Acute Severe Asthma
Acute Severe Asthma
Authors: Ronald M. Perkin, MD, MA, Professor and Chairman, Department of Pediatrics, Brody School of Medicine at East Carolina University, Greenville, NC.
Matthew R. Ledoux, MD, Division Chief, Division of Pediatric Critical Care and Sedation Services, Brody School of Medicine, East Carolina University, Greenville, NC.
Peer Reviewer: Christopher J. Haines, DO, FACEP, FAAP, Associate Professor of Pediatrics and Emergency Medicine, Drexel University College of Medicine, Director, Department of Emergency Medicine, Medical Director, Critical Care Transport Team, St. Christopher's Hospital for Children, Philadelphia, PA.
Asthma is a common problem in the emergency department (ED), with many children having significant exacerbations. The ED physician must be prepared with a versatile approach to rapidly stabilize the patient. This article comprehensively reviews the approach to a child with acute severe asthma, emphasizing management alternatives.
— Ann M. Dietrich, MD, Editor
Introduction
Asthma is the most common chronic pediatric respiratory disease and remains one of the most common reasons children in the United States require hospitalization. Acute exacerbations of asthma can be life-threatening. While most of these acute exacerbations can be managed successfully in the emergency department, a few children with severe exacerbations may require admission to the intensive care unit. These severe exacerbations have been referred to as status asthmaticus, acute severe asthma, life-threatening asthma, or near-fatal asthma.1-5 The following review explores the current understanding of epidemiology, physiology, and treatment of acute severe asthma in children.
Definition
The exact definition of acute severe asthma or status asthmaticus varies between authors.1 For this article, the authors define acute severe asthma as a wheezing patient who does not respond to initial doses of inhaled bronchodilators.
Risk Factors
Although few children have life-threatening asthma episodes, these episodes may be associated with potential mortality, a high morbidity, and a high cost of treatment.3
There appear to be two clinical subsets of children who die from acute severe asthma.3,6 The first group of children with fatal asthma has a long history of poorly controlled asthma, often with a previous history of respiratory failure. In these children, death occurs secondary to acute respiratory failure and asphyxia or complications associated with mechanical ventilation.6-8 Pathologic examination in these children reveals extensive bronchial mucous plugging, edema, and eosinophilic infiltration of the airways. In the second group, the children have only a mild history of asthma, experience the sudden onset of bronchospasm, and can rapidly progress to cardiac arrest and death. If recognized and managed early, these children respond to beta-adrenergic agonists and mechanical ventilatory support better than children with the slow-onset fatal asthma.3,6,9,10 Pathologic examination in this rapidly fatal group of children shows empty airways devoid of mucous plugging with a greater proportion of neutrophils rather than eosinophils.3,6,9,10 Although the existence of this second type of fast onset has been controversial, many studies have shown a high percentage of death occurring within one to two hours of onset of symptoms.3,6-13 Some of these studies reveal an over-representation of obese children and African-American children in this rapidly progressive, type 2 cohort, which invites speculation about an alternative pathophysiology or possible health care disparities in this group of children.3,14 These studies indicate the need to recognize children at risk for sudden asphyxial asthma. Accordingly, several authors have attempted to define risk factors for children who die from asthma.3,6,15,16 (See Table 1.) Patients who have had a life-threatening episode of asthma should receive ongoing care from a physician who specializes in asthma.8,15
Table 1. Risk Factors for Potentially Fatal Asthma

Pathogenesis/Pathophysiology
Asthma is now considered a disease of inflammation. During the past two decades, tremendous gains have been made with regard to the understanding of asthma.6,17-19 Asthma frequently begins during childhood and is often associated with atopy, the genetic susceptibility to produce IgE specific to common airborne allergens. Atopic children respond to these airborne allergens with a T helper type 2 (Th2) immune response. Th2 cells produce interleukin (IL)-4, IL-5, IL-6, IL-9, and IL-13 and are involved in activating B cells for the production of immunoglobulins, particularly IgE. Th2 responses stimulate antibody-mediated responses and activation of mast cells and eosinophils. All of these factors result in the main pathophysiologic components of the asthmatic response — airway inflammation, bronchial airway hyper-responsiveness, airway remodeling, and reversible airway obstruction.
“Severe airway obstruction resulting from inflammation, bronchoconstriction, and excessive mucus production is at the heart of the gas exchange abnormalities and symptomatology in children with acute severe asthma.”6 The increase in airway resistance leads to an increase in the work of breathing and reductions in FEV1 and FEV1/FVC.6,20 In acute severe asthma, transpulmonary pressures greater than 50 cm H2O are not uncommon.19 As the degree of airway obstruction worsens, expiration becomes active rather than passive; air flow rates are low and expiratory times become progressively longer. Inspiration often occurs before termination of the previous expiration, resulting in air-trapping and lung hyperinflation and, because of this, residual volume and functional residual capacity is increased.20
Air-trapping and lung hyperinflation lead to an intrinsic positive end-expiratory pressure (PEEPi), or auto-PEEP, a phenomenon also termed dynamic hyperinflation. Dynamic hyperinflation has several adverse effects in the cardiovascular and respiratory systems. The increased lung volumes shift tidal breathing to a less compliant portion of the pressure volume curve. In addition, flattening of the diaphragm produces additional mechanical disadvantage.21 Dynamic hyperinflation also results in premature closure of the airways, which produces a further increase in air resistance, thereby worsening gas exchange. These factors collectively increase the work of breathing and increase the physiologic dead space. The gas exchange abnormalities produced by dynamic hyperinflation result in ventilation-perfusion mismatch.6
The effects of dynamic hyperinflation on cardiorespiratory interactions are quite complex.22 Right ventricular afterload is increased by a combination of factors, including lung hyperinflation (increased pulmonary vascular resistance), hypoxic pulmonary vasoconstriction, and acidosis. During expiration, the increase of intrathoracic pressure secondary to dynamic hyperinflation impedes systemic venous return, worsening left ventricular preload. During inspiration, the exaggerated negative intrathoracic pressure required to overcome airway resistance increases left ventricular afterload.6,22 These changes can be detected clinically as an increase in the pulsus paradoxus.23,24 Pulsus paradoxus is a valuable clinical sign of disease severity in children with asthma.1,24
Clinical Presentation and Assessment
The onset of asthma symptoms may result from exposure to “triggers” such as exercise, air pollutants, weather conditions, viral infections of the upper respiratory tract (particularly rhinovirus and respiratory syncytial virus), or allergen exposure, all of which increase inflammation in the lower airways.2
The presentation of acute severe asthma varies by severity, asthmatic trigger, and patient age.2 This variability often leads to poor recognition of the severity of illness, which, in turn, results in greater morbidity.25,26 The clinical examination can be misleading, and key clinical features must be taken into consideration when assessing a patient with acute asthma. 5,25-27 (See Table 2.)
Table 2. Markers of Acute Severe Asthma

The immediate assessment of patients with acute asthma exacerbations should include the degree of respiratory distress (ability to speak, respiratory rate, use of accessory muscle, air entry, inspiratory:expiratory ratio), degree of hypoxia (cyanosis, pulse oximetry, level of consciousness), and cardiovascular status (heart rate and rhythm, pulsus paradoxus, blood pressure).
Accessory muscle use, wheeze, and tachypnea might diminish as the patient tires.5 This may be misinterpreted as an improvement, when in fact the patient has worsened. Clinicians must pay close attention to these changes.
There are many “asthma scores” based on these observations that have been developed to assess the severity of the asthma exacerbation; at least 18 different “asthma scores” are available.3,28,29 The Becker asthma score is a quick assessment of severity by using respiratory rate, wheezing, inspiratory:expiratory ratio, and accessory muscle use.30 (See Table 3.) A score greater than 4 is considered moderately severe status asthmaticus, while a patient with a score of 7 or greater may be admitted to the pediatric intensive care unit (PICU). A more detailed asthma score provided by the National Heart, Lung, and Blood Institute is given in Table 4.31 The emergency physician also needs to consider the wide range of potential complications, as attention to these problems when assessing and managing acute asthma might significantly improve outcome.5 (See Table 5.)
Table 3. Assessing Asthma Severity Becker Asthma Score
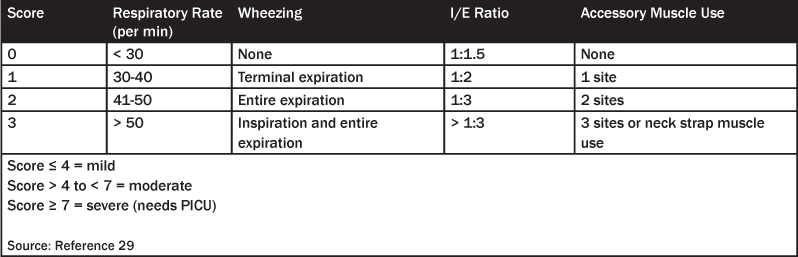
Table 4. Asthma Severity
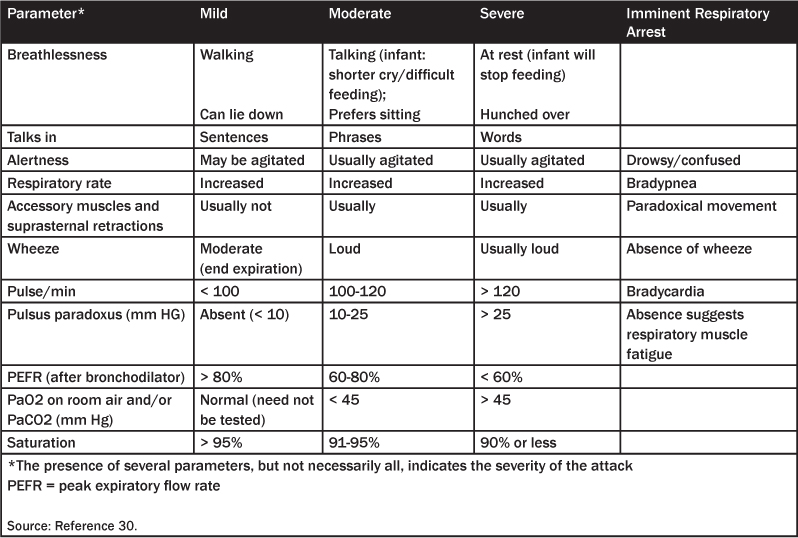
Table 5. Asthma Complications

Pulse oximetry may be useful to differentiate between patients who are likely to improve with therapy and those who are likely to progress to respiratory failure.6 An increase in oxygen saturation following albuterol nebulization predicts patients who are likely to improve, and respiratory failure rarely occurs in patients with an initial oxygen saturation greater than 92%.6 The degree of hypoxemia significantly correlates with the degree of airway obstruction, as determined by FEV1.32 Furthermore, hypoxemia appears to be an independent risk factor for both hospitalization and increased length of stay.33-35
Hypoxemia is multifactorial in origin, resulting from a combination of factors including ventilation-perfusion mismatch, alveolar hypoventilation, and hypercarbia. Significant hypoxemia, however, is relatively uncommon in children with acute severe asthma.6,19,35 Therefore, the presence of significant hypoxemia should alert the physician to search for some additional underlying cause, such as atelectasis secondary to mucous plugging, pneumonia, or pneumothorax.5,6
The degree of airway obstruction is rapidly determined by assessment of pulmonary function using the FEV1 and the peak expiratory flow rate (PEFR). Although FEV1 is considered to be the more reliable test, the PEFR is more easily obtained. Unfortunately, the correct performance of both tests is extremely dependent on the patient’s cooperation and effort; the values obtained may vary widely and may not be reliable. PEFR and FEV1 less than 30-50% of predicted, or of the patient’s personal best, indicate severe airflow obstruction.6,27 If the child is in significant distress, the use of pulmonary function tests may need to be deferred.
Monitoring carbon dioxide levels, invasively or non-invasively, might be useful. Although initial PaCO2 may be below normal levels, a progressive increase in PaCO2 is considered an early warning sign of severe airway obstruction and impending respiratory failure.6
Metabolic acidosis is well described in patients with acute severe asthma.36,37 One reported cause of metabolic acidosis is accumulation of lactic acid, presumably from a prolonged and marked increased work of breathing; additional factors include tissue hypoxia secondary to oxygen supply/demand imbalance in respiratory muscles, dehydration accompanying acute severe asthma from poor oral intake and increased insensible losses, and decreased cardiac output associated with hyperinflation.6 These causes are often labeled as type A and represent impairment in oxygen delivery as the cause of lactic acidosis. Other authors have shown that lactic acidosis is common in acute severe asthma but is primarily type B lactic acidosis, which occurs in the presence of normal oxygen delivery.36 Type B lactic acidosis is often seen with the use of beta-adrenergic agents to treat acute severe asthma, which leads to derangements in glucose metabolism and the development of type B lactic acidosis.36,38,39 Support for this mechanism includes clinical observations demonstrating a resolution of lactic acidosis with decreasing or discontinuation of bronchodilator therapy.36 Lactic acidosis developing during acute severe asthma is important to recognize because the compensatory hyperventilation that ensues to maintain body pH may lead to increased dyspnea. Lactic acid-induced hyperventilation has been mistaken as a sign of worsening airway obstruction, leading to inappropriate escalation of bronchodilator therapy, and has contributed to respiratory failure and the institution of mechanical ventilation.36
The mechanism for lactic acidosis during acute severe asthma is important to determine because effective treatment must address the underlying cause. If it is determined that the lactic acidosis is primarily type B associated with normal oxygen delivery and in the presence of adequate ventilation, a reduction in beta-adrenergic therapy may be appropriate.36
Hypokalemia is the most common electrolyte abnormality in children with acute severe asthma and is a well-recognized complication of beta-agonist administration.38,40,41 In addition, glucocorticoids used in the management of asthma can result in unwanted mineralocorticoid effects, leading to hypokalemia.40,41
Serum potassium levels should be monitored and, if needed, replaced if sufficiently low. Oral supplementation should be avoided due to the patient’s respiratory distress and supplementation should be given intravenously.
Another common abnormality seen is hyperglycemia. Treatment with beta-agonists and systemic steroids has been associated with changes in glucose metabolism.42 The resultant hyperglycemia may result in osmotic diuresis and dehydration; this would be aggravated by utilization of intravenous fluid supplements containing glucose, a common practice during hospitalization of children.
Complicating the hyperglycemia, fluid balance abnormalities are also seen in children with acute severe asthma, and these children are often dehydrated because of increased insensible fluid losses from the respiratory tract, coupled with poor oral intake of fluids. Dehydration may produce thicker, more tenacious bronchial secretions, leading to worsening bronchial mucous plugging. Although the majority of children require intravenous fluid rehydration, an inappropriate type of fluid (hypotonic or glucose-containing) or overzealous fluid administration may lead to fluid retention and pulmonary edema.6,42-45 Children with acute severe asthma may have elevated plasma antidiuretic hormone levels and are at risk for hyponatremia and fluid overload if given large volumes of hypotonic fluid.44-46 In addition, the high negative transpulmonary pressures associated with acute severe asthma promote fluid accumulation around the respiratory bronchioles, leading to pulmonary edema and worsening respiratory status.6,47
Management
Any child with acute severe asthma requires cardiorespiratory monitoring and close attention to fluid balance and neurologic status. A comfortable and supportive environment can ameliorate the situational anxiety, although hypoxemia and anxiety will also lead to agitation and restlessness. Sedatives should be avoided in the nonintubated patient with acute severe asthma, as it may cause hypoventilation and worsen the respiratory distress leading to failure.
There has been a great deal of interest in phenotypic classification of severe or difficult to treat asthma.4 Although childhood asthma is generally an allergic, eosinophilic inflammatory disease, many patients with severe asthma have significant neutrophil inflammation.4,48,49 Neutrophilic inflammation may be due to chronic infection with organisms such as Mycoplasma or Chlamydia.50,51 While antibiotics are not recommended in the therapy of acute asthma, this recent evidence suggests that antibiotic therapy targeted to patients with evidence of atypical bacterial infection may confer additional advantage to standard therapy alone.50,51 Additional use of antibiotics involves using macrolide antibiotics as immunomodulatory drugs that can reduce neutrophil inflammation and decrease asthma severity and the need for corticosteroids.4,50
Oxygen Therapy
Hypoxemia is always present in life-threatening asthma and is a major contributing factor to asthma death.4,52 The administration of supplemental oxygen is considered standard therapy for children with acute severe asthma/status asthmaticus.6,31 (See Table 6.) While oxygen therapy is essential, unregulated oxygen therapy, the provision of high amounts of oxygen beyond what is necessary to obtain physiologic oxygen saturation levels, has been shown to be unnecessary and potentially harmful.4,53,54 Giving 100% oxygen can depress compromised ventilation and increase the partial pressure of carbon dioxide (PaCO2). Therefore, some authors have recommended that only sufficient oxygen be administered to maintain peripheral oxygen saturation (SpO2) between 89-94%.4 It must be remembered that some bronchodilators, particularly the beta-agonists, reduce hypoxic pulmonary vasoconstriction and, thus, worsen hypoxemia by increasing V/Q mismatch.6 Therefore, these medications should be administered concurrently with supplemental oxygen.6,52
Table 6. Grades of Evidence of Interventions in Acute Severe Asthma

Systemic Corticosteroids
With the recognition that airway inflammation plays a prominent role in the pathophysiology of acute severe asthma, corticosteroids are standard treatment for children with acute severe asthma. (See Table 6.) Based on the wealth of available evidence, expert opinion and published guidelines recommend the administration of corticosteroids in the management of acute severe asthma/status asthmaticus within the initial 48 hours of treatment.2,6,31 A 2003 Cochrane Database review supports improved outcomes for children who receive corticosteroids early in their emergency department course.55 Corticosteroids speed the resolution of airflow obstruction, potentiate the effects of beta-agonist therapy, and have the potential to decrease hospitalization for sicker patients.
The optimal dosing of systemic corticosteroids for children with acute severe asthma remains an unresolved issue.6,56 Several studies suggest that high-dose corticosteroid therapy offers few advantages over low-dose corticosteroids in the treatment of acute severe asthma.6 Typically, oral or intravenous corticosteroids (e.g., methylprednisolone) have been given at a dose of 1-2 mg/kg as frequently as every 6 hours during an acute severe asthma attack. Although corticosteroids are often administered intravenously, data do not show that this is more effective than oral steroids, and there are no data that show that giving prednisone 2 mg/kg/day (80 mg maximum for older children and adults) is less effective than higher doses.4 (See Table 7.) Oral steroids are generally not recommended for children with acute severe asthma and impending respiratory failure because of the increased risk of intubating a patient with a full stomach.
Table 7. Common Drugs and Doses in Acute Severe Asthma

Significant side effects may occur with the use of steroids, particularly if used at a high dose; these include hyperglycemia, hypertension, and acute psychosis.2,57 Prolonged steroid use may cause immunosuppression, hypothalamic-pituitary-adrenal axis suppression, osteoporosis, myopathy, and weakness.2,4,58,59
Currently available evidence does not support the use of inhaled steroids in lieu of systemic corticosteroids.6 While it is most common to discontinue inhaled corticosteroids in patients with acute severe asthma, the concomitant use of inhaled steroids along with systemic steroids may provide additional benefit for some, but not all, patients.60,61
Beta-Adrenergic Agonists
Table 7 provides details on the bronchodilators currently used in the management of acute severe asthma. Beta-2-adrenergic receptor agonists have emerged as the single most potent class of bronchodilator available.26,62 Non-selective agonists such as isoproterenol or epinephrine are associated with a higher incidence of significant side-effects (i.e., myocardial ischemia) and are often not used.6,62,63
Subcutaneous epinephrine has been used for decades for the treatment of acute severe asthma.6,62 The use of subcutaneous epinephrine has fallen out of favor because of the widespread availability, ease of administration, and efficacy of the newer beta-adrenergic agonists such as albuterol. However, subcutaneous/IM epinephrine continues to have a role in the treatment of critically ill children with impending respiratory failure secondary to acute severe asthma.64-66 Subcutaneous/IM administration of epinephrine should be considered in children who are rapidly decompensating despite inhaled beta-adrenergic agonists and in children who are unable to cooperate with inhalation therapy secondary to anxiety, altered mental status, or apnea.6 Subcutaneous epinephrine (0.01 mg/kg) may be administered every 20 minutes for three doses. Severe airflow obstruction may be relieved by subcutaneous/IM epinephrine to a degree sufficient to allow adequate delivery of aerosolized beta-adrenergic agonists to the distal airways, thereby allowing these agents to take effect.
For the majority of patients with acute severe asthma, inhaled beta-adrenergic agonists have superior efficacy compared with subcutaneous epinephrine.1,6 Furthermore, although the duration of action appears to be dose-dependent, sequential inhalation of these agents (most commonly albuterol) produces a more rapid, greater improvement in airway obstruction than nebulizing higher doses less frequently.6,67 Based on the available evidence, the consensus is that frequent albuterol nebulization should be considered standard therapy for children presenting with acute severe asthma.6,31 (See Table 6.)
Several studies have compared the efficacy of small-volume nebulizers versus metered-dose inhalers (MDI) with spacers for the treatment of acute asthma exacerbation in children.68 Although nebulizers allow for the concurrent administration of supplemental oxygen, some studies have suggested close to 90% of the drug is lost to the atmosphere.1,6,64 The available evidence suggests that there are no differences between MDIs with spacers compared with nebulizers, and either option appears reasonable in the emergency department setting.68,70 However, evidence is growing that the use of an MDI with a valved holding chamber is more effective than nebulized albuterol in young children with moderate to severe asthma exacerbations.1,4,71
Recent studies have evaluated the use of Breath Actuated Nebulizer (BAN).72,73 These studies have revealed that BANs allow for a lower amount of albuterol while decreasing asthma severity scores, decreasing length of stay in the emergency department, and reducing the rate of hospitalization.73 A BAN may prove to be an acceptable alternative to conventional nebulizer use.73
Continuous albuterol nebulization has been shown to be more effective in children with acute severe asthma and impending respiratory failure.1,62,74,75 Continuous nebulization provides sustained stimulation of the beta-adrenergic receptors in the airways, thereby preventing the rebound bronchospasm that can occur with intermittent nebulization.6 Although continuous nebulization of albuterol can be safely done, it should be done only in a closely monitored setting. Side effects occur with beta-agonists and include hypokalemia, cardiac arrhythmias, cardiac ischemia, tremor, ventilation-perfusion mismatch, lactic acidosis, and increased mucus secretion.1,2,4,6,62 It is important to remember that beta receptors in the lung are down regulated and rapidly saturated with the use of inhaled beta-agonists.4 This may result in no clinically beneficial effect of higher doses of beta-agonists but increasing side effects.4,76
Albuterol exists as a 50:50 mixture of two mirror-image enantiomers: the active R-albuterol and S-albuterol. Previously thought to be an inert compound, S-albuterol may exaggerate airway hyperresponsiveness and result in bronchoconstriction; it may also have a proinflammatory effect.1,70,77 It appears that administration of only the R-enantiomer of albuterol would be an appropriate treatment rationale. Levalbuterol (Xopenex R) is pure R-albuterol and is available for nebulization. Unfortunately, the majority of studies in children with exacerbations of asthma suggest that there is not a clinical benefit to the use of levalbuterol versus racemic albuterol.1,78-80 No recommendation regarding the use of the much more expensive levalbuterol in children with acute severe asthma can presently be made.1,6
Intravenous beta-agonists should be considered in patients unresponsive to treatment with continuous nebulization, as well as those for whom nebulization is not feasible (i.e., intubated patients, patients with prohibitively poor air entry). Decreased tidal volume and/or near complete airway obstruction in severe status asthmaticus may prevent effective aerosolized bronchodilator delivery. Terbutaline is the current intravenous beta-agonist of choice in the United States, while other countries use albuterol intravenously. Most adverse effects of beta-agonists in asthma are of cardiovascular nature, including tachycardia, increased QTc interval, dysrhythmia, hypertension, as well as hypotension. Induced severe hypokalemia may precipitate arrhythmias. Neither albuterol nor terbutaline, however, is known to cause clinically significant cardiac toxicity when used for pediatric status asthmaticus.81,82
The use of intravenous terbutaline is not recommended by the current National Asthma Education Program (NAEP) Expert Panel Report.31
Anticholinergic Therapy
The autonomic nervous system is involved in the regulation of airway smooth muscle tone and mucus secretion. The parasympathetic nerve fibers, which are confined to the larger, central airways, stimulate bronchoconstriction and mucus secretion (mediated through the neurotransmitter acetylcholine).6 In contrast, the sympathetic nerve fibers are distributed more peripherally in the smaller airways and stimulate bronchodilation. This dual-innervation suggests that a therapeutic strategy aimed at both the cholinergic and adrenergic pathways would be beneficial in the treatment of acute severe asthma. Indeed, this is the case, and, when used in conjunction with beta-agonists, anticholinergic agents are now standard of care in the treatment of acute asthma in children.1,4,6 (See Table 6.) A Cochrane review strongly suggests significant clinical benefit to the concomitant use of a beta-agonist and inhaled anticholinergic for treating acute severe asthma, and some studies have indicated an added benefit with minimal risk in the most severely ill patients with asthma.83,84 The most commonly used compound is ipratropium bromide, a quaternary derivative of atropine. (See Table 7.)
Magnesium
Magnesium is safe, and a meta-analysis has shown that when given systematically, it confers additional benefit in the treatment of life-threatening asthma.85 Magnesium might be effective in acute asthma through a variety of mechanisms.5,6
Magnesium administration may serve to replace an underlying magnesium deficiency. Multiple studies have shown that patients with acute severe asthma have underlying hypomagnesemia, and frequent beta-agonist therapy has been demonstrated to result in decreased magnesium levels.6
Magnesium may act as a pharmacologic agent via one of several mechanisms.5,6 It is clear that magnesium acts principally as a calcium antagonist, directly inhibiting calcium uptake in smooth muscle cells, thereby resulting in smooth muscle relaxation. Magnesium acts at the neuromuscular junction by decreasing acetylcholine release, diminishing the depolarization action of acetylcholine, and depressing the excitability of smooth muscle membranes.6,89,90 Magnesium decreases histamine release from mast cells.90-92 Finally, magnesium decreases superoxide production by neutrophils, thereby producing anti-inflammatory properties.90,91
A single dose of intravenous magnesium sulfate administered to patients with severe acute asthma has been shown to be effective.5
Several small, prospective, randomized, controlled trials comparing intravenous magnesium and placebo in children presenting to the emergency department with acute severe asthma have been performed.91,93-96 Not all of these studies demonstrated beneficial effects with magnesium; a subsequent meta-analysis concluded that magnesium sulfate provides additional benefit to children with acute severe asthma when added to a regimen of frequent, nebulized beta-adrenergic agonists and corticosteroids.92 Magnesium sulfate should be tried, at least a single dose, when respiratory failure is impending.26,31 (See Table 6.)
The current dose and frequency of administration have not been adequately defined. (See Table 7.) There is evidence to suggest that increasing the serum magnesium level to greater than 4 mg/dL is necessary to produce bronchodilation.26,97 Onset of action is within minutes and the effects last approximately 2 hours.6,97 Side effects appear dependent upon serum magnesium concentration; mild effects include nausea, vomiting, facial flushing, and dry mouth. At serum magnesium levels greater than 12 mg/dL, loss of deep tendon reflexes, muscle weakness, and respiratory depression, as well as cardiac conduction defects, may be seen.97
Theophylline/Aminophylline
Theophylline has been used as a bronchodilator for the treatment of asthma for many years and was once considered the bronchodilator of choice for the management of acute asthma.100 The therapeutic effects of aminophylline in asthma include bronchodilation and improved diaphragmatic contraction.101 Therapeutic effects occur at blood levels up to 20 mg/dL. Levels above this range may occur and cause toxic effects. These include diuresis, hypokalemia, insomnia, tachycardia, cardiac arrhythmias, convulsions, and sudden death.101,102 In many centers, aminophylline has been supplemented by the use of beta-agonists for the treatment of acute severe asthma.
The NAEP Expert Panel has revised its original guidelines and concluded that methylxanthines are not recommended for treatment of hospitalized children with status asthmaticus.31 However, current recommendations proscribing the use of theophylline are not based on analysis of treatment data in critically ill children with acute severe asthma.6
Theophylline offers several potential advantages for the treatment of acute severe asthma in the PICU population. Theophylline produces bronchodilation and improves air flow without adversely affecting ventilation-perfusion matching.103 Theophylline’s diuretic effect may reduce excess alveolar fluid and microvascular permeability.6,103,104 Finally, theophylline increases respiratory drive, improves mucociliary clearance, reduces pulmonary vascular resistance, and improves contractility of the diaphragm, all of which may benefit the child with acute severe asthma.101,103 For these reasons, there has been renewed interest in intravenous aminophylline.101 Current guidelines assert that there may be a role for aminophylline in particularly severe asthma.101 Aminophylline use in children may be appropriate if children have acute severe asthma and the response to aggressive therapy (inhaled bronchodilators and glucocorticoids) is poor. This recommendation is based on limited data and further work is needed.101,105 Avoidance of toxic theophylline levels is critical. (See Table 7.) One study recommends an intravenous loading dose of 7 mg/kg, followed by age-adjusted rates of 0.5-0.65 mg/kg/hr.105 Theophylline levels are kept between 12-17 mg/mL.
Helium-Oxygen Mixtures
Helium-oxygen mixtures (heliox) are available in concentrations of 80% helium/20% oxygen and 70% helium/30% oxygen. Helium mixtures have a low density compared with air (the 80/20 mixture is approximately one-third the density of air).26 Heliox is a temporary measure to reduce respiratory resistive work and forestall muscle fatigue until airway obstruction improves with conventional therapy.26 Evidence suggests heliox is more effective for the sicker patient.106 The beneficial effects of heliox seem most efficacious when used early in the disease course (less than 24 hours).106 The addition of heliox to the treatment regimen for acute asthma is not warranted for all patients.107 Patients requiring FiO2 greater than 0.30 will not tolerate the use of heliox, as there is no way to titrate beyond the 70/30 mixture.
Heliox may also be effective as the driving gas for nebulized bronchodilators.108,109 The low density of helium improves the deposition of aerosolized particles in the airways, which can lead to a more rapid response to treatment and more significant improvement in airway function.26
In the pediatric patient who requires conventional mechanical support of ventilation, heliox may assist in lowering peak inspiratory pressure (PIP) and improving blood gas pH and partial pressure of CO2.110 This benefit is lost once PIP is less than 30 cm H2O.
Ketamine
Ketamine, a dissociative anesthetic agent, causes bronchodilation by a combination of factors, including drug-induced release of endogenous catecholamines, inhibition of vagal tone, and direct smooth muscle relaxation.111 Although ketamine has been used successfully for the treatment of refractory bronchospasm in intubated patients, there is controversy concerning its use in non-intubated patients because it may cause increased pulmonary secretions and laryngospasm.112-115 Literature exists supporting its use as an efficacious adjunct to standard therapy in the treatment of children with acute severe asthma and impending respiratory failure.116-117Ketamine should be used cautiously in unintubated patients and only in a monitored setting.
Leukotriene-Modifying Agents
The leukotrienes are biologically active fatty acids generated from arachidonic acid by the enzyme 5-lipoxygenase; 5-lipoxygenase generates leukotriene A4 (LTA4) from arachidonic acid. LTA4 is metabolized to LTC4, LTD4, and LTE4, which are referred to as the slow-reacting substances of anaphylaxis.118-120 The leukotrienes produce bronchoconstriction, stimulate mucus secretion, decrease mucociliary clearance, increase vascular permeability, and recruit eosinophils and basophils into the airway, thereby perpetuating airway inflammation.118 The leukotrienes are 1,000 times more potent than either histamine or methacholine in airway challenge tests.121 Activation of the leukotriene pathways during acute asthma exacerbations, as determined by urinary LTE4 levels, appears to correlate with the degree of airway obstruction.122
Several different leukotriene-modifying agents are currently available, each working via different mechanisms. The leukotriene receptor antagonists (LTRA), montelukast and zafirlukast, are the only leukotriene-modifying agents approved for use in children. At present, the main indications for LTRA in pediatric asthma are as add-on therapy to inhaled corticosteroids or as initial controller therapy in children with mild asthma, especially those who cannot or will not use inhaled corticosteroids.120
There has been growing interest in using these agents for the treatment of acute severe asthma, although few studies exist.120,123,124 (See Table 6.)
Mechanical Ventilation
Despite maximum medical therapy, mechanical ventilation may be needed to manage respiratory insufficiency resulting from airway obstruction and respiratory muscle fatigue in acute severe asthma.4,125 Acute severe asthma leading to respiratory failure is an important cause of morbidity and mortality because of the high risk of barotrauma and cardiovascular instability associated with the positive pressure mechanical ventilation in these children.6,126 The goal of mechanical ventilation in acute severe asthma is twofold: to provide sufficient gas exchange to ensure survival until reversal of airway obstruction is accomplished and to minimize complications associated with mechanical ventilation.
In an effort to unload the respiratory muscles during a severe asthma exacerbation, noninvasive mechanical ventilation using bilevel positive airway pressure (BiPAP) may be used.5,127,128 The presence of air-trapping in the alveoli results in auto-positive end-expiratory pressure (auto-PEEP), which requires the patient to generate a higher negative inspiratory force to overcome the auto-PEEP and generate inspiratory flow. The addition of EPAP (end-expiratory pressure) should allow for equilibration of pressure between the mouth and the alveoli, reducing the amount of force the patient needs to generate to achieve inspiratory flow.127 The use of an inspiratory pressure (IPAP) in BiPAP helps to reduce the work of the respiratory muscles.
Early initiation of noninvasive ventilation in addition to standard therapy may be effective in decreasing work of breathing in children with acute severe asthma.127,128 It may be safe and well tolerated by patients for at least 24 hours and may prevent the need for intubation.128 Clinicians using noninvasive ventilation for acute severe asthma must be skilled in noninvasive ventilation and be aware of complications such as barotrauma and hemodynamic instability. These patients must be closely monitored; their condition may abruptly worsen, so the medical team must be ready for immediate intubation.127
There are few absolute indications for tracheal intubation in children with status asthmaticus (e.g., coma, cardiac arrest), although failure to maintain adequate oxygen saturations, a worsening metabolic acidosis, and decreasing mental status are all signs that respiratory arrest is imminent. The decision to tracheally intubate should be based on the clinical examination and not the results of an arterial blood gas. These children should be considered high risk for intubation, and a rapid-sequence intubation technique should be performed by the most experienced physician available. Ketamine (2 mg/kg intravenous [IV]) is an excellent choice for an induction agent because of its bronchodilatory properties.129 Neuromuscular blockade with either succinylcholine (if there are no contraindications to its use) or a nondepolarizing agent such as vecuronium or rocuronium produces acceptable conditions for laryngoscopy and tracheal intubation.129
These children will likely require high inspiratory pressures, so the use of a cuffed endotracheal tube is preferable.6,126,130
More than one-half of the complications in patients requiring mechanical ventilation for acute severe asthma occur at or around the time of tracheal intubation and include hypoxemia, hypotension, and cardiac arrest.130 Hyperventilation should be avoided, and hypotension should improve with volume resuscitation and slowing the respiratory rate to avoid further air-trapping and dynamic hyperinflation. A tension pneumothorax should be considered if these measures fail to relieve hypotension and hypoxemia. In these cases, needle thoracentesis is life-saving.
The goal of mechanical ventilation is to obtain and maintain acceptable oxygenation, avoid complications, and allow time for the corticosteroids and bronchodilators to reduce the bronchospasm and inflammation.
Mechanical ventilation should not be targeted toward the results of an arterial blood gas.6 Utilizing low tidal volumes and respiratory rates and tolerating the resultant hypercapnia dramatically reduces the frequency of barotrauma and death.6,131 In children, strategies that use low tidal volumes (8-10 mL/kg or less), short inspiratory times (0.75-1.5 sec), long expiratory times, and lower than normal respiratory rates result in improved survival.6,126,132 The degree of hypercapnia that can be safely tolerated is not known.6,133
The most appropriate mode of mechanical ventilatory support may differ among patients and their stage of illness. Pressure control, volume control, and pressure support have all been used in children with acute severe asthma.3,19,125 Each mode of ventilation has its advantages and disadvantages. Most authors prefer pressure-controlled ventilation.3,6 The use of positive end-expiratory pressure (PEEP) in these patients is controversial; however, low-level PEEP will minimize dynamic airway collapse and decrease trigger work in spontaneously breathing patients.19,130
The use of high-frequency oscillatory ventilation in children with respiratory failure secondary to status asthmaticus has been reported.134 Extracorporeal life support may be life-saving in children with refractory acute severe asthma.135,136
Finally, inhaled nitric oxide has been utilized in children with refractory status asthmaticus.137,138 The rapid improvement in ventilation described may be related to the action of inhaled nitric oxide as a bronchodilator, direct vascular effect, or both.
Volatile Anesthetics
Inhalational anesthetics are occasionally used for the treatment of status asthmaticus and acute respiratory failure.6 The bronchodilatory properties of these agents are well-known, and proposed mechanisms include direct stimulation of the beta-adrenergic receptor, direct relaxation of bronchial smooth muscle, inhibition of the release and action of bronchoactive mediators (e.g., histamine, acetylcholine), and depression of vagally mediated airway reflexes.139 Halothane appears to be particularly effective, although concerns regarding its potential toxicity, including direct myocardial depression, hypotension, and arrhythmias, have limited its use in this setting.140 These adverse effects may be further potentiated in children with status asthmaticus who will have some degree of hypoxia, hypercapnia, and acidosis and who are frequently managed with the concomitant administration of beta-adrenergic agents and/or theophylline.6
The use of isoflurane in status asthmaticus and acute respiratory failure offers several advantages over halothane.141 Isoflurane produces less myocardial depression and is less arrhythmogenic compared with halothane. Given its role as a general anesthetic, concomitant administration of sedation/analgesia and neuromuscular blockade is not necessary. Isoflurane has been used with some success in both children and adults with status asthmaticus refractory to conventional therapy.141,142
Summary/Conclusion
The burden of childhood asthma continues to rise, with increasing rates of asthma prevalence, severity, and death.143 Background asthma management remains suboptimal in children needing hospitalization. This is despite our improved understanding of the pathophysiology of asthma and the availability of effective anti-inflammatory therapy.
Children will continue to present to the ED with acute severe asthma. The emphasis in treatment remains on the aggressive use of inhaled beta-agonist and steroid therapy to abort the acute attack and decrease both the mortality rate and need for hospitalization. For children who fail to respond, adjunctive therapies and advances in ventilatory management can lead to improved outcomes in these patients.
References
1. Mannix R, Bachur R. Status asthmaticus in children. Curr Opin Pediatr 2007;19:281-287.
2. Saharan S, Lodha R, Kabra SK. Management of status asthmaticus in children. Indian J Pediatr 2010;77: 1417-1423.
3. Newth CJL, Meert KL, Clark AE, et al. Fatal and near-fatal asthma in children: The critical care perspective. J Paediatr Resp Rev 2012;161:214-221.
4. Rubin BK, Pohanka V. Beyond the guidelines: Fatal and near-fatal asthma. Paediatr Resp Rev 2012;13:106-111.
5. Holley AD, Boots RJ. Review article: Management of acute severe asthma and near-fatal asthma. Emergency Medicine Australasia 2009;21:259-268.
6. Wheeler DS, Page K, Shanley TP. Status asthmaticus, in Wheeler DS et al (eds.) The Respiratory Tract in Pediatric Critical Illness and Injury. London, Springer-Verlag, 2009:169-193.
7. Malmstrom K, Kaila M, Kajosaari M, et al. Fatal asthma in Finnish children and adolescents 1976-1998: Validity of death certificated and a clinical description. Pediatr Pulmonol 2007;42:210-215.
8. Triasih R, Duke T, Robertson CF. Outcomes following admission to intensive care for asthma. Arch Dis Child 2011;96:729-734.
9. Maffei FA, van der Jagt EW, Powers KS, et al. Duration of mechanical ventilation in life-threatening pediatric asthma: Description of an acute asphyxial subgroup. Pediatrics 2004;114(3):762-767.
10. Sedik HA, Barr RG, Clark S, et al. Prospective study of sudden-onset asthma exacerbations in children. Pediatr Emerg Care 2007;23(7):439-444.
11. Robertson CF, Rubinfeld AR, Bowes G. Pediatric asthma deaths in Victoria: The mild are at risk. Pediatr Pulmonol 1992;13:95-100.
12. Kravis, LP, Kolski GB. Unexpected death in childhood asthma. Am J Dis Child 1985;139(6):558-563.
13. Flether HJ, Ibrahim SA, Speight N. Survey of asthma deaths in the northern region, 1970-1985. Arch Dis Child 1990;65:163-167.
14. Jensen ME, Collins CE, Gibson PG et al. The obesity phenotype in children with asthma. Paediatric Respiratory Reviews 2011;12:152-159.
15. Bratton SL. Near fatal and fatal asthma in the PICU. AAP Grand Rounds 2012;28:46. 16. Wechsler ME, Shepard JO, Mark EJ. A 20-year-old woman with asthma a cardiorespiratory arrest. N Engl J Med 2007;356(20):2083-2091.
17. McFadden ER. Acute severe asthma. Am J Resp Crit Care Med 2003;168:740-759.
18. Busse WW, Lemanske RF. Asthma. N Engl J Med 2001;344(5):350-362.
19. Bohn D, Kisson N. Acute asthma. Pediatr Crit Care Med 2001;2(2):151-163.
20. Grover S, Jindel A, Bansal A, et al. Acute bronchial asthma. Indian J Pediatr 2011;78(11):1388-1395.
21. McCool FD, Tzelepis GE. Dysfunction of the diaphragm. N Engl J Med 2012;366(10):932-942.
22. Anas, NG, Bronicki RA. Cardiopulmonary interaction. Pediatr Crit Care Med 2009;10(3):313-322.
23. McGregor M. Pulsus paradoxus. N Engl J Med 1979;301(9):480-482.
24. Frey B, Freezer N. Diagnostic value and pathophysiologic basis of pulsus paradoxus in infants and children with respiratory disease. Pediatr Pulmonol 2001;31:138-143.
25. Lugogo NL, MacIntyre NR. Life-threatening asthma: Pathophysiology and management. Respir Care 2008;53(6):726-735.
26. Chipps BE, Murphy KR. Assessment and treatment of acute asthma in children. J Pediatr 2005;147:288-294.
27. Qureshi F. Management of children with acute asthma in the emergency department. Pediatr Emerg Care 1999;15(3):206-214.
28. Gorelick MH, Stevens MW, Schultz TR, et al. Performance of a novel clinical score, the Pediatric Asthma Severity Score (PASS), in the evaluation of acute asthma. Acad Emerg Med 2004;11:10-18.
29. Ducharme FM, Chalut D, Ploynick L, et al. The Pediatric Respiratory Assessment Measure: A valid clinical score for assessing acute asthma severity from toddlers to teenagers. J Pediatr 2008;152:476-480.
30. Becker AB, Nelson NA, Simons FE. The pulmonary index. Assessment of a clinical score for asthma. Am J Dis Child 1984;138:574-576.
31. National Heart, Lung and Blood Institute. Expert panel report 3: Guidelines for the diagnosis and management of asthma-full report 2007. Available at: www.nhlbi.nih.gov/guidelines/asthma.
32. Sok D, Komatsu MK, Curvalho KV, et al. Pulse oximetry in the evaluation of the severity of acute asthma and/or wheezing in children. J Asthma 1999;36:327-333.
33. Morray B, Redding G. Factors associated with prolonged hospitalization of children with asthma. Arch Pediatr Adolesc Med 1995;149:276-279.
34. Keogh KA, MacArthur C, Parkin PC, et al. Predictors of hospitalization in children with acute asthma. J Pediatr 2001;139:273-277.
35. Keahey L, Bulloch B, Becker AB, et al. Initial oxygen saturation as a predictor of admission in children presenting to the emergency department with acute asthma. Ann Emerg Med 2002;40:300-307.
36. Meert KL, McCaulley L, Sarnaik AP. Mechanism of lactic acidosis in children with acute severe asthma. Pediatr Crit Care Med 2012;13:28-31.
37. Yousel E, McGeady SJ. Lactic acidosis and status asthmaticus: How common in pediatrics? Ann Allergy Asthma Immunol 2002;89(6):585-588.
38. Assadi FK. Therapy of acute bronchospasm complicated by lactic acidosis and hypokalemia. Clin Pediatr 1989;28(6):258-260.
39. Koul PB, Minarik M, Totapally BR. Lactic acidosis in children with acute exacerbation of severe asthma. Eur J Emerg Med 2007;14:56-58.
40. Kolski GB, Cunningham AS, Niemec PW, et al. Hypokalemia and respiratory arrest in an infant with status asthmaticus. J Pediatr 1988;112:304-307.
41. Tsai WS, Wu CP, Hsu YJ, et al. Life-threatening hypokalemia in an asthmatic treated with high dose hydrocortisone. Am J Med Sci 2004;377:152-155.
42. Chawla S, Seth D, Cortez J. Asthma and hyperglycemia. Clin Pediatr 2007;46(5):454-457.
43. Beck CE. Hypotonic versus isotonic maintenance intravenous fluid therapy in hospitalized children: A systemic review. Clin Pediatr 2007;46(9):764-770.
44. Arisaka O, Shimura N, Hosaka A, et al. Water intoxication in asthma assessed by urinary arginine vasopressin. Eur J Pediatr 1988;148:137-169.
45. Singleton R, Moel DI, Cohn RA. Preliminary observation of impaired water excretion in treated status asthmaticus. Am J Dis Child 1986;140:59-61.
46. Iikura Y, Odajima Y, Akazama A, et al. Antidiuretic hormone in acute asthma in children: Effects of medication on serum levels and clinical course. Allergy Proc 1989;10(30):197-201.
47. Stalcup SA, Melins RB. Mechanical forces producing pulmonary edema in acute asthma. N Engl J Med 1977;297: 592-596.
48. Fahy JV. Eosinophilic and neutrophilic inflammation in asthma: Insights from clinical studies. Proc Ann Thorac Soc 2009;6:256-259.
49. Zhang X, Moilanen E, Kankaanranta H. Beclomethasone, budesonide and fluticasone propionate inhibit neutrophil apoptosis. Eur J Pharmacol 2001;431:365-371.
50. Blasi F, Consentini R, Tarsia P, et al. Potential role of antibiotics in the treatment of asthma. Curr Drug Targets Inflamm Allergy 2004;3:237-242.
51. Rolins DR, Benther DA, Martin RJ. Update on infection and antibiotics in asthma. Curr Allergy Asthma Rep 2010;10:67-73.
52. Inwald D, Roland M, Kuitert L, et al. Oxygen treatment for acute severe asthma. BMJ 2001;323:98-100.
53. Rodrigo GS, Rodriquez VM, Pereqali V, et al. Effects of short-term 28% and 100% oxygen on PaCO2 and peak expiratory flow rate in acute asthma: A randomized trial. Chest 2003;124:1312-1315.
54. Chien JW, Ciufo R, Novak R, et al. Uncontrolled oxygen administration and respiratory failure in acute asthma. Chest 2001;117:728-733.
55. Smith M, Iqbal SM, Rowe BH, et al. Corticosteroids for hospitalized children with acute asthma. Cochrane Database Syst Rev 2003;1:CD002886.
56. Zhang L, Medoza RA. Doses of systemic corticosteroids in hospitalized children with acute asthma: A systemic review. J Paediatrics and Child Health 2006;42:179-183.
57. Klein-Gitelman MS, Pachman LM. Intravenous corticosteroids: Adverse reactions are more variable than expected in children. J Rheumatol 1998;25: 1995-2002.
58. Mehta R, Fisher LE, Segeleon JE, et al. Acute rhabdomyolysis complicating status asthmaticus in children. Pediatr Emerg Care 2006;22(8):587-591.
59. Goh AYT, Chan PWK. Acute myopathy after status asthmaticus: Steroids, myorelaxants or carbon dioxide. Respirology 1999;4:97-99.
60. Rowe BH, Bota GW, Fabris L, et al. Inhaled budesonide in addition to oral corticosteroids to prevent asthma relapse following discharge from the emergency department: A randomized controlled trial. JAMA 1999;281:2119-2126.
61. Upham BD, Mollen CJ, Scarfone RJ, et al. Nebulized budesonide added to standard pediatric emergency department treatment of acute asthma: A randomized, double-blind trial. Acad Emerg Med 2011;18:665-673.
62. Biarent D. Therapeutic strategies in near fatal asthma in children. Pediatr Pulmonology 2001;23(Suppl):90-93.
63. Baren JM, Zorc JS. Contemporary approach to the emergency department management of pediatric asthma. Emerg Clin N Am 2002;20(1):115-138.
64. Kornberg AE, Zuckerman S, Welliver JR, et al. Effect of injected long-acting epinephrine in addition to aerosolized albuterol in the treatment of acute asthma in children. Pediatr Emerg Care 1991;7:1-3.
65. Safdar B, Cone DL, Phan KT. Subcutaneous epinephrine in the pre-hospital setting. Prehosp Emerg Care 2001;5:200-207.
66. Sharma A, Madan A. Subcutaneous epinephrine vs. nebulized salbutamol in asthma. Indian J Pediatr 2001;68: 1127-1130.
67. Robertson CF, Smith F, Beck R, et al. Response to frequent low doses of nebulized salbutamol in acute asthma. J Pediatr 1985;106:672-674.
68. Amirav I, Newhouse MT. Metered-dose inhaler accessory devices in acute asthma. Arch Pediatr Adolesc Med 1997;151: 876-882.
69. Rubilar L, Castro-Rodriquez JA, Girardi G. Randomized trial of salbutamol via metered-dose inhaler with spacer vs. nebulizer for acute wheezing in children less than 2 years of age. Pediatr Pulmonol 2000;29:264-269.
70. Scarfone RJ, Friedlaender EY. Beta-2 agonists in acute asthma: The evolving state of the art. Pediatr Emerg Care 2002;18:442-447.
71. Castro-Rodriquez JA, Rodrigo GJ. Beta-agonists through metered-dose inhaler with valved holding chamber versus nebulizer for acute exacerbation of wheezing asthma in children under 5 years of age: A systematic review with meta-analysis. J Pediatr 2004;145:172-177.
72. Titus MO, Eady M, King L, et al. Effectiveness of a breath-activated nebulizer device on asthma care in the pediatric emergency department. Clinical Pediatrics 2012;51(12):1150-1154.
73. Arunthari V, Bruinsma RS, Lee AS, et al. A prospective, comparative trial of standard and breath-actuated nebulizer: Efficacy, safety, and satisfaction. Respiratory Care 2012;57(8):1242-1247.
74. Craig VL, Bigos D, Brilli RJ. Efficacy and safety of continuous albuterol nebulization in children with severe status asthmaticus. Pediatr Emerg Care 1996;12(1):1-5.
75. Camargo CA, Spooner C, Rowe BH. Continuous versus intermittent beta-agonists for acute asthma. Cochrane Database Syst Rev 2003;4:CD0001115.
76. Rodrigo GJ, Rodrigo C. Continuous vs intermittent beta-agonists in the treatment of acute adult asthma: A systematic review with meta-analysis. Chest 2002;122:160-225.
77. Johnsson F, Rydberg I, Aberg G, et al. Effects of albuterol enantiomers on in vitro bronchial reactivity. Clin Rev Allergy Immunol 1996;14:57-64.
78. Hardasmalani MD, DeBari V, Bilhoney WG, et al. Levalbuterol versus racemic albuterol in the treatment of acute exacerbations of asthma in children. Pediatr Emerg Care 2005;21:415-419.
79. Ralston ME, Euwema MS, Knecht KR, et al. Comparison of levalbuterol and racemic albuterol combined with ipratropium bromide in acute pediatric asthma: A randomized, controlled trial. J Emerg Med 2005;19:29-35.
80. Qureshi, Zaritsky A, Welch C, et al. Clinical efficacy of racemic albuterol versus levalbuterol for the treatment of acute pediatric asthma. Ann Emerg Med 2005;46:29-36.
81. Chiang VW, Burns JP, Rifai N, et al. Cardiac toxicity of intravenous terbutaline for the treatment of severe asthma in children: A prospective assessment. J Pediatr 2000;137:73-77.
82. Carroll CL, Schramm CM. Protocol-based titration of intravenous terbutaline decreases length of stay in pediatric status asthmaticus. Pediatr Pulmonol 2006;41:350-356.
83. Rodrigo GJ, Castro-Rodriquez JA. Anticholinergics in the treatment of children and adults with acute asthma: A systemic review with meta-analysis. Thorax 2005;60:740-746.
84. Plotnick LH, Ducharme FM. Acute asthma in children and adolescents: Should inhaled anticholinergics be added to beta-agonists? Am J Respir Med 2003;2:109-115.
85. Rowe BH, Camargo CA. The role of magnesium sulfate in the acute and chronic management of asthma. Curr Opin Pulm Med 2008;14:70-76.
86. Kakish KS. Serum magnesium levels in asthmatic children during and between exacerbations. Arch Pediatr Adolesc Med 2001;155(2):181-183.
87. Zervas E, Papatheodoron G, Psathakis K, et al. Reduced intracellular magnesium concentrations in patients with acute asthma. Chest 2003;123:113-118.
88. Bodenhamer J, Bergstrom R, Brown D, et al. Frequent nebulized beta-agonists for asthma: Effects on serum electrolytes. Ann Emerg Med 1992;21:1337-1342.
89. Monem GF, Kisson N, De Nicola L. Use of magnesium sulfate in asthma in childhood. Pediatr Ann 1996;25(3):136-144.
90. Fawcett WJ, Haxby EJ, Male DA. Magnesium: Physiology and pharmacology. British J Anaesthesia 1999;83(2):302-320.
91. Ciaralllo L, Brousseau D, Reinert S. Higher-dose magnesium therapy for children with moderate to severe asthma. Arch Pediatr Adoles Med 2000;154: 979-983.
92. Cheuk DKL, Chau TCH, Lee SL. A meta-analysis on intravenous magnesium sulphate for treating acute asthma. Arch Dis Child 2005;90:74-77.
93. Ciarallo L, Sauer AH, Shannon MW. Intravenous magnesium therapy for moderate to severe pediatric asthma: Results of a randomized, placebo-controlled trial. J Pediatr 1996;129:809-814.
94. Devi PR, Kumar L, Sighi SC, et al. Intravenous magnesium sulfate in acute severe asthma not responding to conventional therapy. Indian Pediatr 1997;34: 389-397.
95. Gurken F, Haspolat K, Bosnak M, et al. Intravenous magnesium sulfate in the management of moderate to severe acute asthmatic children non-responding to conventional therapy. Eur J Med 1999;6: 201-205.
96. Scarfone RJ, Loiselle JM, Jotte MD, et al. A randomized trial of magnesium in the emergency department treatment of children with asthma. Ann Emerg Med 2000;36:572-578.
97. Noppen M, Vanmack L, Impens N, et al. Bronchodilating effect of intravenous magnesium sulfate in acute severe bronchial asthma. Chest 1990;97:373-376.
98. Harker HE, Majcher TA. Hypermagnesemia in a pediatric patient. Anesth Analg 2000;91:1160-1162.
99. McGuire JK, Kulkerni MS, Baden HP. Fatal hypermagnesemia in a child treated with megavitamin/megamineral therapy. Pediatrics 2000;105(2):e18.
100. May C. History of the introduction of theophylline into the treatment of asthma. Clinical Allergy 1974;4:211-217.
101. Mitra A, Bussler D, Goodman K, et al. Intravenous aminophylline for acute severe asthma in children over two years receiving inhaled bronchodilators. Evid-Based Child Health 2006;1:101-146 (Cochrane Library).
102. Emerman CL, Devlin C, Connors AF. Risk of toxicity in patients with elevated theophylline levels. Ann Emerg Med 1990;19(6):643-648.
103. Montserrat JM, Barabera JA, Viegas C, et al. Gas exchange response to intravenous aminophylline in patients with a severe exacerbation of asthma. Eur Respir J 1995;8:28-33.
104. Bell M, Jackson E, Mi Z, et al. Low-dose theophylline increases urine output in diuretic-dependent critically ill children. Intensive Care Med 1998;24: 1099-1105.
105. Rean RS, Loftis LL, Albers GM, et al. Efficacy of IV theophylline in children with severe status asthmaticus. Chest 2001;119:1480-1488.
106. Gupta VK, Cheifetz IM. Heliox administration in the pediatric intensive care unit: An evidence-based review. Ped Crit Care Med 2005;6:204-211.
107. Rodrigo GJ, Pollack CV, Rodrigo C, et al. Heliox for non-intubated acute asthma patients. Cochrane Database of Systematic Reviews 2006;4:CD002884.
108. Kim IK, Phrampus E, Venkataraman S, et al. Helium/oxygen-driven albuterol nebulization in the treatment of children with moderate to severe asthma exacerbations: A randomized, controlled trial. Pediatrics 2005;116:1127-133.
109. Kim IK, Saville AL, Sikes KL, et al. Heliox-Driven albuterol nebulization for asthma exacerbations: An overview. Respir Care 2006;51(6):613-618.
110. Abd-Allah SA, Rogers MS, Terry M, et al. Helium-oxygen therapy for pediatric acute severe asthma requiring mechanical ventilation. Pediatr Crit Care Med 2003;4:353-357.
111. Strube PJ, Hallam PL. Ketamine by continuous infusion in status asthmaticus. Anesthesia 1986;41:1017-1019.
112. Rock MJ, Reyes de la Roche S, L’Hommedieu CS, et al. Use of ketamine in asthmatic children to treat respiratory failure refractory to conventional therapy. Crit Care Med 1986;14(5):514-516.
113. Hemming A, Mackenzie I, Finfer S. Response to ketamine in status asthmaticus resistant to maximal medical treatment. Thorax 1994;49(1):90-91.
114. Nehama J, Pass R, Bechtler-Karsh A, et al. Continuous ketamine infusion for the treatment of refractory asthma in a mechanically ventilated infant: A case report and review of the literature. Pediatr Emerg Care 1996;12(4):294-297.
115. Youseff-Ahmed MZ, Silver P, Ninkott L, et al. Continuous infusion of ketamine in mechanically ventilated children with refractory bronchospasm. Intensive Care Med 1996;22(9):972-976.
116. Sarma VJ. Use of ketamine in acute severe asthma. Acta Anaesthesiol Scand 1992;36(1):106-107.
117. Petrillo TM, Fortenberry JD, Linzer JF, et al. Emergency department use of ketamine in pediatric status asthmaticus. J Asthma 2001;38(8):657-664.
118. Drazen JM, Israel E, O’Byrne PM. Treatment of asthma with drugs modifying the leukotriene pathway. N Engl J Med 1999;340:197-206.
119. Bisqaard H. Leukotriene modifiers in pediatric asthma management. Pediatrics 2001;107:381-390.
120. Dumitra C, Chan SMH, Turcanu V. Role of leukotriene receptor antagonists in the management of pediatric asthma. Pediatr Drugs 2012;14(5):317-330.
121. Adelroth E, Morris MM, Hargreave FE, et al. Airway responsiveness to leukotrienes C4 and D4 and to methacholine in patients with asthma and normal controls. N Engl J Med 1986;315:480-484.
122. Green SA, Malica MP, Tanaka W, et al. Increase in urinary leukotriene LTE4 levels in acute asthma: Correlation with airflow limitation. Thorax 2004;59:100-104.
123. Dempsey OJ, Wilson AM, Sims EJ, et al. Additive bronchoprotective and bronchodilator effects of a single dose of salmeterol and montelukast in asthmatic patients receiving inhaled corticosteroids. Chest 2000;117:950-953.
124. Camargo CA, Smithline HA, Malice MP, et al. A randomized controlled trial of intravenous montelukast in acute asthma. Am J Resp Crit Care Med 2003;167: 528-533.
125. Sarnaik AP, Daphtory KM, Mart KL, et al. Pressure-controlled ventilation in children with severe status asthmaticus. Pediatr Crit Care Med 2004;5:133-138.
126. Cox RG, Barker GA, Bohn DJ. Efficacy, results, and complications of mechanical ventilation in children with status asthmaticus. Pediatr Pulmonol 1991;11(2):120-126.
127. Mayordomo-Colunga J, Medina A, Rey C, et al. Non-invasive ventilation in pediatric status asthmaticus: A prospective observational study. Pediatr Pulmonol 2011;46 949-955.
128. Basnet S, Mander G, Andoh J, et al. Safety, efficacy and tolerability of early initiation of non-invasive positive pressure ventilation in pediatric patients admitted with status asthmaticus: A pilot study. Pediatr Crit Care Med 2012;13:393-398.
129. Staple LE, O’Connell KJ. Pediatric rapid sequence intubation: An in-depth review. Pediatric Emergency Medicine Reviews 2013;18(1):1-11.
130. Werner HA. Status asthmaticus in children: A review. Chest 2001;119: 1913-1929.
131. Dariolo R, Perret C. Mechanical controlled hypoventilation is status asthmaticus. Ann Rev Respir Dis 1984;129:385-387.
132. Malmstrom K, Kaila M, Korhonen R, et al. Mechanical ventilation in children with severe asthma. Pediatr Pulmonol 2001;31:405-411.
133. Edmunds SM, Harrison R. Subarachnoid hemorrhage in a child with status asthmaticus: Significance of permissive hypercapnia. Pediatr Crit Care Med 2003;4:100-103.
134. Duval ELIM, vanVaght AJ. Status asthmaticus treated by high-frequency oscillatory ventilation. Pediatr Pulmonol 2000;30:350-353.
135. Tobias JP, Garrett JS. Therapeutic options for severe, refractory status asthmaticus: inhalational anesthetic agents, extracorporeal membrane oxygenation and helium/oxygen ventilation. Pediatr Anaesth 1997;7(1):47-57.
136. Mikkelsen ME, Pugh ME, Hansen-Flaschan JH, et al. Emergency extracorporeal life support for asphyxia status asthmaticus. Respir Care 2007;52(11):1525-1529.
137. Nakaqawa TA, Johnston SJ, Falkos SA, et al. Life-threatening status asthmaticus treated with inhaled nitric oxide. J Pediatr 2000;137:119-122.
138. Rishani R, El-Khatib M, Mrouch S. Treatment of severe status asthmaticus with nitric oxide. Pediatr Pulmonol 1999;28:451-453.
139. Hirshman LA, Edelstein G, Peetz S, et al. Mechanism of action of inhalational anesthetics on airways. Anesthesiology 1982;56:107-111.
140. O’Rouke PP, Crone PK. Halothane in status asthmaticus. Crit Car Med 1982;10:341-343.
141. Wheeler DS, Clapp CR, Ponaman ML, et al. Isoflurane therapy for status asthmaticus in children: A case series and protocol. Pediatr Crit Care Med 2000;1(1):55-59.
142. Johnston RG, Noseworthy TW, Friesen EG, et al. Isoflurane therapy for status asthmaticus in children and adults. Chest 1990;97(3):698-701.
143. Belessis Y, Dixon S, Thomsen A, et al. Risk factors for an intensive care unit admission in children with asthma. Pediatr Pulmonol 2004;37:201-209.
Asthma is a common problem in the emergency department (ED), with many children having significant exacerbations. The ED physician must be prepared with a versatile approach to rapidly stabilize the patient. This article comprehensively reviews the approach to a child with acute severe asthma, emphasizing management alternatives.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
