple laboratory abnormalities. The most striking of these abnormalities was a profoundly elevated eosinophil count. The newly described illness was called eosinophilia-myalgia syndrome. The FDA first issued a public advisory against using L-tryptophan supplements in November of 1989, and later effectively removed supplements containing manufactured L-tryptophan from the market. It is still not entirely clear if the eosinophilia-myalgia syndrome was caused by L-tryptophan itself or impurities in the L-tryptophan-containing supplements. Ultimately, 1,500 cases of eosinophilia-myalgia syndrome and 38 deaths were reported to the CDC.6
In the past, nutritional supplements often contained ingredients that were not listed on the label. Doses of active ingredients can vary wildly, and contaminants like lead and mercury may also be present in some preparations.3 Traditional Chinese and Indian remedies are particularly prone to this type of contamination. In one study, one in five ayurvedic (traditional Indian) herbal medicinal products imported from south Asia and purchased in Boston were found to be contaminated with mercury, lead, or arsenic.7 A California-based study of Chinese traditional medications showed that 32% of medications tested were contaminated with undeclared pharmaceutical agents or heavy metals.8 When considering the possible toxic effects of herbal medications and supplements, it is important to consider the possibility of contamination with heavy metals and other pharmacologic agents. Likewise, if unusual toxidromes are seen in patients, it is essential to ask about use of herbal medications and supplements.
Figure 1:
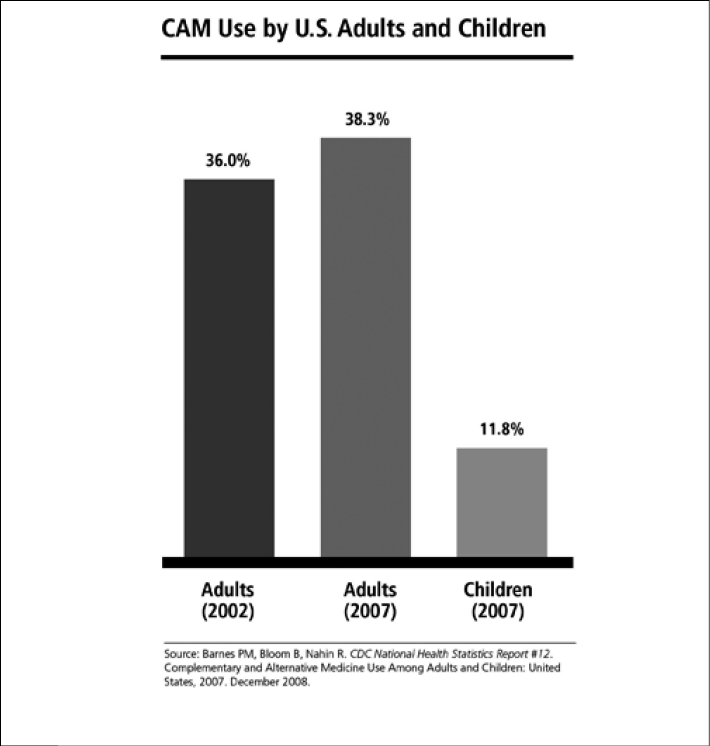
Solvents used in herbal remedies can also have significant toxicity. Tinctures or extracts of herbal medicines may contain ethyl alcohol. Patients taking medications like disulfiram and metronidazole can experience significant drug interactions if they inadvertently ingest a supplement or herbal product that contains ethyl alcohol. In spite of the tragic lesson of the Elixir Sulfanilamide poisonings, diethylene glycol has episodically emerged as a cause of mass poisonings. This has usually occurred in products produced outside of the United States.9
The FDA has taken steps to address the problem of adulterants in herbal and supplemental medications. In 2007, the FDA issued the Current Good Manufacturing Practices to ensure that supplement manufacturers have controls in place to ensure consistent processing and quality standards. These rules were phased in from 2008-2010.10 In addition to improving quality control in manufacturing, these rules require manufacturers of herbal medications and supplements to properly label supplements and not make misleading or false claims about their products.
The regulation of ephedra illustrates the FDA’s role in the oversight of herbal supplements. Ma huang (Ephedra sinica) and several other plants contain ephedrine.11 Ephedrine acts as a sympathomimetic drug by increasing the release of norepinephrine and epinephrine. This causes a wide range of physiologic effects, including bronchodilation, increased blood pressure, increased cardiac output, appetite suppression, and stimulation of the central nervous system. For these reasons, ma huang has long been used in Chinese medicine to treat asthma, allergies, and several other conditions. As an herbal supplement, ephedra was also marketed as an aid for weight loss, energy, and athletic performance. It was frequently mixed with caffeine and/or aspirin and marketed as a weight-loss and body-building supplement. In the late 1990s, a body of evidence emerged that use of ephedra-containing supplements could cause cardiovascular and neurologic complications. From 1995 to 1997, the FDA received more than 900 reports of adverse reactions to ephedra-containing products, including 37 cases of stroke, heart attack, and sudden death.11
Because ephedra was not a new product, the FDA did not have to approve it as an ingredient in herbal medications to be sold in the United States.5 However, when evidence emerged that ephedra was dangerous, the FDA had the authority to regulate ephedra-containing herbal supplements. In 1997, the FDA proposed that products containing ephedra should carry a warning, and that the supplement should be used for no more than seven days. As further evidence emerged, the FDA tightened regulations on ephedra-containing supplements. In 2003, the death of 23-year-old professional baseball player Joshua Bechler was linked to use of ephedra.12 In 2004, the FDA banned the sale of herbal supplements containing ephedra.11 In spite of this, the FDA seized ephedra alkaloids from supplement manufacturers as recently as 2011.
The upshot of this is that while FDA oversight of herbal and supplemental medications has gradually increased during the past 20 years, herbal and supplemental medications still do not have to meet the safety and efficacy standards of drugs, and data on toxicity, dosing, and efficacy are often sparse. Even with increased oversight and regulation, supplements may contain banned ingredients like ephedra. Traditional medications that are illicitly produced or imported may contain heavy metals, pharmaceutical agents, or other adulterants.
Figure 2:
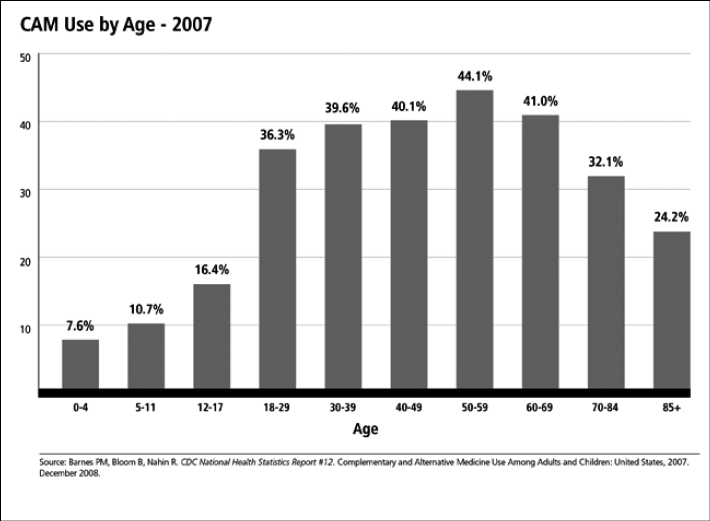
The most commonly used supplements are listed in descending order:
Supplements in Medicine
While large numbers of patients take supplements, they frequently do not disclose this to their doctors.3 The reasons for this vary, but they include a belief that their doctor does not support the use of supplements, a belief that natural drugs are benign and unlikely to interact with medications and health conditions, and a fear that their physician may ridicule them for using supplements. Frequently, patients do not discuss herbal and supplemental medications because they are not asked about use of these agents.
Supplements frequently contain pharmacologically active ingredients, so patients and providers alike need to communicate about the use of these products. Providers should ask in a non-judgmental fashion about the use of vitamins, minerals, and herbal supplements. If the patient does take a supplement, try to find out the dose of the pharmacologically active agents in the supplement. If the patient doesn’t know the name of a supplement or the ingredients of a supplement, encourage the patient to bring the container to future visits. Even if the provider advises against the use of a supplement, it is important to maintain a non-judgmental tone so that the patient will openly communicate with the provider their herbal and supplemental medication use. This information may help providers avoid drugs that interact with herbal medications and address complications of herbal and supplemental medications. Because the FDA’s safety role in the regulation of herbal and supplemental medications is frequently reactive, it is also important that providers document case reports of unusual reactions to herbal and supplemental medications.
There are several excellent information sources concerning the safety and efficacy of supplements. The FDA maintains databases of unsafe plants and information on the use of supplements. The National Center for Complementary and Alternative Medicine (NCCAM) is a branch of the National Institutes of Health that also maintains online resources about the safety and efficacy of herbal medications and supplements. Herbs at a Glance on the NCCAM web site is a database with quick links to basic information and references about herbal supplements.11 UpToDate.com has thorough and helpful articles about the toxicity of herbal medications and supplements. The Professional’s Handbook of Complementary and Alternative Medicines13 is another well-organized and useful resource. As with any other part of medicine, it is essential for health care providers to read about herbal and supplemental medications if patients are taking these agents or if these agents are thought to be causing or contributing to a patient’s illness. A basic understanding of the more important toxicities of herbal medications is essential to the practice of primary care medicine.
Herbal Supplements
The most commonly used supplements in the United States are listed in descending order in Table 1.14
| Table 1: Most Commonly Used Supplements in the United States14 |
|---|
|
In pure form, most of these supplements are relatively well-tolerated. Most supplements can cause gastrointestinal distress, and allergic reactions are possible with virtually all supplements. The combination of education and regulatory oversight has reduced the usage of toxic herbal supplements. The sale of dangerous herbal supplements has been restricted or banned in the United States. Likewise, regulation of manufacturing processes has reduced the likelihood that herbal medications are contaminated with heavy metals, infectious agents, pharmaceutical drugs, or other unintended agents. Use of St. John’s wort declined precipitously after reports of significant side effects and interactions with other medications were publicized.15 In spite of these developments, herbal supplements still cause a number of significant complications. These complications can span multiple organ systems, but are listed below by the primary or most significant toxicity.
Figure 3: CAM Use by Disease/Condition
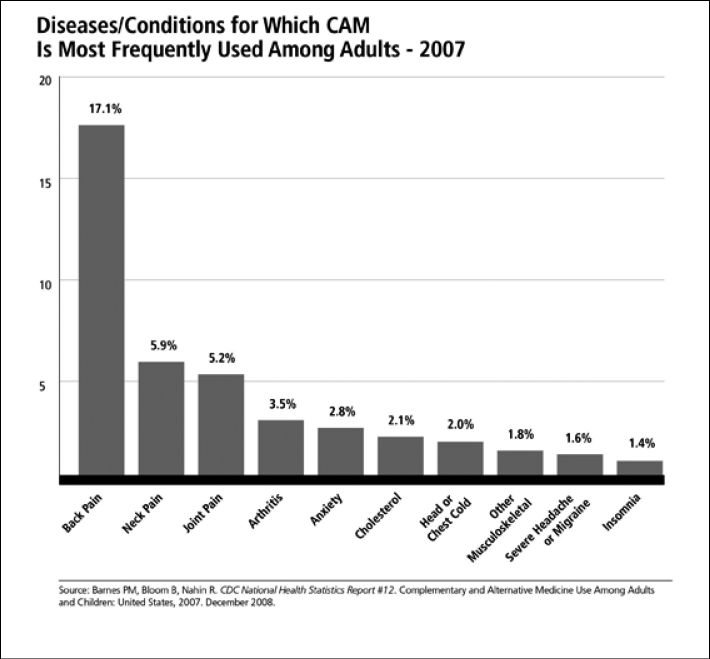
Gastrointestinal Toxicity
Liver toxicity is a common serious complication of herbal and supplemental medications.16 While data are relatively sparse, it is estimated that herbal remedies and supplements may play a role in 10-68% of cases of drug-induced liver injury (DILI). Tinctures and extracts may also contain ethyl alcohol, a well-known liver toxin. Many plants contain toxic compounds called pyrrolizidine alkaloids. These alkaloids can cause damage to the hepatic vein endothelium. This, in turn, leads to veno-occlusive disease, portal hypertension, and liver failure. Patients present acutely with jaundice, elevated liver function tests, ascites, and hepatomegaly. Chronic exposure presents with cirrhosis and portal hypertension. The timetable of the toxicity is highly variable, with death occurring between two weeks and two years after exposure. Several traditional herbal remedies contain these alkaloids, including comfrey, greater celandine, bush teas, germander, and other herbs.16
Comfrey (Symphytum officinale, Symphytum asperum, and Symphytum x uplandicum) is a particularly important herbal cause of hepatotoxicity. Comfrey has a long history of use, both for oral ingestion and topical use on wounds and injuries. Its long tradition of topical use is the reason it has been called knitbone and bruisewort.13 Traditionally, comfrey has been ingested as a tea to treat a wide range of gastrointestinal ailments. Ingestion of comfrey is not recommended, but comfrey is still used as a topical therapy. It is not clear if topical application of comfrey can result in liver injury. The toxicity of comfrey has caused it to be removed from the market in France.16 However, comfrey remains widely available, in part because it is commonly used as a fertilizer in organic gardening.
Butterbur (Petasites hybridus) root is used for the prevention of migraine headaches. It is also used for the treatment of allergic conjunctivitis, allergic rhinitis, and gastrointestinal disorders.13 The root contains pyrrolizidine alkaloids, but these alkaloids are removed in commercially available extracts. If a patient is ingesting butterbur root or improperly prepared butterbur extracts, it is possible that the patient may be exposed to pyrrolizidine alkaloids.11,13
The use of many other herbal products may cause liver injury. Chaparral, which is derived from the creosote bush, is used for a number of conditions, such as arthritis, bowel disorders, and cancer.13,16 It is especially popular among patients with HIV, and is thought to have a wide range of anti-infective properties. Patients with chaparral toxicity have elevated liver function tests and jaundice.16 Chaparral toxicity can progress to liver failure. It is essential to recognize chaparral hepatotoxicity because the herb is marketed as a liver tonic.
Kava kava, mistletoe, pennyroyal, skullcap, and Chinese green tea have all been implicated in cases of liver toxicity.16 Kava kava has been used in traditional Polynesian medicine for centuries. It has been used as a treatment for seizures, psychiatric conditions, pain, sexually transmitted diseases, and to aid wound healing. Drinks made from kava kava and other ingredients are used socially in Polynesian cultures.13 Kava kava contains a number of pharmacologically active compounds called kavalactones. Kavalactones act as local anesthetics and muscle relaxants. In humans, kava kava causes mild euphoria, analgesia, sedation, and hyporeflexia. Mistletoe has been used to treat cardiovascular disease, psychiatric illnesses, infertility, and many other conditions.13 It is also used as an aphrodisiac. Pennyroyal is used as a flavoring in some herbal teas. Pennyroyal can cause gastrointestinal upset and neurologic injury, and ingestion of less than 15 mL of pennyroyal oil can be fatal.16
Fiber supplements, flax seeds, and fish oil are widely used for a number of conditions. Flax seeds contain both fiber and polyunsaturated fatty acids.11 Fiber is used to treat and prevent constipation and diarrhea. Fiber products that contain soluble fiber can reduce serum cholesterol.17 Fish oil is used to treat hypercholesterolemia, especially hypertriglyceridemia.17 Fish oil is used for treatment of arthritis, psychiatric conditions, and the prevention of sudden cardiac death. Fish oil can cause nausea, flatulence, and esophageal reflux. Fiber can also cause flatulence and bezoars, if taken with an inadequate amount of water.
Cardiovascular and Hematologic Toxicities
Supplements and herbal medications can produce damage to the cardiovascular system. As mentioned above, ma huang was removed from the market because of cardiovascular toxicity. Ma huang was often replaced in herbal remedies by bitter orange (Citrus aurantium). Bitter orange contains synephrine, which is an adrenergic agonist. In addition to being an appetite suppressant, it can increase blood pressure, and there have been case reports of heart attack and stroke in patients taking bitter orange.11 Ma huang may still be illicitly added to supplements that are made in the United States, or in illegally imported supplements. Ephedra causes the release of epinephrine and norepinephrine, which act as both alpha and beta adrenergic agonists. This can cause a wide range of cardiovascular and neurologic complications, including hypertension, angina, myocardial infarction, sudden cardiac death, ischemic and hemorrhagic stroke, agitation, and delirium. Ephedrine is also available and regulated as a drug in asthma preparations such as Bronkaid® and Primatene® tablets.
A number of supplements are sold for the treatment of erectile dysfunction or under the vague rubric of "male enhancement." These supplements often contain L-arginine and a wide range of other herbs, vitamins, and minerals. L-arginine is thought to increase blood flow through the release of nitric oxide. More worrisome, a significant number of products for erectile dysfunction/male enhancement contain phosphodiesterase (PDE) inhibitors.18 While PDE inhibitors are generally safe and well-tolerated when taken appropriately, they can cause catastrophic reductions in blood pressure when co-administered with nitrates. The combination of nitrates and PDE inhibitors can be fatal. PDE inhibitors can also cause priapism and vision changes. Enzyte®, an herbal combination product marketed for male enhancement, has also been linked to hepatotoxicity.16 Nitrates should be used with caution in patients taking herbal supplements for erectile dysfunction and male enhancement.
Ginkgo biloba, garlic, ginseng, and ginger have all been linked to an increased risk of bleeding.19 The actual magnitude of this risk is not clear, but some experts advise stopping these medications prior to surgery to avoid excessive bleeding. These supplements should also be used with caution in combination with aspirin, warfarin, and other anti-coagulants. Ginkgo biloba may reduce the activity of monoamine oxidase. For this reason, it may contribute to serotonin syndrome. Ginkgo biloba has been used to treat or prevent memory loss, sexual dysfunction, dementia, and many other conditions. There is at least some evidence to support a role for ginkgo biloba in the treatment of a number of conditions.19 Garlic has been used to lower cholesterol, but it is not clear that it is effective in this role, or that it is effective in the prevention of cardiovascular disease.17 Ginger has been used to treat infections, arthritis, and several other conditions. Ginger is most frequently used as an anti-emetic, and it is at least modestly effective in that role.11,13
A number of plants carry the name ginseng. These include Asian ginseng (Panax ginseng), American ginseng (Panax quinquefolius), and Siberian ginseng (Eleutherococcus senticosus). Siberian ginseng is not a true ginseng. Ginseng has been used traditionally to support health and general well-being, and to treat conditions ranging from infectious diseases to diabetes and erectile dysfunction. It is not clear how effective ginseng is for these applications. In addition to a risk of bleeding and allergic reactions, ginseng may reduce serum glucose levels.11,13
Licorice (Glycyrrhiza glabra) and Chinese licorice (Glycyrrhiza uralensis) are herbal remedies that are most well known as candy flavoring. Licorice has been used to treat bronchitis, stomach ulcers, infections, and sore throat.11 In large doses, imported licorice can cause a syndrome similar to aldosteronism. This syndrome includes hypertension and hypokalemia. This can occur in patients who ingest licorice as a medication or in individuals who eat a large amount of candy containing licorice root. Hypokalemia caused by licorice ingestion can be exacerbated by loop diuretics and thiazide-type diuretics.11
Willow bark and other components of the willow tree have been used for pain, fever, and other ailments for several millennia. Willow contains salicylate-like compounds that have pharmacologic effects that are similar to other nonsteroidal anti-inflammatory (NSAID) medications.11 Willow bark and other willow products may have side effects that are similar to other medications, including gastrointestinal upset, gastritis, gastric ulcers, and gastrointestinal bleeding. Like other NSAIDs, willow products may interact with a wide range of other medications, and can cause fluid retention, tinnitus, renal injury, exacerbation of asthma, and allergic reactions. Salicylate concentrations vary widely in different parts of the willow tree.11
Indian snakeroot (Rauwolfia serpentina) has been used in India for more than 2,000 years. Rauwolfia has been used as a tranquilizer and as a treatment for hypertension, mental illnesses, and a number of other conditions. It contains several pharmacologically active alkaloids, including reserpine.13 Reserpine reduces blood pressure by depleting neurons of catecholamines and, thus, reducing sympathetic tone in the cardiovascular system. Reserpine has a wide range of side effects, including orthostatic hypotension, depression, and impotence. Reserpine has largely been replaced by newer anti-hypertensive medications with more acceptable side-effect profiles.
Red yeast rice has been used in Chinese medicine for more than 1,000 years. It is also a widely used ingredient in Asian cuisine. Red yeast rice contains a number of biologically active compounds, including lovastatin.17 The amount of lovastatin can vary wildly in different preparations of red yeast rice. Lovastatin is an effective and relatively safe drug for the treatment of hyperlipidemia. The other biologically active compounds in red yeast rice are not as well studied as lovastatin.17 Lovastatin is known to cause myalgias, myositis, and rhabdomyolysis. Patients with these conditions should be asked about use of lovastatin. Patients taking red rice yeast should also be counseled to avoid taking medications that are known to interact with lovastatin.
Allergic and Dermatologic Complications
Echinacea purpurea, or the purple coneflower, is used for the treatment of colds and other infections. It has also been used for wound healing, urinary tract infections, and the treatment of cancer.11,13,20 There is conflicting evidence about the efficacy of echinacea in the treatment of colds. Echinacea is generally well-tolerated, but it can cause allergic reactions. In one study, 7% of children receiving echinacea developed a rash, compared with 3% of children receiving a placebo.20
Bloodroot (Sanguinaria canadensis) is an herb that contains alkaloids that are highly toxic to human tissue.13 It is occasionally applied as a salve or solution to warts and skin cancers. It produces tissue necrosis and leads to eschar formation. In spite of claims to the contrary, bloodroot does not spare normal tissues. The efficacy of bloodroot in treating skin cancer is not well-established, and treatment with bloodroot can be disfiguring. Bloodroot is also used as an anti-plaque agent in herbal toothpastes and mouthwashes. Bloodroot has been marketed under the name "black salve," which has been promoted as a treatment of non-dermatologic conditions, including infections and internal malignancies. Bloodroot is listed as one of the FDA’s "187 Fake Cancer Cures’ Consumers Should Avoid."21
Neurologic and Renal Complications
St. John’s wort (Hypericum perforatum) has been used in clinical medicine for more than 1,000 years.15 It has primarily been used to treat mood disorders because of its ability to inhibit the re-uptake of serotonin into the presynaptic terminals, just like the selective serotonin re-uptake inhibitors, or SSRI agents. It is also thought to possess wound-healing and anti-infective properties.11,13,15 Studies of St. John’s wort have shown mixed results, but there is at least some evidence to support a role for St. John’s wort in the treatment of depression. St. John’s wort has a wide range of drug interactions, and it is a potent inducer of the cytochrome p-450 system. This induction increases the rate of metabolism of a wide range of pharmaceutical agents. Treatment failures with digoxin, oral contraceptives, anti-viral medications, and immunosuppressives have been observed in patients taking St. John’s wort. Photosensitivity and anorgasmia have also been reported in patients taking St. John’s wort.15
St. John’s wort is one of several herbal medications that can contribute to serotonin syndrome.22 Serotonin syndrome is a clinical diagnosis that should be suspected in patients taking one or more serotonergic agents. Symptoms include agitation, restlessness, tremor, confusion, and gastrointestinal upset. Signs of serotonin syndrome include hypertension, tachycardia, hyperthermia, muscle rigidity, and hyperreflexia. In severe cases, rhabdomyolysis, renal failure, and acute respiratory distress syndrome (ARDS) may occur.22 Treatment of serotonin syndrome involves discontinuation of serotonergic agents and supportive care. The details of treating serotonin syndrome are beyond the scope of this article. A wide range of drugs can contribute to serotonin syndrome, including multiple anti-depressants, ondansetron, cyclobenzaprine, meperidine, amphetamines, levodopa, cocaine, tramadol, dextromethorphan, buspirone, and multiple other medications. Herbal and supplemental medications that may also contribute to serotonin syndrome include L-trypotphan, Syrian rue, and gingko biloba.19,22
Jimsonweed (Datura stramonium) is a potent anti-cholinergic herbal medication. It has been used for asthma, Parkinsonism, cough, and muscle spasm. As with other anti-cholinergic medications, side effects limit the use of jimsonweed in these conditions. Jimsonweed is also consumed as a hallucinogen. Side effects of jimsonweed include dry mouth, urinary retention, dry, flushed skin, ataxia, delirium, hypertension, tachycardia, and other typical signs of anti-cholinergic poisoning. Seizures and death may occur with ingestion of about 50-100 seeds.13
Creatine is a supplement frequently taken by competitive athletes and body builders. Phosphocreatine is an important reservoir of energy in skeletal muscles, and is derived from creatine.23 Creatine may have some benefit for exercises that involve short bursts of strength. Creatine use is linked to weight gain and renal disease.24 Body-building and weight-loss supplements also frequently contain caffeine. Caffeine is present in coffee, tea, energy drinks, guarana, supplements, and pharmaceutical drugs. The side effects of caffeine include tremor, anxiety, panic attacks, agitation, gastrointestinal distress, and tachycardia.25 It may be advisable for patients with serious cardiac arrhythmias to avoid caffeine.26
Vitamins and Minerals
Vitamins are organic chemicals that are metabolic co-factors essential for human health. Minerals are elements that are essential for the function of the human body. In appropriate doses, vitamins and minerals are generally well-tolerated. The generally accepted dose of a vitamin that is sufficient for the general population is known as the recommended daily allowance (RDA). The RDA is the quantity of a vitamin or mineral that is adequate to prevent a deficiency state in 97-98% of the population.27 A number of vitamins and minerals are used to treat and prevent a broad range of conditions. These uses often extend well beyond treating or preventing the conditions directly caused by vitamin deficiency. In traditional medicine, high doses of vitamins are sometimes used. Niacin is used in doses that are often more than 100 times the RDA to treat hyperlipidemia. Vitamin A derivatives have been used in high doses to treat acne and acute myelogenous leukemia. Riboflavin is occasionally used in doses several hundred times above the RDA to prevent migraine headaches.28 In complementary and alternative medicine, several vitamins and minerals are given in doses that far exceed the RDA. High doses of selenium, zinc, vitamin C, vitamin E, and many other agents have been promoted for the treatment and prevention of disease.
The side effects of vitamins and minerals vary greatly in significance. Thiamin (vitamin B1), riboflavin (vitamin B2), pantothenic acid (vitamin B), and biotin are hydrophilic vitamins that are relatively non-toxic, even in high doses.29 Vitamin C is hydrophilic and can cause gastrointestinal distress in high doses. More significantly, large doses of vitamin C can cause false-negative fecal occult blood tests. Niacin (vitamin B3) is hydrophilic and causes a wide range of toxicities in the doses used to treat hyperlipidemia. The most common side effect is flushing, but less common side effects include pruritis, hyperglycemia, hyperuricemia, and hepatotoxicity. The hepatotoxicity can include massive hepatocellular necrosis. Pyridoxine (vitamin B6) is a hydrophilic vitamin that can cause nausea, neuropathy, and dermatologic problems when supra-therapeutic doses are taken.29 In one double-blind, placebo-controlled trial involving more than 3,000 patients, supratherapeutic doses of vitamin B6 resulted in a 17% increase in myocardial infarction.30
Vitamin B12 is a hydrophilic vitamin that is absorbed in the ileum when it is bound to intrinsic factor. Reduced stomach acid production, inadequate production of intrinsic factor, celiac disease, and parasitic infection can all cause vitamin B12 deficiency.31 Vitamin B12 is well-tolerated, even in supratherapeutic oral doses. Folic acid is a hydrophilic vitamin that is relatively well-tolerated in supratherapeutic doses. Folic acid supplementation can reverse the macrocytic anemia normally seen in B12 deficiency. This can delay the diagnosis of B12 deficiency, allowing neurologic complications to progress.
Vitamin A is a lipophilic vitamin essential for cell development and vision.32 The primary dietary source of vitamin A is the provitamin beta-carotene, an orange pigment found in a wide range of plants. In general, vitamin A is more toxic than beta-carotene. This is because the body regulates the conversion of beta-carotene to active vitamin A. Nonetheless, long-term beta-carotene use is not entirely benign. In two well-designed studies, long-term beta-carotene supplementation increased the risk of lung cancer in smokers.33 Large doses of beta-carotene can also cause carotenemia, an orange discoloration of the skin that may be mistaken for jaundice.
Excessive vitamin A ingestion can produce both acute and chronic toxicity. Vitamin A overdose can occur both by massive ingestion of supplements and ingestion of liver, which is rich in vitamin A. Acute vitamin A overdose can cause gastrointestinal distress, vertigo, and visual disturbances. Chronic excessive ingestion of vitamin A can lead to hepatotoxicity, pseudotumor cerebri, nausea, headache, bone pain, and multiple other symptoms.32
Naturally occurring vitamin K is a lipophilic vitamin that is generally well-tolerated, even in high doses.34 Vitamin K can antagonize the anti-thrombotic activity of warfarin. Patients taking warfarin should be counseled about vitamin K intake. If a patient who was previously therapeutic on warfarin has a sub-therapeutic international normalized ratio (INR), the patient should be queried about dietary and supplemental vitamin K intake.
Vitamin D is obtained both from dietary sources and exposure to sunlight. Vitamin D is often co-administered with calcium in products that are marketed for bone health. Very high doses of vitamin D can lead to hypercalcemia.35
Vitamin E functions as an anti-oxidant in cells. Vitamin E has been widely studied as a possible preventative agent for chronic diseases, including cancer and cardiovascular disease. In general, the results of these studies have been inconsistent.36 Vitamin E is generally well-tolerated, but some toxicity has been observed in very high doses. Doses of vitamin E in excess of 400 international units daily have been associated with increased mortality. Vitamin E, especially in high doses, seems to increase the risk of bleeding, including hemorrhagic stroke.33,36 Very large doses of vitamin E may also impair the absorption of other nutrients.
A wide range of minerals are essential for human health. The body is relatively good at regulating uptake of minerals, and within usual dosage ranges, minerals are well-tolerated. Mineral toxicity can arise from increased oral intake or from transdermal or respiratory exposures. Massive exposure to minerals can occur in industrial settings and is beyond the scope of this article. Likewise, metabolic conditions that result in mineral overload states such as Wilson’s disease (copper) and hemochromatosis (iron) are beyond the scope of this article.
Calcium has several important roles within the human body. Calcium is essential to the structure of bones, and acts as a signal in many cellular processes. Calcium-containing agents are often taken for bone health and the treatment of gastrointestinal conditions like reflux esophagitis. Calcium is generally well-tolerated, but can cause constipation. If taken in large doses along with a free base, calcium supplements can cause hypercalcemia and the milk-alkali syndrome.37 The free base can either be a component of the supplement (as in calcium carbonate) or a basic substance like sodium bicarbonate that is taken separately. In the absence of a free base, calcium absorption is generally well-regulated, and hypercalcemia is less likely.
With excessive vitamin D, hypercalcemia occurs due to mobilization of calcium from the bones and, thus, bone mineral density is reduced. In milk-alkali syndrome, the source of the additional calcium is dietary, and bone mineral density is unchanged. Hypercalcemia can lead to mental status changes, psychiatric disturbances, kidney stones, polyuria, metastatic calcification, and renal failure. Hypercalcemia is also frequently seen in malignancy, whether by direct tumor effects, as in multiple myeloma, or by paraneoplastic syndromes as in production of PTH-related protein in squamous cell lung cancer.37 Serious causes of hypercalcemia should be carefully considered before attributing hypercalcemia to supplement use.
Magnesium also plays a vital role in multiple cellular processes. Oral magnesium-containing agents are used to treat constipation, prevent migraine headaches, and treat hypomagnesemia.28 Intravenous magnesium sulfate is used to treat eclampsia, hypomagnesemia, and torsades de pointes or polymorphic ventricular tachycardia in Advanced Cardiac Life Support.38 Epsom salts are widely available and contain magnesium sulfate. Epsom salts are used both topically and internally. Diarrhea and gastrointestinal distress are the most common side effects of magnesium therapy. In the setting of renal failure, massive oral ingestion, or intravenous administration of magnesium, more serious toxicity can occur. Magnesium toxicity can cause loss of reflexes, confusion, sedation, hypotension, arrhythmias, respiratory depression, and death.39
Chromium, manganese, and zinc are generally well-tolerated, and overdose is unlikely.40 Very high doses of zinc can cause copper deficiency and gastrointestinal distress. Toxicities from chromium and manganese are generally seen in industrial exposures or intravenous administration, and not with oral supplementation.
Selenium is a mineral that is involved in the body’s free-radical scavenging glutathione system. Selenium is incorporated into an amino acid called selenocysteine. Selenocysteine is a vital component of the enzyme glutathione peroxidase, which is vital for the proper function of glutathione.23 Selenium is also present in a number of other proteins. Topical selenium compounds are used to treat seborrheic dermatitis, tinea versicolor, and dandruff.
Selenium has a complex role in human health.40,41 Low selenium levels are a risk factor for cancer, cardiomyopathy, and other conditions. In parts of China, very low levels of dietary selenium were linked to cardiomyopathy prior to a selenium supplementation program. The RDA for selenium ranges from 15 micrograms per day for infants to 70 micrograms per day for lactating women. Selenium doses as high as 3,000 micrograms per day have been advocated in the alternative medical literature for the treatment and prevention of disease. Selenium appears to have a relatively narrow therapeutic window. The tolerable upper intake level of selenium in adults is 400 micrograms per day. Brazil nuts contain a large amount of selenium, and consumption of as few as five nuts per day may result in dietary intake of selenium in excess of 400 micrograms per day.41 In 2008, a selenium product inadvertently contained 200 times the labeled amount of selenium. Among patients taking the product, the median dose of selenium was estimated to be more than 40,000 micrograms.42
Symptoms of selenium overdose include a garlic-like breath odor, brittle hair and nails, gastrointestinal distress, rash, fatigue, and anxiety. Massive selenium ingestion can cause severe neurologic, cardiovascular, gastrointestinal, dermatologic, and respiratory illness. Selenium overdose has been linked to ARDS, myocardial infarction, heart failure, and death.41
Summary
Primary care physicians will commonly encounter patients taking herbal and supplemental medications. Many of these supplements have significant pharmacologic effects and drug interactions. Physicians should ask patients about use of herbal and nutritional supplements in an open, non-judgmental fashion. Herbal and supplemental medications can cause complications by interacting with other drugs or by direct toxicity. Clinicians should be alert to the more common toxicities of herbal and supplemental medications, and should be comfortable accessing the medical literature when unsure of the pharmacology of a particular supplement. Supplements are neither useless placebos nor magically benign. Medical practitioners and patients alike need to understand and communicate the risks and benefits of herbal and supplemental medications.
References
- Shulz H. Herbal supplement sales rose 5.5% in US in 2012, ABC says. Nutraingredients-usa.com. http://www.nutraingredients-usa.com/Markets/Herbal-supplement-sales-rose-5.5-in-US-in-2012-ABC-says?nocount, accessed 8/23/2013. William Reed Business Media. Aug. 19, 2013.
- World Health Organization. Traditional medicine fact sheet. December 2008. http://www.who.int/mediacentre/factsheets/fs134/en/. Accessed 8/25/2013.
- Saper RB. Overview of herbal medicine and dietary supplements. UpToDate.com. Accessed 8/25/2013.
- Meadows M. Promoting safe and effective drugs for 100 years. FDA Consumer. January-February 2006. Food and Drug Administration web site. http://www.fda.gov/AboutFDA/WhatWeDo/History/CentennialofFDA/CentennialEditionofFDAConsumer/ucm093787.htm. Accessed 8/25/2013.
- Q&A on Dietary Supplements. Food and Drug Administration. http://www.fda.gov/Food/DietarySupplements/QADietarySupplements/default.htm#FDA_role. Accessed 8/25/2013.
- Staff College, Center for Drug Evaluation and Research, Food and Drug Administration. The clinical impact of adverse event reporting. October 1996. Food and Drug Administration. http://www.fda.gov/downloads/safety/medwatch/ucm168505.pdf. Clinical synopsis 3. Accessed 9/16/2013.
- Sasper RB, et al. Heavy metal content of ayurvedic herbal medicine products. JAMA 2004;292:2868.
- Ko RJ. Adulterants in Asian patent medicines. N Engl J Med 1998;339:847.
- Schep LJ, et al. Diethylene glycol poisoning. Clin Toxicol (Phila) 2009;47:525-535.
- Dietary supplement Current Good Manufacturing Practices (CGMPs) and Interim Final Rule (IFR) facts. Food and Drug Administration. June 22, 2007. http://www.fda.gov/Food/GuidanceRegulation/CGMP/ucm110858.htm. Accessed 8/26/2013.
- Herbs at a glance. National Center for Complementary and Alternative Medicine (NCCAM). National Institutes of Health. http://nccam.nih.gov/health/herbsataglance.htm. Accessed 9/16/2013.
- Bodley H. Medical examiner: Ephedra a factor in Bechler death. USA Today March 13, 2013. http://usatoday30.usatoday.com/sports/baseball/al/orioles/2003-03-13-bechler-exam_x.htm. Accessed 9/16/2013.
- Fetrow CW, Avila JR. Professional’s Handbook of Complementary and Alternative Medicines. Springhouse, PA: Springhouse Corporation; 1999.
- Barnes PM, et al. Complementary and alternative medicine use among adults and children: United States, 2007. National Health Statistics Reports. Dec. 10, 2008. http://nccam.nih.gov/sites/nccam.nih.gov/files/news/nhsr12.pdf. Accessed 8/26/2013.
- Saper RB. Clinical use of St. John’s wort. UpToDate.com. Accessed 9/16/2013.
- Larson AM. Hepatotoxicity due to herbal medications and dietary supplements. UpToDate.com. Accessed 9/16/2013.
- Tangney CC, Rosenson RS. Lipid lowering with diet or dietary supplements. UpToDate.com. Accessed 9/16/2013.
- Martin KA. Treatment of male sexual dysfunction. UpToDate.com. Accessed 9/16/2013.
- Saper RB. Clinical use of ginkgo biloba. UpToDate.com. Accessed 9/16/2013.
- Saper RB. Clinical use of echinacea. UpToDate.com. Accessed 9/17/13.
- Food and Drug Administration. 187 fake cancer "cures" consumers should avoid. http://www.fda.gov/drugs/guidancecomplianceregulatoryinformation/enforcementactivitiesbyfda/ucm171057.htm. Accessed 9/17/13.
- Boyer EW. Serotonin syndrome. UpToDate.com. Accessed 9/17/13.
- Lehninger AL, Nelson DL, Cox MM. Principles of Biochemistry. New York: Worth, 1993.
- Baron D. Non-hormonal performance enhancement. UpToDate.com. Accessed 9/17/13.
- Kabagambe EK, Wellons MF. Benefits and risks of caffeine and caffeinated beverages. UpToDate.com. Accessed 9/17/13.
- Giardina EG. Cardiovascular effects of caffeine. UpToDate.com. Accessed 9/17/13.
- Nutrient recommendations: Dietary reference intakes (DRI). Office of Dietary Supplements. National Institutes of Health. http://ods.od.nih.gov/Health_Information/Dietary_Reference_Intakes.aspx. Accessed 9/17/13.
- Bajwa ZH, Sabahat A. Preventative treatment of migraine in adults. UpToDate.com. Accessed 9/17/13.
- Pazirandeh S, Lo CW, Burns DL. Overview of water-soluble vitamins. UpToDate.com. Accessed 9/17/13.
- Bonaa KH, et al. Homocysteine lowering and cardiovascular events after acute myocardial infarction. N Engl J Med 2006;354:1578-1588.
- Schrier SL. Etiology and clinical manifestations of vitamin B12 and folate deficiency. UpToDate.com. Accessed 9/17/13.
- Pazirandeh S, Burns DL. Overview of vitamin A. UpToDate.com. Accessed 9/17/13.
- Nutrient recommendations: Dietary reference intakes (DRI). Office of Dietary Supplements. National Institutes of Health. http://ods.od.nih.gov/Health_Information/Dietary_Reference_Intakes.aspx. Accessed 9/17/13.
- Pazirandeh S, Burns DL. Overview of vitamin K. UpToDate.com. Accessed 9/17/13.
- Pazirandeh S, Burns DL. Overview of vitamin D. UpToDate.com. Accessed 9/17/13.
- Pazirandeh S, Burns DL. Overview of vitamin E. UpToDate.com. Accessed 9/17/13.
- Shane E. Etiology of hypercalcemia. UpToDate.com. Accessed 9/17/13.
- Yu ASL, Gupta A. Causes and treatment of hypermagnesemia. UpToDate.com. Accessed 9/17/13.
- Agus ZS. Symptoms of hypermagnesemia. UpToDate.com. Accessed 9/17/13.
- Pazirandeh S, et al. Overview of dietary trace minerals. UpToDate.com. Accessed 9/17/13.
- Dietary Supplement Fact Sheet: Selenium. Office of Dietary Supplements. National Institutes of Health. http://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/#en64. Accessed 9/17/13.
- MacFarguhar JK, et al. Acute selenium toxicity associated with a dietary supplement. Arch Intern Med 2010;170:256-261.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
