Preventing ICU Infections: An Effective Application of An Old Public Health Strategy
Preventing ICU Infections: An Effective Application of An Old Public Health Strategy
Abstract & Commentary
By Michael Young, MD, Pulmonary and Critical Care, Wake Forest University Health Sciences Medical Center, Winston-Salem, NC. Dr. Young reports no financial relationships relevant to this field of study.
Synopsis: Despite better compliance with hand hygiene and screening, use of isolation, and other techniques, ICUs remain notorious breeding grounds for hospital-acquired infections. A universal decolonization strategy reduces the total number of ICU bloodborne infections.
Source: Huang SS, et al. Targeted versus universal decolonization to prevent ICU infection. N Engl J Med 2013;368:2255-2265.
Introduction
In the 1840s, the Hungarian physician Ignaz Semmelweis demonstrated that maternal mortality was reduced by 90% when physicians washed their hands with a chlorinated solution prior to doing pelvic exams on women in labor. The physician community at that time was harshly skeptical of Semmelweis’s observations and recommendations. Women of the time were aware that something was lethal about having babies in the hospital. Some women in the 1840s preferred having their babies literally in the street vs the high-mortality obstetrical wards in teaching hospitals. A century and a half later, hand hygiene is widely accepted as a technique to reduce transmission of infections among hospitalized patients. Uniform compliance with recommended hygiene practices remains disappointing.
About 100 years after Semmelweis’s discovery that hand hygiene really matters, ICUs came into existence. It made sense to clinicians to cluster the sickest patients in one area of the hospital to more conveniently apply new technologies such as mechanical ventilation, central venous lines, and the rapidly evolving skill sets of clinicians who focused on managing critically ill patients. Unfortunately, those life-preserving technologies and the ICU population of patients and their clinicians became reservoirs of methicillin-resistant Staphylococcus aureus (MRSA) as early as the 1960s. By 2005, it was estimated that hospitals see more than 18,000 deaths/year from MRSA — more deaths than those reported from AIDS and breast cancer combined. The ICU was no small contributor to the rising death toll from MRSA infections.
In the past two decades, a number of strategies have come into use to prevent the spread of MRSA and other infectious agents in the ICU. The effectiveness of some of these strategies, such as screening patients for MRSA, isolation, contact precautions, and decolonization, has been uncertain. The study by Huang and colleagues provides clinicians, nurses, and infection control specialists much needed guidance.
Abstract
The authors conducted the study in 43 Hospital Corporation of America hospitals, including 74 ICUs. A 12-month baseline period was followed by an 18-month intervention period in which participating hospitals were randomized, using a stratified technique, to one of three infection control groups. In group 1, > 90% of ICU patients received screening and isolation if their nasal swab was positive for MRSA. Contact precautions were used among patients who screened positive for MRSA. In group 2 (“targeted decolonization”), MRSA screening and isolation occurred as in group 1. MRSA-positive patients underwent a 5-day decolonization process. In group 3 (“universal decolonization”), patients were not screened on admission to the ICU. All ICU patients in group 3 received twice-daily intra-nasal mupirocin for 4 days, plus daily baths with chlorhexidine-soaked cloths during the patients’ entire ICU stay.
Adherence to the infection control interventions in each of the three groups was encouraged by monthly educational teleconferences and use of on-site hospital personnel. Infections/pathogens were defined using Centers for Disease Control and Prevention (CDC) criteria. Compliance rates of 85% were cited.
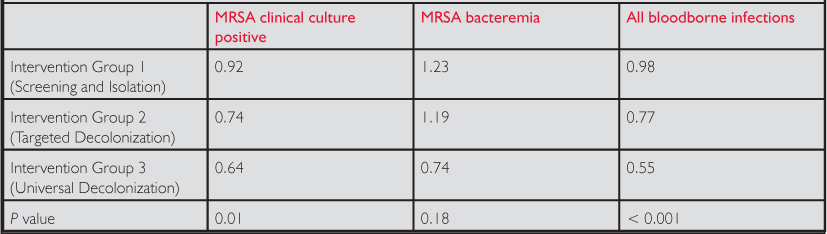
There were nearly 100,000 patients enrolled in each of the three groups and the groups had largely similar baseline demographic characteristics. MRSA-positive isolates and the risk of developing a bloodborne pathogen were significantly reduced by universal decolonization. With universal decolonization, the number needed to treat to prevent one bloodstream infection was 54 ICU patients. The rate of blood-borne infections from MRSA was not reduced by either targeted or universal precautions (see Table.)
Table. Adjusted Hazard Ratios for Major Outcomes
Commentary
Limitations of the study include lack of an explicit and detailed protocol for each of the three groups. In addition, it remains uncertain if routine use of chlorhexidine and mupirocin will lead to resistance. Severity of illness on ICU admission was not reported. Most of the hospitals included in the study were community hospitals, so it remains unknown if similar results will be seen in large teaching hospitals. It is also unclear why a decrease in MRSA colonization rates, the primary outcome, was not associated with a decrease in MRSA bacteremia.
Study strengths are considerable. Implementing universal decolonization was undertaken with existing personnel without apparent increased costs. The sample size was substantial. Randomization ensured similar baseline characteristics. The size of the reduction in MRSA colonization and positive blood cultures was both statistically and clinically significant. Finally, use of universal decolonization techniques in ICUs is becoming a more common practice.
Should this study change standard infection control measures in our ICUs? I believe that the answer to this question is an emphatic yes. As this study demonstrates, isolation and contact precautions lack effectiveness at preventing infection, and they also reduce clinician bedside presence. Arguably, it is intensivist bedside presence that is at least partly responsible for the improved patient outcomes seen with the intensivist “high intensity” model. In addition, from the use of “bundles” in the ICU over the past 10 years, such as those used for ventilator-associated pneumonia, central line infection reduction, and early resuscitation for sepsis, we have learned that ICU outcomes dramatically improve when a practice becomes universal and applied to all rather than applied selectively or simply at physician discretion.
Will many or all ICUs make the change to universal decolonization soon? This seems less certain, but my forecast is largely positive for the following reasons. We recognize that changes in hospital infection control practices involve complicated organizational and cultural shifts. However, physicians and hospitals change practice patterns over a period of months rather than over years if certain forces align, including: a convincing level of medical evidence; minimal cost to implementing the intervention; the intervention makes “life” easier for the clinicians; and authoritative groups such as the Society of Critical Care Medicine, the American Thoracic Society, and the CDC endorse the change in practice. Finally, the “stick” used by regulatory bodies such as the Centers for Medicare & Medicaid Services can provide powerful incentives to accelerate change in practice.
In summary, this is the time for us to develop a new infection control bundle in the ICU that includes universal decolonization. If Semmelweis were alive today, he would surely agree with this approach. On the other hand, he might be distraught to learn that in 2013 we are still struggling to achieve better compliance with hand hygiene.
Despite better compliance with hand hygiene and screening, use of isolation, and other techniques, ICUs remain notorious breeding grounds for hospital-acquired infections. A universal decolonization strategy reduces the total number of ICU bloodborne infections.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
