Drug Criteria & Outcomes: Palonosetron (Aloxi) Formulary Evaluation
Drug Criteria & Outcomes: Palonosetron (Aloxi) Formulary Evaluation
Part 2: Clinical Trial Data, Adverse Effects, and Cost Analysis
By Brian Watkins,
PharmD Candidate
Harrison School of Pharmacy
Auburn (AL) University
(Editor’s note: This second part of the palonosetron formulary evaluation addresses the available clinical trial, adverse effects, and cost analysis data. For information on mechanism of action, pharmacokinetics, indications, dosage, and administration of palonosetron, please consult the March issue of Drug Formulary Review's Drug Criteria & Outcomes.)
Moderately emetogenic chemotherapy
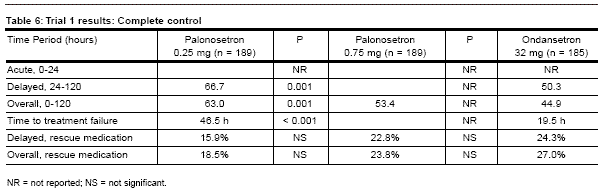
Trial 1: Palonosetron improves prevention of chemotherapy-induced nausea and vomiting (CINV) following moderately emetogenic chemotherapy: Results of a double-blind randomized Phase III trial comparing single doses of palonosetron with ondansetron.
Objective: To compare the efficacy and tolerability of single, fixed intravenous doses of palonosetron with a single intravenous dose of ondansetron in the prevention of acute and delayed CINV following administration of moderately emetogenic therapy.
Study Design: Multicenter, randomized, controlled, double-blind, double-dummy, stratified, Phase III study, noninferiority study.
Methods:
- 563 patients randomized; palonosetron 0.25 mg (n = 189), palonosetron 0.75 mg (n = 189), ondansetron 32 mg (n = 185).
- Patients had to be 18 years of age or older with histologically or cytologically confirmed malignant disease. The study included patients who were chemotherapy-naïve as well as those who had been treated previously with chemotherapy; all were scheduled to receive moderately emetogenic chemotherapy (any dose of carboplatin, epirubicin, idarubicin, ifosfamide, irinotecan, or mitoxantrone; methotrexate > 250 mg/m2; cyclophosphamide < 1500 mg/m2; doxorubicin > 25 mg/m2; or cisplatin < 50 mg/m2).
- Patients were excluded if they were unable to understand or cooperate with study procedures, used any drug with antiemetic activity within 24 hours prior to treatment and until day five, had evidence of a seizure disorder requiring anticonvulsants, experienced anticipatory nausea and vomiting, or were scheduled for radiation of the upper abdomen or cranium on days 2-6.
- Patients were randomized to receive a single, fixed intravenous dose of either palonosetron 0.25 mg, palonosetron 0.75 mg, or ondansetron 32 mg.
Results
- No patient received pretreatment with corticosteroids. Metoclopramide was the most commonly administered rescue medication.
- The primary efficacy
endpoint was the proportion of patients achieving a complete response
(CR; defined as no emetic episode and no use of rescue medication) during
the first 24 hours following chemotherapy administration (see Table 5).

- Secondary endpoints
examined were the proportion of patients with a CR during the delayed 24-120
hour time period, the cumulative overall 0-120 hour period, and CR rates during
successive 24 hour time periods. The proportion of patients achieving complete
control (CC; defined as no emetic episode, no need for rescue medication,
and no more than mild nausea) for the 0-24 hour, 24-120 hour, and 0-120 hour
intervals (see Table 6). Time to first emetic episode, severity of
nausea measured daily, time to treatment failure, time to administration and
need for rescue medication, patient’s global satisfaction with antiemetic
therapy, as well as quality of life also were secondary endpoints.
- To demonstrate the noninferiority of at least one dose of palonosetron to ondansetron, the lower boundary of the two-sided 97.5% confidence interval for the difference (palonosetron ondansetron) between the proportion of patients achieving a CR during the first 24 hours after chemotherapy had to be greater than the preset threshold of -15% difference.
- Palonosetron 0.25 mg was consistently superior to ondansetron (P < 0.05) in the number of emetic episodes (during the acute, delayed, and overall intervals as well as on study days 2 and 3), proportion of patients with no emetic episodes (during delayed and overall intervals as well as on study days 1-3), and the proportion of nausea-free patients (on study days 3-5).
- Palonosetron 0.75 mg was superior to ondansetron in the proportion of patients with no emetic episodes (during delayed and overall intervals as well as on study day 3) and the proportion of patients with no nausea (on study days 4 and 5).
Trial strengths
- Large multicenter, randomized, double-blind trial.
- Study population included chemotherapy-naïve patients and those who previously had received chemotherapy.
- Study reproduced CR rates for ondansetron similar to those found in other studies.
Trial weaknesses
- Lack of consistency in presentation of the results.
- Results section does not mirror efficacy/ outcome measures section.
Authors’ conclusions:
- Palonosetron 0.25 and 0.75 mg are similar in overall efficacy.
- A single intravenous dose of palonosetron results in prolonged protection against nausea and emesis following moderately emetogenic chemotherapy.
- Palonosetron proved superior to ondansetron in preventing both acute and delayed CINV following moderately emetogenic chemotherapy. Thus, palonosetron is a significant and important addition to antiemetic therapy.
Trial 2: Palonosetron is active in preventing acute and delayed emesis following moderately emetogenic chemotherapy: Results of a Phase III trial.
Objective: To assess the safety and efficacy of single intravenous doses of palonosetron vs. dolasetron for the prevention of moderately emetogenic CINV.
Study design: Multicenter, randomized, double-blind, parallel, stratified, noninferiority trial.
Methods:
- 569 patients randomized; palonosetron 0.25 mg (n = 189), palonosetron 0.75 mg (n = 189), and dolasetron 100 mg (n = 191).
- Patients were randomized to receive a single, fixed intravenous dose of either palonosetron 0.25 mg, palonosetron 0.75 mg, or dolasetron 100 mg.
- Following an amendment, patients were allowed to receive 20 mg dexamethasone or 125 mg methylprednisolone prior to chemotherapy at investigator discretion.
- The primary endpoint
was 24-hour CR (see Table 7).
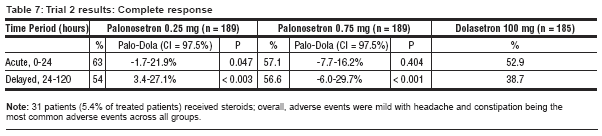
- Secondary endpoints included daily and cumulative CR rates for the delayed 24- to 120-hour interval.
Authors’ conclusions
- Palonosetron has demonstrated significant activity in preventing both acute and delayed emesis with a single intravenous dose in patients receiving moderately emetogenic chemotherapy.
- Palonosetron was safe and well tolerated.
Highly emetogenic chemotherapy
Trial 3: The efficacy and safety profile of palonosetron in a Phase II study involving chemotherapy-naïve patients undergoing highly emetogenic chemotherapy.
Objective: To assess the safety and efficacy of palonosetron administered over a range of single intravenous doses for the prevention of highly emetic CINV.
Study design: Randomized, double-blind, multicenter, dose-ranging Phase II trial.
Methods
- 161 patients randomized to one of five dose groups, which involved six-dose levels of palonosetron (0.3-1, 3, 10, 30, or 90 mcg/kg). Patients received a single intravenous injection of palonosetron 30 minutes prior to receiving highly emetogenic chemotherapy (cisplatin > 70 mg/m2).
- Dexamethasone was not administered to these patients.
- Safety and efficacy evaluations
(CR and CC, see Table 8 for results) were recorded in a patient diary
during the first 24 hours and then daily for the next six days following palonosetron
administration.

Adverse events
- Most adverse drug reactions were mild (83.9%) and considered unrelated to palonosetron (86%).
- The most common adverse events related to study medication were headache (19.3%), constipation (8.7%), dizziness (2.5%), and abdominal pain (2.5%).
Authors’ conclusion: Palonosetron was safe and effective as a monotherapy in treating chemotherapy-induced emesis and warranted further study as an antiemetic in patients receiving highly emetogenic chemotherapy.
Trial 4: Palonosetron is effective in preventing acute and delayed CINV in patients receiving highly emetogenic chemotherapy.
Objective: To assess the efficacy and safety of single IV doses of palonosetron 0.25 mg or 0.75 mg vs. ondansetron 32 mg administered 30 minutes prior to highly emetogenic chemotherapy.
Study design: Multicenter, randomized, double-blind, stratified Phase III study.
Methods
- Patients were randomized to receive a single, fixed intravenous dose of either palonosetron 0.25 mg, palonosetron 0.75 mg, or ondansetron 32 mg 30 minutes prior to highly emetogenic therapy (cisplatin > 60 mg/m2).
- 67% of patients in each group also received prophylactic corticosteroids.
- The primary endpoint
was 24-hour CR (see Table 9 for results).
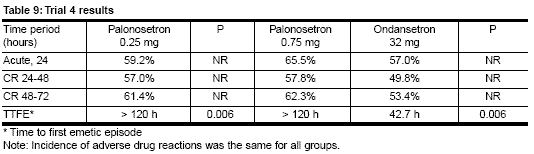
Author’s conclusion: A single dose of palonosetron prior to highly emetogenic chemotherapy is as effective as a single dose of ondansetron 32 mg for prevention of acute CINV and maybe of greater benefit in preventing delayed CINV after highly emetogenic chemotherapy.
Adverse drug reactions
To date, palonosetron appears to have a side effect profile similar to other currently available setrons; however, post-marketing surveillance should reveal any side effects that recent clinical trials may not have demonstrated. Also, one potential limitation of palonosetron is its ability to prolong QT and QTc intervals, although the drug has been administered safely to 192 patients with pre-existing cardiac impairment.
Cost analysis
Currently, palonosetron is the most expensive setron at Huntsville Hospital when comparing equivalent single doses; the formulary drug granisetron is the least expensive of the four available agents (ondansetron, dolasetron, granisetron, and palonosetron). The price difference per dose between granisetron and the other agents varies from $60 to $100 per dose.
Recommendations
Nausea and vomiting can be a strong predictor of a patient’s decision to continue an efficacious chemotherapy regimen. Palonosetron’s pharmacodynamics gives way to a binding affinity 100 times greater than that of any other setron. Kinetically, this drug has an elimination half-life of approximately 40 hours. Together, these properties yield efficacy in preventing both acute and delayed CINV. However, drugs in other classes are generally used first for delayed nausea and vomiting.
Palonosetron has not been compared to other regimens used for CINV, including oral and intravenous granisetron (the HH formulary drug). Benefits of palonosetron and other setron drugs include rapid and durable clinical effectiveness, ease of administration, single fixed dosing, and a favorable side effect profile. Additionally, palonosetron has no dosage adjustment in renal or hepatic impairment, and a low potential for drug interactions. Palonosetron is considerably more expensive than granisetron oral or IV in equivalent dosages (see cost summary section). As with other setrons, potential limitations of palonosetron include the ability to prolong the QT and QTc intervals.
Palonosetron should not be added to the formulary at this time. To date, palonosetron appears at least comparable to other setrons in the prevention of CINV, and shows positive results in preventing delayed symptoms.
However, greater experience with this drug through post-marketing surveillance will give a clearer picture with respect to the safety and efficacy of this new therapeutic option. Orders for palonosetron should be converted to granisetron according to the following dosage equivalents: palonosetron 0.25 mg IV for CINV be interchanged to either granisetron 1 mg IV or 2 mg oral tablet.
Resources
- MGI Pharma. Palonosetron (Aloxi) [formulary information request] Bloomington, MN; 2003.
- Gralla R, Lichinitser M, Van der Vegt S, et al. Palonosetron improves prevention of chemotherapy induced nausea and vomiting following moderately emetogenic chemotherapy: Results of a double-blind randomized Phase III trial comparing single doses of palonosetron with ondansetron. Ann Oncol 2003;14:1570-1577.
- Lacy CF, Armstrong L, Goldman MP, et al. Drug Information Handbook. 11th ed. Hudson, OH: Lexi-Comp; 2003.
- Markman MR, Peterson G, Kulp B, et al. Effectiveness of serotonin-receptor antagonist antiemetic therapy over successive courses of carboplatin-based chemotherapy. Gynecol Oncol 2002;85:435-437.
- Perez EA, Chawla SP, Kaywin PK, et al. Efficacy and safety of oral granisetron vs. IV ondansetron in prevention of moderately emetogenic chemotherapy-induced nausea and vomiting: A poster presentation. Proffered Papers 1997;S18.
- Roscoe J, Morrow GR, Hickok J, et al. Nausea and vomiting remain a significant clinical problem: Trends over time in controlling chemotherapy-induced nausea and vomiting in 1,413 patients treated in community clinical practices. J Pain Symptom Manage 2003;20:113-121.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
