Calcium and Magnesium for Premenstrual Syndrome
By Judith L. Balk, MD, FACOG
In 400 BC, Hippocrates described tempestuous premenstrual behavior, and he attributed the symptoms to agitated blood seeking escape from the womb.1 Over the past 2,000 years, we have made substantial progress in understanding premenstrual syndrome (PMS), but this condition still is not understood completely.
Definition and Prevalence
PMS is defined as "the cyclic occurrence of symptoms that are of sufficient severity to interfere with some aspects of life and that appear with consistent and predictable relationship to the menses." Although the symptoms themselves are fairly common, the restriction of the symptoms to only the luteal phase is pathognomonic of PMS.2 Premenstrual symptoms are common and often are considered to be a normal aspect of menstrual cycles, with surveys finding that up to 85% of menstruating women report one or more premenstrual symptoms. However, the definition of PMS requires that the symptoms be of sufficient severity that they interfere with aspects of life, and only 5-10% of menstruating women meet this criteria.2 More recently, the fourth edition of the Diagnostic and Statistical Manual of Mental Disorders includes premenstrual dysphoric disorder (PMDD), which differs from PMS in the severity of the emotional symptoms that a patient experiences.
Etiology
No one etiology has been found for PMS. Since the description of PMS decades ago, many etiologies have been suggested, from hormonal factors to nutritional factors to central nervous system factors. Most likely, there are interactions between these factors. For instance, there may be central nervous system-mediated neurotransmitter interactions with sex steroids.2 Also, sex steroid hormones have been found to modulate serum ionized magnesium and calcium levels throughout the menstrual cycle, possibly causing PMS in vulnerable women.3
Diagnosis
Four key elements are required to make the diagnosis of PMS.2 Diagnosis must be made by maintaining a prospective diary for at least two menstrual cycles. The key elements are shown in Table 1. Symptoms consistent with PMS are shown in Table 2.
Table 1: Key elements to diagnosis of PMS

Table 2: PMS symptoms
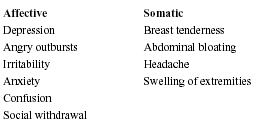
Treatment
Because the etiology is not precisely understood, the treatment options for PMS are many. Both lifestyle and pharmacologic management have been studied and commonly are used. A standard approach is to begin lifestyle modification prior to pharmacologic management, reserving pharmacologic management for those who fail to benefit from lifestyle modification. Lifestyle modifications include mind-body approaches, aerobic exercise, and supplementation with vitamins, minerals, and complex carbohydrates. Pharmacologic management includes selective serotonin reuptake inhibitors, anxiolytics, diuretics, GnRH analogs, and oral contraceptive pills. Because many studies use different diagnostic criteria and heterogeneous patient populations, making firm conclusions about which patients benefit from certain approaches is difficult.
Many types of dietary supplements have been advocated for the reduction of PMS symptoms.4 Conflicting results have been found with trials of vitamin B6. Similarly, evening primrose oil has conflicting results; however the most rigorous studies demonstrated no benefit. Supplements for which there is limited evidence for efficacy include magnesium, vitamin E, and carbohydrate supplements. The one supplement that has been shown to have significant benefit is calcium.
Calcium, Magnesium, and PMS
Ovarian hormones influence calcium, magnesium, and vitamin D metabolism. In a small prospective study, 10 women with regular menstrual cycles had concentrations of hormones, ionized calcium, and magnesium levels measured in five different stages of the menstrual cycle: menstrual phase, early follicular, late follicular, ovulatory/early luteal, and luteal phases.3 In each woman, calcium and magnesium levels consistently changed during the menstrual cycle. The authors conclude that the changes could produce premenstrual symptoms in certain women.
Clinical trials of magnesium supplementation are not highly convincing. One study was a small, double-blind, randomized clinical trial of 32 women with PMS by Moos Menstrual Distress Questionnaire (MMDQ), a validated tool for measuring PMS.5 Patients were randomly assigned to receive either magnesium pyrrolidone carboxylic acid (360 mg magnesium) or placebo, three times per day from day 15 of the menstrual cycle to the onset of menses. The MMDQ scores for pain were reduced in both groups, whereas magnesium treatment significantly reduced total MMDQ scores and assessment of negative affect. However, the groups appeared to differ at baseline with respect to the total score. Also, studies of PMS typically show a placebo response, at least initially, but this study demonstrated no placebo effect at the end of the first two months. However, once the placebo group began the active treatment, scores did decrease.
A double-blind crossover study over four menstrual cycles also investigated the effects of magnesium supplementation on PMS.6 Premenstrual symptoms were grouped into four categories: anxiety-related, craving-related, depression-related, and hydration-related, (i.e., swelling, bloating, and breast tenderness). Subjects received either placebo or 200 mg of magnesium, as magnesium oxide, for two cycles and then crossed over to receive the other, either magnesium or placebo for two more cycles. In the second month of treatment, there was a statistically significant reduction in symptoms within the hydration-related subgroup with magnesium compared to placebo. No changes were seen in the other subgroups.
Calcium has been studied extensively for PMS, and the results are fairly convincing. As Thys-Jacobs notes, there is striking similarity between the symptoms and PMS and hypocalcemia.7 A literature review of Medline from 1967 to 1999 concluded that calcium supplementation of 1,200-1,600 mg/d, unless contraindicated, should be considered a sound treatment option in women who experience premenstrual syndrome.8
A large, multicenter study enrolled healthy, premenopausal women with moderate-to-severe PMS.9 Symptoms were prospectively documented over two menstrual cycles with a daily rating scale that had 17 core symptoms and four symptom factors (negative affect, water retention, food cravings, and pain). Almost 500 women enrolled in this study, with 466 valid for the efficacy analysis. Subjects were randomized to receive either placebo or 1,200 mg elemental calcium per day in the form of calcium carbonate for three menstrual cycles. The primary outcome was the 17-variable symptom complex score. Groups were similar at baseline. Compared to those in the placebo group, subjects in the calcium-treated group had lower symptom complex scores for both the second and third treatment cycles. By the third treatment cycle, calcium effectively resulted in an overall 48% reduction in total symptom scores from baseline compared with a 30% reduction in the placebo group. All four symptom factors were also significantly reduced by the third treatment cycle. The authors note that calcium supplementation may act by repleting an underlying physiologic deficit, suppressing parathyroid hormone secretion, and ultimately reducing neuromuscular irritability and vascular reactivity.
Contraindications and Dosage
The safe, tolerable maximum dosage of calcium is 2,500 mg per day. Calcium should not be taken at the same time as iron because calcium blocks iron absorption. The recommended dosage for PMS is 1,200-1,600 mg per day calcium, in divided doses. It is difficult to achieve this dosage by food intake alone. (See Table 3 for food sources of calcium.)
Table 3: Calcium content of foods

Patients with vitamin D toxicity and those with hypercalcemia and risk factors for hypercalcemia should not use supplemental calcium. Low-dose magnesium, such as 200 mg of magnesium oxide, can be used to reduce mild symptoms of fluid retention. High doses of magnesium can cause diarrhea. Patients with renal disease should use magnesium only under the supervision of a physician.
Conclusion
Calcium supplementation has strong evidence for efficacy in improving PMS symptoms. Less evidence exists for using magnesium for PMS. Calcium has other benefits besides treating PMS, such as beneficial effects on bone. Also, low calcium intake has been implicated in the development of hypertension and colon cancer.10 Calcium can be a first-line approach to treating PMS, once the diagnosis is clearly made using prospective charting. If calcium is not beneficial in improving PMS symptoms, the other lifestyle and pharmacologic approaches can be tried.
Dr. Ball, Assistant Research Professor University of Pittsburgh Pittsburgh, PA, is on the Editorial Advisory Board of Alternative Therapies in Women’s Health.
References
1. Rapkin A. A review of treatment of premenstrual syndrome and premenstrual dysphoric disorder. Psychoneuroendcrinology 2003;28(Suppl 3):39-53.
2. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin: Premenstrual Syndrome. 2000:Number 15.
3. Muneyvirci-Delale Z, et al. Sex steroid hormones modulate serum ionized magnesium and calcium levels throughout the menstrual cycle in women. Fertil Steril 1998;69:958-962.
4. Bendich A. The potential for dietary supplements to reduce premenstrual syndrome (PMS) symptoms. J Am Coll Nutr 2000;19:3-12.
5. Facchinetti F, et al. Oral magnesium successfully relieves premenstrual mood changes. Obstet Gynecol 1991;78:177-181.
6. Walker A, et al. Magnesium supplementation alleviates premenstrual symptoms of fluid retention. J Womens Health 1998;7:1157-1165.
7. Thys-Jacobs S. Micronutrients and the premenstrual syndrome: The case for calcium. J Am Coll Nutr 2000; 19:220-227.
8. Ward M, Holimon T. Calcium treatment for premenstrual syndrome. Ann Pharmacother 1999;33: 1356-1358.
9. Thys-Jacobs S, et al. Calcium carbonate and the premenstrual syndrome: Effects on premenstrual and menstrual symptoms. Premenstrual Syndrome Study Group. Am J Obstet Gynecol 1998;179:444-452.
10. Power M, et al. The role of calcium in health and disease. Am J Obstet Gynecol 1999;181:1560-1569.
Balk JL. Calcium and magnesium for prementrual syndrome. Altern Ther Women's Health 2004;6(5):33-36.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
