"I'm Swollen": Evaluation of Peripheral Edema in the Emergency Department
"I'm Swollen": Evaluation of Peripheral Edema in the Emergency Department
Authors:
Halleh Akbarnia, MD, FACEP, Infinity Health Care, St. Francis Hospital, Evanston, IL; Resurrection Emergency Medicine Program, Chicago, IL.
Viet Vo, MD, Resurrection Emergency Medicine Program, Chicago, IL.
Shu Chan, MD, FACEP, Resurrection Emergency Medicine Program, Chicago, IL.
Peer Reviewer:
Frank LoVecchio, DO, Department of Emergency Medicine, Maricopa Medical Center, Phoenix, AZ.
Case Examples
Case 1. A 53-year-old female presents to the emergency department with a 5-day history of generalized edema, primarily noted in her lower extremities. She states that she started noticing some swelling in her arms for the past 24 hours, which prompted her to come and be evaluated. The patient's history includes poorly controlled hypertension and borderline diabetes. She states that she was having trouble walking up more than one flight of stairs, and has been unable to walk more than one block, which is different from her previous status. The patient also states that she has been more anxious with her symptoms, has been worried about the increased cough she has been noticing, and has been having trouble sleeping at night. On physical exam, she is tachycardic and tachypnic, sitting upright, appearing a bit diaphoretic and anxious. She is hypertensive at 172/96, and on exam, her lungs appear to have fine rales at the bases.
Case 2. A 42-year-old male presents to the emergency department after a 7-day history of progressive symptoms, he was previously healthy, and has lower extremity pitting edema, dyspnea, fatigue, and a syncopal episode. Of note, the patient is a businessman and just returned 10 days ago from a long trip to Japan.
Case 3. A 29-year-old female presents to the emergency department with a history of being 37 weeks pregnant. Her blood pressure has been within the normal range during pregnancy; however, today she is 190/110 and also is appearing to have peripheral edema in both her lower extremities and her hands.
Introduction
The concept of edema has evolved through the ages. The ancient Greeks described all processes involving edema as a single entity called "dropsy" or "hydropsy" that appeared to collect in various parts of the body; we can appreciate this today as generalized edema, anasarca, and ascites.1
Edema is defined as a clinically apparent increase in interstitial fluid volume, which typically requires several liters before the abnormality is evident. It is primarily caused by the loss of equilibrium between the oncotic gradient that draws the fluid into the intravascular space and the hydrostatic forces that push the fluids into the various tissue spaces available. Anasarca is defined as gross, generalized edema — edema in its most severe form. Ascites and hydrothorax refer to fluid accumulation in the peritoneum and pleural cavities, respectively. Depending on the mechanism, edema can be localized or generalized in distribution.2
Peripheral edema is a very nonspecific and common finding in the emergency department and it can represent a benign condition or a rather serious condition that warrants intervention and admission, such as when associated with cardiac, pulmonary, and/or renal dysfunction.
Basic Physiology
The body contains both intracellular and extracellular fluid. About one-third of total body water is in the extracellular space. The extracellular fluid is further subdivided into interstitial fluid and plasma. About 75% of the extracellular fluid is interstitial fluid, and the remaining fluid is in the plasma compartment. Starling forces are the primary regulatory mechanism behind the distribution of fluid between the extracellular compartments. There are two primary Starling forces: hydrostatic pressure and oncotic pressure. Hydrostatic pressure within the vasculature and oncotic pressure within the interstitial fluid tend to promote movement of fluid into the extravascular space. Hydrostatic pressure in the interstitial fluid and the colloid oncotic pressure contributed by the plasma proteins tend to promote movement of fluid into the vasculature.3 There is a net movement of water and diffusible solutes from the vascular space at the arteriolar end of capillaries. In contrast, there is a general return of fluid from the interstitium to the venous end of the capillaries by the lymphatic system. Lymph flow will increase with an increase of movement of fluid from the vascular compartment to the interstitium. Unless blockage of lymphatics or other pathology arises, then there is a steady state between the interstitium and the vasculature.
Pathophysiology: Where It Can All Go Wrong
Hydrostatic pressure in the intravascular space is always greater than that of the interstitium. If it is increased in any way, then an increased net movement of fluid into the extracellular space will take place, resulting in edema. Correspondingly, decreases in colloid oncotic pressure, due to hypoalbuminemia, severe malnutrition, liver disease, loss of protein in urine, or a severe catabolic state, will favor net movement of fluid into the interstitial space.3,4 (See Figure 1.)
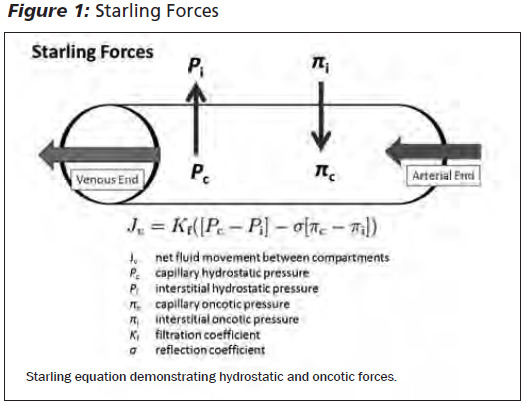
Homeostatic factors play a large role in attempting to return the body to a steady state. In many derangements that lead to edema, there is a reduction of effective arterial volume. This reduction is caused by diminished cardiac output or systemic vascular resistance. Reduced effective arterial volume can lead to physiologic activation to attempt to restore or retain volume. This response leads to retention of salt and water that eventually manifests as edema. Sodium retention, essential in the development of generalized edema, is caused by the diminished blood flow to the juxtaglomerular cells in the kidney in an arterial volume-depleted state. This, in turn, causes a cascade of renin release, and angiotensinogen is released, then angiotensin I, which is then converted to angiotensin II. The activation eventually leads to vasoconstriction on the renal efferent arterioles with resultant enhanced salt and water reabsorption in the kidneys. Angiotensin II enters the systemic circulation, stimulating aldosterone release by the zona glomerulosa of the adrenal cortex. Aldosterone will further enhance sodium reabsorption and potassium excretion and its release is increased in many edematous states such as heart failure.3,4
Fluid accumulation does not become clinically apparent until the interstitial volume has increased by an amount that approaches plasma volume, which may be up to 2.5 liters to 3 liters.
Another cause of an altered gradient is capillary damage and leak. Endothelial damage can result in increased permeability, which permits transfer of proteins into the interstitium. This can be the result of drugs, viral or bacterial agents, thermal or mechanical trauma, or a consequence of hypersensitivity reaction. This kind of edema is usually inflammatory in nature and usually non-pitting, localized, and associated with erythema, heat, and tenderness.3
Differential Diagnoses for Peripheral Edema
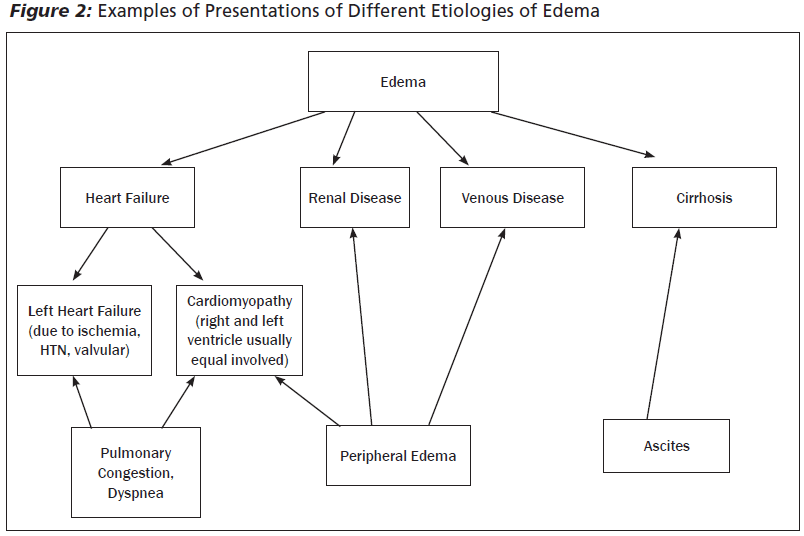
There should be a differentiation between cardiac and non-cardiac etiologies of peripheral edema. (See Table 1.) These different etiologies can be distinguished by patient presentation, vital signs, history, physical examination, imaging, and biomarkers. (See Figure 2.)

Acute Heart Failure. Acute heart failure syndrome can be best understood as a set of physiological mechanisms that may produce signs and symptoms that are primarily related to elevated left heart pressures, resulting in vascular congestion.5 Impaired systolic emptying of ventricles and/or poor diastolic relaxation promotes accumulation of blood in the venous circulation, effectively decreasing arterial volume and initiating the sequence of compensatory events. With right ventricular impairment or failure, pressures in the venous system rise, which augments fluid transmission into the interstitium, increasing the likelihood of peripheral edema. Likewise, with left ventricular impairment, there are increased pressures in the pulmonary vasculature that will affect right ventricular emptying or forward flow. With an impairment of right ventricular emptying, diastolic, central, and systemic venous pressures increase, again resulting in an increased likelihood of edema.3 As a result, with a perception of decreased arterial volume, the body initiates a neurohormonal cascade that in turn promotes increased sodium retention.
Constrictive Pericarditis and Restrictive Cardiomyopathy. Constrictive pericarditis is a condition in which the pericardium is inflamed, scarred, or calcified. In these conditions, the heart has limited diastolic filling and results in diastolic failure but still has preserved systolic function. In contrast, in restrictive cardiomyopathy, once known as "constrictive cardiomyopathy,"6 the myocardium is rigid and causes a major decrease in effective compliance of the muscle itself. A history of cardiac surgery or systemic illness that may affect the pericardium tends to favor pericarditis. The most common symptoms are related to either fluid overload (peripheral edema, ascites, etc.) or decreased cardiac output (shortness of breath, weakness, exercise intolerance, etc.).7 Diagnostic studies include heart catheterization, transthoracic echocardiography, and possible tissue biopsy.2
Venous Insufficiency/Venous Occlusion. This condition is found more commonly in the elderly and can be a cause of both unilateral and bilateral edema or swelling. However, it is more commonly unilateral. Within the superficial veins, venous wall abnormalities cause dilatation that promotes valvular incompetence and leads to persistent venous hypertension. In the deep veins, thrombosis may cause venous valvular incompetence that leads to increased venous hypertension. Residual thrombus is replaced by fibrous tissue that can obstruct outflow or impede proper function. Valvular damage and wall distortion as a consequence of healing alter valve function and cause subsequent valvular incompetence and reflux.5
Nephrotic Syndrome. In nephrotic syndrome, patients present with proteinuria, hypoalbuminemia, hypercholesterolemia, and peripheral edema. Increased glomerular permeability to large molecules, mostly albumin, is the essential pathophysiology. The edema is thought to be primarily due to decreased colloid oncotic pressure due to substantive loss of protein in the urine. Sodium and water spill into the interstitium due to an imbalance of the Starling forces. Additionally, total effective arterial volume is decreased and initiates the neurohormonal mechanisms that will further contribute to the formation of edema.8
Pulmonary Hypertension. Patients with pulmonary hypertension typically present to the emergency department with shortness of breath, fatigue, syncope, and peripheral edema, especially after any kind of exertion. The history of presentation and symptoms are important, especially to determine if there is a familial factor involved. Patients will demonstrate decreased exercise tolerance during the start of this severe disease process.
Pulmonary hypertension has been classified by the World Health Organization into five different types, including arterial, venous, thromboembolic, hypoxic, and miscellaneous based on the causes of the disease.9 Each classification has different presentations and symptoms.
In WHO group I, arterial pulmonary hypertension, the pulmonary vessels tend to constrict and lead to the vessels becoming fibrotic. With this extra workload, right ventricular hypertrophy can occur and eventually leads to cor pulmonale, right heart failure. With the decreased blood flow to the left side of the heart, the heart pumps less oxygen to the rest of the body, and this contributes to hypoxia and increased exercise intolerance.
With group II, pulmonary venous hypertension, there is no obstruction to blood flow in the lungs and this is where pulmonary edema can be noted, along with pleural effusions, when the left side of the heart fails to pump blood efficiently. When patients present with hypoxic pulmonary hypertension (WHO group III) based on lung etiology, the low levels of oxygen cause vasoconstriction of the pulmonary arteries, which also can lead to similar features as arterial pulmonary hypertension. With thromboembolic pulmonary hypertension (WHO group IV), pulmonary embolism in the pulmonary arteries, or the presence of other matter such as tumor or infection, can cause blockage, which again presents with similar features as above.9,10
Cirrhosis. Cirrhosis is chronic liver disease characterized by hepatocyte death, resulting in inflammation and eventually fibrosis. As fibrosis develops causing progressive loss of liver function, portal hypertension arises. There is an overall sense of decreased arterial volume that is due in part to splanchnic vasodilation and accumulation due to hepatic outflow obstruction. Also, reduced intravascular volume will result in initiation of the neurohormonal cascade, resulting in an increase in sodium retention and water reabsorption by the kidneys. Most of the edema is accumulated proximally (peritoneal fluid) as ascites. As the disease process progresses further, peripheral edema becomes more apparent, especially with severe hypoalbuminemia due to a failing liver.11
Hypoproteinemia. Albumin is essential in maintaining a plasma oncotic pressure, and with levels below 2 g/dL, edema is often the result.2 The etiologies that can contribute to a low serum protein include severe nutritional deficiency, protein losing enteropathies, and severe hepatic disease. Kwashiorkor is a well-known protein deficient state that has a normal weight for age, significant generalized edema, skin changes, hypopigmented hair, abdominal distension, and hepatomegaly. The edema here is usually a result of low serum albumin, elevated cortisol, and the inability to activate antidiuretic hormone.12
Drugs. Medications can cause or worsen peripheral edema. The most common medications implicated as causing edema are hypertensive medications, which include calcium channel blockers, clonidine, hydralazine, methyldopa, and beta-blockers. It is thought that direct vasodilators, such as minoxidil, diazoxide, and nifedipine, have a two-fold effect on blood pressure. They have a direct effect on vasculature as well as an indirect effect on both the renin-angiotensin-aldosterone and sympathetic systems, causing increased sodium retention. With the calcium channel blockers, the dihydropyridine class (which is often used to reduce systemic vascular resistance and arterial pressure) has more of an edematous effect due to more selective arteriolar vasodilatory characteristics than phenylalkylamine or benzothiazepine (verapamil and diltiazem). ACE inhibitors rarely cause dependent edema.2
Hormonal medications such as corticosteroids, estrogen, progesterone, and testosterone also can cause edema, as well as many other medications such as nonsteroidal anti-inflammatory drugs (NSAIDs), pioglitazone, rosiglitazone, and MAO inhibitors. Those with underlying heart disease may be more susceptible to having edema with the thiazolidinediones such as rosiglitazone or pioglitazone by way of sodium retention and resorption by activating sodium channels also stimulated by aldosterone.
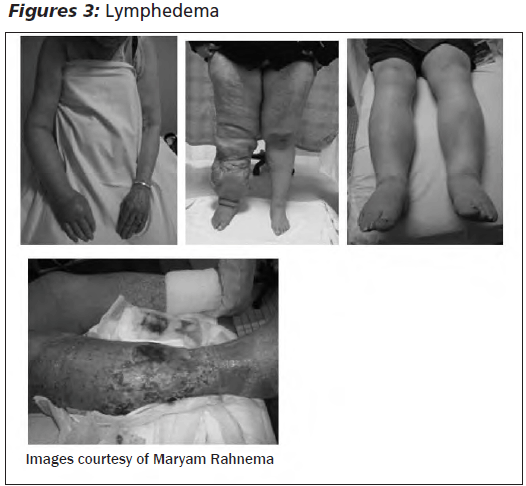
Lymphedema. While lymphedema has a different pathophysiology than edema, lymphedema should remain on the differential when encountered due to the similar presentation. In contrast to edema, lymphedema is the result of protein-rich interstitial fluid overload secondary to lymphatic dysfunction with intact capillary function.13,14 This impaired lymphatic transport leads to the pathologic accumulation of protein-rich lymphatic fluid in the interstitium found mostly in the extremities. (See Figure 3.) Secondary lymphedema is the most common form. Common causes in industrialized nations are malignancy and malignancy-associated treatments such as radiotherapy and surgery (e.g., radical lymph node dissection). Worldwide, the primary cause is the parasitic tropical disease filariasis caused by thread-like nematodes (roundworms) Wuchereria bancrofti.14
Myxedema. Myxedema can be found with both hypothyroidism and hyperthyroidism, but most often is part of long-standing hypothyroidism. Hypothyroidism is a potentially serious and often overlooked diagnosis. It affects almost all organ systems due to a slower metabolic state. Initially it can present with nonspecific constitutional and psychiatric complaints. However, with long-standing progression, hypothyroidism can lead to heart failure, psychosis, and coma. Unfortunately, it is a great mimicker of many common complaints encountered in the emergency department. In overt hypothyroidism, peripheral edema is among the multiple clinical findings, including periorbital edema. Although the causes of edema associated with myxedema are still not fully appreciated, it is thought that within the capillaries, there is an increase in permeability resulting in the accumulation of proteins and mucopolysaccharides in the interstitium, followed by sodium and water and a concomitant expansion in total body water and sodium.2,15
Pregnancy. Edema in pregnancy can be seen in up to 80% of pregnancies due to factors that elevate venous pressures, particularly in the lower extremities. During pregnancy there are many physiological changes that favor edema formation that include elevated progesterone levels which act to increase venous distensibility, increased plasma volume, sodium retention, decreased plasma protein concentration, increased capillary hydrostatic pressure late in pregnancy due to mechanical compression of vena cava and iliac veins, increased antinatriuretic hormones such as aldosterone and desoxycorticosterone, and the activation of renin-angiotensin-aldosterone system.2 These factors, in addition to the decreased venous return from the inferior vena cava, result in edema, varicose veins, and hemorrhoids.16 It should be noted that while generalized edema is often present in cases of preeclampsia, it is no longer part of the diagnostic criteria.17
Idiopathic Edema. Although the frequency of idiopathic edema is not known, as of late researchers have claimed that it is becoming quite common in the premenopausal female. This syndrome includes fluid retention, causing swelling of the limbs, trunk, face, feet, and hands. Typically, the patient does not exhibit any signs of cardiac, renal, or hepatic disease; however, psychological symptoms do present along with this form of edema. Obesity and diabetes often accompany this syndrome as well. It is important to note that many of these women may present with an attempt to control their obesity and attempt weight loss with the use of diuretics, purging behaviors such as forced vomiting, and laxatives.18 This fact has many authors questioning whether idiopathic edema truly is an independent syndrome and one of exclusion, or one caused by the patient's habits and behaviors.
Three major theories have been put forth regarding how edema occurs, including capillary leak described above, diuretic-induced edema, and refeeding. With capillary leak, patients develop somewhat of a plasma volume depletion and pool fluid in their lower extremities while in the upright position. During the daytime, there can be a weight gain of 0.5 kg to 1.5 kg, along with a fall in urinary sodium excretion for normal patients; however, with patients exhibiting idiopathic edema, the change can be up to 5 kg in severe cases because more fluid is lost into the vascular space with standing, and the body produces more renin, ADH, and norepinephrine, which causes this increased fluid change from morning to evening.19
Another idea is that idiopathic edema could be secondary to refeeding. These patients are very conscious of their weight and will go through dramatic changes in their food intake to try to lose weight. When they end their fasting period and start to ingest food again, they can have refeeding edema, which may be related to increased insulin release and activation of the renin-angiotensin-aldosterone pathway. Sodium is retained and hypokalemia is seen secondary to the movement of potassium into the cell.19
A third possibility for the genesis of idiopathic edema is from the chronic intake of diuretics and diuretic-induced edema. Patients initially may be started on diuretics when they are diagnosed with fluid retention. As they continue their therapy, they can see diuretic-induced hypovolemia, which activates the retention of sodium and again the renin-angiotensin-aldosterone pathway. Once a patient stops taking the diuretic, the pathway may not shut down and rapid edema can occur, which will mistakenly make the patient believe that more diuretics are needed. With time, this passes, and after 1-3 weeks, the edema will improve if the patient refrains from taking the medications.19,20
Idiopathic edema is a diagnosis of exclusion when other possibilities have been evaluated and there is no evidence of renal, hepatic, or cardiac disease. This is also different than premenstrual edema, which tends to be self-limiting and resolves shortly after menses begins. Premenstrual edema is thought to be mostly caused by estrogens, which is a transient change.
Treatment of idiopathic edema needs to include changes in diet, including a consistent low sodium diet, along with cessation of the diuretics that may be causing the edema in the first place, which may take 3-4 weeks to resolve. Because patients can be resistant to conservative treatment of temporary cessation of diuretics and diet changes, other modalities may be necessary. Renal injury can be seen with high-dose loop diuretics, so these should be avoided. However, ACE inhibitors have been shown to have some effectiveness in blocking the renin-angiotensin-aldosterone system, and can be attempted. Some patients may benefit from dopamine agonist agents such as bromocriptine or a combination of levodopa/carbidopa.21
Patient Assessment: History and Physical Examination
If the patient is in distress, necessary interventions such as oxygen, peripheral IV placement, and cardiac monitors should be placed. If reports of chest pain or an indication of cardiac etiology is suggested, an ECG should be obtained. If an emergent condition presents or is discovered, the interview and exam should be delayed and necessary intervention is indicated. Once stabilized, a more thorough interview and assessment may proceed. (See Table 3.)

A thorough history is essential. A list of the patient's medications with both current and recent changes is important. Substance abuse-related questions need to be asked cautiously, as it is just as important to note cocaine abuse as it may be knowing if a patient is taking NSAIDs or estrogen, or is taking a calcium channel blocker for their hypertension.22,23 A dietary history, especially including fluid intake and salt consumption, can be useful.
The patient will need to be thoroughly examined, and it is essential that an accurate weight is obtained and documented. Documentation of the location of the edema, symmetry, pitting or non-pitting, skin appearance, and whether the patient's skin exhibits signs of tenderness is important. Are there ulcerations present and is the quality and color of the skin altered in any way? It is important to look for signs of ascites and fluid waves.24 Testing and laboratory evaluation should be guided by the history and examination. Thyroid testing should be done if thyroid dysfunction is a possibility. ECG, chest radiographs, transthoracic echocardiography, and venous duplex of the lower extremities are also possibilities for further testing.
With simple inquiry, some information can be gleaned right away, such as medication history, with new medications, current medications, and any changes made recently. Additionally, if a female patient is of childbearing age and not obviously gravid, it may be appropriate to gather an OB/GYN history and/or a pregnancy test.
Regarding edema, essential to the history is the duration, onset, and location of the edema. The onset and duration can determine whether this is new, old, or an exacerbation of an ongoing problem. Whether it is symmetrical, asymmetrical, localized, or generalized can aid in formulating a differential. More symmetrical and generalized edema suggests a systemic or more central process is occurring, such as in heart failure, cirrhosis, or nephrotic syndrome. If there is a more chronic, localized, and asymmetrical picture, then the differential should shift to a more confined etiology such as venous insufficiency or lymphedema.25
The finding of pitting edema is significant and important to document accurately. Pitting edema refers to the edema noted when the examiner's thumb is pushed against a bony structure such as the tibia or sacrum, and the resulting "pit" remains visible for a short period of time. It is graded on a scale of 1 to 4 (from very slight to more apparent to deep pitting that persists for longer than 2 minutes).
Establishing a new diagnosis of heart failure is not always easy. The most common symptom reported by patients in acute heart failure syndrome is dyspnea or breathlessness. Historically, a patient may notice weight gain and/or difficulty performing daily activities. During the patient exam, signs consistent with poor cardiac output show up as shortness of breath, jugular venous distention, hepatojugular reflex, and an S3 heart sound. Right-sided failure patients will have peripheral edema and, when severe, can show anasarca and ascites along with their dyspnea. Most often the peripheral edema is due to a measured increase in central venous pressure caused by increases in pressures in the subclavian vein and right atrium.26 The patient can have radiographic abnormalities that are highly specific in indicating pulmonary vascular congestion, such as cephalization, interstitial edema, and alveolar edema (96%, 98%, and 99% specificity, respectively). However, in long-standing or chronic heart failure, the body can adapt to this volume-overloaded state, possibly making these radiographic findings absent.27-29 ECG can identify acute conditions that may be the cause of an exacerbation such as atrial fibrillation or ischemia while revealing other associated findings suggestive of heart failure syndrome (i.e., strain, hypertrophy, conduction abnormalities).29 B-type natriuretic peptide is most helpful with very low (< 80-100 pg/mL) or very high (> 400-500 pg/mL) values in conjunction with an intermediate pre-test probability. Intermediate values require further clinical assessment.
Constrictive pericarditis and restrictive cardiomyopathy may be indistinguishable from other etiologies that can cause right heart failure. It can be observed that jugular venous pressures are always elevated; Kussmaul sign and a pericardial knock both are considered specific for constrictive pericarditis. On chest radiography, pericardial calcifications best appreciated on a lateral view would be indicative of pericarditis. In a recent review, only 25% of patients were found to have calcifications apparent on radiography, so this lack of finding does not exclude constrictive pericarditis.7 Nonspecific ST-segment and T-wave changes are common features of constrictive pericarditis, whereas low QRS voltage and isolated repolarization abnormalities are more typical in constrictive pericarditis. Depolarization abnormalities, such as bundle branch block, ventricular hypertrophy, pathologic Q waves, and impaired atrioventricular conduction, strongly favor restrictive cardiomyopathy. Late-stage atrial fibrillation can occur in both conditions. If available, echocardiography is the usual diagnostic imaging study in patients with suspected constrictive pericarditis.7
Cirrhosis can develop in an insidious, indolent manner, and a patient can present to the ED with the first signs if medical care was neglected long enough.
This progression is most characteristic of hepatitis C-induced cirrhosis, which often remains clinically silent while destroying up to 70% of the liver. The development of portal hypertension signals depletion of the substantial physiologic reserve of the liver that allows it to compensate for so long. Signs and symptoms include anorexia, nausea, vomiting, abdominal discomfort, weakness, asterixis, jaundice, palmar erythema, spider angiomata, ascites, splenomegaly, a firm liver, dilated abdominal wall veins, esophageal varices, and portosystemic encephalopathy.30,31 Initially, fluid can collect in the form of ascites, but with long-standing progression, it can manifest as peripheral edema as well.2 Patients with tense ascites will have upward pressure on the diaphragm and can have increased intrathoracic pressure. The central venous pressure (CVP) will be elevated initially, but as soon as any abdominal fluid is removed, the CVP will decrease very rapidly.21
The history is key in nephrotic syndrome. The number one cause of nephrotic syndrome is now diabetic nephropathy.32 A history of diabetes mellitus or other systemic illnesses that commonly affect the kidneys, recent infections, medication use or changes, drug use, or even a prior history of proteinuria, hypertension, edema, or renal disease can be helpful. Also, it may prove useful to take a family history because the syndrome has several congenital causes.8,33
The examination should include evaluation of the blood pressure and assessment for signs of systemic disease or renal insufficiency. Edema associated with nephrotic syndrome is often apparent peripherally and periorbitally and can become increasingly more severe. Patients may develop edema in the lower extremities and genitals, ascites, pleural effusions, and even pericardial effusions.8 Patients can also present with severe hypoalbuminemia, high proteinuria (greater than 3.5 grams per day), and have a normal to high CVP.21
In young female patients, the possibility of pregnancy should be kept in mind because pregnancy can exacerbate previously unknown renal disease; in late pregnancy, proteinuria may be the first sign of preeclampsia.
Historically, lymphedema can be teased out with questions pertaining to family history, past medical history of malignancy, procedures secondary to malignancy, or even living outside of the United States in tropical locations such as Africa, Asia, and even South America. The initial presentation may start with unilateral painless swelling that starts on the dorsum of the foot, usually with a squared-off appearance. It can start out as a pitting edema, but eventually could become fibrotic, giving a non-pitting brawny appearance. If not treated, the process could become circumferential. Eventually the skin may get hyperkeratotic, papillomatous, or verrucous, rough, and covered in wart-like projections — this thickening, peau d'orange skin, is the hallmark of lymphedema. The patient will also present with an inability to tent the skin at the base of the digits in the affected extremity, called "Stemmer sign." The skin is predisposed to recurrent infection and becomes dysfunctional with a risk of fluid leakage, further impairing the healing process.14 Depending on the etiology, the edema may be unilateral or bilateral. Even when bilateral, it is common for the lymphedema to be asymmetric in severity.13 (See Figure 3.)
If venous insufficiency is thought to be the problem, then inquire about any history of previous episodes of thrombosis, lower extremity cellulitis, leg ulcers, surgery of the lower extremities or pelvis, and arterial insufficiency symptoms. Other risk factors associated should be noted, including diabetes mellitus, smoking, and hyperlipidemia. In addition to lower extremity edema, symptomatology includes leg aching, heaviness, and venous claudication.34 There are a number of physical findings that may be present depending on chronicity. The more chronic changes include varicosities, induration, fibrosis, ulceration, and hemosiderin deposition in the lower extremities. Clinicians should look for unilateral findings, which typically would not be seen in generalized edematous states. In these patients, the central venous pressure would be normal, and diuretics would not help with the edema associated with this process.13,34
In hypothyroidism, signs and symptoms may be subtle and often difficult to recognize. Symptoms may be ignored or tolerated due to such an insidious or gradual onset.
Localized edema of the eyelids, face, and dorsum of the hand are noted more frequently in these patients. It can be apparent as puffiness of the face, most notably the periorbital areas of the face, or persistent indentation of the skin, or even having jewelry such as rings fitting more snugly or the inability to remove them.
Myxedema is typically non-pitting edema of the face and extremities. Its non-pitting nature is due to glycosaminoglycans. The disease process is most commonly found in hypothyroidism and, clinically, the patient may exhibit slow mentation and speech, thinning and brittleness of the hair and nails, skin changes that result in dryness and scaling, sinus bradycardia, QT prolongation, constipation, hypothermia, and delayed deep tendon reflexes. A number of complaints may be noted as well, such as impaired ability to concentrate, decreased memory, cold intolerance, fatigue, shortness of breath, and weight gain with decreased appetite.
Idiopathic edema should be considered after excluding other diagnoses, such as cardiac, hepatic, and renal conditions, and should be considered in menstruating women only.19,20
Management and Disposition
Initial treatment can be as benign as limiting salt intake and restricting fluids — this is the mainstay of non-pharmacologic treatment. Pharmacologic treatment often will consist of diuretics, but the clinician must consider the effects of fluid removal to the patient's overall status — in cases of life-threatening emergencies such as acute pulmonary edema, quick action with diuretics can be lifesaving. A slower removal of fluids may be more appropriate for those with more chronic conditions and a more stable clinical picture. Prior to initiation of diuretic therapy, the questions that should be considered include what the consequences of removal of fluid would be and how rapidly can/should the fluid be removed. If constrictive pericarditis is strongly suspected and/or diagnosed, surgical consultation should be considered because most require surgical pericardiectomy, which can be performed with a very acceptable risk at experienced centers.7
Pregnancy obviously involves numerous physiological changes that produce generalized edema in a large percentage of patients; however, other causes should be considered.
Treatment of cirrhosis in the emergency department tends to be conservative and limited to correcting electrolyte and hematologic abnormalities and supplementing nutritional and vitamin deficiencies. If the patient does not show signs of peripheral edema, the excess ascitic fluid will be mobilized by the peritoneal capillaries on their own — approximately 300 to 500 mL/day is the maximum that is mobilized. If diuresis is done more rapidly, the ascitic fluid cannot replenish the volume in the plasma, and the patient can experience azotemia, significant hypokalemia, and metabolic alkalosis, and hepatorenal syndrome can be precipitated, as well as instability in their vital signs including hypotension.35 Paracentesis for relief from respiratory distress or general discomfort may be necessary, however, removal of large quantities of ascitic fluid can provoke azotemia and hypotension. Most patients can be discharged into the care of their primary care provider or referred to a general internist or specialist for further evaluation and treatment.32
Like cirrhosis, emergency department treatment of nephrotic syndrome is limited. In these patients, if excessive and too rapid of fluid removal takes place through aggressive diuretic therapy, further renal injury can take place because of the intravascular volume depletion created by the clinician.32
In a severe case of hypothyroidism or myxedema coma, ventilatory and cardiovascular support is important, in addition to thyroid hormone replacement. In contrast, a new diagnosis of hypothyroidism generally does not need treatment and can be referred to or discussed with a primary care provider for treatment options and confirmation of thyroid function abnormality. If the patient is symptomatic or a TSH is found to be greater than 10, then hormone replacement may be indicated.32
Since the mainstay pharmacologic treatment for edema includes diuretics, this will be discussed briefly. Diuretics include loop diuretics, thiazide agents, and potassium-sparing medications. Furosemide, a loop diuretic, still is considered one of the most effective — its half-life is short (approximately 1.5 to 2 hours) and, therefore, it can be used in the emergency department when prompt diuresis is desired. Most patients will have a maximum amount of medication that will work with them — a threshold level. For patients with renal failure, it may be necessary to give larger doses to get maximum effect. At times, adding either a potassium-sparing diuretic (i.e., spironolactone) or a thiazide (i.e., hydrochlorothiazide) may help reduce sodium reabsorption in the distal renal nephron when loop diuretics do not work on their own.36
In many patients, lifestyle modifications will be necessary to help prevent further fluid retention and edema. Compression stockings aid patients with venous stasis by helping increase venous return to the heart, which will decrease their lower extremity edema. Patients with sleep apnea and hypoxia secondary to obstructive pulmonary disease may need oxygen therapy at night, including continuous positive airway pressure machines.
Case Conclusions
All three of the patients described at the beginning of this article presented to the emergency department with some form of peripheral edema and other various findings leading to their diagnosis.
References
- Stoycheff, N, et al. Nephrotic syndrome in diabetic kidney disease: An evaluation and update of the definition. Am J Kidney Dis 2009;54:840-849.
- Cho S, Atwood J. Peripheral edema. Am J Med 2002;113:580–586.
- Harrison TR, Fauci AS. Harrison's Principles of Internal Medicine, 17th ed. New York: McGraw-Hill; 2008:290-295.
- Taylor AE. Capillary fluid filtration. Starling forces and lymph flow. Circ Res 1981;49:557.
- Braunwald E. Braunwald: Heart Disease a Textbook of Cardiovascular Medicine, 9th ed. Philadelphia: W.B. Saunders; 2011:517-527.
- Hancock EW. Differential diagnosis of restrictive cardiomyopathy and constrictive pericarditis. Heart (British Cardiac Society) 2001;86(3):343–349.
- Khandaker MH, et al. Pericardial disease: Diagnosis and management. Mayo Clinic Proceedings 2010;85:572-593.
- Hull RP, Goldsmith DJ. Nephrotic syndrome in adults. BMJ 2008;336:
1185-1189. - Simonneau G, Galiè N, Rubin LJ, et al. "Clinical classification of pulmonary hypertension." J Am Coll Cardiol 2004;43 (12 Suppl S):5S–12S.
- Barst RJ, McGoon M, Torbicki A, et al. Diagnosis and differential assessment of pulmonary arterial hypertension. J Am Coll Cardiol 2004;43(12 Suppl S):
40S–47S. - Lefton HB, Rosa A, Cohen M. Diagnosis and epidemiology of cirrhosis. Med Clin North Am 2009;93:787-799.
- Grover Z, Ee LC. Protein energy malnutrition. Pediatr Clin North Am 2009;56:1055-1068.
- Ely J, Osher J, et al. Approach to leg edema of unclear etiology. J Am Board Family Med 2006;19;148-160.
- Kerchner K, Fleischer A, Yosipovitch G. Lower extremity lymphedema. J Am Acad Dermatology 2008;59:324-331.
- Chen Ying-Ju, et al. Diagnosis of unrecognized primary overt hypothyroidism in the ED. Am J Emerg Med 2010;28:866-870.
- Hill CC, Pickinpaugh J. Physiologic changes in pregnancy. Surg Clin North Am 2008;88:391-401.
- Leeman L, Fontaine P. Hypertensive disorders of pregnancy. Am Fam Physician 2008;78:1.
- Dorhout Mees EJ. Idiopathic edema: Definition of the syndrome and an approach to management. Renal Physiol and Path; Nephrol Dial Transplant 1994;9(supp):1093.
- Dewardener HE. Idiopathic edema: Role of diuretic abuse. Kidney Int 1981;19:881.
- MacGregor GA, Markandu ND, Roulston JE, et al. Is "idiopathic" edema idiopathic? Lancet 1979;1:397.
- Edwards OM, Dent RG. Idiopathic edema. Lancet 1979;1:1188.
- Russell RP. Side effects of calcium channel blockers. Hypertension 1988;11(3 pt 2):1142-1144.
- Clive DM, Stoff JS. Renal syndromes associated with nonsteroidal antiinflammatory drugs. N Engl J Med 1984;310(9):563-572.
- Bates B. A Guide to Physical Examination and History Taking. 5th ed. Philadelphia, Pa: Lippincott; 1991.
- Gorman WP, Davis KR, Donnelly R. ABC of arterial and venous disease. Swollen lower limb-1: General assessment and deep vein thrombosis. BMJ 2000;320:1453-1456.
- Rose BD. Clinical manifestations and diagnosis of edema in adults. In: UpToDate. 2012. Accessed from: http://www.uptodate.com/home/index.html.
- Pang PS. Acute heart failure syndromes: Initial management. Emerg Med Clin North Am 2011;29:675-688.
- Jessup, M, Brozena S. Medical Progress: Heart failure. N Engl J Med 2003;348:2007-2018.
- Collins S, et al. Beyond pulmonary edema: Diagnostic, risk stratification, and treatment challenges of acute heart failure management in the emergency department. Ann Emerg Med 2008;51:45-57.
- Starr SP, Raines D. Cirrhosis: Diagnosis, management, and prevention. Am Fam Physician 2011;84:12.
- Mushlin PS, et al, eds. Miller's Anesthesia. Philadelphia: Churchill Livingstone; 2009.
- Marx JA, Hockberger RS, Walls RM. Rosen's Emergency Medicine: Concepts and Clinical Practice, 7th ed. St. Louis: Mosby; 2009:1160-1161; 1666-1671; 2110-2110.
- Orth SR, Ritz E. Medical Progress: The nephrotic syndrome. N Engl J Med 2006;338:1203-1211.
- Word R. Medical and surgical therapy for advanced chronic venous insufficiency. Surg Clin North Am 2010;90:1195-1214.
- Pockros PJ, Reynolds TB. Rapid diuresis in patients with ascites from chronic liver disease: The importance of peripheral edema. Gastroenterology 1986;90:1827.
- Brater DC. Diuretic therapy. N Engl J Med 1998;339(6):387-395.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
