IRB protocols, workloads rise in 2012
IRB protocols, workloads rise in 2012
2013 will be another busy year
The last 12 months have been packed for IRB members. Respondents to IRB Advisor’s annual salary survey report that their workloads and working hours are on the rise.
When asked how much workloads have increased, 75% saw a jump in 2012, up 5% from 2011. Forty-two percent say that they work 41-45 hours per week — a 12% increase from 2011. Nearly 6% reported working 65 or more hours weekly.
The salary survey also found:
- Most report working in a hospital or academic program — 47% and 49%, respectively.
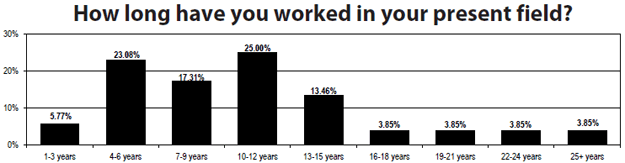
- A majority of respondents report 4-12 years in the field.
- Sixty-nine percent report no change in staff, while 27% say staff was lost.
- Almost 45% have graduate degrees.
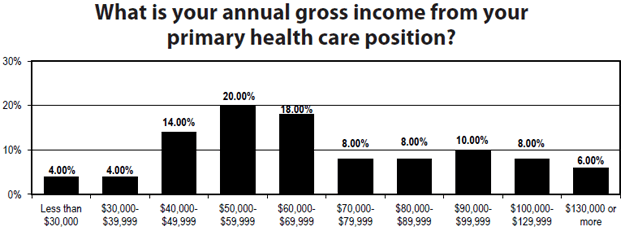
- Twenty percent make between $50,000-$59,000, 18% between $60,000-$69,000, and 10% between $90,000-$99,000.
- Eighty-eight percent of respondents were female, most age 46-55.
“Overall, the survey shows that there are a lot of people with quite a few years of experience in the field — and there are more people with graduate degrees,” says Mark Schreiner, MD, chairman of the committee for protections of human subjects at Children’s Hospital of Philadelphia (CHOP), and member of the IRB Advisor editorial board.
Schreiner is also seeing younger people beginning to emerge in IRBs. “A lot of attendees at national meetings are younger people,” he says. “About 80% of our IRB office is made up of people in their late 20s or early 30s.”
The CHOP IRB is also seeing a growing workload, with a 10% increase in expedited protocol reviews and 3.1% increase in full board reviews last year, continuing the trend of the past 4 or 5 years.
Another busy year
The next 12 months may not be any easier. Schreiner believes the coming year will see greater emphasis on cooperative IRB agreements for multicenter studies and issues related to advances in genomic research.
“One of the things we’re starting to see is a greater movement toward cooperative agreements for multicenter studies,” Schreiner says. Funding agencies, he says, are one of the factors behind the shift. “The NIH [National Institutes of Health] seems to be driving this. For several research consortium proposals, our investigators have had to agree in principle to participate with a central IRB in order to be eligible for the grant.”
One program in which could serve as a model is the NIH-funded IRBshare, a new shared review model for multisite studies. Institutions use shared review documents and review process under one centralized master agreement and Web portal.
Another driving factor for a shift to multisite studies is to save time and get things going quickly. “Sites can get through the IRB process in as little as a month if they’re using a central or fast academic IRB and they have a good contracting department, or it could take as long as 6-9 months or more at other sites,” he says. “Nine months is an awful lot of wasted time. I think that if everybody agrees to have a cooperative IRB agreement, we could shorten the time and reduce wasted effort.”
There are pros and cons to having a single IRB approve a multicenter study. “A lot of times, one IRB will raise an issue that was missed at other sites,” Schreiner says. “If we think a study is a flawed, we need to decide if we are not going to participate, or just accept the flaw. However, when a central IRB finds a problem, it can insist on changes to a flawed study and it is more likely that the study will be fixed.”
Hot topic: genome research
The 2013 research spotlight will be on areas related to the advances in genomics, Schreiner predicts. “I think the big discussion will be about genomics and safeguards for genetic information and,” he says. Debate will be swirling around confidentiality issues and also trying to define what constitutes a clinically significant result.
“What is a clinically significant result? Is it actionable, and will it help somebody? Will it help someone to know they have a 10% increase in getting a rare cancer by age 80? Researchers and IRBs will have to decide which results could benefit participants’ lives, and which make them worse. I think that this is going to be one of the hot area in research ethics.”
Confidentiality concerns
Confidentiality of data will also be a big issue in genome research, and clinical research in general, Schreiner says. Concerns will center around what information will be clinically useful and what should be shared — and is a debate, he says, that requires perspective. “The cost of whole-genome sequencing is rapidly dropping and we need to define whether this information requires special protections or not. In the age of Facebook and Twitter, people share all kinds of things. We have given up tons of privacy already and we need to understand what research participants want rather than what we think they want.”
The last 12 months have been packed for IRB members. Respondents to IRB Advisors annual salary survey report that their workloads and working hours are on the rise.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
