Skin and Soft-tissue Infections
Diagnostic Studies
Wound Cultures. Skin infection with purulent drainage should be cultured if antibiotics will be used. Culture is becoming increasingly critical in this era of CA-MRSA as resistance increases to antibiotic agents, including clindamycin.9,13 Needle aspiration or a swab of purulent drainage should be sent for culture in high-risk patients or situations in which the diagnosis is unclear to guide antibiotic management. Aspiration of cellulitis has a low yield, but should be considered if the infection has not responded to appropriate empiric treatment or the patient has significant underlying medical conditions.4 Aspirate taken from the area of greatest inflammation rather than the leading edge provides a better yield.
Blood Work. Laboratory testing is not routinely indicated for SSTIs; however, testing may be indicated if bacteremia is suspected, as in the case of an ill-appearing child. Blood culture should be performed whenever bacteremia is suspected. CBC and inflammatory markers (ESR and CRP) may be helpful when complicated infection is suspected, for example deep tissue infection, septic arthritis, or osteomyelitis. An elevated white blood cell count (WBC) with left shift in a febrile child has some predictive value for bacteremia.18 Cellulitis of the face (e.g., periorbital or buccal) has a high rate of associated bacteremia.
Imaging. Ultrasound has been shown to be a useful tool in detecting drainable fluid collections. In a recent study, bedside ultrasound showed a higher sensitivity and specificity for detecting a drainable fluid collection than clinical judgment alone.19 (See Figures 1 and 2.) Ultrasound can also help to identify foreign bodies, which might not be visible upon radiography. (See Figures 3 and 4.) Ultrasound may also be used to evaluate lymph nodes and assist with early diagnosis of lymphadenitis and recognition of an abscess within a lymph node. (See Figure 5.)
Plain radiographs are not routinely indicated. Plain radiographs cannot exclude deep infections such as fasciitis, and their use should not delay surgical evaluation and antibiotic treatment if deep tissue infection is suspected on physical exam.
Figure 1. Cellulitis
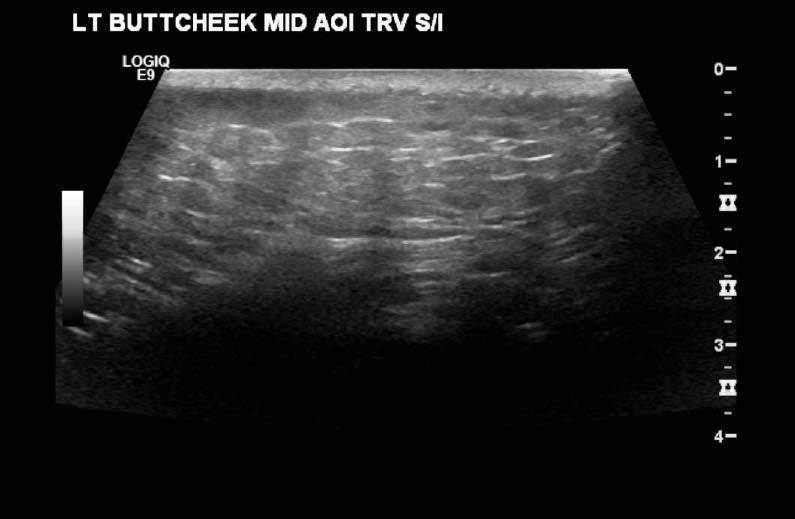
Figure 2. Thigh Abscess

Figure 3. Abscess Foreign Body
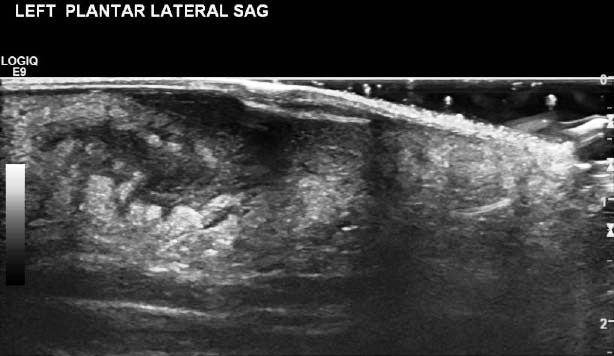
Figure 4. Foreign Body Foot
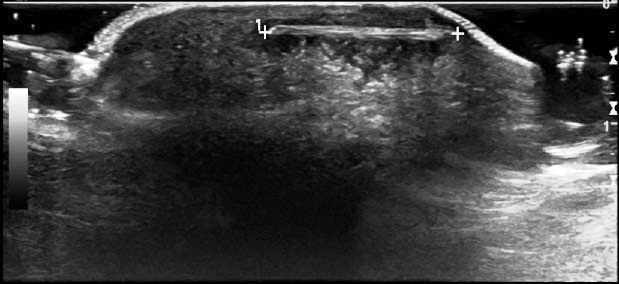
Figure 4. Foreign Body Foot
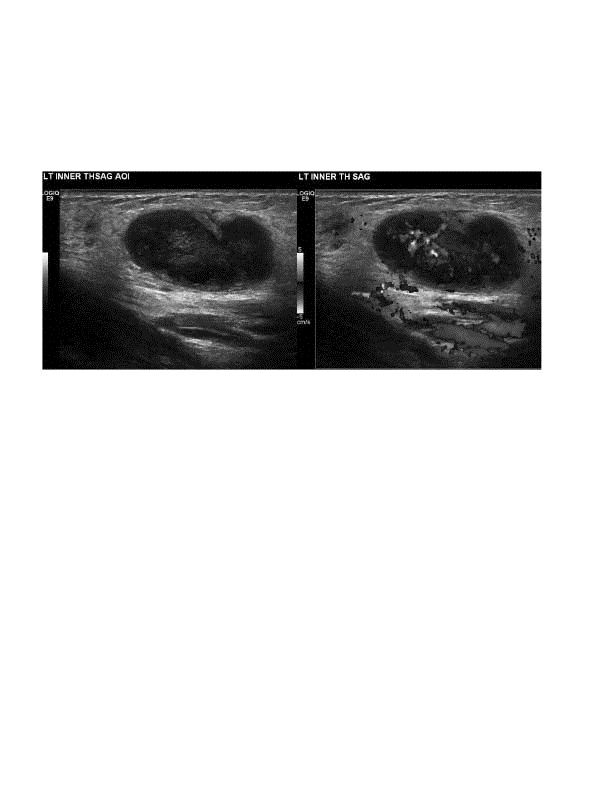
|
Oral Antibiotics |
Dose (mg/kg/day) |
Adult/max dose (per day) |
Frequency |
MSSA |
MRSA |
Group A Strep |
|
++ good coverage + some resistance reported, prevalence may vary by community ¨C no coverage * Recommended for treatment of infection associated with human or animal bite wounds. Covers Eikenella and anaerobes. Red Book p. 206. **Trimethoprim-sulfamethoxazole is not FDA approved for treatment of Staphylococcus aureus infections. |
||||||
|
Clindamycin |
30-40 |
0.9-1.8 g |
Q8 hours |
+ |
+ |
++ res 1-2% (Red Book p. 676) |
|
Doxycycline (age ≤ 8 years) |
2-4 |
200 mg |
Q12-q day |
+ |
+ |
¨C |
|
Trimethoprim-Sulfamethoxazole** |
8-12 tmp |
320 mg tmp |
Q12 hours |
++ |
++ |
- |
|
Dicloxacillin |
12-25 |
0.5-1 g |
Q6 hours |
++ |
¨C |
++ |
|
Cephalexin |
25-50 |
1-2 g |
Q6 or q12 hours |
++ |
¨C |
++ |
|
Amoxicillin-clavulanate (7:1)* |
25-45 amox |
1750 mg |
Q12 |
++ |
¨C |
++ |
Management
Folliculitis can be treated with warm compresses applied several times a day. If folliculitis fails to resolve spontaneously or with application of warm compresses and attention to proper hygiene, topical antibiotics should be used. For non-pseudomonal folliculitis, use topical mupirocin 2% TID for 7 to 10 days. A small furuncle can also be adequately treated by application of warm compresses to promote drainage.20
Impetigo, both bullous and non-bullous, can be treated with topical antibiotics. Topical antibiotics improve cure rates over skin cleansing. Uncomplicated impetigo should be treated with topical mupirocin 2% TID for 7 to 10 days.21 In the case of extensive impetigo, or if underlying cellulitis is present, oral antibiotics should be given.
Cellulitis and erysipelas infections can typically be treated with outpatient oral antibiotics. Erysipelas can be treated with penicillin or amoxicillin; however, if the infection cannot be distinguished from cellulitis, the antibiotic choice should also cover Staphylococcus aureus.
Non-purulent cellulitis is frequently caused by beta-hemolytic Streptococcus. Treatment should include coverage for beta-hemolytic Streptococcus and MSSA, such as with a first-generation cephalosporin (cephalexin). Empiric treatment for CA-MRSA should be considered as well, as many cases of cellulitis are now caused by CA-MRSA.4 Clindamycin is a good first-line choice for cellulitis due to adequate coverage of all common pathogens: MRSA, MSSA, and beta-hemolytic Streptococcus. In geographic areas with a low prevalence of CA-MRSA, cephalexin alone may still be an effective course of treatment.
Purulent cellulitis should be treated for suspected MRSA with clindamycin or trimethoprim-sulfamethoxazole (TMP-SMX); coverage for beta-hemolytic Streptococcus is not necessary.15 Treatment for 7 to 10 days is usually adequate.
Paronychia. Small superficial infections can be treated with warm compresses. Incision and drainage and oral antibiotics should be used for deep lesions/paronychial abscesses. Amoxicillin/clavulanic acid or clindamycin should be used for coverage of anaerobic oral flora.21
Abscess. Primary treatment for a skin/soft-tissue abscess is incision and drainage.15 Controversy exists over the use of packing and oral antibiotics after incision and drainage15. In adults, a randomized trial has shown that incision and drainage without packing does not increase the risk of treatment failure.22
Only one small randomized trial of packing versus no packing has been conducted in children. It concluded that packing is not necessary in superficial skin abscesses.23
Several recent pediatric studies have evaluated the use of antibiotics for abscesses in addition to incision and drainage. In adults, studies have shown that most abscesses will resolve without antibiotic treatment,3,24,25 although retrospective studies did show a small benefit of antibiotic therapy.26,27 Antibiotics may prevent the development of new lesions in the short term.25 In children, use of TMP-SMX prevented development of new lesions but did not improve outcomes for abscesses treated by incision and drainage.16 Two other pediatric studies saw favorable results with incision and drainage, even when the prescribed antibiotic was not effective against the isolated organism.28,29
Antibiotics are indicated if an abscess cannot be adequately drained, multiple concurrent lesions are present, significant cellulitis is present (e.g., > 5 cm), or when the child is febrile.15 Antibiotics should also be considered for children younger than 1 year of age; there have been higher treatment failure rates without antibiotic use.28 A 7- to 10-day course is standard, although a 5-day course has been effective in adults.15
Incision and drainage of an abscess in a child may require sedation. After application of a topical anesthetic and infiltration with lidocaine, the abscess should be incised with a #11 blade along the length of the abscess following skin lines. Smaller incisions can be used for cosmetic reasons. Use a hemostat to break up loculations, then irrigate with saline. If warranted, pack the abscess. Contraindications to incision and drainage include abscesses in the middle third of the face (due to the risk for septic thrombosis) or paronychia associated with herpetic whitlow.18
A potential alternative to incision is the application of anesthetic cream to aid in abscess drainage. A significant number of abscesses will spontaneously drain after topical application of lidocaine cream covered with an occlusive dressing.30 In a trial of this method, several patients did not require further intervention after spontaneous abscess drainage, and fewer patients required sedation. However, this study did not look at treatment failure rates.30 Further study is needed to determine if expression of a spontaneously draining abscess is comparable to incision and drainage.
Oral Antibiotics. Penicillinase-resistant penicillins (e.g., dicloxacillin) and first-generation cephalosporins (cephalexin) cover S. pyogenes and MSSA for impetigo and cellulitis; however, they should not be used as a first-line agent for SSTI if the community prevalence of MRSA is significant. Dosing for either medication in 25 mg/kg/day PO divided q6 hours.20
Clindamycin has been studied in children and is effective for treatment of SSTI31,32 and invasive staphylococcal infections.15 If community resistance to clindamycin is low, clindamycin is a good first-line agent for cellulitis, with good coverage of Streptococcus and most MRSA and MSSA. Resistance is increasing, however.9,13 Soft-tissue and abscess penetration is excellent. Oral dosing for clindamycin is 30 mg/kg/day divided TID.15 One limitation in the use of clindamycin is its poor palatability.33 The most common side effect is diarrhea, and clindamycin has been associated with development of Clostridium difficile colitis.34
TMP-SMX is an appealing option for treatment of MRSA-related SSTIs, as nearly all isolates are susceptible in vitro. TMP-SMX is not FDA approved for treatment of S. aureus infections; however, it has been used successfully in adults for cellulitis and other SSTIs.27,35,36 Its efficacy in clinical practice is not well established in children. A retrospective study showed slightly higher failure rates for TMP-SMX-treated SSTI than clindamycin.37 TMP-SMX should not be used alone for non-purulent cellulitis, as it lacks streptococcal coverage, but may be used in combination with a beta-lactam antibiotic. Common side effects include nausea. Although rare, Stevens Johnson syndrome can occur with TMP-SMX use. Patients with G6PD deficiency should not be prescribed TMP-SMX.
Tetracyclines such as doxycycline are effective for uncomplicated SSTIs. Doxycycline can be used to treat SSTIs caused by CA-MRSA, although inducible resistance has been reported. Tetracyclines should not be used in children younger than the age of 8 years due to the possibility of tooth enamel discoloration.
Linezolid is effective for treating MRSA SSTIs, but is rarely used as a first-line agent due to its high cost. Oral availability is 100%. Dosing is 600 mg PO BID for patients who are 12 years of age or older, or 30 mg/kg/day divided q8 hours for patients younger than 12 years of age.15
Hygiene. Patients should be instructed on proper hygiene measures to prevent the spread of MRSA and other infections. Infected draining areas should be kept covered with clean dressings, and hand washing after contact with the infected area should be encouraged. To prevent spread among members of a household, patients should not share items such as towels, and surfaces in the home that contact skin frequently should be cleaned. Prophylactic antibiotics are not indicated for family members; antibiotics should only be used for an active infection.15
Complications
Bacteremia. Aside from facial cellulitis with Streptococcus pneumoniae or Haemophilus influenzae, which are almost always associated with bacteremia, the incidence of bacteremia secondary to cellulitis or cutaneous abscess is low. Fasciitis is often associated with bacteremia.
Lymphatic Involvement. SSTIs, particularly cellulitis, can spread via lymphatics. Lymphangitis is recognized by streaking erythema from the area of infection, and often progresses rapidly. Infection of local lymph nodes can occur without evidence of lymphangitis. Treatment is with IV antibiotics.
Deep Tissue Infections. Cellulitis can progress to osteomyelitis, septic arthritis, and necrotizing fasciitis.21 Necrotizing soft-tissue infection is characterized by rapid progression of infection with tissue necrosis, and can involve subcutaneous tissue and superficial fascia, or deep fascia and muscle. Immediate surgical intervention and antibiotic therapy is necessary to control the infection and remove necrotic tissue. Immunocompromised children are at higher risk for necrotizing infections; however, these infections can also develop in normal hosts following skin trauma or varicella infection.
Venous Thrombosis. Thrombophlebitis can complicate cellulitis, particularly if other risk factors for thrombosis are present. A rare complication of SSTI of the central face is cavernous sinus thrombosis, which presents with orbital pain, proptosis, and oculomotor nerve palsies.
Toxic Shock Syndrome/Toxin-mediated Disease. Staphylococcus and Streptococcus can both produce exotoxins, which can cause a spectrum of illness ranging from rash alone to shock. Patients are usually febrile, have a diffuse erythrodermal rash, and may have multi-organ system dysfunction/failure, including circulatory collapse. Often the focus of infection is not identified; however, toxic shock syndrome can result from an identified SSTI. Treatment is with antibiotics and supportive therapy in an appropriate inpatient setting.
Post-streptococcal Glomerulo-nephritis. Certain strains of Streptococcus pyogenes can lead to glomerulonephritis, which presents an average of 2-3 weeks after skin infection. Antibiotics do not prevent post-streptococcal glomerulonephritis. Skin infection with S. pyogenes does not cause rheumatic fever.
Disposition
Indications for Admission. Most infections can be managed as an outpatient unless bacteremia or deep tissue infection is suspected. Children younger than 1 year of age or with a compromised immune system should be considered for admission unless treating a superficial skin infection.18 Signs of infection warranting admission include an ill- or toxic-appearing child, fever greater than 39°C with leukocytosis or extensive infection, and facial cellulitis with fever.18 Lymphangitis or rapid spread of infection are also indications for admission.
Parenteral antibiotic selection depends on the extent of infection. If bacteremia is suspected, vancomycin should be used. For children without bacteremia, appropriate choices include clindamycin 40 mg/kg/day divided q6-8 hours or linezolid 600 mg IV/PO BID for patients 12 years of age or older, or 30 mg/kg/day divided q8 hours for patients younger than 12 years of age.
Neonates. Neonates require hospitalization and IV antibiotics for most SSTIs. Omphalitis and mastitis can occur in the first few weeks of life. Antibiotic therapy should cover S. aureus, S. pyogenes, and group B streptococci for neonatal SSTIs.
Assessing Therapeutic Response. Follow-up at approximately 48 hours is appropriate to assess the response to initial therapy. If incision and drainage was performed, packing should be removed and replaced if necessary. Persistent fluid collections warrant drainage, and antibiotics should be considered. Most abscesses heal within 1-2 weeks. SSTIs such as cellulitis treated with appropriate antimicrobial therapy should show signs of improvement within 24-48 hours. If cellulitis has not improved or has progressed, the antibiotic choice should be re-evaluated. If an antibiotic lacking activity against CA-MRSA was used initially, it should be changed to an antibiotic effective against CA-MRSA, such as clindamycin, TMP-SMX, or linezolid. Culture data and susceptibilities should be used to select an appropriate antibiotic whenever available.
Prognosis
Uncomplicated SSTIs in children have a good prognosis. However, recurrent infections are problematic, particularly in children colonized with CA-MRSA. Decolonization regimens involving dilute bleach baths and nasal mupirocin have been shown to have benefits for some patients with recurrent MRSA infections.15
Summary
Skin and soft-tissue infections are common in children. Diagnosis can typically be made on physical exam alone. Primary treatment includes topical antibiotics for superficial infection, oral antibiotic for cellulitis, and incision and drainage for abscesses, resulting in resolution without sequelae in the majority of cases.
References
1. Edelsberg J, Taneja C, Zervos M, et al. Trends in US hospital admissions for skin and soft tissue infections. Emerging Infectious Diseases 2009;15(9):1516.
2. Qualls ML, Mooney MM, Camargo CA, et al. Emergency department visit rates for abscess versus other skin infections during the emergence of community-associated methicillin-resistant Staphylococcus aureus, 1997¨C2007. Clinical Infectious Diseases. 2012;55(1):103-105.
3. Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006;355(7):666-674.
4. Wylie FP, Kaplan SL, Mason EO, Allen CH. Needle aspiration for the etiologic diagnosis of children with cellulitis in the era of community-acquired methicillin-resistant Staphylococcus aureus. Clinical Pediatrics 2011;50(6):503-507.
5. Magilner D, Byerly MM, Cline DM. The prevalence of community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) in skin abscesses presenting to the pediatric emergency department. NC Med J 2008;69(5):351-354.
6. Huang JT, Abrams M, Tlougan B, Rademaker A, Paller AS. Treatment of Staphylococcus aureus colonization in atopic dermatitis decreases disease severity. Pediatrics 2009;123(5):e808-e814.
7. Fritz SA, Epplin EK, Garbutt J, Storch GA. Skin infection in children colonized with community-associated methicillin-resistant Staphylococcus aureus. Journal of Infection 2009;59(6):394-401.
8. Faden H, Lesse AJ, Trask J, et al. Importance of colonization site in the current epidemic of staphylococcal skin abscesses. Pediatrics 2010;125(3):e618-e624.
9. Kaplan SL, Hulten KG, Gonzalez BE, et al. Three-year surveillance of community-acquired Staphylococcus aureus infections in children. Clinical Infectious Diseases 2005;40(12):1785-1791.
10. Centers for Disease Control and Prevention. S. aureus and MRSA Surveillance Summary. http://www.cdc.gov/mrsa/statistics/MRSA-Surveillance-Summary.html. Accessed 3/3/2013.
11. Johnson PN, Rapp RP, Nelson CT, Butler J, Overman S, Kuhn RJ. Characterization of community-acquired Staphylococcus aureus infections in children. Ann Pharmacotherapy 2007;41(9):1361-1367.
12. Hasty MB, Klasner A, Kness S, et al. Cutaneous community-associated methicillin-resistant Staphylococcus aureus among all skin and soft-tissue infections in two geographically distant pediatric emergency departments. Academic Emergency Medicine 2007;14(1):35-40.
13. Pickett A, Wilkinson M, Menoch M, Snell J, Yniguez R, Bulloch B. Changing incidence of methicillin-resistant Staphylococcus aureus skin abscesses in a pediatric emergency department. Pediatric Emergency Care 2009;25(12):831.
14. Jeng A, Beheshti M, Li J, Nathan R. The Role of beta-hemolytic Streptococci in causing diffuse, nonculturable cellulitis: A prospective investigation. Medicine 2010;89(4):217-226.
15. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clinical Infectious Diseases 2011;52(3):e18-e55.
16. Duong M, Markwell S, Peter J, Barenkamp S. Randomized, controlled trial of antibiotics in the management of community-acquired skin abscesses in the pediatric patient. Annals of Emergency Medicine 2010;55(5):401-407.
17. Giovanni JE, Dowd MD, Kennedy C, Michael JG. Interexaminer agreement in physical examination for children with suspected soft tissue abscesses. Pediatric Emergency Care 2011;27(6):475-478.
18. Fleisher GR, Ludwig S. Textbook of Pediatric Emergency Medicine. Lippincott Williams & Wilkins; 2010.
19. Sivitz AB, Lam SHF, Ramirez-Schrempp D, Valente JH, Nagdev AD. Effect of bedside ultrasound on management of pediatric soft-tissue infection. J Emerg Med 2010;39(5):637-643.
20. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clinical Infectious Diseases 2005;41(10):1373-1406.
21. Long SS, Pickering LK, Prober CG. Principles and Practice of Pediatric Infectious Diseases, 3rd ed. Churchill Livingstone; 2009.
22. O’Malley GF, Dominici P, Giraldo P, et al. Routine packing of simple cutaneous abscesses is painful and probably unnecessary. Academic Emergency Medicine 2009;16(5):470-473.
23. Kessler DO, Krantz A, Mojica M. Randomized trial comparing wound packing to no wound packing following incision and drainage of superficial skin abscesses in the pediatric emergency department. Pediatric Emergency Care 2012;28(6):514-517.
24. Rajendran PM, Young D, Maurer T, et al. Randomized, double-blind, placebo-controlled trial of cephalexin for treatment of uncomplicated skin abscesses in a population at risk for community-acquired methicillin-resistant Staphylococcus aureus infection. Antimicrobial Agents and Chemotherapy 2007;51(11):4044-4048.
25. Schmitz GR, Bruner D, Pitotti R, et al. Randomized controlled trial of trimethoprim-sulfamethoxazole for uncomplicated skin abscesses in patients at risk for community-associated methicillin-resistant Staphylococcus aureus infection. Annals of Emergency Medicine 2010;56(3):283-287.
26. Ruhe JJ, Smith N, Bradsher RW, Menon A. Community-onset methicillin-resistant Staphylococcus aureus skin and soft-tissue infections: Impact of antimicrobial therapy on outcome. Clinical Infectious Diseases 2007;44(6):777-784.
27. Szumowski JD, Cohen DE, Kanaya F, Mayer KH. Treatment and outcomes of infections by methicillin-resistant Staphylococcus aureus at an ambulatory clinic. Antimicrobial Agents and Chemotherapy 2007;51(2):423-428.
28. Chen AE, Carroll KC, Diener-West M, et al. Randomized controlled trial of cephalexin versus clindamycin for uncomplicated pediatric skin infections. Pediatrics 2011;127(3):e573-e580.
29. Lee MC, Rios A, Aten MF, et al. Management and outcome of children with skin and soft tissue abscesses caused by community-acquired methicillin-resistant Staphylococcus aureus. The Pediatric Infectious Disease Journal 2004;23(2):123-127.
30. Cassidy-Smith T, Mistry RD, Russo CJ, et al. Topical anesthetic cream is associated with spontaneous cutaneous abscess drainage in children. The American Journal of Emergency Medicine 2012;30(1):104-109.
31. Hyun DY, Mason EO, Forbes A, Kaplan SL. Trimethoprim-sulfamethoxazole or clindamycin for treatment of community-acquired methicillin-resistant Staphylococcus aureus skin and soft tissue infections. The Pediatric Infectious Disease Journal 2009;28(1):57-59.
32. Purcell K, Fergie J. Epidemic of community-acquired methicillin-resistant Staphylococcus aureus infections: A 14-year study at Driscoll Children’s Hospital. Archives of Pediatrics & Adolescent Medicine 2005;159(10):980.
33. Steele RW, Russo TM, Thomas MP. Adherence issues related to the selection of antistaphylococcal or antifungal antibiotic suspensions for children. Clinical Pediatrics 2006;45(3):245-250.
34. Raveh D, Rabinowitz B, Breuer GS, Rudensky B, Yinnon AM. Risk factors for Clostridium difficile toxin-positive nosocomial diarrhoea. International Journal of Antimicrobial Agents 2006;28(3):231-237.
35. Khawcharoenporn T, Tice A. Empiric outpatient therapy with trimethoprim-sulfamethoxazole, cephalexin, or clindamycin for cellulitis. The American Journal of Medicine 2010;123(10):942-950.
36. Cenizal MJ, Skiest D, Luber S, et al. Prospective randomized trial of empiric therapy with trimethoprim-sulfamethoxazole or doxycycline for outpatient skin and soft tissue infections in an area of high prevalence of methicillin-resistant Staphylococcus aureus. Antimicrobial Agents and Chemotherapy 2007;51(7):2628-2630.
37. Williams DJ, Cooper WO, Kaltenbach LA, et al. Comparative effectiveness of antibiotic treatment strategies for pediatric skin and soft-tissue infections. Pediatrics 2011;128(3):e479-e487.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
