Extreme Measures: Procedures and Therapies for Life-threatening Situations
September 8, 2013
Reprints
Extreme Measures: Procedures and Therapies for Life-threatening Situations
Authors:
Dennis Hanlon , MD, FAAEM, Vice Chairman, Operations, Associate Professor of Emergency Medicine, Allegheny General Hospital, Pittsburgh, PA.
Michael Yeh, MD, Senior Resident, Combined EM/IM Program, Allegheny General Hospital, Pittsburgh, PA.
Jennifer Nelson, MD, Senior Resident, Emergency Medicine Residency, Allegheny General Hospital, Pittsburgh, PA.
Peer Reviewer:
Jay Menaker, MD, FACEP, Associate Professor of Surgery, Associate Professor Emergency Medicine, University of Maryland School of Medicine, Baltimore.
Introduction
The purpose of this article is to review procedures and therapies used in the difficult, life-threatening clinical circumstances. The authors review five procedures that are rarely used in extreme situations.
Emergency Department Thoracotomy
Emergency department thoracotomy (EDT) has become a well-accepted procedure in penetrating trauma within certain parameters. Its role in blunt trauma remains a matter of great controversy.1
Indications for ED Thoracotomy. The primary goals of EDT are to release pericardial tamponade, control hemorrhage, evacuate air emboli, and perform cardiac massage and internal defibrillation in patients sustaining blunt force and penetrating trauma who are too unstable for transport to the operating room. (See Table 1.) Indications to perform EDT are based on the physiological status of the patient, injury mechanism, and evidence of ongoing massive hemorrhage despite standard resuscitation measures.2-7

Blunt or Penetrating Trauma with Recent Loss of Signs of Life. Certain physiologic criteria must be present to warrant EDT. These include the presence of signs of life at the trauma scene, with loss of signs of life on arrival or in the ED. In penetrating trauma, the loss of signs of life can be extended up to 15 minutes prior to ED arrival, assuming appropriate initiation of CPR without a significant response to volume resuscitation, as evidenced by a systolic blood pressure (SBP) less than 70 mmHg.2-7 The American College of Surgeons Committee on Trauma defines signs of life as pupillary response, spontaneous ventilation, measurable or palpable blood pressure, and/or cardiac electrical activity.8 The literature expresses a general consensus that no signs of life in the field and on presentation to the ED is an independent contraindication to EDT; the risk to the health care provider and cost outweigh the benefit.2,5-7
Pericardial Tamponade. Regardless of the type of trauma, EDT should be considered in pulseless electrical activity (PEA), asystole, or inadequate cardiac output due to suspected pericardial tamponade.2,7 This is independent of when the patient lost pulses, given that evacuation may result in return of spontaneous circulation despite greater than 15 minutes of CPR. Anoxic brain injury is likely in this scenario, but should not be the sole factor precluding EDT. Interestingly, according to the American College of Surgeons Committee on Trauma, asystole or PEA upon arrival to the ED is a contraindication to EDT, regardless of the suspicion of pericardial tamponade. However, the Western Trauma Association supports EDT in the setting of pericardial tamponade.2,7 Prior to performing EDT, a tension pneumothorax should be excluded as the cause of decreased cardiac output. This is done with needle decompression of one or both pleural spaces. The highest survivability of pericardial tamponade relieved with thoracotomy is seen in patients with penetrating injuries such as stab wounds or small lacerations of the heart.6
Massive Intrathoracic Hemorrhage. Conversion to open EDT is warranted in patients who undergo tube thoracostomy for evacuation of hemothorax with an initial output of greater than 1500 mL or ongoing output of 200 mL/hr in the first two hours.9 Based on weight, this volume is defined as > 15 mL/kg initially or > 4 mL/kg/hr in the first two hours.10 This is indicative of an injury to one or more of the great vessels of the thorax or penetrating injury to the heart. Each hemithorax can accumulate almost half of the patient's circulating blood volume in minutes.7 EDT is required to visualize and control the source of bleeding. If the patient can survive transport to the operating room, open thoracotomy in the operating room is preferred.
Evacuation of Bronchovascular Air Embolism. In the setting of thoracic trauma, an alveolarvascular or bronchovascular communication can occur. After initiation of positive pressure ventilation, air emboli move down the pressure gradient to the pulmonary venous system. This can impede venous return to the left side of the heart. Propagation of air emboli to the coronary arteries causes obstruction with resultant ischemia and possible arrest. EDT with cross clamping of the pulmonary hilum prevents further propagation of air emboli. Furthermore, with the patient in the Trendelenburg position, air can be aspirated from the apex of the left ventricle.7,11,12 Manual cardiac massage restores 55% of the normal cardiac output in the arrested heart and may also break up larger air pockets in the coronary arteries.13,14
Hemorrhagic Shock and/or Subdiaphragmatic Hemorrhage Control. Thoracic clamping of the descending aorta after EDT serves to redistribute blood flow to the coronary and cerebral arteries. Clamping of the thoracic aorta may reduce subdiaphragmatic blood loss from an abdominal injury. This provides a limited amount of time for surgical repair of the hemorrhage source. As soon as adequate blood volume has been restored and/or the site of hemorrhage has been controlled, the aortic clamp should be removed.7,15,16
Surgical Technique. The patient's left arm should be secured above the head to allow the largest area for surgical access. This is assuming that the source of injury is not isolated to the right hemithorax. The patient should be supine unless air embolism is suspected, in which case the patient is placed in the Trendelenburg position to minimize the risk of propagation. The thoracotomy incision starts just to the right of the sternum. If contralateral thoracotomy is required, this saves time in resection of the right chest. The excision should follow the natural curvature between the fourth and fifth ribs. The cut should extend through all the layers of the subcutaneous tissue and intercostal muscles in a single attempt. Once the pleural space is entered, the teeth of the rib retractors are inserted inside the pleural cavity. The long end of the handle should be directed posteriorly and caudal to the patient in case a right-sided thoracotomy is also warranted. Once the thoracic cavity is opened, rapid identification of the injury source should be made.7,9
The lung should be reflected superiorly to identify the pericardium. Alternatively, some have advocated intentional right mainstem intubation to deflate the left lung.17 The phrenic nerve must be identified and avoided. The pericardium should be grasped with toothed forceps or hemostats with one hand, and the pericardium should be opened with blunt-tip scissors anterior and parallel to the phrenic nerve. Evacuation of the clot and isolation of the source of bleeding can be difficult to visualize in the beating heart. Therefore, a manual approach is often used. If injury to the ventricle is encountered, digital pressure can be used to attempt to control the bleeding. Several methods may be used to more definitely provide control. Sutures should be buttressed with pledgets to control cardiac surface bleeding when passing running or horizontal mattress sutures.7,9 Staples can be used on the ventricle to rapidly control hemorrhage and minimize exposure risk.9,18 A Foley catheter can be placed through the defect. Once in place, the Foley bulb can be inflated inside the ventricle and gentle traction can be applied to control bleeding.9 Vascular clamps should be used on the more delicate tissue of the atrium or great vessels. Avoid suturing over the coronary arteries if possible. Of note, in the non-beating heart, cardiorrhaphy should be done prior to internal defibrillation or direct cardiac massage.
The indications for defibrillation are the same as those for non-traumatic cardiac arrest. Internal defibrillation paddles are positioned on the surface of the heart anteriorly and posteriorly, with care to avoid the coronary arteries or phrenic nerve, if possible. Defibrillation begins at 10 joules and can be increased up to 50 joules in increments of 10 joules per defibrillation attempt.19 Manual cardiac massage is done using a two-handed approach. This is done with both wrists touching, followed by a sequential clapping motion of the hands as they come together from the palms to the fingers. Intra-cardiac epinephrine may be warranted and is injected using a long, thin needle directly into the left ventricle, taking care to avoid the left anterior descending artery and its large diagonal branches.7,13,14
If the primary injury is to the descending aorta or one of the major intraabdominal branches, occlusion of the descending aorta is done to maximize coronary and cerebral perfusion. The inferior hilar ligament tethers the lung to the mediastinum and serves as a reflection point of the lung. Prior to isolation of the aorta, the mediastinal pleura must be incised carefully to avoid further aortic, hilar, or esophageal injury. This is best accomplished by palpation of the spine just superior to the diaphragm. The first structure anterior to the spine is the aorta. Blunt dissection is made through the pleura and around the aorta to release it from the spine posteriorly and esophagus anteriorly. This can be done with blunt hemostats or one's fingers. An orogastric tube can serve as a tactile reference to avoid clamping the esophagus. If possible, gentle pressure should be applied on the aorta during its isolation. While keeping one hand in the dissection plane to serve as a guide, the other hand inserts vascular clamps around the aorta. Once clamped, distal perfusion to the body is impaired and will convert to anaerobic metabolism, leading to accumulation of vasoactive agents and lactic acid, thus increasing morality with every minute of occlusion. Mortality following aortic cross clamping increases exponentially after approximately 30 minutes.7,15,16
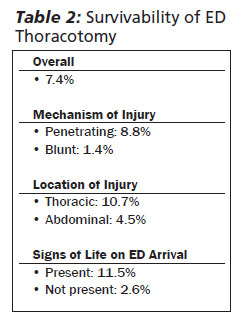
Outcomes. Survivability following ED thoracotomy is based on several factors, including mechanism of injury, location of major injury, and the physiologic status of the patient on arrival to the ED.20 Rhee et al addressed the question of EDT survival via a meta-analysis involving 4,620 cases at 24 institutions. They found that overall survival following EDT was 7.4%, with most surviving patients reaching a full neurological recovery. Based on mechanism of injury, 8.8% of patients survived following penetrating injury, whereas 1.4% survived following blunt injury. Based on location, a survival rate of 10.7% was seen in those patients with thoracic injuries, and a rate of 4.5% was seen for abdominal injuries.6 This number dropped to less than 1% for patients with multiple injuries. Finally, if signs of life were noted on arrival to the ED, survival rates following thoracotomy were 11.5% in contrast to 2.6% if signs of life were absent.6 (See Table 2.) The decision to perform an EDT is complex. One EDT can cost on average $100,000 and substantial hospital resources.6 Studies involving pre-hospital thoracotomies and thoracotomy in pediatric trauma, as well as continued observation of techniques and survival rates, are underway to further contribute to our understanding of this potentially life-saving but high risk procedure.21-24
Perimortem Cesarean Section
Perimortem cesarean section was described in literature dating as early as 800 BC.25 Yet, emergent perimortem cesarean section was first described in the medical literature in the mid 1980s.26 The literature on the indications, timing, and process of perimortem cesarean section during cardiac or traumatic maternal arrest comes largely from case reports and observational outcomes. In addition to trauma (blunt or penetrating), causes of cardiac arrest in the pregnant patient include venous thromboembolism, preeclampsia, eclampsia, anaphylactoid syndrome of pregnancy (previously known as amniotic fluid embolism), hemorrhage from placental abruption or previa, allergic reactions, hypermagnesemia, congenital or acquired structural heart disease, or infection.27 Fortunately, it is still very rare.28 If a gravid female presents in extremis and is not actively in labor, every effort should be made to transport the patient to the closest obstetrical unit following adequate resuscitation in the ED. Once pulses are lost and ACLS/ATLS protocols are initiated, an attempt to improve venous return to the heart is made by rolling the patient to the left lateral decubitus position with a wedge pillow. This reduces the gravitational compression of the inferior vena cava by the gravid uterus.
Rees et al studied the effective force of chest compressions when done at varying angles. They found that the minimum loss of compression force is at a 27° decubitus angle.29 At this angulation, the maximal compressive force is 80% of the force generated in the supine position. 30 From this determination, the Cardiff resuscitation wedge was constructed. An alternative option is to use a caretaker's thighs as the person kneels against the patient's back to provide support during CPR. Also, the patient may be kept supine with someone manually displacing the uterus to the left.
If the gestational age is not known, rapid measurement of the fundal height can be assessed using the "finger breadth rule" in which the fundus is one finger-width above the umbilicus for every two weeks past 20 weeks gestation.31 A fundus with two finger-widths above the umbilicus is consistent with a 24-week pregnancy.31 The consensus opinion states that perimortem cesarean section should be considered only if the estimated gestational age is 24 weeks or greater, despite the fact that there are case reports of fetal survival at less than 24 weeks.31 This is based on the likelihood of fetal survival and the fact that at 24 weeks, venocaval compression and anatomical changes often cause inadequate chest compressions and generation of pulses is rarely reported.33 Perimortem cesarean section allows for more effective CPR and redistribution of up to 30% of the stroke volume away from the uterus and back to systemic circulation.33,34 Based on the outcomes of several hundred maternal arrests, if perimortem section is to be performed, it should be initiated within four minutes of loss of pulses.26,32 The goal is delivery of the fetus within five minutes.25,27,35,36 An exponential decline in survival rates is seen at time intervals greater than five minutes. However, successful maternal resuscitation and viable fetuses have been delivered at longer time intervals.25,26,34,37,38
The following equipment should be available: sterile suction catheter tip on suction canister, scalpel, several hemostats, sterile towels and surgical sponges, retractors, blunt-tip scissors, basin for the placenta, large absorbable sutures, and suction bulbs. A classic long, vertical incision from the xiphoid to the symphysis pubis is performed. Care should be taken to avoid bowel or bladder injury, if possible. The bladder can be evacuated with a Foley catheter if time allows. Another midline vertical incision is made gently into the upper segment of the uterus and can be extended caudally with blunt scissors. Blunt-tip scissors are advisable for the chorionic membrane as well to avoid injury to the fetus. If the placenta is anterior, it must also be transected.
Once the fetus is delivered, it is held below the level of the mother until the cord is doubly clamped and cut. The placenta should then be evacuated gently with blunt manual dissection. Large, locking running sutures should be placed over the uterus to prevent bleeding once cardiac output is restored. This is followed by closure of the abdominal cavity in the same fashion. Some advocate for sterile abdominal packing if hemorrhage control is adequate to prevent abdominal compartment syndrome caused by bowel ileus and edema following ischemia.35,36 CPR should be continued during this entire procedure.35,36 Once delivered, a separate team, preferably the neonatal intensive care unit (NICU) team, will direct the resuscitation of the infant. The mother's vital signs need to be immediately reassessed as the resuscitation continues. Delivery can improve the maternal response to resuscitative efforts.26,27
Fibrinolytics for Pulmonary Embolus
The use of fibrinolytic agents for treatment of pulmonary embolism (PE) remains controversial. A massive PE is defined as acute PE with SBP < 90 for at least 15 minutes or requiring inotropic support not due to other causes of hypotension.39 Cardiac arrest due to PE may occur via multiple mechanisms, including obstructive shock with right ventricular systolic dysfunction, arrhythmias induced by cardiac strain, release of mediators of pulmonary vasospasm, or respiratory failure. A submassive PE is defined as acute PE without hypotension but demonstrating evidence of either right ventricular dysfunction or myocardial necrosis.39
Fibrinolytic treatment appears to improve frequency of return of spontaneous circulation (ROSC) in patients with cardiac arrest due to massive PE, although it is unclear whether there is a definite improvement in long-term survival.40,41 Early bolus dosing of tenecteplase was associated with ROSC in one small pilot study of 35 patients.40 A retrospective cohort study of 66 patients with massive PE-induced cardiac arrest showed more frequent ROSC among those treated with thrombolysis, but no statistically significant survival to discharge.41 In a meta-analysis restricted to placebo-controlled, randomized trials of fibrinolysis for massive PE, 9.4% of patients treated with fibrinolytics had recurrent PE or death, compared to 19% for those treated with heparin alone (OR = 0.45, 95% CI 0.22-0.9). The number needed to treat was 10.42 The number needed to harm has been estimated at 8 to 17.42,43
The 2010 American Heart Association ACLS Guidelines state that fibrinolytic therapy may be reasonable for cardiac arrest patients with presumed or known pulmonary embolism (class IIa, level of evidence B recommendation).44,45 In the emergency department setting, bedside echocardiography may be useful for detecting a dilated, hypokinetic right ventricle suggestive of right heart strain and the presence of thrombus.
For cardiac arrest without known PE, empiric fibrinolytic treatment has not been shown to be of clear benefit.44 The use of tPA during CPR in pulseless electrical activity arrest does not improve survival to hospital discharge.44 There was no difference in return of spontaneous circulation, survival, or improved neurologic outcome in one multicenter trial in which adult patients with witnessed out-of-hospital cardiac arrest were randomized to receive tenecteplase or placebo.47 However, in one retrospective cohort analysis of out-of-hospital, non-traumatic cardiac arrest of all causes, recombinant tPA use during CPR was associated with increased ROSC (70.4% vs. 51% in controls) and improved 24-hour survival (48.1% vs. 32.9%).48 A meta-analysis of thrombolysis in CPR did show improved ROSC, 24-hour survival, survival to discharge, and long-term neurological function, although patients treated with thrombolytics had increased risk of severe hemorrhage.49
In cases of acute PE without cardiac arrest, the value of fibrinolytics depends on whether the PE is massive, submassive, or low risk. Low-risk pulmonary emboli are those without hypotension, normal biomarkers (BNP, proBNP), and no evidence of right ventricular dysfunction. The benefit of thrombolysis does not outweigh the potential complications in patients with low risk PE.39 Fibrinolytics appear to decrease death or recurrent PE in hemodynamically unstable patients or massive PE. Despite these benefits, fibrinolytics are used in only 30% of eligible patients.50 This lack of thrombolytic use is greater in the elderly and patients with comorbidities.51 Patients in these groups who were treated with thrombolytic therapy had a lower case fatality rate, regardless of age or comorbid conditions; therefore, the cautious approach may not be the safest approach.51 Although fibrinolytic treatment is associated with more rapid recovery of right ventricular motion and restoration of pulmonary perfusion, these findings are not linked to decreased mortality in patients with submassive PE.42,43,52,53 Whether this quicker recovery prevents long-term thromboembolic-induced pulmonary hypertension is unclear. Currently, there are two ongoing randomized, controlled trials to determine if there is a role for fibrinolytics in patients with submassive PE. At the present time, the decision to treat with thrombolytics must be made on a case-by-case basis.54
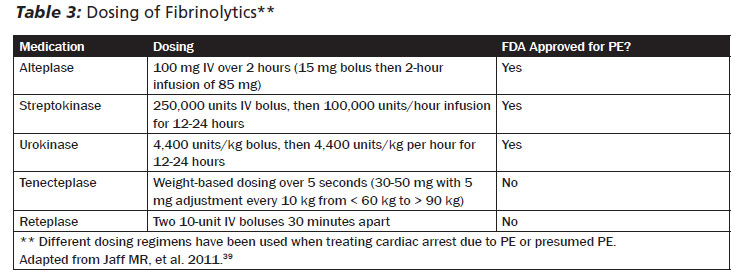
Currently, the U.S. Food and Drug Administration has approved three agents for treatment of PE: alteplase, streptokinase, and urokinase. Although not currently approved, clinical trials have been performed using tenecteplase and reteplase as well. Alteplase and tenecteplase have greater resistance to plasminogen-activating enzymes, and thereby have a longer duration of action.39
Absolute contraindications to fibrinolysis include CNS malignancy, prior intracranial hemorrhage, ischemic stroke within three months, active bleeding, suspected aortic dissection, bleeding diathesis, recent surgery near the spinal canal or brain, or head trauma. Other factors and relative contraindications include chronic anticoagulation, advanced age, pregnancy, and uncontrolled hypertension.39 The use of thrombolytics and duration of CPR are not associated with a statistically significant increase in major bleeding complications with patients in extremis.44,55,56
Although intracranial hemorrhage is an absolute contraindication to fibrinolytics, there has been a case report of a patient with a recent intracerebral hemorrhage who received tPA with no rebleeding or new neurologic deficits.57
Percutaneous thromboembolectomy during active CPR has been described in several cases, with restoration of pulmonary circulation.58 Emergency surgical embolectomy with cardiopulmonary bypass may be an option for patients for whom fibrinolysis is contraindicated, or who have refractory shock.39,59-62
Induced Hypothermia
Therapeutic hypothermia has been shown to improve neurological outcome in adult patients with witnessed out-of-hospital cardiac arrest due to ventricular fibrillation (VF) or pulseless ventricular tachycardia (VT) who remain comatose after restoration of spontaneous circulation. Hypothermia appears to limit reperfusion injury caused by systemic inflammation, formation of reactive oxygen species, and release of excitatory neurotransmitters after successful initial resuscitation.63
In one European multicenter, randomized trial, comatose survivors of VF cardiac arrest who were cooled were more likely to have a favorable neurologic outcome compared to the normothermia control group (55% vs. 39%, RR = 1.4; 95% CI: 1.08-1.81). Mortality at six months was also slightly lower among those in the hypothermia group (41% vs. 55%, RR = 0.74, 95% CI: 0.58-0.95).64 An Australian trial of 77 out-of-hospital cardiac arrest patients randomized to hypothermia versus normothermia showed improved survival to discharge with sufficiently good neurologic function to return home or to a rehabilitation facility. Among those in the hypothermia group, 49% were discharged home or to rehab compared to 26% of those assigned to normothermia.64
The evidence of benefit for therapeutic hypothermia in patients presenting with non-shockable rhythms is less clear, mostly limited to observational studies with substantial risk of bias. Multiple studies with historical controls suggest that the beneficial effects of therapeutic hypothermia may be associated with any initial arrest rhythm.66-71 Two non-randomized trials using concurrent controls also suggested that hypothermia may be of benefit for cardiac arrest with non-VF initial rhythms.70,71 However, the neuroprotective effect of hypothermia in non-shockable rhythms is less clear. One recent meta-analysis of 12 non-randomized and two randomized trials with patients resuscitated from a non-shockable rhythm showed lower in-hospital mortality but no statistically significant difference in neurologic outcome.72
More research is needed to further characterize the benefits of inducing hypothermia in patients with initial rhythms other than VF or VT. A Cochrane Systematic Review in 2012 found that patients treated with hypothermia were more likely to have good cerebral recovery as well as survival to hospital discharge, compared to those with standard normothermic treatment.73 The 2010 Guidelines for Advanced Cardiac Life Support recommend initiation of therapeutic hypothermia for all comatose adult post-cardiac arrest patients.45
It is unclear whether the timing of hypothermia induction results in better neurologic outcomes, although it may be ideal to initiate cooling as soon as possible after return of spontaneous circulation. Animal studies in mice, rats, and dogs suggest that early initiation of hypothermia within less than 20 minutes appeared more beneficial than when hypothermia was delayed.74-76
One case series of 986 patients showed no significant association between time to initiation of cooling and time to achieving target temperature within 3-6 hours, with improved neurologic outcome after discharge.77 Another case study of 49 comatose out-of-hospital post-arrest patients showed that time to target temperature did not independently predict neurologic outcome.78
Induction of therapeutic hypothermia by EMS providers has not been shown to improve neurologically intact survival to discharge.79,80
Therapeutic hypothermia has been used with success in neonates with hypoxic ischemic encephalopathy, but there are few data regarding its use in cardiac arrest. A recent Cochrane Systematic Review showed no difference in survival or neurologic outcome. Therefore, the authors were unable to make specific recommendations for clinical practice.81
Therapeutic hypothermia may be considered for any patient who lacks meaningful response to verbal commands after return of spontaneous circulation. Potential contraindications include severe hemorrhage, intracranial bleeding, and hypotension refractory to multiple pressors.63
Induction may be performed via multiple methods, including intravascular heat exchange catheters and surface cooling devices. An initial IV bolus of 500 mL to 30 mL/kg ice-cold (4°C) normal saline or lactated Ringer's solution is a simple and effective method for lowering core temperature by up to 1.5°C.82 Cold IV fluids can decrease core temperature more rapidly than endovascular catheters and may be administered in the pre-hospital setting.83 External cooling may be more labor intensive, but may be achieved with cooling blankets, a cooling helmet, wet towels, fanning, or ice packs to the neck, axillae, and groin areas. External cooling may also be complicated by the normal vasoconstriction of blood vessels in the skin at around 36.5°C, thereby reducing heat exchange.84 Patients receiving therapeutic hypothermia should be cooled to a target temperature between 32°C and 34°C for 12-24 hours.45,85
Although central venous temperature is the gold standard, esophageal temperature may be the most accurate and feasible surrogate in the emergency department. Changes in rectal temperature may be delayed compared to acute core temperature fluctuations, and bladder temperature may be misleading if urine output is low.45,86
Shivering can slow down the cooling process and impede therapeutic hypothermia.87-89 It can be prevented with neuromuscular blocking agents and sedation. However, paralytics may also mask seizure activity, and EEG may be indicated. In one of the landmark trials, pancuronium 0.1 mg/kg was given to prevent shivering.64 Propofol may be used as well for sedation. Magnesium sulfate given intravenously can increase the shivering threshold.63,84
Low body temperature causes a mild coagulopathy, as clotting factors and platelets function less effectively. In one retrospective review of 69 cardiac arrest patients who underwent therapeutic hypothermia, 22% of patients had bleeding.89 However, significant bleeding requiring transfusion occurred in only 4% of 986 patients studied in a Swedish case registry from 2004 to 2008, with higher risk among patients who had percutaneous interventions.77
Therapeutic hypothermia has become a class one recommendation by the American Heart Association for VF/VT arrests and is certainly not considered an extreme measure, although it may have been at one time.90 The question now is what other clinical circumstances will benefit from therapeutic hypothermia. Will it be nonshockable rhythms (PEA or asystole), strokes, heart attacks without arrest, or some other condition?
Emergency Cardiopulmonary Bypass: Extracorporeal Membrane Oxygenation
Extracorporeal membrane oxygenation (ECMO) provides temporary cardiorespiratory support for patients in respiratory or cardiac failure. Veno-venous (VV) circuit ECMO provides gas exchange only, and is used to support patients with acute lung injury such as ARDS. Cardiac ECMO utilizes a veno-arterial (VA) circuit that propels blood through the body to maintain circulation in addition to gas exchange.91
Deoxygenated blood is transported by a pump through a membrane oxygenator where gas exchange takes place. A heat exchanger in the device maintains normal blood temperature, which may potentially be used for therapeutic hypothermia in the setting of cardiac arrest. Vascular access can be established peripherally with percutaneous cannulation of the femoral vessels by a Seldinger technique or cutdown procedure.91,92
Complications of ECMO include bleeding, especially at cannulation sites, which may be exacerbated by thrombocytopenia and the need for systemic anticoagulation with heparin to prevent thrombus formation. Massive gas embolism from air in the circuit and tubing rupture may be catastrophic. Decreased blood flow through the circuit is the most common complication, and is usually due to hypovolemia.91 With femorofemoral ECMO in the setting of cardiac arrest, oxygenated blood travels in a retrograde fashion up the aorta. In the absence of effective left ventricular output, this may result in elevated left ventricular pressures and pulmonary edema or hemorrhage.92,93
Extracorporeal cardiopulmonary resuscitation (E-CPR) is an emerging application of ECMO as an adjunct to conventional ACLS for refractory cardiac arrest patients. Most studies to date on E-CPR have reported survival benefit among patients who have in-hospital cardiac arrests. In 1986, one hospital in San Diego created a program for rapid mobile initiation of ECMO for in-hospital cardiac arrests. Case-series data collected over 20 years showed 46% overall survival and a 30-day survival rate of 27.7%.94 One retrospective analysis of adults suffering witnessed cardiac arrest in the hospital showed that patients treated with ECMO had improved survival with minimal neurologic impairment at discharge and at six months.95 A prospective, observational study of 975 in-hospital cardiac arrest patients who had CPR for longer than 10 minutes showed that ECMO was associated with improved survival to discharge (28% vs. 12.3%) and one-year survival (18.6% vs. 9.7%).96
Duration of ECMO does not appear to be associated with survival. Even with prolonged CPR times in one study, with a mean of 47.6 minutes (+/- 13.4 minutes), 66.7% of patients eventually were able to be weaned off ECMO. Survival discharge was seen in 31.6%. Among survivors, only 5.6% had severe neurologic deficit.97 Duration of CPR prior to initiation of ECMO, however, is associated with survival. In one study of 135 adult in-hospital cardiac arrest patients, probability of survival for CPR duration of 30, 60, or 90 minutes was 50%, 30%, or 10%, respectively. Favorable neurologic outcomes were reported in 89% of survivors.98
Among pediatric patients with in-hospital cardiac arrest treated with ECMO, one study of 27 patients showed a 41% survival rate. Nonsurvivors had higher lactate levels, longer duration of CPR, higher incidence of renal failure, and longer time to ECMO initiation.99
The role of ECMO in treating patients with out-of-hospital cardiac arrest is not well established. Neurologic outcomes were generally poor. In the 1990s, ED initiation of ECMO was reported in a case series of 10 patients. Although vascular access and circulatory support was achieved, the mean survival was two days, with no long-term survivors.100 A French study of 51 consecutive patients with witnessed out-of-hospital refractory cardiac arrest who received ECMO immediately upon arrival showed only 4% survival with favorable neurologic outcome at day 28.101 In Japan, a retrospective study of adult patients with witnessed out-of-hospital cardiac arrest found three-month survival of 22.7% among patients treated with cardiopulmonary bypass, versus 9.9% with standard resuscitation. There was no statistically significant difference in neurologic outcome.102
Another study of 162 adult patients with witnessed cardiac arrest who received CPR for more than 20 minutes showed 29.2% survival in the ECMO group compared to 8.3% of those receiving standard treatment. Pupil diameter greater than 6 mm was associated with poor neurological survival, but duration of CPR was not a significant factor.103
More recently, there have been encouraging results in several reports with small numbers of patients. Shinar and colleagues reported a case of successful ECMO initiation in the ED for an adult patient with refractory ventricular fibrillation secondary to acute myocardial infarction. This patient subsequently underwent cardiac catheterization, and a left anterior descending artery occlusion was visualized and stented. He was discharged on hospital day 9 with no neurologic sequelae.92 A subsequent study from this institution reported eight patients who had successful E-CPR in the ED, with five surviving to hospital discharge neurologically intact.93 Preliminary data from the SAVE-J multicenter, nonrandomized prospective cohort study based in Japan compared 180 patients treated with E-CPR with 134 patients who received standard treatment. These were mostly witnessed, out-of-hospital arrests, with half of the patients receiving lay-rescuer CPR at the scene. Average time to arrival at the ED was approximately a half hour. Survival with favorable neurologic outcome was 12.4% in the E-CPR group, compared to 1.6% among the controls.104
Other future applications of ECMO for cardiac arrest may include field resuscitation. This has been attempted in France and Germany, where the EMS systems emphasize direct provision of medical care by physicians. The first case of field initiation of ECMO was reported in France in an adult athlete who had refractory cardiac arrest during a road race. ECMO was started in the ambulance within one hour of the arrest. The patient was found to have a right coronary artery occlusion, which was treated with angioplasty. The patient was successfully weaned off ECMO within 48 hours.105 One recent case report of a 9-year-old pediatric drowning patient in cardiac arrest described the field application of a new portable mini-ECMO system in Germany. Although effective circulation and gas exchange was established, the patient did not survive.106
The decision to initiate ECMO in the ED depends on identification of the appropriate patient. More research is needed to define predictors of favorable outcomes and specific indications for treatment in order to identify good candidates and to prevent futile treatment. Cost is also a consideration in the decision to start ECMO. In 2005, a cost-utility analysis of pediatric patients at Emory University requiring cardiac ECMO for a mean duration of 5 days revealed a median cost of $156,324 per patient.107 However, as the use of ECMO in cardiac arrest care is still not widely used, the true costs have not been well studied. The 2010 ACLS guidelines state that ECMO may be considered for patients with a potentially reversible cause of cardiac arrest, such as a drug overdose, and if the time period without blood flow is brief.108 Survival among patients on ECMO was seen mostly among patients with a condition amenable to definitive intervention, such as pulmonary embolism or a respiratory process.109,110 In such cases, ECMO is an important bridge to survival rather than a definitive treatment itself. Future studies will clarify the indications and optimal use of ECMO. Its use is clearly not a solitary endeavor and will require a multidisciplinary team with appropriate administrative and ancillary support.111
References
1. Khorsandi M, Skouras C, Shah R. Is there any role for resuscitative emergency department thoracotomy in blunt trauma? Interactive Cardiovascular and Thoracic Surgery 2013;16:509-516.
2. Moore E, Knudson M, Burlew C, et al. Defining the limits of resuscitative emergency department thoracotomy: A contemporary Western Trauma Association perspective. J Trauma 2011;70:334-339.
3. Hunt P, Greaves I, Owens W. Emergency thoracotomy in thoracic trauma — a review. Injury 2006;37:1-19.
4. Kirkpatrick A, Ball C, D'Amours S, et al. Acute resuscitation of the unstable adult trauma patient: Bedside diagnosis and therapy. Can J Surg 2008;51:57-69.
5. Powell D, Moore E, Cothren C, et al. Is emergency department resuscitative thoracotomy futile care for the critically injured patient requiring prehospital cardiopulmonary resuscitation? J Am Coll Surg 2004;199:211-215.
6. Rhee P, Acosta J, Bridgeman A, et al. Survival after emergency department thoracotomy: Review of published data from the past 25 years. J Am Coll Surg2000;190:288-298.
7. Cothren C, Moore E. Emergency department thoracotomy for the critically injured patient: Objectives, indications, and outcomes. World J Surgery 2006:1;
1-13.
8. Working Group, Ad Hoc Subcommittee on Outcomes, American College of Surgeons — Committee on Trauma. Practice management guidelines for emergency department thoracotomy. J Am Coll Surg 2001;193:303-309.
9. Van Waes O, Van Riet P, Van Lieshout E, et al. Immediate thoracotomy for penetrating injuries: Ten years' experience at a Dutch level 1 trauma center. Eur J Trauma Emerg Surg 2012;38:543-551.
10. LoCicero J III, Mattox K. Epidemiology of chest trauma. Surg Clin North Am 1989;69:15-19.
11. King M, Aitchison J, Nel J. Fatal air embolism following penetrating lung trauma: An autopsy study. J Trauma 1984;24:753.
12. Thomas A, Stevens B. Air embolism: A cause of morbidity and death after penetrating trauma. J Trauma 1974;14:633.
13. Boczar ME, Howard MA, Rivers EP, et al. A technique revisited: Hemodynamic comparison of closed- and open-chest cardiac massage during human cardiopulmonary resuscitation. Crit Care Med 1995;23:498-503.
14. Jackson RE, Freeman SB. Hemodynamics of cardiac massage. Emerg Med Clin North Am 1983;1:501-513.
15. Connery C, Geller E, Dulchavsky S, et al. Paraparesis following emergency room thoracotomy: Case report. J Trauma 1990;30:362-363.
16. Kavolius J, Golocovsky M, Champion H. Predictors of outcome in patients who have sustained trauma and who undergo emergency thoracotomy. Arch Surg 1993;128:1158-1162.
17. Lui RC, Johnson FE. Selective right lung ventilation during emergency department thoracotomy. Crit Care Med 1989;17:1057-1059.
18. Macho J, Markison R, Schecter P. Cardiac stapling in the management of penetrating injuries of the heart: Rapid control of hemorrhage and decreased risk of personal contamination. J Trauma 1993;34:711-715.
19. Tintinalli J, et al. ED Thoracotomy. In: Tintinalli's Emergency Medicine, 7th edition. McGraw-Hill Publishing: 2011.
20. Passos EM, Engels PT, Doyle JD, et al. Societal costs of inappropriate emergency department thoracotomy. J Am Coll Surg 2012;214:18-25.
21. Keogh S, Wilson A. Survival following pre-hospital arrest with on-scene thoracotomy for a stabbed heart. Injury 1996;27:525-527.
22. Morrison J, Mellor A, Midwinter M, et al. Is pre-hospital thoracotomy necessary in the military environment? Injury 2011;42:469-473.
23. Davies G, Lockey D. Thirteen survivors of pre-hospital thoracotomy for penetrating trauma: A prehospital physician-performed resuscitation procedure that can yield good results. J Trauma 2011;70:
75-78.
24. Coats T, Keogh S, Clark H, et al. Pre-hospital resuscitative thoracotomy for cardiac arrest after penetrating trauma: Rationale and case series. J Trauma 2001;50:670-673.
25. Atta E, Gardner M. Cardiopulmonary resuscitation in pregnancy. Obstet Gynecol Clin North Am 2007;34:585-597.
26. Katz V, Balderston K, DeFreest M. Perimortem cesarean delivery: Were our assumptions correct? Am J Obstet Gynecol 2005;192:1916-1921.
27. Farinelli CK, Hameed AB. Cardiopulmonary resuscitation in pregnancy. Cardiol Clin 2012;30:453-461.
28. Department of Health and Human Services, Northern Ireland. Why mothers die: Report on confidential inquiries into maternal deaths in the United Kingdom 2000-2002. London. The Stationary Office. 2004.
29. Rees G, Willis B. Bioimpedance measurement of cardiac output. Eur J Obstet Gynecol Reprod Biol 1990;36:11-17.
30. Mallampalli A, Powner D, Gardner M. Cardiopulmonary resuscitation and somatic support of the pregnant patient. Crit Care Clin 2004;20:747-761.
31. Katz V. Perimortem cesarean delivery: It's role in maternal mortality. Seminars in Perinatology 2012;36:68-72.
32. Katz V, Dotters D, Droegemueller W. Perimortem cesarean delivery. Obstetrics and Gynecology 1986;68:571-576.
33. Kerr M. The mechanical effects of the gravid uterus in late pregnancy. J Obstet Gynaen Brit Commonw 1965;72:
513-529.
34. Selden B, Burke T. Complete maternal and fetal recovery following prolonged cardiac arrest. Ann Emerg Med 1988;17:346-349.
35. Strong T, Lowe R. Perimortem Cesarean section. Am J Emerg Med 1989;7:
489-483.
36. Stallard T, Burns B. Emergency delivery and perimortem c-section. Emerg Med Clin North Am 2003;21:679-693.
37. DePace N, Betesh S, Kotter MN. "Postmortem" Cesarean section with recovery of both mother and offspring. JAMA 1982;248:971.
38. Capobianco G, Balata A, Mannazzu M, et al. Perimortem Cesarean section delivery 30 minutes after laboring patient jumped from a fourth floor window: Baby survives and is normal at 4 years. Am J Obstet Gynecol 2008;198:15-16.
39. Jaff MR, McMurtry S, Archer SL, et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: A scientific statement from the American Heart Association. Circulation 2011;123:
1788-1830.
40. Fatovich DM, Dobb GJ, Clugston RA. A pilot randomised trial of thrombolysis in cardiac arrest (The TICA trial). Resuscitation 2004;61:309-313.
41. Janata K, Holzer M, Kurkciyan I, et al. Major bleeding complications in cardiopulmonary resuscitation: The place of thrombolytic therapy in cardiac arrest due to massive pulmonary embolism. Resuscitation 2003;57:49-55.
42. Wan S, Quinlan DJ, Agnelli G, et al. Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: A meta-analysis of the randomized controlled trials. Circulation 2004;110:744-749.
43. Thabut G, Thabut D, Myers RP, et al. Thrombolytic therapy of pulmonary embolism: A meta-analysis. J Am Coll Cardiol 2002;40:1660-1667.
44. Vanden Hoek TL, Morrison LJ, Shuster M, et al. Part 12: Cardiac arrest in special situations: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122(18 Suppl 3):
S829-861.
45. Peberdy MA, Callaway CW, Neumar RW, et al. Part 9: Post-cardiac arrest care: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122(18 Suppl 3):S768-786.
46. Abu-Laban RB, Christenson JM, Innes GD, et al. Tissue plasminogen activator in cardiac arrest with pulseless electrical activity. N Engl J Med 2002;346:
1522-1528.
47. Böttiger BW, Arntz HR, Chamberlain DA, et al. Thrombolysis during resuscitation for out-of-hospital cardiac arrest.
N Engl J Med 2008;359:2651-2662.
48. Lederer W, Lichtenberger C, Pechlaner C, et al. Recombinant tissue plasminogen activator during cardiopulmonary resuscitation in 108 patients with out-of-hospital cardiac arrest. Resuscitation2001;50:
71-76.
49. Li X, Fu Q, Jing X, et al. A meta-analysis of cardiopulmonary resuscitation with and without the administration of thrombolytic agents. Resuscitation2006;70:31-36.
50. Stein PD, Matta F. Thrombolytic therapy in unstable patients with acute pulmonary embolism: Saves lives but underused.
Am J Med 2012;125:465-470.
51. Stein PD, Matta F. Treatment of unstable pulmonary embolism in the elderly and those with comorbid conditions. Am J Med 2013;126:304-310.
52. Dong BR, Hao Q, Yue J, et al. Thrombolytic therapy for pulmonary embolism. Cochrane Database Syst Rev 2006(2):CD004437.
53. Agnelli G, Becattini C, Kirschstein T. Thrombolysis vs heparin in the treatment of pulmonary embolism: A clinical outcome-based meta-analysis. Arch Int Med 2002;162:2537-2541.
54. Tapson VF. Treatment of pulmonary embolism: Anticoagulation, thrombolytic therapy, and complications of therapy. Crit Care Clin 2011;27:825-839.
55. Bottiger BW, Bode C, Kern S, et al. Efficacy and safety of thrombolytic therapy after initially unsuccessful cardiopulmonary resuscitation: A prospective clinical trial. Lancet 2001;357:
1583-1585.
56. Bailen MR, Cuadra JA, Aguayo De Hoyos E. Thrombolysis during cardiopulmonary resuscitation in fulminant pulmonary embolism: A review. Crit Care Med 2001;29:2211-2219.
57. Koroneos A, Koutsoukou A, Zervakis D, et al. Successful resuscitation with thrombolysis of a patient suffering fulminant pulmonary embolism after recent intracerebral haemorrhage. Resuscitation2007;72:154-157.
58. Fava, M, Loyola S, Bertoni, et al. Massive pulmonary embolism: Percutaneous mechanical thrombectomy during cardiopulmonary resuscitation. J Vascular & Interventional Radiology 2005;16:
119-123.
59. Dauphine C, Omari B. Pulmonary embolectomy for acute massive pulmonary embolism. Ann Thoracic Surgery 2005;79:1240-1244.
60. Meneveau N, Seronde MF, Blonde MC, et al. Management of unsuccessful thrombolysis in acute massive pulmonary embolism. Chest 2006;129:1043-1050.
61. Stein PD, Alnas M, Beemath A, et al. Outcome of pulmonary embolectomy. Am J Cardiol 2007;99:421-423.
62. Leacche M, Unic D, Goldhaber SZ, et al. Modern surgical treatment of massive pulmonary embolism: Results in 47 consecutive patients after rapid diagnosis and aggressive surgical approach. J Thorac Cardiovasc Surg 2005;129:1018-1023.
63. Scirica BM. Therapeutic hypothermia after cardiac arrest. Circulation 2013;127:244-250.
64. Nikolow NM, Cunnungham AJ. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med 2002;346:549-556.
65. Bernard SA, Gray TW, Buist MB, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med 2002;346:557-563.
66. Sunde K, Pytte M, Jacobsen D, et al. Implementation of a standardised treatment protocol for post resuscitation care after out-of-hospital cardiac arrest. Resuscitation2007;73:29-39.
67. Don CW, Longstreth, Jr WT, Maynard C, et al. Active surface cooling protocol to induce mild therapeutic hypothermia after out-of-hospital cardiac arrest: A retrospective before-and-after comparison in a single hospital. Crit Care Med 2009;37:3062-3069.
68. Storm C, Steffen I, Schefold JC, et al. Mild therapeutic hypothermia shortens intensive care unit stay of survivors after out-of-hospital cardiac arrest compared to historical controls. Crit Care 2008;12:R78:1-8.
69. Oddo M, Schaller MD, Feihl F, et al. From evidence to clinical practice: Effective implementation of therapeutic hypothermia to improve patient outcome after cardiac arrest. Crit Care Med 2006;34:1865-1873.
70. Holzer M, Mullner M, Sterz F, et al. Efficacy and safety of endovascular cooling after cardiac arrest: Cohort study and Bayesian approach. Stroke 2006;37:
1792-1797.
71. Arrich J. Clinical application of mild therapeutic hypothermia after cardiac arrest. Crit Care Med 2007;35:1041-1047.
72. Kim YM, Yim HW, Jeong SH, et al, Does therapeutic hypothermia benefit adult cardiac arrest patients presenting with non-shockable initial rhythms? A systematic review and meta-analysis of randomized and non-randomized studies. Resuscitation 2012;83:188-196.
73. Arrich J, Holzer M, Havel C, et al. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database Syst Rev 2012;9:CD004128.
74. Abella BS, Zhao D, Alvarado J, et al. Intra-arrest cooling improves outcomes in a murine cardiac arrest model. Circulation 2004;109:2786-2791.
75. Kuboyama K, Safer P, Radovsky A, et al. Delay in cooling negates the beneficial effect of mild resuscitative cerebral hypothermia after cardiac arrest in dogs: A prospective, randomized study. Crit Care Med 1993;21:1348-1358.
76. Takata K, Takeda Y, Sato T, et al. Effects of hypothermia for a short period on histologic outcome and extracellular glutamate concentration during and after cardiac arrest in rats. Crit Care Med 2005;33:1340-1345.
78. Wolff B, Machill K, Schumacher D, et al. Early achievement of mild therapeutic hypothermia and the neurologic outcome after cardiac arrest. Int J Cardiol 2009;133:223-228.
77. Nielsen N, Hovdenes J, Nilsson F, et al. Outcome, timing and adverse events in therapeutic hypothermia after out-of-hospital cardiac arrest. Acta Anaesthesiol Scand 2009;53:926-934.
79. Castren M, Nordberg P, Svensson L, et al. Intra-arrest transnasal evaporative cooling: A randomized, prehospital, multicenter study (PRINCE: Pre-ROSC IntraNasal Cooling Effectiveness). Circulation 2010;122:729-736.
80. Bernard SA, Smith K, Cameron P, et al. Induction of therapeutic hypothermia by paramedics after resuscitation from out-of-hospital ventricular fibrillation cardiac arrest: A randomized controlled trial. Circulation 2010;122:737-742.
81. Scholefield B, Duncan H, Davies P, et al. Hypothermia for neuroprotection in children after cardiopulmonary arrest. Cochrane Database of Syst Rev 2013;2:CD009442.
82. Moore TM, Callaway CW, Hostler D. Core temperature cooling in healthy volunteers after rapid intravenous infusion of cold and room temperature saline solution. Ann Emerg Med 2008;51:153-159.
83. Kim F,Olsufka M, Longstreth WT Jr, et al. Pilot randomized clinical trial of prehospital induction of mild hypothermia in out-of-hospital cardiac arrest patients with a rapid infusion of 4 degrees C normal saline. Circulation 2007;115:3064-3070.
84. Polderman KH, Herold I. Therapeutic hypothermia and controlled normothermia in the intensive care unit: Practical considerations, side effects, and cooling methods. Crit Care Med 2009;37:
1101-1120.
85. Nolan JP, Morley PT, Hoek TLV, et al. Therapeutic hypothermia after cardiac arrest: An advisory statement by the advanced life support task force of the International Liaison Committee on Resuscitation. Circulation 2003;108:
118-121.
86. Robinson J, Charlton J, Seal R, et al. Oesophageal, rectal, axillary, tympanic and pulmonary artery temperatures during cardiac surgery. Can J Anaesth 1998;45:317-323.
87. Hostler D, Northington WE, Callaway CW. High-dose diazepam facilitates core cooling during cold saline infusion in healthy volunteers. Appl Physiol Nutr Metab 2009;34:582-586.
88. Badjatia N, Strongilis E, Gordon E, et al. Metabolic impact of shivering during therapeutic temperature modulation: The Bedside Shivering Assessment Scale. Stroke 2008;39:3242-3247.
89. Jarrah S, Dziodzio J, Fraser GL, et al. Surface cooling after cardiac arrest: Effectiveness, skin safety, and adverse events in routine clinical practice. Neuro Crit Care 2011;14:382-388.
90. O'Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2013;127:e362-425.
91. Hung M, Vuylsteke A, Valchanov K. Extracorporeal membrane oxygenation: Coming to an ICU near you. J Intensive Care Society 2012;13:31-38.
92. Shinar Z, Bellezo J, Paradis N, et al. Emergency department initiation of cardiopulmonary bypass: A case report and review of the literature. J Emerg Med 2012;43:83-86.
93. Bellezzo JM, Shinar Z, Davis DP, et al. Emergency physician-initiated extracorporeal cardiopulmonary resuscitation. Resuscitation 2012;83:966-970.
94. Chillcott S, Stahovich M, Earnhardt C, et al. Portable rapid response extracorporeal life support: A center's 20-year experience with a registered nurse-run program. Crit Care Nurs Q 2008;31:211-215.
95. Shin TG, Choi JH, Jo IJ, et al. Extracorporeal cardiopulmonary resuscitation in patients with inhospital cardiac arrest: A comparison with conventional cardiopulmonary resuscitation. Crit Care Med 2011;39:1-7.
96. Chen YS, Lin JW, Yu HY, et al. Cardiopulmonary resuscitation with assisted extracorporeal life-support versus conventional cardiopulmonary resuscitation in adults with in-hospital cardiac arrest: An observational study and propensity analysis. Lancet 2008;372:
554-561.
97. Chen YS,Chao A, Yu HY, et al. Analysis and results of prolonged resuscitation in cardiac arrest patients rescued by extracorporeal membrane oxygenation. J Am Coll Cardiol2003;41:197-203.
98. Chen YS, Yu HY, Huang SC, et al. Extracorporeal membrane oxygenation support can extend the duration of cardiopulmonary resuscitation. Crit Care Med 2008;36:2529-2535.
99. Huang SC, Wu CT, Chen YS, et al. Extracorporeal membrane oxygenation rescue for cardiopulmonary resuscitation in pediatric patients. Crit Care Med 2008;36:1607-1613.
100. Martin GB, Rivers EP, Paradis NA, et al. Emergency department cardiopulmonary bypass in the treatment of human cardiac arrest. Chest 1998;113:743-751.
101. Le Guen M, Nicolas-Robon A, Carreira S, et al. Extracorporeal life support following out-of-hospital refractory cardiac arrest. Crit Care 2011;15:R29.
102. Tanno K, Itoh Y, Takeyama Y, et al. Utstein style study of cardiopulmonary bypass after cardiac arrest. Am J Emerg Med 2008;26:649-654.
103. Maekawa K, Tanno K, Hase M, et al. Extracorporeal cardiopulmonary resuscitation for patients with out-of-hospital cardiac arrest of cardiac origin: A propensity-matched study and predictor analysis. Crit Care Med 2013;41:1186-1196.
104. Sakamoto TA, Asai Y, Nagao K, et al. Abstract 18132: Multicenter non-randomized prospective cohort study of extracorporeal cardiopulmonary resuscitation for out-of hospital cardiac arrest: Study of Advanced life support for Ventricular fibrillation with Extracorporeal circulation in Japan (SAVE-J). Circulation 2011;124(A18132).
105. Lebreton G, Pozzi M, Luyt CE, et al. Out-of-hospital extra-corporeal life support implantation during refractory cardiac arrest in a half-marathon runner. Resuscitation 2011;82:1239-1242.
106. Arlt M, Philipp A, Voekel S, et al. Out-of-hospital extracorporeal life support for cardiac arrest: A case report. Resuscitation2011;82:1243-1245.
107. Mahle WT, Forbess JM, Kirschbom PM, et al. Cost-utility analysis of salvage cardiac extracorporeal membrane oxygenation in children. J Thorac Cardiovasc Surg 2005;129:1084-1090.
108. Dworschak M. Is extracorporeal cardiopulmonary resuscitation for out-of-hospital cardiac arrest superior compared with conventional resuscitation? Crit Care Med 2013;41:1365-1366.
109. Younger JG, Schreiner RJ, Swaniker F, et al. Extracorporeal resuscitation of cardiac arrest. Acad Emerg Med 1999;6:
700-707.
110. Maggio P,Hemmila M, Haft J, et al. Extracorporeal life support for massive pulmonary embolism. J Trauma 2007;62:570-576.
111. Gaieski DF, Boller M, Becker LB. Emergency cardiopulmonary bypass: A promising rescue strategy for refractory cardiac arrest. Crit Care Clin2012;28:211-229.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
