Abdominal Aortic Aneurysm — The Great Imitator in the Elderly Patient
Abdominal Aortic Aneurysm—The Great Imitator in the Elderly Patient
Authors: Robert L. Rogers, MD, Departments of Medicine and Surgery, University of Maryland School of Medicine, Baltimore, MD; Amal Mattu, MD, Assistant Professor of Surgery, Division of Emergency Medicine, Co-Director of Internal Medicine/Emergency Medicine Combined Residency Program, University of Maryland School of Medicine, Baltimore, MD.
Peer Reviewer: Gary Hals, MD, PhD, Attending Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC.
A ruptured abdominal aortic aneurysm (AAA) is one of the most lethal conditions encountered in emergency medicine practice. Even when a diagnosis is made rapidly in the emergency department (ED) and the patient quickly is taken to the operating room, mortality rates in excess of 70% are reported.1,2 The rate of initial misdiagnosis or delays in diagnosis are reported to be 30-60%;1-3 this further complicates the care of these patients. Despite recent advances in diagnostic imaging modalities, including bedside ultrasound and helical computed tomography (CT), misdiagnosis and mortality rates remain high. This is, in large part, due to the frequency with which patients with ruptured AAAs report "atypical," or non-classic symptoms.
Classic teaching has been that the patient with a ruptured AAA presents with the triad of abdominal pain, hypotension, and a pulsatile abdominal mass. In reality, this triad is present in fewer than one-half of patients. Abdominal pain is present in only 70-80% of these patients, hypotension in as few as 70%, and a pulsatile abdominal mass in as few as 26% in some studies.1,2 In contrast, many patients will present with atypical symptoms and signs, including isolated back or flank pain, testicular pain, groin or thigh pain, gastrointestinal bleeding, or lower extremity neuropathy. Patients also rarely present with evidence of peripheral embolization or spinal cord ischemia. A particularly common misdiagnosis is renal colic because of the frequency with which these patients present with flank pain and microscopic hematuria. Diverticulitis is another common misdiagnosis because of frequent left lower quadrant pain. Both of these misdiagnoses can be avoided through the use of current radiological imaging studies.
The most common radiological imaging studies used in the evaluation of patients with suspected AAAs are plain radiographs, ultrasound, and CT scan. Plain radiographs of the abdomen have limited utility in the diagnosis of AAAs. Although they may demonstrate aortic calcifications and, therefore, suggest the presence of an aneurysm, absence of calcifications cannot exclude the diagnosis. Plain films will miss approximately 30% of aneurysms. Furthermore, plain films cannot identify the presence or absence of rupture.
The use of bedside ultrasound by emergency physicians has increased significantly during the past several years. Ultrasound is proving to be an important adjunct in the rapid evaluation of these patients. It has excellent sensitivity for detecting the presence of AAAs, although it is insensitive for detecting rupture. Nevertheless, the visualization of an aneurysm by ultrasound in an unstable patient with abdominal or back pain often provides enough evidence to justify emergent surgery.
CT has been the most common imaging study used to evaluate patients with suspected AAAs. There are several advantages to CT. Aneurysm size can be determined reliably, as can the presence of rupture. Advanced generation "helical" CT scan allows three-dimensional reconstruction of images, providing excellent anatomic details for vascular surgeons in planning surgery. However, the patient must leave the ED for a CT scan, which is not feasible in unstable patients.
Patients who are found to have unruptured, asymptomatic AAAs during a work-up of another medical disorder should be referred to a vascular surgeon for elective repair. There is significant controversy regarding the optimal timing of elective surgery. Follow-up and surgical planning should be discussed with the surgeon and the primary care physician.
The AAA represents a "high-risk" clinical entity for emergency physicians. Any elderly patient who presents to the ED complaining of abdominal pain, back pain, or symptoms consistent with renal colic or diverticulitis should be considered to have a ruptured AAA until proven otherwise. Misdiagnosis and delays in diagnosis are catastrophic. Emergency physicians must maintain vigilance for this entity and obtain early surgical consultation to achieve the best possible outcome for these patients. — The Editor
Introduction
Evaluation of abdominal pain in the elderly population can be a challenging and time-consuming endeavor for the emergency physician. Physicians who care for older patients are well aware that patients older than ages 55-60 who present with a complaint of acute abdominal pain often have serious intra-abdominal pathology, with significant, associated morbidity and mortality rates. One of the most lethal disease entities in this patient population is a ruptured AAA. Mortality rates in excess of 70% often are reported, even with rapid surgical intervention.1,2 The frequency with which delays in diagnosis or misdiagnosis occur (often in excess of 30%) further complicates the care of these patients.1 Patients with suspected or documented ruptured AAAs must be managed aggressively. Large bore intravenous access should be obtained, and patients should be volume-resuscitated if they are unstable. Blood should be sent for type and crossmatch early in the patient’s presentation. A vascular surgeon should be consulted immediately and operating room personnel should be mobilized rapidly. The prevalence of AAAs can be expected to grow as the median age of our society increases. Emergency physicians, therefore, must have a heightened sense of awareness for this catastrophic disease. Through familiarity with common and uncommon presentations of AAAs, emergency physicians will be better able to improve the outcome in elderly patients with ruptured or leaking AAAs.
Anatomy and Pathogenesis
The aorta enters the abdominal cavity at the level of the 12th thoracic vertebra. Intra-abdominal aortic branches include the celiac trunk, superior and inferior mesenteric arteries, and the renal arteries. The abdominal aorta then courses inferiorly along the retroperitoneum and bifurcates into the common iliac arteries at the level of the umbilicus. Most abdominal aneurysms are located below the renal arteries. Many structures, including the intestines, ureters, nerves, and bony structures, are in close proximity to the aorta. The proximity of these structures to the aorta is responsible for many of the atypical symptoms associated with AAAs (e.g., renal colic, gastrointestinal bleeding, neuropathy, etc.).3
The normal intra-abdominal aorta is approximately 2 cm in diameter, and is considered aneurysmal if it is greater than 3 cm. An alternative definition that allows for anatomic variability among patients is to define an aneurysm as anything greater than 1.5 times the adjacent normal aorta. Most AAAs are true aneurysms (i.e., they involve all layers of the vessel wall; see Figure 1).
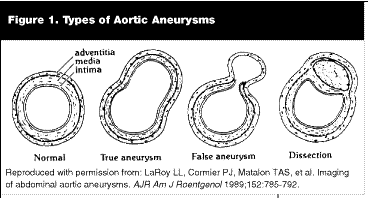 |
There are three primary layers of the aortic wall: the intima, the media, and the adventitia. The media is composed of connective tissue components such as collagen, elastin, and various enzymes. Increasing evidence suggests that the pathophysiology behind AAA formation, expansion, and rupture relate to the complex interplay between these structural components and their degradation by matrix-degrading enzymes known as metalloproteinases.4 Atherosclerosis was formerly thought to be the major risk factor for the development of aneurysm formation. However, recent evidence indicates that atherosclerosis plays a lesser role.5-8 The literature from the last several years indicate that increased metalloproteinase activity in the aortic wall leads to connective tissue—especially elastin—breakdown and eventual aneurysm formation.9,10 Hypertension is another contributing factor, as elevated blood pressure places a greater burden on the collagen once it is weakened by connective tissue degradation.11
Prevalence, Natural History, and Mortality
The prevalence of AAAs increases with age. As the median age of our population increases, emergency physicians will be faced with this challenging disease with increasing frequency. It is estimated that AAAs are four-fold as common and 10-fold more fatal in men than in women.8,12 Women tend to develop AAAs an average of 10 years later than their male counterparts. AAAs typically occur in the sixth and seventh decades of life.12 The reported prevalence of AAA in men older than age 65 ranges from 4% to 8%.9,13 The average age range at the time of diagnosis is 65-70 years.14
The risk of rupture of an AAA is related to size, and the risk dramatically increases as the aneurysm grows in diameter. It should be emphasized, however, that small aneurysms can and do rupture. For aneurysms less than 4 cm in diameter, the yearly risk of rupture is about 0.5-1.0%. For AAAs of 4-5 cm in diameter, the yearly risk is 5-20%. For aneurysms greater than 10 cm in diameter, the yearly risk of rupture is as high as 60%. The rate of aneurysm expansion also correlates with the aneurysm size. The rate of expansion usually is estimated to be 3-5 mm per year, but there is individual variation. Factors that increase the risk of expansion and rupture include hypertension, tobacco use, and the presence of peripheral vascular disease.15-17
Both morbidity and mortality are high in patients with an AAA. It is estimated that rupture of an AAA leads to 10,000-15,000 deaths annually in the United States—ranking AAA as the 13th leading cause of death.18 Nearly 40% of patients die before they reach the hospital.19 Several studies show an average mortality rate of about 80% for ruptured AAAs. High morbidity and mortality rates in patients undergoing emergent repair are attributable to perioperative and postoperative complications, such as myocardial infarction (MI), renal failure, and sepsis.20,21
Risk Factors
Several risk factors have been shown to be associated with AAA formation. (See Table 1.) These include male sex, advanced age (mean age of diagnosis, 67 years) hypertension, tobacco use, and family history.22
| Table 1. Risk Factors for the Development of Abdominal Aortic Aneurysm7 | |
| • | Male sex |
| • | Age older than 60 years |
| • | Hypertension |
| • | Tobacco smoking |
| • | Coronary artery disease |
| • | Marfan’s syndrome |
| • | Ehlers-Danlos syndrome |
| • | First-degree relative with AAA |
Patients with connective tissue disorders such as Marfan’s disease are rare, but are at high risk for AAA development. Connective tissue laxity in these patients leads to AAA development at younger ages than the classic elderly AAA patient.23,24 Although chronic obstructive pulmonary disease was once thought to be a risk factor, it seems more likely that comorbid conditions, including hypertension, tobacco use, and peripheral vascular disease, play a greater role. There does appear to be a familial tendency for development of AAAs. As many as 20% of first-degree relatives of patients with an AAA also will have an AAA. The prevalence of AAAs is higher in the brothers of patients with aneurysms, with a concurrent prevalence as high as 25%. It has been proposed that patients in this high-risk group undergo periodic screening to detect aneurysm formation.25
Clinical Presentations
Unruptured Aneurysms. Unruptured abdominal aneurysms represent a large portion of clinical presentations. Unruptured AAAs may be asymptomatic or symptomatic. Asymptomatic AAAs often are discovered during a routine screening examination of the asymptomatic patient, or during the work-up of a patient who presents with abdominal or back complaints and eventually is diagnosed with an alternative condition.26-29 It is important to note that the diagnosis of alternative conditions as the cause of the patient’s symptoms is best left to a vascular consultant. In other words, any patient with an AAA and symptoms possibly related to it should be treated as if they have a rapidly expanding or ruptured AAA. Surgical intervention may be needed to truly rule out whether AAA is the cause of his or her symptoms.
Unruptured abdominal aneurysms may cause painful symptoms, and this usually indicates rapid expansion and impending rupture. Patients with symptomatic, unruptured AAAs typically present with abdominal, back, or flank pain. They also may present with a sensation of pulsations in the abdomen or abdominal "fullness."27 It should be emphasized that isolated back pain is a common presentation; therefore, the absence of abdominal pain should not exclude consideration of an AAA. Abdominal pain may be diffuse and poorly localized, or it may be localized and simulate other intra-abdominal conditions, such as cystitis, peptic ulcer disease, renal colic, and diverticulitis. Symptoms may be vague and nonspecific. As renal colic is the most frequent misdiagnosis for symptomatic AAA, emergency physicians should have a high index of suspicion for a rapidly expanding or ruptured AAA when evaluating anyone older than age 55 years who presents with flank pain and hematuria. These patients often are misdiagnosed by the unwary physician with renal colic, despite the absence of any prior history of nephrolithiasis.
The physical examination in patients with AAAs is associated with a high percentage of both false-positive and false-negative findings, and thus frequently can be misleading. Patients without an AAA may be noted to have a pulsatile aorta. Conversely, patients with an AAA may be noted to have a completely normal abdominal exam. The aorta should be non-tender in patients with asymptomatic AAAs. Tenderness is indicative of either rapid expansion or rupture. Palpation in asymptomatic patients can be performed without fear of precipitating rupture.30 Classically, it is taught that the abdominal exam in patients with an AAA should be notable for the presence of a pulsatile, abdominal mass. However, small aneurysms or aneurysms in obese patients may not be easily detected. Further complicating matters, thin patients and patients with tortuous aortas often are mistakenly diagnosed with an AAA based on physical exam. The most clinically useful aspect of the physical exam probably is the detection of abnormal widening of the aortic pulsation.31 Although abdominal palpation of the aorta has only a moderate sensitivity in the detection of AAA, the sensitivity increases as the diameter of the aorta increases. The sensitivity of physical exam is reported to be 29% for AAAs of 3.0-4.0 cm in diameter, 50% for AAAs of 4.0-4.9 cm, and 76% for AAAs of 5.0 cm or greater.31 Other studies note an even higher physical exam sensitivity.32
Abdominal bruits occur in approximately 10% of patients with AAA. Pulses in the femoral region as well as the entire lower extremity should be examined. Ilioaortic occlusive disease, a ruptured AAA, or aortic thrombosis may lead to diminished pulses. Physicians should, however, realize the limitations of this part of the examination. Normal pulses do not exclude the presence of an AAA.33 In one study, the presence of abdominal or femoral bruits and the absence of femoral pulses had no predictive value for the presence of AAAs. Bruits may be caused by stenosis of renal or mesenteric arteries. The performance of these physical exam maneuvers is important in the detection of complications of an AAA; they may lead to the discovery of other etiologies, such as renal artery stenosis, but they do not seem to be helpful for the detection of AAAs.28
Ruptured Aneurysms. It is estimated that approximately 40% of patients with AAAs present for the first time with frank rupture of the aneurysm.34-36 Only a minority of patients with a ruptured AAA will have been diagnosed previously with an AAA.8 It is classically taught that patients with ruptured AAAs present with abdominal pain and distension, a pulsatile abdominal mass, and hypotension. In reality, only 30-50% of patients will present with this triad.37 Physicians should be wary of the patient who presents with abdominal pain and reports a history of diaphoresis and/or syncope. Abdominal pain and syncope in the older patient represent a high-risk clinical encounter; this presentation should be considered an AAA rupture until proven otherwise. Early consideration and rapid evaluation of a suspected AAA based on history may help minimize morbidity and mortality. (See Table 2.)
|
Table 2. History and Physical Exam Findings in Symptomatic AAA34 |
|
| • | Prominent, pulsatile aorta |
| • | Abdominal bruit |
| • | Unexplained hypotension or "sepsis" appearance |
| • | Hematuria |
| • | Signs of peripheral embolization ("blue toe syndrome") |
| • | Syncope |
| • | Diaphoresis |
| • | Left lower quadrant abdominal mass |
AAAs also can present with acute onset of back or flank pain, especially when associated with retroperitoneal rupture. Hypotension initially may be absent when rupture occurs into the left retroperitoneal space because hematoma expansion may be contained by tamponade. Nearly 65-70% of patients initially may be normotensive, presumably from this mechanism.34 Retroperitoneal rupture is more common than intraperitoneal rupture. Intraperitoneal rupture leads to rapid hemodynamic deterioration causing most patients to die before reaching the hospital. Retroperitoneal bleeding occurs in about 80% of cases, and typically causes back pain. Patients may present in extremis from rapid bleeding or may present days to weeks later with a chronic, contained retroperitoneal rupture.
There may be a delay in diagnosis of chronic, contained ruptures due to consideration of more common diagnoses such as herniated lumbar discs and renal colic. Patients with chronic, contained ruptures may present with back pain due to erosion into lumbar vertebral bodies; lower extremity neuropathy due to impingement on peripheral nerves; and rarely, obstructive jaundice due to compression of biliary vessels.21 Thus, some patients may present without a history of acute abdominal or back pain. Even when the pain is sudden and severe in onset, the pain may radiate to the inguinal region, testicles, or thigh, simulating renal colic.38,39 All of these presentations frequently lead to misdiagnosis, with resulting increased morbidity and mortality.
Misdiagnosis
Misdiagnosis of ruptured AAAs is quite common, with some studies reporting misdiagnosis rates as high as 60%.40-42 Patients who initially are misdiagnosed have almost twice the risk of mortality as patients who are correctly diagnosed.8,20 Ruptured AAAs are well known for producing atypical symptoms and signs, which are responsible for the high rate of misdiagnosis. (See Tables 3 and 4.)
|
Table 3. Atypical Presentations of AAA |
|
| • | Flank pain mimicking renal colic |
| • | Left lower quadrant pain mimicking diverticulitis |
| • | Testicular pain |
| • | Isolated back pain |
| • | Groin/thigh pain |
| • | Spinal cord ischemia |
| • | Aortic thrombosis with embolization |
| • | Lower extremity radiculopathy |
| • | Gastrointestinal bleeding |
|
Table 4. Common Misdiagnoses of AAA41 |
|
| Misdiagnosis |
Percentage |
| Renal colic | 23% |
| Gastrointestinal bleed | 13% |
| Diverticulitis | 12% |
| Musculoskeletal pain | 9% |
| Acute myocardial infarction | 9% |
| Sepsis | 7% |
| Other GI disorders (e.g., perforated viscus, appendicitis, pancreatitis) | 7% |
The classic triad of abdominal pain, pulsatile abdominal mass, and hypotension is present in fewer than 50% of patients.37 Only a minority of these patients will have a known diagnosis of AAA when they present with an acute rupture.42 As a result, many physicians will tend to attribute the patient’s presentation to a more common and perhaps more benign diagnosis. For example, hypotension may be attributed to sepsis,41 and not AAA rupture. Because imaging modalities are accurate for the presence of AAAs in patients, the key to avoiding misdiagnosis often is to think of and look for AAAs in elderly patients being diagnosed with renal colic, diverticulitis, appendicitis, pancreatitis, acute coronary syndromes, or musculoskeletal back pain. None of the alternative diagnoses need treatment as aggressive as that required for a ruptured AAA—so the worst diagnosis should be ruled out first in these patients.
Further confounding diagnostic efforts, many patients present without acute abdominal pain. A large number of patients will present with acute back pain, leading the unwary physician to make a diagnosis of musculoskeletal pain. Patients also may present with acute flank pain. When the aneurysm compresses the ureter, the pain may radiate toward the groin and also may be associated with microscopic hematuria, simulating ureterolithiasis. Indeed, renal colic is the most common misdiagnosis in patients with ruptured or rapidly expanding AAAs. An elderly patient who presents with symptoms that are typical of renal colic should be considered to have a ruptured AAA until proven otherwise.
Ruptured AAAs also are frequently misdiagnosed as various gastrointestinal disorders. Because abdominal pain from AAAs often occurs in the left lower quadrant, patients often are thought to have diverticulitis. Right lower quadrant pain may lead the physician to make the diagnosis of appendicitis. If an aortoenteric fistula forms, patients may develop gastrointestinal bleeding, which also may mislead the treating physician. The sudden nature of the pain may simulate a perforated viscus. The severity of the pain may simulate pancreatitis.
Ruptured AAAs may be misdiagnosed as acute cardiac conditions as well. Patients with a ruptured AAA may present with syncope. As a result, these patients are sometimes misdiagnosed as having acute coronary syndromes or acute dysrhythmias. The additional presence of hypotension and/or tachycardia also may mislead the treating physician into believing the patient has an acute cardiac condition. However, some patients may have both—an acute coronary syndrome precipitated by acute AAA rupture. Hypotension from the leaking AAA can cause acute coronary ischemia. Often the acute MI will be diagnosed first, but patients complaining of abdominal or back pain should have AAA rupture or thoracic aortic dissection ruled out as a cause of their MI.
AAA also can cause neuromuscular symptoms and signs. Expansion, rupture, or thrombosis of the aneurysm may cause spinal cord ischemia in the T10-T12 region due to interruption of blood flow to the greater radicular artery (artery of Adamkiewitz).43 Patients rarely may present with a femoral neuropathy caused by compression of the femoral nerve against the iliopsoas muscle by retroperitoneal blood. This may cause hip or thigh pain. Femoral nerve dysfunction also may cause quadricep weakness, decreased sensation over anteromedial thigh, and a diminished patellar reflex.44,45
Complications of Unruptured Aneurysms
Unruptured AAAs may be associated with several rare complications that generally are relevant to the vascular surgeon during the operative repair. However, emergency physicians should be aware of two specific complications that may cause atypical presentations: peripheral embolization and gastrointestinal bleeding.
Intramural thrombus within the aneurysm may cause peripheral embolization and the "blue toe syndrome." This mechanism, surprisingly, may be responsible for up to 10% of all peripheral emboli. Nearly 5% of patients with an AAA will present with evidence of peripheral embolization.8 Any patient who presents with embolic phenomenon should be evaluated for the presence of an AAA.
In some patients, erosion of an aneurysm into the intestines may lead to massive upper or lower gastrointestinal (GI) bleeding. It is important to note that some patients will present with massive GI bleeding, but that others will have minimal bleeding. Any patient with GI bleeding and a history of AAA repair should be consulted and evaluated for aortoenteric fistula as the cause. The duodenum is the area of intestine most commonly involved, but this may occur at any point in the gastrointestinal tract, including the esophagus. These "aortoenteric fistulas" usually occur following surgical repair of a ruptured AAA. Misdiagnosis and delays in diagnosis contribute to the greater than 50% mortality associated with this complication.8,22 Treatment requires hemodynamic support for the unstable patient and prompt surgical consultation for graft replacement.
Radiological Evaluation
Plain Abdominal X-Rays. Plain films have been used for years as an initial screening tool for the detection of many abdominal diseases, including AAA. It is estimated that evidence of an AAA is seen in 55-68% of plain x-rays.8 Calcification of the aortic wall is the most common abnormality found in plain radiographs. (See Figure 2, x-ray of AAA calcifications.)
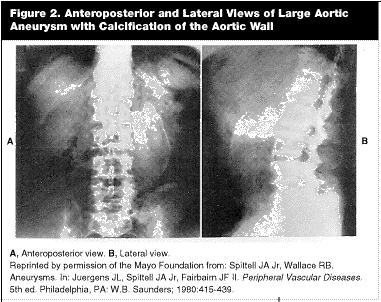 |
Physicians should remember, however, that the outer rim of the aneurysm might not be calcified. Failure to account for this may lead to an underestimation of aneurysm size. The presence of a paravertebral soft-tissue mass and loss of the psoas or renal shadows are other abnormalities associated with AAA on plain radiographs.
Advantages of plain radiographs include low cost and rapid availability. The studies also can be obtained without requiring the patient to leave the ED. The main disadvantage is the low sensitivity. In addition, plain x-rays are unable to detect aneurysm rupture and are unable to delineate detailed aortic branch anatomy. Plain radiographs should be obtained in the elderly patient with severe abdominal pain to evaluate for the presence of other causes of intra-abdominal disorders (e.g., ruptured viscus, bowel obstruction). The presence of aortic calcifications should strongly suggest the diagnosis, but their absence also clearly does not exclude the diagnosis.46-50 Thus, plain films are not indicated in patients who are considered to have high suspicion of AAA rupture unless ultrasound or CT is not readily available.
Ultrasound. Ultrasound has been used to detect AAAs for many years. Ultrasound scanning for AAAs is rapid, highly sensitive, inexpensive, noninvasive, and relatively easy for even an inexperienced user to perform. Ultrasound appears to have a strong role as an adjunctive imaging modality in the diagnosis of AAA. It can be used as a screening tool in patients who are considered to be high-risk and in patients who present with symptoms that are compatible with an AAA. Recently, an increasing number of EDs and emergency medicine residency programs across the country have been using ultrasound for the detection of AAAs. In one study, interpretation of 68 total scans was 100% accurate in the detection of AAAs.51 In another, similar study, 31 of 32 patients were correctly identified as having an AAA. In the study, rapid, bedside ultrasound was performed by ultrasonographers who were readily available to the ED.52 Evidence is mounting that even relatively new ultrasound users can accurately identify AAAs.51 In addition to its speed and ease of use, ultrasound also is desirable because it can be performed at the patient’s bedside and will not interfere with ongoing resuscitative efforts. Ultrasound also is very accurate for measuring the size of the aneurysm. (See Figure 3, ultrasound of AAA.)
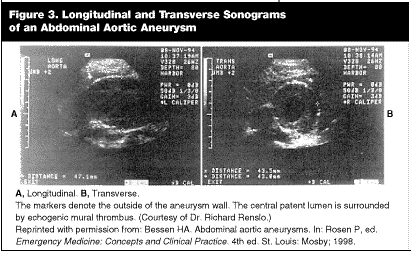 |
Although ultrasound has excellent sensitivity for detecting the presence of an AAA, it has poor sensitivity for detecting the presence of rupture. Difficulty with obtaining adequate images in obese patients or in patients with excessive bowel gas is another drawback of ultrasound. Other vascular structures adjacent to an AAA and the detailed aortic branch anatomy, both important to vascular surgeons, also are not visualized.51-55
Despite these limitations, however, performance of rapid bedside ultrasound is becoming more common in the evaluation of elderly patients who present with abdominal, back, or flank pain. Ultrasonography serves as an excellent screening test for patients with a suspected AAA. Demonstration of an aneurysm by ultrasound in the hypotensive patient with abdominal pain often is enough to prompt rapid mobilization of the operating team and an emergency surgery. Early bedside ultrasound, therefore, should be used when it is available for all patients with AAA risk factors who present with either abdominal or back pain and hypotension, or in stable patients with unexplained abdominal or back pain.54
Computerized Tomography. Abdominal CT scanning is the most reliable diagnostic modality for detection of an AAA. (See Figure 4, CT of AAA.) It is highly sensitive for detecting aneurysms (nearly 100%) and rupture (> 90%). In most centers, it is easy to obtain as well.
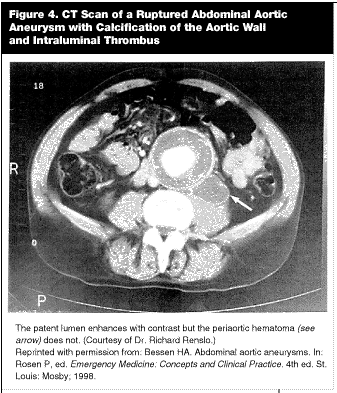 |
There are many other advantages to using CT scanning in patient management. Recent technological improvements in abdominal CT scanners have made them very fast. These new "helical" CT scanners can provide three-dimensional reconstruction of images, allowing vascular surgeons to see detailed anatomy and involvement of aortic branches.55,56 Problems inherent to ultrasound, such as interpreting abnormalities in the presence of excessive bowel gas, do not exist with CT scanning.
There are, however, several disadvantages to using CT for the detection of an AAA. CT is more expensive than plain radiography or ultrasound. Accurate delineation of aortic branch involvement requires administration of intravenous contrast material, with the inherent risk of contrast nephrotoxicity and allergic reactions to the dye. CT also can produce false-negative results. Case reports of patients who had scans that showed no rupture or hematoma but who later decompensated from a ruptured AAA are scattered throughout the vascular surgery literature.57 Thus, with these patients in mind, all patients with symptomatic AAAs but no evidence of rupture on CT scan should have emergency consultation by a vascular surgeon. Surgical intervention may be required to truly rule out AAA rupture. Perhaps the greatest disadvantage to performing a CT scan on the patient is that it requires the patient to physically leave the ED and travel to the radiology department or CT suite for the study. The patient, therefore, must have relatively stable vital signs before transport. Ideally, the patient also should be accompanied by a physician or experienced nurse with monitoring and resuscitation equipment. Despite this risk, CT scanning still is the most commonly used diagnostic modality for detection of an AAA, detection of rupture, and preoperative planning.55,57
Angiography. Angiography has been a traditional imaging modality performed by vascular surgeons for years, but it is not a useful screening tool in the ED. Angiography provides good anatomic detail, and is helpful in the evaluation of aortic branch involvement and surgical planning. However, angiography has several drawbacks. This modality has a sensitivity for detection of aneurysm and rupture less than that of CT (80-90%). Because mural thrombus formation is common in patients with an AAA, angiography can underestimate aortic diameter. Other disadvantages include the high cost, the invasive nature of the procedure, a long set-up time, and the risk of contrast nephrotoxicity and allergic reactions.3,8,47
Magnetic Resonance Imaging. Magnetic resonance imaging (MRI) is an accurate imaging modality for the detection and evaluation of AAAs. The sensitivity of MRI for detecting aneurysms approaches 100%, and the accuracy for detecting rupture also is greater than 90%.8 Furthermore, MRI is reported to be superior to ultrasound and CT for detection of aortic branch vessel involvement and for detection of visceral involvement.42 MRI is noninvasive and exposes the patient to no ionizing radiation or contrast dye.
There are several important disadvantages to the use of MRI for evaluation of the suspected AAA. As with CT, patients must be transported out of the ED to the MRI suite for the study, making MRI impractical in the unstable patient. In addition, the study takes significantly more time to perform than either ultrasound or CT. Claustrophobic patients require sedatives to tolerate the procedure. These medications may compromise an already tenuous blood pressure. MRI also is significantly more expensive than other imaging studies, and is less widely available. Finally, patients who have implanted metallic objects (e.g. pacemaker, surgical clips) or are connected to metallic monitoring equipment cannot be imaged. From a practical standpoint, the use of MRI generally is reserved for the stable outpatient undergoing preoperative evaluation for elective repair of an AAA.42,58
Management
Patients who present with acute onset of back, flank, or abdominal pain should have a rapid bedside ultrasound (if available). If an aneurysm is detected by ultrasound in such a patient, the patient should be assumed to have a ruptured AAA and a vascular surgeon should be consulted immediately. Patients with a previous, known history of an AAA with suspicious symptoms also should be assumed to have a ruptured AAA. In EDs that do not have available rapid bedside ultrasound, patients with stable vital signs and no obvious evidence of an abdominal aneurysm by physical exam may be sent for radiographic imaging (e.g., abdominal CT). The patient should receive continuous monitoring, have well-established intravenous access, and have airway equipment nearby in case deterioration ensues. Physicians should consult a vascular surgeon as soon as strong clinical suspicion of an AAA is entertained, even before diagnostic imaging has confirmed the diagnosis.
Ruptured Aneurysms. Management of the patient who presents with a ruptured AAA often begins in the prehospital arena. Paramedics may arrive on the scene and obtain information regarding a previously known diagnosis of an AAA. For symptomatic patients with a known history of an AAA, or for patients with a pulsatile distended abdomen, hypotension, and abdominal or back pain, a vascular surgeon should be notified immediately and operating room personnel should be mobilized. This prehospital alert allows ED and operating room staff to be ready for the patient’s arrival.
Treatment in the ED should begin with rapid assessment of the airway and respiratory status. Supplemental high-flow oxygen should be administered to maintain good arterial saturation. Early stabilization of the airway with intubation is warranted in patients who are rapidly decompensating. Two large bore intravenous lines should be obtained, and blood should be typed and cross-matched for at least 10 units of blood. Rapid measurement of hematocrit and other preoperative labs should be performed. Intravenous fluids and blood, when available, should be administered to maintain a sufficient blood pressure. Adequate blood pressure will be different in each patient, and should be measured clinically by signs of end organ perfusion. Goals include maintenance of mentation and prevention of cardiac ischemia. Lower blood pressures are advantageous as less bleeding will occur from the aneurysm, whereas higher pressures will increase blood loss. Unstable patients should be rapidly transported to the operating room without delay. It is both unwise and dangerous to transport hemodynamically unstable patients to the CT suite in cases in which the diagnosis is fairly certain. Unstable patients have an increased mortality rate when surgery is delayed for preoperative testing.22,59
Hemodynamically stable patients with suspected ruptured or expanding AAA generally are diagnosed definitively with an imaging study before operative repair. However, a vascular surgeon should be notified promptly so that arrangements can be made for immediate surgical repair. This surgical consultation should be made as soon as the diagnosis is strongly considered, even before definite diagnosis is made. Physicians should not be reassured by temporary hemodynamic stability; these patients can deteriorate rapidly, and should be treated as potentially unstable.
Unruptured Aneurysms. Many patients present to the ED or a physician’s office and are found to have asymptomatic AAAs that are detected by radiographic imaging during the evaluation of another complaint. Patients with asymptomatic AAAs can be referred to a vascular surgeon and their primary care physician for close follow-up. Strict instructions about warning signs that should prompt emergent return to the ED should be given. Other patients are found to have unruptured aneurysms during the evaluation for abdominal or back pain or urologic complaints. Proper management involves urgent consultation with a vascular surgeon. As no test is 100% accurate for identification of rupture or leak in an AAA, all symptomatic AAAs should be treated carefully. Evidence of rupture may not be found until the time of surgery, and the vascular surgeon needs to be involved in patient care at an early stage. Unruptured aneurysms that are considered to be expanding are treated very differently than asymptomatic, unruptured aneurysms. The decision to surgically repair the aneurysm generally is based on the aneurysm size and the patient’s symptoms. Because of the presence of other comorbid medical problems such as coronary artery disease and obstructive lung disease, further preoperative testing may be necessary.
Controversy still exists regarding the size of the aneurysm at which elective surgical repair is indicated.50,53 Some surgeons advocate early repair for even small aneurysms (3-5 cm) based on the probability that they will increase in size and eventually rupture. They argue that waiting until the aneurysm grows in size is associated with greater surgical risk because of the increase in the patient’s age.60 Others argue that these patients should be followed with serial ultrasound examinations; elective repair should occur when the aneurysm diameter is 5-6 cm, if acute expansion is suspected, or if the patient develops symptoms.61,62 Most aneurysms have an average expansion rate of 3-5 mm per year.15 The timing of surgery generally is left to the discretion of the consulting vascular surgeon.
Surgical Repair
Most patients who are found to have ruptured aneurysms or rapidly expanding aneurysms will undergo open abdominal repair, which involves cross clamping of the aorta and interposition of a vascular graft. Mortality estimates vary, but the risk of death with emergent surgical repair generally is estimated to be approximately 50%.63 One study found intraoperative mortality to be 21% and overall mortality to be 57%.36 Factors related to an increased mortality were age, severity of initial shock, amount of blood transfused, and location of rupture. Although surgeons traditionally have used open abdominal aneurysm repair, endovascular repair is becoming more common and has a growing role in patients who are very unstable or have many comorbid medical conditions.64-66
Complications of Surgical Repair. Although the estimated five-year survival rate after repair is approximately 75%, patients may present weeks, months, or years after repair with certain complications of which physicians should be aware.67 Patients may present with new aneurysm formation. The most common site tends to be above the level of the repair.
Patients also may present with graft infection. This should be suspected in patients who recently have undergone repair and who present with fever and/or sepsis. This entity has a high mortality rate without prompt recognition and treatment. Antibiotic therapy aimed at the most common pathogen, Staphylococcus epidermidis, and prompt graft excision are required.
One of the most dreaded complications is the development of an aortoenteric fistula. This complication occurs due to graft erosion into a portion of the gastrointestinal tract and manifests as gastrointestinal bleeding. Bleeding can be minimal or extensive. Aortoenteric fistula should be strongly suspected in patients who have undergone repair that present with any upper or lower gastrointestinal bleeding.46 The fistula usually involves the duodenum, but may involve any part of the gastrointestinal tract, including the esophagus. Treatment requires initial supportive care followed by emergent graft replacement. Long-term survival for these patients, even after operative repair, is poor.22,62,67
Future Treatment
Research in the last several years has shown that the formation and rupture of AAAs is related to connective tissue degradation by normal tissue enzymes called metalloproteinases. An interesting approach to the prevention and treatment of AAAs is the use of medications that inhibit these degradative enzymes found in the aortic wall. Doxycycline currently is being investigated for use in the prevention of AAA growth and expansion. This medication is thought to work by inhibiting metalloproteinase mRNA synthesis.68 Data from animal studies, human tissue experiments, and limited human studies show that treatment with doxycycline may benefit patients. One recent study found that patients pretreated with doxycycline before operative repair of established AAAs had decreased metalloproteinase production and activity as measured in their postoperative AAA tissue samples.68 Prospective, randomized clinical trials are needed to clarify the role of doxycycline and other similar medications in the early treatment of AAAs.
Risk Management Issues
AAAs represent a "high-risk" clinical entity for the emergency physician. That is, the ruptured AAA is one of the few presentations in emergency medicine in which prompt recognition and treatment can be associated with a good outcome, whereas failure to diagnose or delays in diagnosis have catastrophic results for the patient. In terms of medicolegal risk to the emergency physician, a study showed that failure to diagnose (or delays in diagnosis of) AAA represents 3% of all money paid out by emergency physicians and 2% of total claims.40 Table 5 lists key points that emergency physicians should remember to minimize both risk to the patient and malpractice risk.
| Table 5. Risk Management Pearls22 | |
| • | Abdominal, flank, or back pain in an elderly patient is an AAA until proven otherwise. |
| • | Common misdiagnoses include renal colic and diverticulitis. |
| • | An elderly patient presenting with apparent renal colic (flank pain and hematuria) has an AAA until proven otherwise. |
| • | Urologic symptoms are present in approximately 10% of patients. |
| • | Ruptured AAAs may present with urinary retention. |
| • | Patients may present with vague abdominal pain or back pain with histories of muscle strain. |
| • | AAA must be ruled out when an elderly patient presents with hypotension or syncope. |
| • | Patients with symptomatic AAAs may deteriorate rapidly, regardless of initial hemodynamic stability. |
| • | Physical examination is unreliable in excluding the diagnosis of an AAA. |
| • | Patients who present with signs of peripheral emboli should be evaluated for an AAA. |
| • | Bedside ultrasound is a rapid and reliable way to screen for the presence of an AAA. |
Summary
As our population ages, we can expect vascular emergencies such as AAAs to become increasingly common. Ruptured AAAs are associated with a significant morbidity and mortality. Delays in diagnosis and misdiagnosis contribute significantly to poor outcome. Only sound knowledge of both the common and the atypical presentations of AAAs will increase the likelihood of early diagnosis. Bedside ultrasound is being utilized increasingly to aid in early diagnosis of this deadly entity.
Treatment of the patient with a symptomatic or ruptured AAA requires aggressive supportive care. Large bore intravenous lines should be placed and blood should be typed and crossmatched. A vascular surgeon should be consulted as soon as the diagnosis is strongly suspected. Prognosis is dependent on rapid surgical repair.
Debate exists regarding the optimum timing of surgical repair in patients with asymptomatic, incidentally found AAAs. Proper management, whether as an inpatient or outpatient, should be coordinated with the consulting vascular surgeon and the patient’s primary care physician.
Recent advances have been made in surgical repair techniques for symptomatic AAAs, as well as in possible medical treatments for asymptomatic AAAs. For now, however, the best possible patient outcome remains in the hands of emergency physicians. Only through prompt recognition, aggressive supportive treatment, and early surgical consultation can these patients’ lives be preserved.
References
1. McNamara RM. Acute abdominal pain. In: Sanders AB, ed. Emergency Care of the Elder Person. St. Louis: Beverly Cracom Publications; 1996:219-243.
2. Sanson TG, O’Keefe KP. Evaluation of abdominal pain in the elderly. Emerg Med Clin North Am 1996;14:615-627.
3. Ouriel K, Green RM. Arterial disease. In: Schwartz SI, Shires GT, Spencer FC, et al, eds. Principles of Surgery. 7th ed. New York: Mcgraw-Hill; 1999:931-1005.
4. Grange JJ, Davis V, Baxter BT. Pathogenesis of abdominal aortic aneurysm: An update and look toward the future. Cardiovas Surg 1997;5:256-265.
5. Crowther M, Goodall S, Jones JL, et al. Localization of matrix metalloproteinase 2 within the aneurysmal and normal aortic wall. Br J Surg 2000;87:1391-1400.
6. Cohen JR, Keegan L, Sarfati I, et al. Smooth muscle cell elastase, atherosclerosis, and abdominal aortic aneurysms. Ann Surg 1992;216:327-332.
7. Blanchard JF, Armenian HK, Friesen PP. Risk factors for abdominal aortic aneurysm: Results of a case-control study. Am J Epidemiol 2000;151:575-583.
8. Rothrock SG, Green SM. Abdominal aortic aneurysm: Current clinical strategies for avoiding disaster. Emerg Med Reports 1994;15:125-136.
9. Patel MI, Melrose J, Ghosh P, et al. Increased synthesis of matrix metalloproteinases by aortic smooth muscle cells is implicated in the etiopathogenesis of abdominal aortic aneurysms. J Vasc Surg 1996;24:82-92.
10. Limet R, Sakalihasan N, Lapiere CM. Mechanism of the growth and rupture of abdominal aortic aneurysm. Bull Mem Acad R Med Belg 1997;152:295-302.
11. MacSweeney ST, Powell JT, Greenhalgh RM. Pathogenesis of abdominal aortic aneurysm. Br J Surg 1994;81:935-941.
12. Bessen HA. Abdominal aortic aneurysms. In: Rosen P, ed. Emergency Medicine: Concepts and Clinical Practice. 4th ed. St. Louis: Mosby-Year Book, Inc; 1998:1806-1819.
13. Beebe HG, Kritpracha B. Screening and preoperative imaging of candidates for conventional repair of abdominal aortic aneurysms. Semin Vasc Surg 1999;12:300-305.
14. Sternbergh WC 3rd, Gonze MD, Garrard CT, et al. Abdominal and thoracoabdominal aortic aneurysm. Surg Clin North Am 1998;78: 827-843.
15. van der Vliet JA, Boll AP. Abdominal aortic aneurysm. Lancet 1997;349:863-866.
16. Pleumeekers HJ, Hoes AW, van der Does E, et al. Aneurysms of the abdominal aorta in older adults: The Rotterdam Study. Am J Epidemiol 1995;142:1291-1299.
17. Simoni G, Pastorino C, Perrone R, et al. Screening for abdominal aortic aneurysms and associated risk factors in a general population. Eur J Vasc Endovasc Surg 1995;10:207-210.
18. Gillum RF. Epidemiology of aortic aneurysm in the United States. J Clin Epidemiol 1995;48:1289-1298.
19. Mackiewicz Z, Molski S, Szpinda M, et al. J Mal Vasc 1998;23:368-370.
20. Johansen K, Kohler TR, Nicholls SC, et al. Ruptured abdominal aortic aneurysm: The Harborview experience. J Vasc Surg 1991;13:240-245.
21. Dorrucci V, Veraldi GF, Dushi R, et al. Chronic rupture of abdominal aortic aneurysms. Ann Ital Chir 2000;71:247-50.
22. Salkin MS. Abdominal aortic aneurysm: Avoiding failure to diagnose. ED Legal Letter 1997;8:67-78.
23. Wilmink AB, Quick CR, Hubbard CS, et al. The association between connective tissue laxity and the risk of an abdominal aortic aneurysm. Eur J Vasc Endovasc Surg 2000;20: 290-295.
24. Rehm JP, Grange JJ, Baxter BT. The formation of aneurysms. Semin Vasc Surg 1998;11:193-202.
25. Verloes A, Sakalihasan N, Koulischer L, et al. Aneurysms of the abdominal aorta: Familial and genetic aspects in three hundred thirteen pedigrees. J Vasc Surg 1995;21:646-655.
26. Walker JS, Dire D. Vascular abdominal emergencies. Emerg Med Clin North Am 1996;14:571-592.
27. Fielding JWL, Black J, Ashton F, et al. Diagnosis and management of 528 abdominal aortic aneurysms. BMJ 1981;283: 355-359.
28. Lederle FA, Walker JM, Reinke DB. Selective screening for abdominal aortic aneurysms with physical examination and ultrasound. Arch Intern Med 1988;148:1753-1756.
29. Beede SD, Ballard DJ, James EM, et al. Positive predictive value of clinical suspicion of abdominal aortic aneurysm. Arch Intern Med 1990;150:549-551.
30. Gore I, Hirst AE Jr. Arteriosclerotic aneurysms of the abdominal aorta: A review. Prog Cardiovasc Dis 1973;16:113-150.
31. Lederle FA, Simel DL. Does this patient have abdominal aortic aneurysm? JAMA 1999;281:77-82.
32. Fink HA, Lederle FA, Roth CS, et al. The accuracy of physical examination to detect abdominal aortic aneurysm. Arch Intern Med 2000;160:833-836.
33. Galland RB, Simmons MJ, Torrie EP. Prevalence of abdominal aortic aneurysm in patients with occlusive peripheral vascular disease. Br J Surg 1991;78:1259-1260.
34. Rutherford RB, McCroskey BL. Ruptured abdominal aortic aneurysm: Special considerations. Surg Clin North Am 1989;69: 859-868.
35. Treska V. Etiopathogenesis, diagnosis, and treatment of aneurysms of the abdominal aorta in the year 2000. Rozhl Chir 2000;79: 157-161.
36. Martinez R, Garces D, Podeur L, et al. Ruptured abdominal aortic aneurysm: A ten year experience. J Cardiovasc Surg 1997;38: 1-6.
37. Banerjee A. Atypical manifestations of ruptured abdominal aortic aneurysms. Postgrad Med 1993;69:6-11.
38. Balsano N, Cayten CG. Abdominal aortic aneurysms. In: Schwartz GR, et al, eds. Principles and Practice of Emergency Medicine. Philadelphia: Lea & Febiger; 1992:1370-1375.
39. Colucciello SA, Lukens TW, Morgan DL. Assessing abdominal pain in adults: A rational, cost-effective, and evidence-based strategy. Emerg Med Practice 1999;1:1-20.
40. Karcz, A, Holbrook J, Auerbach BS, et al. Preventability of malpractice claims in emergency medicine: A closed claims study. Ann Emerg Med 1990;19:865-873.
41. Marston WA, Ahlquist R, Johnson G Jr., et al. Misdiagnosis of ruptured abdominal aortic aneurysms. J Vasc Surg 1992;16: 17-22.
42. Tennant WG, Martnell G, Baird RN, et al. Radiologic investigation of aortic aneurysm disease: Comparison of three modalitites in staging and the detection of inflammatory change. J Vasc Surg 1993;17:703-709.
43. Szilagyi DE, Hageman JH, Smith RF, et al. Spinal cord damage in surgery of the abdominal aorta. Surgery 1978;83:38-56.
44. Merchant RF, Cafferata HT, DePalma RG. Ruptured aortic aneurysm seen initially as acute femoral neuropathy. Arch Surg 1982;117:811-813.
45. Edrees WK, Lee B. A rare case of radiculopathy in an elderly man. Postgrad Med 2000;76:586, 591-592.
46. Loughran CF. A review of the plain abdominal radiograph in acute rupture of abdominal aortic aneurysms. Clin Radiol 1986;37: 383-387.
47. Brewster DC, Darling RC, Raines JK, et al. Assessment of abdominal aortic aneurysm size. Circulation 1977;56(supp 2): 164-169.
48. LaRoy LL, Cornier RJ, Matalon TA, et al. Imaging of abdominal aortic aneurysms. Am J Roentgenol 1989;152:785-792.
49. Wears RL. Abdominal aortic aneurysm. In: Harwood-Nuss AL Wolfson AB, Linden C, et al, eds. The Clinical Practice of Emergency Medicine. 2nd ed. Philadelphia: Lippincott-Raven; 1996:179-181.
50. Hals G, Pallaci M. The clinical challenges of abdominal aortic aneurysm: Rapid, systematic detection and outcome-effective management, part I. Emerg Med Reports 2000;21:121-132.
51. Kuhn M, Bonnin RL, Davey MJ, et al. Emergency department ultrasound scanning for abdominal aortic aneurysm: Accessible, accurate, and advantageous. Ann Emerg Med 2000;36:219-213.
52. Shuman WP, Hastrup W Jr., Kohler TR, et al. Suspected leaking abdominal aortic aneurysm: Use of sonography in the emergency room. Radiology 1988;168:117-119.
53. Ernst CB. Abdominal aortic aneurysm. N Engl J Med 1993;328: 1167-1172.
54. Frazee BW. The abdominal aorta. In: Simon BC, Snoey ER, eds. Ultrasound in Emergency and Ambulatory Medicine. St. Louis: Mosby-Year Book, Inc; 1997:190-202.
55. Bernstein EF. Computed tomography, ultrasound, and magnetic resonance imaging in the management of aortic aneurysm. In: Hobson RW, Williams RA, Veith FJ, et al, eds. Vascular Surgery: Principles and Practice. 2nd ed. New York: McGraw-Hill; 1994.
56. Zarnke MD, Gould HR, Goldman MH. Computed tomography. Surgery 1988;103:638-642.
57. Greatorex RA, Dixon AK, Flower CD, et al. Limitations of computed tomography in leaking abdominal aortic aneurysms. BMJ 1988;297:284-285.
58. Durham JR, Hackworth CA, Tober JC, et al. Magnetic resonance angiography in the preoperative evaluation of abdominal aortic aneurysms. Am J Surg 1993;166:173-177.
59. Donaldson MC, Rosenberg JM, Bucknam CA. Factors affecting survival after ruptured abdominal aortic aneurysm. J Vasc Surg 1985;2:564-570.
60. Katz DA, Littenberg B, Cronenwett JL. Management of small abdominal aortic aneurysms: Early surgery vs watchful waiting. JAMA 1992;268:2678-2686.
61. Brown PM, Pattenden R, Gutelius JR. The selective management of small abdominal aortic aneurysms: The Kingston study. J Vasc Surg 1992;15:21-25.
62. Hals G, Pallaci M. The clinical challenges of abdominal aortic aneurysm: Rapid, systematic detection and outcome-effective management. Part II. Emerg Med Reports 2000;21:133-140.
63. Hollier LH, Taylor LM, Ochsner J. Recommended indications for operative treatment of abdominal aortic aneurysms: Report of a subcommittee of the Joint Council of the Society for Vascular Surgery and the North American Chapter of The International Society for Cardiovascular Surgery. J Vasc Surg 1992;15:1046-1056.
64. Zarins CK, Wolf YG, Lee WA, et al. Will endovascular repair replace open surgery for abdominal aortic aneurysm repair? Ann Surg 2000;232:501-507.
65. Laheij RJ, van Marrewijk CJ. Endovascular stenting of abdominal aortic aneurysm in patients unfit for elective open surgery. Lancet 2000;356:832.
66. de Virgilio C, Bui H, Donayre C, et al. Endovascular vs open abdominal aortic aneursym repair: A comparison of cardiac morbidity and mortality. Arch Surg 1999;134:947-951.
67. Moriyama Y, Toyohira H, Saigenji H, et al. A review of 103 cases with elective repair of abdominal aortic aneurysm: An analysis of the risk factors based on postoperative complications and long-term follow up. Surg Today 1994;24:591-595.
68. Curci JA. Preoperative treatment with doxycycline reduces aortic wall expression and activation of matrix metalloproteinases in patients with abdominal aortic aneurysms. J Vasc Surg 2000;31:325-334.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
