High Altitude Illness
High Altitude Illness
Author: Jonathan Glauser, MD, Department of Emergency Medicine, MetroHealth Medical Center, Cleveland, OH.
Peer Reviewer: Michael Caudell, MD, FACP, FAWM, Professor of Emergency Medicine, Medical Director, Wilderness and Survival Medicine, Georgia Regents University, Augusta, GA.
Introduction
Clinical syndromes due to altitude can manifest in susceptible individuals with elevations as low as 1500 meters (5000 feet) above sea level. For otherwise healthy adults, altitudes of 2350 meters (8000 feet) are considered the arbitrary cutoff for placing one at risk for more serious syndromes, such as acute mountain sickness (AMS). While decreased ambient oxygen pressure is the most evident change at high altitude and presumably is responsible for most of the pathophysiologic derangements, other factors may have medical implications as well, such as decreased ambient temperature, increased exposure to ultraviolet radiation, and lower humidity.
People travel to high altitudes for religious, business, military, and recreational reasons. It has been estimated that 30 million people travel to altitude in the western United States each year that places them at risk for altitude-related illnesses.1 An estimated 250,000 people annually visit the summit of Pike’s Peak (4302 meters [14,114 feet]) in Colorado. Astronomers stationed at several telescopes located at the summit of Mauna Kea in Hawaii (4205 meters or 13,796 feet) would develop an arterial PO2 of about 54 mmHg if they underwent full acclimatization.2 This is comparable to a patient with chronic obstructive pulmonary disease (COPD) at sea level who is entitled to continuous oxygen therapy with an arterial PO2 of less than 55 mmHg.
In 2003, it was estimated that 140 million people worldwide, including about 30 million people in the Andes of South America, live permanently above 2500 meters (8200 feet).3 Soldiers in the Indian army have been posted at altitudes of more than 6000 meters (19,685 feet) for months at a time. Between 30,000-50,000 workers worked on the railroad constructed to Lhasa, Tibet, at an altitude of approximately 3380 meters (11,000 feet). At the most extreme, the inspired PO2 on the summit of Mount Everest is less than 30% of that at sea level.2 (See Table 1.) Clearly, altitude illness is not restricted to a handful of mountain climbers or recreational skiers.
Table 1: Sample Locations, Altitudes, and PIO22,3,5,6,82
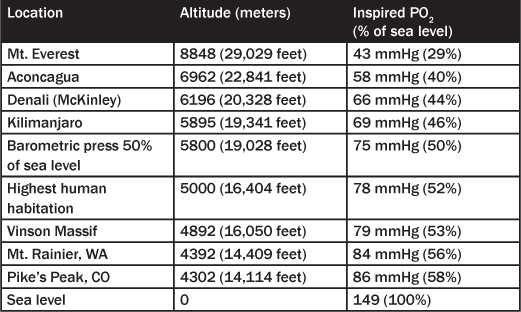
With increasing numbers of reasons for people to travel to high altitudes for pleasure or for business, physicians may encounter more travelers with preexisting conditions or who are taking a variety of medications. Even moderate altitudes may lead to splenic infarctions in healthy athletes with sickle cell trait.4
High altitude illness (HAI) refers to the cerebral and pulmonary syndromes that are specific to exposure to high elevations. The cerebral abnormalities include AMS and high altitude cerebral edema (HACE). The pulmonary syndrome is high altitude pulmonary edema (HAPE). Acclimatization may forestall some problems related to altitude, while some problems are idiosyncratic to the individual and, therefore, not predictable.
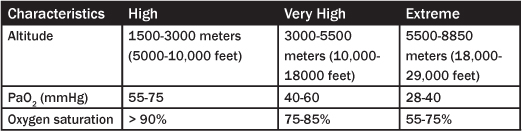
Exposure to extreme altitude (above 5500 meters or about 18,000 feet) (see Table 2) is characterized by hypoxemia, alkalosis, and deterioration of physiologic function. No permanent human habitation occurs at these altitudes. However, exceptional tolerance to the stress of high altitude for short periods of time is possible. For example, it was not expected that the summit of Mount Everest could be reached without supplemental oxygen, yet this occurred in May 1978.3
Table 2: High Altitude Categories
Physiologic Changes with Altitude
The physiologic response to high altitude is influenced by a variety of factors; the primary ones are the rate of ascent, the altitude attained, individual physiology, and the altitude at which the affected person slept night before.
The partial pressure of inspired air (PIO2) drives diffusion of oxygen from inspired air into the blood.
PIO2 = FIO2 x (Pb- 47 mmHg), where Pb is the barometric pressure and 47 mmHg is the partial pressure of water at 37° C. The concentration of oxygen remains constant at 21% (more precisely, 20.94%) regardless of altitude, extending into the upper troposphere, which includes all altitudes that may be attained standing on the earth’s surface. As the barometric pressure (Pb) declines with altitude, the partial pressure of all inhaled gases such as nitrogen or oxygen declines proportionately. Diminished PIO2 at altitude is the result of lower barometric pressure and results in hypobaric hypoxia.
At sea level, the barometric pressure is 760 mmHg, so the ambient PO2 is approximately 159 mmHg. At the summit of Mount Everest, the Pb is 252.7, making the ambient PO2 only 53 mmHg, similar to breathing 6% oxygen at sea level. The degree of hypoxia and hyperventilation at this altitude has produced some extraordinary blood gas values measured on the summit of Everest: PaCO2 of 7-8 mmHg, PaO2 of 35 mmHg, and a consequent pH of more than 7.70 from the respiratory alkalosis.2
The ambient barometric pressure is not simply a matter of altitude. Locations near the equator have relatively higher barometric pressures because solar radiation causes upwelling of the atmosphere. Most of the high-altitude areas frequented for recreational purposes, including the Andes, the Himalayas, and Kilimanjaro, therefore, have higher barometric pressures than otherwise predicted by standard atmosphere and altitude.2 Climbers in locations further from the equator, in locations such as Denali in Alaska or Vinson Massif in Antarctica, therefore, would be expected to breathe a lower PIO2 than corresponding altitudes in the tropics.
Special Populations. The physiologic changes with altitude can induce complications in patients with underlying pulmonary, cardiovascular and hematologic disorders. (See Table 3.) Pregnant patients with hypertension or preeclampsia should not travel to high altitude, especially after 20 weeks gestation.5 Patients with sickle cell disease are at increased risk for splenic infarcts and painful crises at altitude greater than 1500 meters (4920 feet).6 Any condition associated with pulmonary hypertension is a contraindication to high altitude exposure.
Table 3: Problems Potentially Exacerbated by High Altitude

Acclimatization
With ascent, progressive tissue hypoxia occurs and ambient pressure falls. Compensatory responses to hypobaric hypoxia occur over minutes to weeks, and are collectively termed acclimatization. This process involves multiple organ systems that interact to optimize delivery of oxygen at the cellular level. There is marked variation between individuals regarding adaptation to hypoxia. Some of the variation may be genetic, and some may be related to rate of ascent and can be modified by use of alcohol or of prescribed medications.
The process of acclimatization begins within minutes of ascent, but may take several weeks to complete. Observations indicate that both limiting the elevation gained each day and periods of rest facilitates acclimatization, reducing the incidence of altitude sickness.
Increased alveolar ventilation is the most important compensatory mechanism in improving oxygen delivery at altitude, and the response begins at altitudes of approximately 1500 meters. Peripheral chemoreceptors in the carotid and aortic bodies detect hypoxia and stimulate the hypoxic ventilatory response (HVR).9 The HVR increases in sensitivity over several days spent at altitude. At the same time, the PCO2 level at which ventilation is stimulated is lowered, resulting in a reduced threshold for the hypercapneic ventilatory response (HCVR). The result of both processes is increased ventilatory response at high altitude. Minute ventilation increases, and alveolar PCO2 is reduced, increasing alveolar oxygen concentration. Ventilation is maximized after several days to one week at high altitude and results in a chronic respiratory alkalosis.
The HVR may be sensitive to caffeine and acetazolamide, and depressed by alcohol or sedative-hypnotics.10,11 Progesterone as a respiratory stimulant may increase HVR.12
To partially offset the respiratory alkalosis, increased renal excretion of bicarbonate over 1 to 2 days results in partial metabolic compensation.
Increased hemoglobin concentration occurs with acclimatization. Initially, hemoglobin concentration increases due to decreased plasma volume, but within hours, there is increased erythropoietin production in the kidneys. While erythropoietin secretion is enhanced within two hours of ascent, new red blood cells are not produced in increased quantities for 4-5 days.13 It takes several days to develop a polycythemia that is detectable, and the process is not complete for several weeks.2
Pathophysiology of High Altitude Illness
The cause of AMS is postulated to be related to hypoxia-induced cerebral vasodilatation, with production of nitric oxide producing the headache. The early symptoms of AMS may be due to mild cerebral edema. Variation in clinical symptoms between individuals may be due to a relationship between brain volume and cerebrospinal fluid (CSF) volume; those with greater CSF volume might better compensate for brain swelling.7 Also, there is evidence for a genetic predisposition to AMS, presumably due to polymorphism of dozens of suspect genes.8
High altitude pulmonary edema (HAPE) is a non-cardiogenic pulmonary edema associated with elevated pulmonary artery pressure. The pathogenesis of HAPE is complex and involves the interaction of increased sympathetic activity over-expression of vasoconstrictors such as endothelin-1 and thromboxane B2, or underexpression of vasodilators such as nitric oxide. Individuals who have previously experienced HAPE are more likely to have a recurrence, so HAPE may have a genetic basis.8 For example, visitors to altitude who contract HAPE have lower NO levels than their healthy counterparts.14
Other factors, including cold ambient temperature, which induces pulmonary vasoconstriction and increases pulmonary artery pressure, may predispose to HAPE. Mild pulmonary hypertension may be worsened with exercise at altitude to the point that pulmonary pressure may approach that of systemic blood pressure.13 Along with exaggerated pulmonary hypertension, defective alveolar fluid clearance may be important as well.15
Acute Mountain Sickness
AMS entails a constellation of symptoms, all nonspecific: headache, nausea, lightheadedness, dizziness, or insomnia. (See Table 4.) The incidence of AMS brought on by rapid ascent to altitudes of 2000-2500 meters (6500-8200 feet) is approximately 25%.16-18 AMS is common in mountainous ski areas, as the skiers typically stay at lodging altitudes of 2000-3000 meters (6500-10,000 feet) and then exercise at altitudes higher than 3500 meters (11,500 feet) without acclimatization. With increasing rapidity of ascent and higher altitude, the incidence of AMS rises to approximately 60% at 4550 meters (14900 feet) in people not previously exposed to altitude within the preceding three months.19 About 85% of travelers who fly directly to an altitude of 3740 meters (12270 feet) in the Himalayas develop AMS.20 The symptoms of AMS typically occur within 6-12 hours of gaining altitude. A useful clinical observation is that a rapid rise in respiratory rate within one hour of ascent is likely to herald severe AMS.21
Table 4: Lake Louise Criteria for Mountain Illness22,23,83

The Lake Louise Consensus Group has defined AMS as the presence of headache in an unacclimatized person, along with the following: recent arrival at an altitude of greater than 2500 meters (8200 feet); and a combination of nausea, vomiting, anorexia, insomnia, lassitude, fatigue, or subjective dizziness.22 (See Table 4.) There are no specific diagnostic physical findings. Although AMS and HACE are often considered a continuum, an important distinction is that AMS does not entail ataxia or encephalopathy.
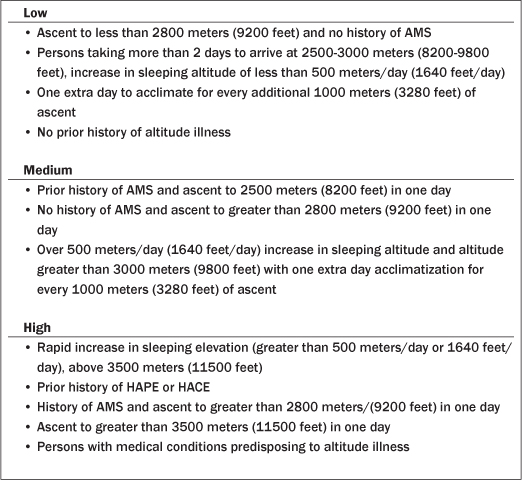
Risk factors for AMS include residing at or near sea level prior to ascent, a prior history of AMS, and vigorous exertion before acclimatization. (See Table 5.) As noted, the rate of ascent is proportional to the rate of development of AMS. Obesity may be a risk factor for AMS.13 Poor physical fitness, cardiovascular disease, and chronic pulmonary disease do not seem to confer any increased risk.7 People older than the age of 50 appear to be less susceptible to AMS than younger persons.23 Pregnancy, hypertension, coronary artery disease, and diabetes do not appear to increase the likelihood of high altitude illness.7 Prior history of migraine or desaturation at exercise appear to confer increased risk for altitude illness,24 as does any entity causing pulmonary hypertension.
Table 5: Relative Risks for Developing AMS27
Patients with coronary artery disease can go to altitudes of 3000-3500 meters (9800-11500 feet) safely so long as they have stable disease, preserved left ventricular function, and good exercise capacity. It has been recommended that people with coronary heart disease avoid altitudes above 4500 meters (14800 feet) due to severe hypoxia at that altitude.25 Patients with pre-existing pulmonary hypertension may risk further elevations in pulmonary artery pressure during sojourns at high altitude. It has been proposed that people whose systolic pulmonary artery pressure is greater than 50 mmHg or whose mean pulmonary artery pressure is greater than 35 mmHg should avoid travel to altitudes higher than 2000 meters (6500 feet).26
Prevention and Treatment of Acute Mountain Sickness
Adequate acclimatization via controlled ascent remains the most important factor in the prevention of altitude sickness. Wilderness Medical Society consensus guidelines recommend that sleeping elevation increase be limited to no more than 500 meters (1600 feet)per day above an altitude of 3000 meters (9800 feet).27 If practical, one rest day for every one to two ascent days may be incorporated as well. An extra day of acclimatization — during which activity or hiking may occur without gain in sleeping altitude — or rest may be added for every 600-1200 meters (2000-4000 feet) of altitude increase.7 These guidelines are developed to minimize the risk of AMS in reasonably health travelers, but despite adherence to recommendations for altitude gain and sleeping, illness can develop.
Impaired sleep is common at higher altitudes. For patients with underlying sleep disturbances who go to altitude during the day, it is recommended to descend about 1000 meters (3300 feet) prior to sleep, consistent with the adage “climb high, sleep low.”
There are instances in which gradual ascent is impossible, as with military teams or rescue personnel who may have to ascend rapidly to altitudes greater than 3500 meters (11500 feet). In this case, pharmacologic prophylaxis is appropriate to prevent AMS.
Early studies on prevention of AMS and how to improve tolerance to hypoxia emphasized aviation and pilot performance. Factors such as diet were investigated for their influence in altering adaptation to high altitude. A high carbohydrate diet, for example, was shown to decrease symptoms of AMS in one study,28 but not in another.29
Current recommendations for prophylaxis of AMS with medication include acetazolamide, dexamethasone, or both together (see below for doses).30 Ginkgo biloba has been reported in small studies to be effective in the prevention of AMS,31,32 but its use has not been sufficiently studied to recommend its use. Other prophylactic medications reported to be effective in preventing AMS include salmeterol, nifedipine, and phosphodiesterase inhibitors such as sildenafil and tadalafil.33 A recent study indicated that ibuprofen alone was superior to placebo in preventing AMS.34 An earlier study had concluded that ibuprofen was non-inferior to acetazolamide in its ability to prevent high-altitude headache.35 In view of the prominence of headache in diagnosing AMS, it may be that NSAIDs such as ibuprofen merely mask the headache of AMA while other pathophysiology continues rather than preventling AMS altogether.
Management of AMS does not necessarily include descent. However, further ascent should be avoided until symptoms have resolved. Any symptoms of HACE mandate descent and oxygen therapy.
High Altitude Cerebral Edema (HACE)
High-altitude cerebral edema is a clinical diagnosis characterized by ataxia, altered consciousness, or both in someone at altitude. An affected individual may have symptoms of AMS and can have HAPE as well. Hypoxemia, especially in the presence of HAPE, may promote the progression from AMS to HACE.36 Clinical findings in HACE may include papilledema, retinal hemorrhage, global encephalopathy, hallucinations, confusion, vomiting, mood changes, hemiparesis, and cranial nerve palsy.9 Encephalopathy may manifest as stupor or drowsiness, possibly with focal neurologic findings, seldom as seizures, with death due to brain herniation.37 Computerized tomography may show compression of sulci with white matter attenuation. Magnetic resonance imaging will show changes consistent with vasogenic cerebral edema, with diffusion-weighted imaging often showing the most striking abnormalities.3
Ataxia is a key finding in HACE, which is presumably related to hypoxia affecting the basal ganglia and hindbrain. Ataxia is frequently cited as a differentiator between AMS and HACE.
Ataxia is assessed by the heel-to-toe tandem walk.38 In addition to gait ataxia, a positive Romberg test evaluating vision, truncal coordination, and proprioception is a useful diagnostic test in HACE.
There is some evidence by MRI that all people have some degree of brain swelling on ascent to high altitude, including those with only clinical AMS.13,39,40 Why some patients progress to HACE is unknown.
With HACE, descent is imperative and the top priority in treatment. The patient may only need to descend 150-300 meters (500-1000 feet) for significant relief. If urgent descent is not possible, a portable hyperbaric chamber at a pressure of 2 psi can mimic a descent that is dependent on the ambient altitude. For example, at 4250 meters (14000 feet), inflation inside the portable hyperbaric chamber of 2 psi is equivalent to an altitude of 2100 meters (7000 feet).
In addition to descent, management of HACE includes oxygen and dexamethasone. Dexamethasone 8 mg (or 0.15 mg/kg in children) may be given IV, IM, or orally as initial dose, followed by 4 mg every 6 hours until symptoms resolve. As with any critically ill patient, airway management and bladder drainage may be necessary.13 Other entities causing altered mentation should be considered, such as electrolyte abnormalities, drug or alcohol use, hypoglycemia, meningitis, or hypothermia, for example.
High Altitude Pulmonary Edema (HAPE)
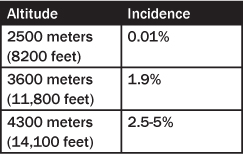
High altitude pulmonary edema accounts for most deaths from high altitude.1,13 HAPE may be related to rate of ascent, individual susceptibility, exertion, and cold temperature. HAPE typically affects previously healthy individuals who travel to altitude greater than 3000 meters (9800 feet), and usually develops within 2-4 days of ascent. (See Table 6.) Individuals with conditions affecting the pulmonary vasculature, such as Down syndrome, pulmonary hypertension, congenital absence of a pulmonary artery, or pulmonary emboli, may be predisposed to HAPE.9 A rapid ascent to 4500 meters (14,800 feet) was associated with a 10% incidence of HAPE, as opposed to an incidence of only 1-2% with gradual ascent. Re-ascent pulmonary edema may be seen in residents of high altitudes who travel to a lower altitude and then return.2
Table 6: Incidence of HAPE by Altitude84
The initial symptoms of HAPE are nonspecific, making the early diagnosis of HAPE difficult to differentiate from the more common high altitude cough and bronchitis. Dry cough and diminished performance are suggestive of, but not pathognomonic for, HAPE. Pink, frothy, or bloody sputum are later findings, but gross hemoptysis is uncommon.7 Many people with HAPE have AMS and HACE as well. The patient with HAPE usually exhibits dyspnea at rest, tachycardia, rales, tachypnea, and possibly cyanosis. Weakness may be severe.
A chest radiograph typically shows a normal-sized heart with patchy infiltrates confined to the right middle and lower lobes in mild cases, and diffusely in both lungs in more severe cases.7 ECG may show right axis deviation or right bundle branch block with right ventricular strain. Pulmonary artery pressure elevations to 36-51 mmHg are characteristic,41 but pulmonary wedge pressures are normal, establishing that HAPE is not a form of left heart failure.42
An abnormal rise in pulmonary artery pressure during ascent may be associated with HAPE, possibly related to a defect in nitric oxide synthesis during hypoxia or to vasoconstrictor factors such as endothelin-I, a potent pulmonary vasoconstrictor, in regulating pulmonary vascular tone.3 There is also some evidence that HAPE may be related to abnormalities in the fibrinolysis and coagulation system. One study found that patients with HAPE had higher D-dimer, fibrinogen, fibrin degradation products, and t-PA levels than controls.43 The disruptions in capillary endothelium and alveolar epithelium seen in HAPE disappear rapidly on descent. Various gene polymorphisms have been identified as risk factors for HAPE, including HLA alleles.8,44,45
HAPE may possibly be prevented utilizing certain measures. Gradual ascent, nifedipine, the beta-agonist salmeterol, tadalafil, and dexamethasone have all been proposed to prevent HAPE.27
Treatment of HAPE includes oxygen 4-6 liters/minute, if available, and descent of about 1000 meters (3300 feet) as soon as possible. Breathing supplemental oxygen may reduce pulmonary artery pressure by more than 30%.13,46 A portable hyperbaric chamber should be used if descent is not possible.3,7 Nifedipine should be given. The vasodilator nitric oxide may be considered in hospitalized settings, but is generally not a feasible option in the field.7
If neurologic deterioration occurs, dexamethasone should be administered. Continuous positive airway pressure may be useful in the treatment of HAPE; a portable device has been developed for use at altitude.47 If needed, respiratory support may require intubation and mechanical ventilation. HAPE developing at altitudes of less than 2500 meters (8200 feet) may benefit from echocardiography to evaluate for shunts, pulmonary hypertension, or valvular disease. Salmeterol and the phosphodiesterase-5 inhibitors sildenafil and tadalafil have been used to treat HAPE, but supplemental oxygen and descent are the recommended treatments of choice.48,49 HAPE is a non-cardiogenic pulmonary edema, and patients are generally hypovolemic, so loop diuretics should be avoided.9 Patients should be kept warm, as cold stress increases pulmonary artery pressure.13
Medications to Prevent High Altitude Illness
Medications used to prevent high altitude illness are presented in rough order by frequency of use. (See Table 7.) In most cases, the optimal dosage and duration of treatment have not been established from controlled studies.
Table 7: Medications for High Altitude Illness
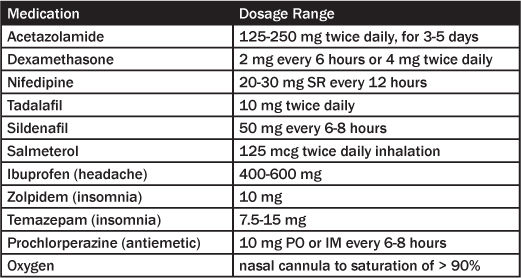
Acetazolamide. Traditionally, the carbonic anhydrase inhibitor acetazolamide has been the mainstay for prevention of AMS. While acetazolamide contains a sulfonamide and has generally been contraindicated in patients with a sulfa allergy, recent analysis indicates that acetazolamide need not be withheld in such individuals.50 Presumably it would be advisable to undergo trial or test dose of acetazolamide prior to the trip to manage allergy or anaphylaxis if one should occur. As a carbonic anhydrase inhibitor, acetazolamide causes a bicarbonate diuresis and respiratory stimulation. It reduces production of cerebrospinal fluid and promotes ion transport across the blood-brain barrier.7 In addition, acetazolamide prevents hypoxic pulmonary vasoconstriction.41
Typical prophylaxis for AMS includes acetazolamide 250 mg daily or 125 mg twice daily, starting one day prior to ascent and continuing for at least two days at high altitude.52 Acetazolamide doses of 500 or 750 mg daily are also effective in preventing AMS, but these higher doses increase the risk of polyuria and of taste disturbances.53
Acetazolamide provides approximately 48% relative risk-reduction for development of AMS.54 The increase in ventilation may prevent the periodic breathing seen in sleep at high altitude, as well as preventing nocturnal hypoxia seen during sleep.9,38 The aversion to carbonated beverages in those taking acetazolamide may be due to inhibition of salivary carbonic anhydrase. It tends to induce a flat and unpleasant taste to carbonated drinks, and may cause loss of appetite and nausea. One report indicates that acetazolamide may be ineffective in the prevention of AMS in extremely cold, arid conditions, such as at the South Pole.55
Patients with renal insufficiency, including a GFR of 10-50 mL/minute should take a lower dose of acetazolamide, and patients with a GFR less than 10 mL/minute should not use the drug.33 Since acetazolamide can cause an acidosis, patients with a pre-existing metabolic acidosis should avoid acetazolamide. In patients with limited ventilatory reserve, such as those with COPD, CO2 excretion can be impaired, and the dose should be limited to 125 mg daily or withheld altogether.33
Urinary alkalinization may cause urinary calcium-phosphate stone formation, especially in patients with a history of hypercalcemia or nephrolithiasis. Since acetazolamide alkalinizes the urine, ammonium ions may accumulate in the bloodstream. It has been recommended, therefore, that people with any significant liver disease avoid taking this drug.33 Acetazolamide is a kaliuretic drug and may act in concert with other diuretic drugs to lower serum potassium. Acetazolamide is listed as pregnancy class C and may cause neonatal jaundice if taken in the last month of pregnancy.
Dexamethasone. Dexamethasone is an alternative to acetazolamide in the prevention of AMS.56,57 Since it is not contraindicated in renal insufficiency, it is likely the drug of choice in patients with a low GFR for the prevention of AMS. Dexamethasone may also be used in cirrhotic patients since, unlike acetazolamide, there are no contraindications to its use in liver disease.33 The dexamethasone dose does not need to be adjusted for renal or hepatic insufficiency. In patients with diabetes, the dose of oral hypoglycemic agents or insulin may need to be adjusted at altitude for patients on steroids.
Recommended doses for adults are 4 mg every 12 hours for prevention of AMS. In general, avoid usage for more than 10 days to prevent adrenal suppression.27
The adverse effects of this medication are those common for steroids in general, such as the gastrointestinal bleeding in patients at risk for peptic ulcer disease and elevated blood glucose levels in diabetic patients. The incidence of upper gastrointestinal bleeding may be enhanced in people concurrently taking NSAIDs, especially at altitude.58
People with certain infections, such as amebiasis or strongyloidiasis, should avoid steroids. Traveler’s diarrhea is commonly treated with fluoroquinolones (ciprofloxacin or levofloxacin), and the risk of tendon rupture may be higher in patients taking corticosteroids and quinolones.59
Nifedipine. This calcium channel antagonist inhibits pulmonary artery vasoconstriction from hypoxia and may prevent HAPE. Typical dosages to prevent HAPE are 20 mg of the sustained-release preparation by mouth every 12 hours, and the dose of the sustained-release version should be halved for people with liver disease. Its actions may be additive with those of other antihypertensive medications.13
Phosphodiesterase Inhibitors: Tadalafil, Sildenafil. Phosphodiesterase inhibitors are pulmonary vasodilators and have been used to prevent HAPE. Typical doses include tadalafil 10 mg twice daily and sildenafil 50 mg every 8 hours.33 Doses should be lowered in the presence of renal insufficiency or hepatic insufficiency; a maximum of 10 mg daily of tadalafil should be given. Patients with coronary artery disease taking nitrates or alpha blockers should avoid these medications concurrently. A recent study found a greater benefit with tadalafil in the prevention of severe high altitude illness compared to acetazolamide.60
Salmeterol. Salmeterol is a long-acting beta-agonist that has been used to prevent HAPE. Typical dosing is 125 mcg by inhalation every 12 hours. No reduction in dosage is required in patients with renal insufficiency. Since any beta-agonist lowers serum potassium, caution should be used if this drug is given with acetazolamide. It is recommended that concurrent usage of monoamine oxidase inhibitors or tricyclic antidepressants be avoided.33 Beta agonists may facilitate enhanced alveolar fluid clearance and lower pulmonary artery pressure.9
Other Agents. Other agents have been proposed with variable and largely unproven success; they are mentioned for completeness and do not represent current standard of care in the prevention of altitude illness.
Gabapentin at doses of 300 to 600 mg per day has been found in small studies to reduce the headache severity associated with AMS, but its use has not gained wide accpetance.61-63
Ginkgo biloba 80-120 mg twice daily has been proposed to prevent AMS, although there are conflicting reports of its effect on the incidence and severity of AMS. The clinical results may depend upon the source and composition, as the active ingredients are unknown, and the amounts of specific chemicals, such as flavonoids and terpene ginkgolides, can vary considerably.13,64
Intravenous iron (200 mg iron(III)-hydroxide sucrose) has been used with some success in the prevention of AMS and, possibly, pulmonary hypertension, presumably due to its ability to influence cellular oxygen sensing pathways.65,66
Treatment Modalities for Established High Altitude Illness
Once illness has set in, the medications utilized in treatment are the same as those used in prevention.
Oxygen, if available, is indicated in the treatment of all high-altitude illnesses. Dosage should be titrated until arterial oxygen saturation is greater than 90%. Oxygen decreases pulmonary artery pressure, reduces cerebral blood flow and blood volume, and may relieve headache in AMS within minutes. It may be lifesaving for HAPE; mild to moderate HAPE has been treated with rest and supplemental oxygen for 48-72 hours.67 As noted above, breathing supplemental oxygen lowers pulmonary arterial pressure by 30-50%.
Descent is of utmost importance in the management of HAPE or HACE. Unfortunately, the physical work of descent may entail increasing cardiac output and pulmonary artery pressure if the patient is required to walk. In addition, descent may not always be feasible.
Ibuprofen in a dose of 400-600 mg may be effective in relieving high-altitude headaches in 60-70% of individuals.68,69 Some reports indicate that ibuprofen decreases the rate of AMS, although this may be related to headache relief alone, as noted earlier.34,35 Aspirin 325 mg every 4 hours may also prevent headache via its prostaglandin-inhibitory action.
Acetazolamide can be used to treat AMS, reducing the severity of symptoms in one report by 74% within 24 hours. The typical treatment dose is 250 mg twice per day until symptoms resolve, or 5 mg/kg/day in children.7
Dexamethasone may be used in the treatment of AMS or HACE. The dose for treatment of AMS is suggested to be 4 mg every 6 hours orally, IM, or IV, whereas the initial dosage for HACE has been suggested to be 8 mg initially, with a subsequent dose of 4 mg every 6 hours.7
Nifedipine may be used in the treatment of HAPE, as it reduces pulmonary artery pressure. Initial dosing is 10 mg, followed by 20-30 mg of extended-release formulation every 12 hours by mouth. Nifedipine may decrease pulmonary artery pressure but may have little effect on oxygenation.70 Nifedipine may induce a reflex tachycardia or hypotension.
Zolpidem has been proposed for treatment of insomnia at high altitude. At doses of 10 mg, it appears not to depress ventilation.7 Other sleep aids proposed include triazolam 0.125-0.25 mg or temazepam 15 mg. Patients with periodic breathing may have sleep disturbance at altitude, and acetazolamide 62.5 mg to 125 mg at bedtime may be efficacious. It is suggested that diazepam may cause hypoventilation at high altitude, and diphenhydramine has not been well-studied in the environment of high altitude.71
Theophylline at low doses has been proposed to reduce the symptoms of AMS. One report indicated effectiveness of 300 mg daily theophylline in alleviating events of periodic breathing and oxygen desaturations, characterized by rapid breathing alternating with apnea.72
Promethazine 25-50 mg by suppository or orally may be useful for nausea and vomiting.1 Prochlorperazine 10 mg orally or IM has been proposed as well, but may cause sedation and extrapyramidal reactions.13 Ondansetron has been used for nausea associated with altitude sickness.73
A portable hyperbaric bag or chamber may be employed for all high-altitude illness. The patient with HACE or HAPE is placed inside the bag, and the pressure is increased with a foot pump to reduce the effective altitude. The intent is to increase ambient pressure 2-4 psi for at least two hours to increase PaO2 and simulate descent. These fabric pressure bags weigh less than 7 kg.13 This modality does not require supplemental oxygen, although this can be added by nasal cannula or mask. There is a potential rebound effect after removing the person from the chamber. There are a number of models, and the effects are equivalent to the administration of low-flow oxygen.7 The disadvantages to the use of portable hyperbaric chambers include the need for constant oversight by care providers. As well, they present difficulties for the vomiting or claustrophobic patient. Descent, when feasible, is preferable.27 However, these bags are useful when weather or injury may preclude descent.9
The sleep disorder associated with AMS is thought to be related to hyperventilation and the resultant decrease in arterial PCO2 level. One study demonstrated an improvement in sleep-disordered breathing by adding 500 mL of dead space through a custom-designed full face mask.74
High Altitude Retinopathy
Hypoxia may result in dilatation of ocular vessels and capillary membrane hyperpermeability. Capillary bursts then may result in retinal hemorrhages. These are quite common at altitudes above 5000 meters (16,400 feet) and generally cause no visual impairment, but scotomata have been described. They typically resolve on return to low altitude. One report of 200 trekkers indicated that 4% had retinal hemorrhages when evaluated at 4243 meters (13,920 feet).75 If there is macular involvement of the hemorrhage, loss of visual acuity and permanent paracentral scotomata have been reported.76
Other Health Considerations at High Altitude
A high altitude renal syndrome has been described in people who live permanently at high altitude. Renal responses to life at high altitude include proteinuria, hypertension, polycythemia, and hyperuricemia. GFR appears to be preserved, and ACE inhibitors appear to be effective at reducing proteinuria.77
Erythrocytosis and high-altitude pulmonary hypertension are well-known aspects of chronic mountain sickness characterized by headaches, dizziness, fatigue, anorexia, breathlessness, and sleep disturbance in individuals who reside at greater than 3000 meters (9800 feet) for many years. Hematocrit values of greater than 80% have been recorded.78 The clinical significance of such high hematocrits is unclear, but relief of headache, fatigue, depression, or somnolence has been reported with phlebotomy.2
Ultraviolet keratitis, or “snow blindness,” may result from corneal injury and ultraviolet exposure. This may be preventable with UV-protectant sunglasses and side shields when exposed to snow at high altitudes. Treatment may include ophthalmic topical antibiotics, cycloplegics, and NSAIDs for comfort.79
People who have undergone radial keratotomy are at risk of refractive shift.44 Climbs to altitude entail measurable changes in intraocular pressure that are modulated by systemic oxygen saturation. However, these changes do not appear to be vision-threatening.80
High altitude bronchitis and pharyngitis may manifest as sore throat and cough, and are quite common. Mouth breathing may be necessitated by nasal congestion. Steam inhalation and nasal decongestant spray are likely to be more effective therapies than antibiotics.13
Hypothermia and frostbite must always be a consideration with any ascent. Temperatures, as a rule, decline 2.0°C (3.6°F) for every 300 meters (1000 feet) of elevation.13
A variety of neurologic symptoms can develop from high altitude exposure. Transient global amnesia has been reported, with recurrent memory loss. Mental impairment has been demonstrated, with reduced attention span, increased mental fatigue, and an increased number of arithmetic errors on testing.2 Focal neurologic findings may include transient hemiplegia or hemiparesis, aphasia, scotomata, and cortical blindness. The latter may respond to breathing carbon dioxide — a cerebral vasodilator — or oxygen.13 Cortical blindness should be managed as with any other neurologic dysfunction at altitude: descent, oxygen, or hyperbaric bag recompression pending descent.76
Carbon monoxide poisoning is a danger at high altitude. Tents are designed to be small and windproof. Cooking inside snow shelters or small closed tents is clearly a risk for developing carbon monoxide toxicity.
There appears to be an increased risk of gastrointestinal bleeding associated with high altitude.81
Conclusion
High altitude exposure is no longer an esoteric or rare phenomenon. Many people live at high altitudes, and many more work professionally or serve in rescue or the military at high altitude. Others visit high altitude sites for recreation or tourism. Knowledge of high altitude illness is useful not only for physicians who practice in these areas, but also for physicians who have patients traveling to or returning from high altitude trips.
References
1. Klocke DL, Decker WW, Stepanek J. Altitude-related illness. Mayo Clin Proc 1998;73:988-993.
2. West JB. The physiologic basis of high-altitude diseases. Ann Intern Med 2004;141:789-800.
3. Basynat B, Murdoch DR. High-altitude Illness. Lancet 2003;361:1967-1974.
4. Schommer K, Menold E, Subudhi AW, Bartsch P. Health risk for athletes at moderate altitude and normobaric hypoxia. Br J Sports Med 2012;11:828-832.
5. Moore JD. Travel to high altitude during pregnancy: frequently asked questions and recommendations for clinicians. High Alt Med Biol 2012;13:73-81.
6. Green RL, Huntsman RG, Serjeant GR. The sickle-cell and altitude. Br Med J 1971;4:595-595.
7. Hackett PH, Roach RC. High-Altitude Medicine. In: Auerbach PS. Wilderness Medicine, 5th ed. Mosby; 2007:2-36.
8. Luo Y, Chen Y, Zhang Y, et al. Association of endothelial nitric oxide synthase G894T polymorphism with high altitude pulmonary edema susceptibility: A meta-analysis. Wilderness Environ Med 2012;23:270-274.
9. Schoene RB. Illnesses at high altitude. Chest 2008;134:402-416.
10. León-Velarde F, Richalet JP. Respiratory control in residents at high altitude: Physiology and pathophysiology. High Alt Med Biol 2006;7:125-137.
11. Bukowskyj M, Nakatsu K. Theophylline reassessed. Ann Intern Med 1984;101: 63-73.
12. Macnutt MJ, De Souza MJ, Tomczak SE, et al. Resting and exercise ventilatory chemosensitivity across the menstrual cycle. J Appl Physiol 2012;112:737-747.
13. Hackett PH, Roach RC. Current concepts: High-altitude illness. N Engl J Med 2001;345:107-114.
14. Beall CM, Laskowski D, Erzurum SC. Nitric oxide in adaptation to altitude. Free Radic Biol Med 2012;52(7):1123-1134.
15. Scherrer U, Rexhaj E, Javet PY, et al. New insights in the pathogenesis of high-altitude pulmonary edema. Prog Cardiovasc Dis 2010;52:485-492.
16. Honigman B, Theis MK, Koziol-McLain J, et al. Acute mountain sickness in a general tourist population at moderate altitudes. Ann Intern Med 1993;118: 587-592.
17. Murdoch D, Curry C. Acute mountain sickness in the Southern Alps of New Zealand. N Z Med J 1998;111:168-169.
18. Montgomery A, Mills J, Luce JM. Incidence of acute mountain sickness at intermediate altitude. JAMA 1989;261:732-734.
19. Schneider M, Bernasch D, Weymann J, et al. Acute mountain sickness: Influence of susceptibility, preexposure and ascent rate. Med Sci Sports Exerc 2002;34:1886-1891.
20. Murdoch DR. Altitude illness among tourists flying to 3740 meters elevation in the Nepal Himalayas. J Travel Med 1995;2:255-256.
21. Jafarian S, Gorouhi F, Ghergherechi M, Lotfi J Respiratory rate within the first hour of ascent predicts subsequent acute mountain sickness severity. Arch Iran Med 2008;11:152-156.
22. Roach RC, Bartsch P, Oelz O, Hackett PH. Lake Louise AMS Scoring Consensus Committee. The Lake Louise acute mountain scoring system. 1993, available at http://www.thepeakinc.com/assets/PDFs/LakeLouisescore_001-1.pdf. Accessed Feb 4, 2013.
23. Roach RC, Houston CS, Honigman B, et al. How well do older persons tolerate higher altitude? West J Med 1995;162: 32-36.
24. Richalet JP, Larmignat P, Poitrine E, et al. Physiological risk factors for severe high-altitude illness: A prospective cohort study. Am J Respir Crit Care Med 2012;185:192-198.
25. Dehnert C, Bartsch P. Can patients with coronary heart disease go to high altitude? High Alt Med Biol 2010;11: 183-188.
26. Luks AM. Can patients with pulmonary hypertension travel to high altitude? High Alt Med Biol 2009;10:215-219.
27. Luks AM, McIntosh SE, Colin K, et al. Wilderness Medical Society Consensus Guidelines for the Prevention and Treatment of Acute Altitude Illness. Wilderness and Environmental Medicine 2010;21:146-155.
28. Consolazio CF, Matoush LO, Johnson HL, et al. Effects of high carbohydrate diets on performance and clinical symptomatology after rapid ascent to high altitude. Proc Fed Am Soc Exp Biol 1969;28:937-943.
29. Swenson ER, MacDonald A, Vatheuer M, et al. Acute mountain sickness is not altered by a high carbohydrate diet nor associated with elevated circulating cytokines. Aviation, Space and Environmental Medicine 1997;68:499-503.
30. Bernhard WN, Schalick LM, Delaney PA, et al. Acetazolamide plus low-dose dexamethasone is better than acetazolamide alone to ameliorate symptoms of acute mountain sickness. Aviat Space Environ Med 1998;69:883-886.
31. Maakestad K, Leadbetter G, Olson S, et al. Ginkgo biloba reduced incidence and severity of acute mountain sickness. Wilderness Environ Med 2001;12:51.
32. Roncin JP, Schwartz F, D’Arbigny P. EGb 761 in control of acute mountain sickness and vascular reactivity to cold exposure. Aviat Space Environ Med 1996;67: 445-452.
33. Luks AM, Swenson ER. Medication and dosage considerations in the prophylaxis and treatment of high-altitude sickness. Chest 2008;133:744-755.
34. Lipman GS, Kanaan NC, Holck PS et al. Ibuprofen prevents altitude illness: A randomized controlled trial for prevention of altitude illness with nonsteroidal anti-inflammatories. Ann Emerg Med 2012;59:484-490.
35. Gertsch JH, Lipman GS, Holck PS, et al. Prospective, double-blind randomized placebo-controlled comparison of acetazolamide versus ibuprofen for prophylaxis against high altitude headache: The headache evaluation at altitude trial (HEAT). Wildern and Env Med 2010;21:236-243.
36. Richalet JP, Larmignat P, Poitrine E, et al. Physiological risk factors for severe high-altitude illness: A prospective cohort study. Am J Respir Crit Care Med 2012;185:192-198.
37. Deweber K, Skorza K. Return to activity at altitude after high-altitude illness. Sports Health 2010;2(4):291-300.
38. Grissom CK. Ataxia is still an important clinical finding in severe high altitude illness. Wilderness & Environ Med 2011;22:105-106.
39. Muza SR, Lyons TP, Rock PB, et al. Effect of altitude exposure on brain volume and development of acute mountain sickness (AMS). In: Roach RC, Wagner PD, Hackett PH, eds. Hypoxia: Into the Next Millennium. Advances in experimental medicine and biology. New York. Kluwer Academic/Plenum 1999: 414.
40. Zavasky DM, Hackett PH. Cerebral etiology of acute mountain sickness: MRI findings. Wild Environ Med 1995;6: 229-230.
41. Maggiorini M. Prevention and treatment of high-altitude pulmonary edema. Prog Cardiovasc Dis 2010:500-506.
42. Penaloza D, Sime F. Circulatory dynamics during high altitude pulmonary edema. Am J Cardiol 1969;23:369-378.
43. Ren Y, Cui F, Lei Y, et al. High-altitude pulmonary edema is associated with coagulation and fibrinolytic abnormalities. Am J Med Sci 2012;344(3):186-189.
44. Luo Y, Chen Y, Zhang Y, et al. Association of endothelial nitric oxide synthase G894T polymorphism with high altitude pulmonary edema susceptibility: A meta-analysis. Wilderness Environ Med 2012;23:270-274.
45. MacInnes MJ, Koehle MS, Rupert JL. Evidence for a genetic basis for altitude illness: 2010 update. High Alt Med Biol 2010 Winter 11(4):349-368.
46. Hultgren HN, Lopez CE, Lundberg E, Miller H. Physiologic studies of pulmonary edema at high altitude. Circulation 1964;29:393-408.
47. Davis PR, Kippax J, Shaw GM, et al. A novel continuous positive airways pressure (CPAP) device for use at high altitude. High Alt Med Biol 2002;3:101.
48. Fiore DC, Hall S. Altitude illness: Risk factors, prevention, presentation, and treatment. Am Fam Pract 2010;82(9):1103-1110.
49. Fagenholz PJ, Gutman JA, Murray AF, Harris NS. Treatment of high altitude pulmonary edema at 4240 m in Nepal. High Alt Med Biol 2007;8(2):139-146.
50. Kelly TE, Hackett PH. Acetazolamide and sulfonamide allergy: A not so simple story. High Alt Med Biol 2010;11: 319-323.
51. Teppema LJ, Balanos GM, Steinback CD, et al. Effects of acetazolamide on ventilatory, cerebrovascular, and pulmonary vascular responses to hypoxia. Am J Respir Crit Care Med 2007;175:277-281.
52. Low EV, Avery AJ, Gupta V, et al. Identifying the lowest effective dose of acetazolamide for the prophylaxis of acute mountain sickness: Systematic review and meta-analysis. BMJ 2012;345.
53. Kayser B, Dumont L, Lysakowski C, et al. Reappraisal of acetazolamide for the prevention of acute mountain sickness: A systematic review and meta-analysis. High Alt Med Biol 2012;13(2):82-92.
54. Ritchie ND, Baggott AV, Todd WT. Acetazolamide for the prevention of acute mountain sickness — a systematic review and meta-analysis. J Travel Med 2012;19(5):298-307.
55. Anderson PJ, Miller AD, O’Malley KA, et al. Incidence and symptoms of high altitude illness in South Pole workers: Antarctic study of altitude physiology. Clin Med Insights Circ Respir Pulm Med 2011;5:27-35.
56. Murdoch D. Altitude sickness. Clinical Evidence, BMJ Publishing Group 2010;3:1209-1218.
57 Fiore DC, Hall S, Shoja P. Altitude illness: Risk factors, prevention, presentation, and treatment. Am Fam Physician 2010;82:1103-1110.
58. Wu T, Liu J. Alcohol and aspirin in combination with dexamethasone causes gastrointestinal bleeding at high altitude. Wilderness Environ Med 2006;17:69-71.
59. Levofloxacin (Levaquin): Full U.S. prescribing information. Raritan, N.J. Ortho-McNeil 2000.
60. Leshem E, Caine Y, Rosenberg E, et al. Tadalafil and acetazolamide versus acetazolamide for the prevention of severe high-altitude illness. J Travel Med 2012;19(5):308-310.
61. Jafarian S, Abolfazli R, Gorouhi F, et al. Gabapentin for prevention of hypobaric hypoxia-induced headache: Randomized double-blind clinical trial. J Neurol Neurosurg Psychiatry 2008:321-323.
62. Jafarian S, Gorouhi F, Salimi S, et al. Low-dose gabapentin in treatment of high-altitude headache. Cephalalgia 2007;27:1274-1277.
63. Seupaul RA, Welch JL, Malka ST, et al. Pharmacologic prophylaxis for acute mountain sickness: A systematic shortcut review. Ann Emerg Med 2012;59: 307-317.
64. Leadbetter G, Keyes LE, Maakestad KM, et al. Gingko biloba does — and does not — prevent acute mountain sickness. Wilderness Environ Med 2009;20(1): 66-71.
65. Talbot NP, Smith TG, Privat C, et al. Intravenous iron supplementation may protect against acute mountain sickness: A randomized, double-blinded placebo-controlled trial. High Alt Med Biol 2011;12(3):265-269.
66. Smith TG, Talbot NP, Privat C, et al. Effects of iron supplementation and depletion on hypoxic pulmonbary hypertension: Two randomized controlled trials. JAMA 2009;302(13):1444-1450.
67. Hultgren HN. High altitude pulmonary edema at a ski resort. West J Med 1996;164:222-227.
68. Broome JR, Stoneham MD, Beeley JM, et al. High altitude headache: Treatment with ibuprofen. Aviat Space Environ Med 1994;65:19-20.
69. Burtcher M, Likar R, Lachbauer W, et al. Ibuprofen versus sumatriptan for high-altitude headache. Lancet 1994;344:1445.
70. Oelz O, Maggiorini M, Ritter M, et al. Nifedipine for high altitude pulmonary edema. Lancet 1989;2:1241-1244.
71. Luks AM. Which medications are safe and effective for improving sleep at high altitude? High Alt Med Biol 2008;9(3): 195-198.
72. Kupper TE, Strohl KP, Hoefer M, et al. Low-dose theophylline reduces symptoms of acute mountain sickness. J Travel Med 2008;15:307-314.
73 Shapiro R. Ondansetron for the treatment of nausea associated with altitude sickness. Wilderness Environ Med 2008;19: 317-318.
74. Lovis A, De Riedmatten M, Greined D, et al. Effect of added dead space on sleep disordered breathing at high altitude. Sleep Med 2012;13:663-667.
75. Hackett PH, Rennie D. Rales, peripheral edema, retinal hemorrhage and acute mountain sickness. Am J Med 1979;67:214-218.
76. Butler FK. The eye in the wilderness. In: Auerbach P. Wilderness Medicine, 5th ed. Mosby; 2007:617-618.
77. Arestegui AH, Fuquay R, Sirota J, et al. High altitude renal syndrome (HARS). J Am Soc Nephrol 2011;11:1963-1968.
78. Hurtado A. Chronic mountain sickness. JAMA 1942;120:1278-1282.
79. Ellerton JA, Zuljan I, Agazzi G, et al. Eye problems in mountain and remote areas: Prevention and onsite treatment — official recommendations of the International Commission for Mountain Emergency Medicine ICAR MEDCOM. Wilderness Environ Med 2009;20:169-175.
80. Bosch MM, Barthelmes D, Merz TM, et al. Intraocular pressure during a very high altitude climb. Invest Ophthalmol Vis Sci 2010;51(3):1609-1613.
81. Wu TY, Ding SQ, Liu JL, et al. High-altitude gastrointestinal bleeding: An observation in Qinghai-Tibetan railroad construction workers on Mountain Tanggula. World J Gastroenterol 2007;13:774-780.
82. http://en.wikipedia.org/wiki/List_of_mountains_by_elevation
83. Lake Louise Criteria Available at http://www.high-altitude-medicine.com/AMS-LakeLouise.html. Accessed March 26, 2013.
84. Hall DP, Duncan K, Baillie JK. High altitude pulmonary edema. JR Army Med Corps 2011;157:68-72.
Clinical syndromes due to altitude can manifest in susceptible individuals with elevations as low as 1500 meters (5000 feet) above sea level. For otherwise healthy adults, altitudes of 2350 meters (8000 feet) are considered the arbitrary cutoff for placing one at risk for more serious syndromes, such as acute mountain sickness (AMS). While decreased ambient oxygen pressure is the most evident change at high altitude and presumably is responsible for most of the pathophysiologic derangements, other factors may have medical implications as well, such as decreased ambient temperature, increased exposure to ultraviolet radiation, and lower humidity.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
